Abstract
Rotavirus (RV) is an important viral pathogen causing diarrhea in piglets and other mammals worldwide. We describe 34 cases from 4 diarrheal outbreaks caused by RV in unvaccinated farrowing units in southern Brazil from 2011 to 2013. We performed autopsy, histologic examinations, bacterial culture, RV immunohistochemistry (IHC), and enteric virus detection through molecular assays for rotavirus A, B, and C, transmissible gastroenteritis virus, porcine epidemic diarrhea virus, sapovirus, norovirus, and kobuvirus. Histologically, villus atrophy (29 of 34) and epithelial vacuolation (27 of 34) occurred in all 4 outbreaks. Cell debris in the lamina propria occurred in 20 cases, mostly from outbreaks A (8 of 11), C (4 of 6), and D (7 of 11). IHC was positive for RV in 21 of 34 samples. RT-PCR was positive for RV in 20 of 30 samples; RV-C was the most frequently detected RV (
Introduction
Diarrhea is among the most prevalent health problems in swine, causing economic losses related to death and reduced weight gain. Among the viral pathogens, rotavirus (RV) is one of the most frequent causes of diarrhea in suckling piglets.9,19
Swine that are naive to RV are more susceptible to viral infection before weaning, particularly neonates, given their slower enterocyte turnover rate. 12 Usually, clinical signs are observed 19–24 h post-infection (hpi) and are characterized by anorexia, vomiting, apathy, and profuse diarrhea that can persist for 10 d, causing up to 15% weight loss. 8 RV is classified in different groups or species based on viral protein 6 (VP6) antigenic- and nucleotide-related features, and RV from types A (RV-A), B (RV-B), C (RV-C), E (RV-E), and H (RV-H) have been detected in pigs. 34 However, the importance of RV-C has increased in swine.25,26
Grossly, RV causes emaciation, gas-distended bowels, and liquid-to-pasty intestinal contents in the spiral colon and cecum. 8 The histologic lesions observed in infected swine intestines include reduction in the villus-to-crypt length ratio, villus loss and fusion, cuboidal-to-flat epithelial cells at the tips of villi, epithelial vacuolation, and epithelial desquamation in the small intestine.6,8
There are several methods for the detection of RV, including electron microscopy (EM) and polyacrylamide gel electrophoresis (PAGE), but to date, reverse-transcription (RT)-PCR is the method of choice for RV detection because it has high sensitivity, and various protocols for this technique allow differentiation of rotavirus strains. 32 Immunohistochemistry (IHC) can be used for RV infection detection and has the advantage of associating the agent with the histologic lesion, making it a highly specific technique. IHC stains RV-positive granules in the cytoplasm of enterocytes, with a positive staining peak at 31–48 hpi. 12 The staining is more intense in the distal and middle portions of the small intestine, although there can be positive enterocytes in the duodenum, cecum, and colon, without differences related to the age of the animal. 12 The disadvantage of this technique is that for an agent with high diversity, such as RV,5,34 specific antibodies are required.
Other viruses that can cause villus atrophy and epithelial vacuolation in swine are included in the differential diagnoses. Porcine epidemic diarrhea virus (PEDV), transmissible gastroenteritis virus (TGEV; species
Materials and methods
Sample selection and autopsies
We analyzed 34 suckling piglets with diarrhea during 4 outbreaks (A–D) that occurred in cities from the eastern central and northern regions of Rio Grande do Sul (RS) state and from the southern region of Santa Catarina (SC) state, both in the south of Brazil (Table 1). Outbreaks occurred in single farms or in several neighboring farms affecting pigs in the same age group, with similar clinical signs. The team from the Veterinary Pathology Sector of the Universidade Federal do Rio Grande do Sul (UFRGS) performed autopsies on 32 affected piglets that were severely dehydrated and in poor body condition.
Clinical data of affected piglets per farm in rotaviral outbreaks in southern Brazil, 2011–2013 (

RS = Rio Grande do Sul; SC = Santa Catarina.
In total, 28 severely affected piglets were euthanized with a barbiturate overdose, and intestinal tissue samples were taken in 10% buffered formalin 10 min after euthanasia (these samples were the first to be taken in these cases). One piglet from outbreak A and 3 piglets from outbreak D died before the veterinary pathology team arrived at the farms; therefore, these pigs were not included in the enteric viral assay and bacterial culture because they were in a more advanced state of autolysis compared to piglets sampled immediately after euthanasia. Nevertheless, these 4 piglets were included in IHC and histologic analysis.
Additionally, veterinary practitioners who attended outbreak B euthanized 2 piglets with a barbiturate overdose, immediately performed autopsies, and collected and sent refrigerated and 10% buffered formalin–fixed intestinal samples to the Veterinary Pathology Sector before the team visited the affected farm. The enteric viral assays using RT-PCR and bacterial culture and isolation were performed on 30 fecal samples taken directly from intestinal lumens that had been kept refrigerated for 48 h.
Histology
Samples of brain, lung, liver, kidney, heart, stomach, skeletal muscle, mesenteric lymph nodes, spleen, cecum, and colon, plus random sections of jejunum and ileum, were fixed in 10% buffered formalin and routinely processed for histologic (H&E) evaluation in 32 cases. In the 2 cases sent by field veterinary practitioners from outbreak B, only samples from small intestine, cecum, and colon were processed for histologic analysis. A total of 34 samples of small and large intestines underwent histologic and IHC evaluation. Histologic evaluation included description of lesions observed and detection of infectious agents that can be confirmed through this method, such as coccidia or
Immunohistochemistry
Sections of jejunum, ileum, and colon from all 34 piglets, including autolyzed sections, underwent IHC staining using streptavidin–biotin linked to peroxidase. Endogenous peroxidase blocking was performed through incubation of the sections in 10% hydrogen peroxide in methanol solution for 15 min. Citrate buffer (pH 6.0) was applied for antigen recovery at 125°C for 7 min.
Antibody against RV-A VP6 protein (Rotavirus Omnitope polyclonal goat serum, ViroStat, Portland, ME) was diluted 1:100 in phosphate-buffered saline (PBS) and applied to the slides in a humid chamber overnight at 4°C. Subsequently, slides were incubated with secondary biotinylated antibody linked to streptavidin–peroxidase (K0690, LSAB-HRPTM, Dako North America, Carpinteria, CA) for 20 min for each solution. The reaction was then incubated with 3-amino-9-ethylcarbazole (AEC; K3464, AEC substrate-chromogen, Dako). The slides were counterstained using Mayer hematoxylin and mounted in an aqueous medium (S1964, Ultramount permanent mounting medium, Dako). A slide of pig intestinal sections that tested positive previously for RV-A in both PCR and IHC was used as the positive control. Histologic sections of the positive control incubated with PBS were used as negative controls. Additionally, 2 positive samples from outbreaks B and C underwent the same protocol using an antibody against
Positive intestinal sections were examined (at a magnification of 400×), and a positive cell per field (CPF) count was established using the average of 10 fields. Sections with a CPF count <1 were considered mild IHC labeling, whereas a CPF count of 1–1.5 was considered moderate, and a CPF >1.5 was considered marked.
Enteric viral assay
Intestinal loops with contents from 30 piglets were submitted for the detection of enteric viruses using molecular methods. RT-PCR techniques were applied for RV-A, RV-B, RV-C, coronavirus (TGEV and PEDV), enteric calicivirus (SaV and NoV), and KoV. Viral nucleic acid was extracted from 10–20% (w/v) fecal suspensions in Tris-Ca++ buffer (50 mM Tris-HCl, 10 mM NaCl, 1.5 mM 2-mercaptoethanol, 3 mM CaCl2). 2 Fecal samples that were positive in molecular assays and confirmed by sequence analysis were used as positive controls for RV-A, RV-B, and RV-C, 24 SaV, 3 NoV, 33 and KoV 31 in all procedures. Sterile ultrapure water was used as a negative control. Because porcine fecal samples positive for most viruses evaluated were included in the specific RT-PCR assays, we did not use a reference gene control.
RT-PCR assays were performed with consensus primers for amplification of 876- and 1,062-bp products from
The RT-PCR assays to detect coronavirus (TGEV and PEDV) were carried out using specific primers to amplify the spike protein coding region (859 bp) and the membrane protein gene (377 bp), respectively.20,21 The RT-PCR assay for SaV was performed using primers
18
that detect enteric caliciviruses (319- or 331-bp fragment from
Bacterial culture
Bacterial culture and isolation was performed on 30 fecal samples from the autopsied piglets. Samples from loops of colon and small intestine with contents underwent aerobic and anaerobic culture according to standard laboratory protocols.27,28
Results
The diarrhea outbreaks occurred from May 2011 to August 2013. All piglets evaluated had liquid-to-pasty diarrhea, dehydration, and weight loss, and none of the herds was vaccinated against RV-A. The piglets evaluated were from birth to 20-d-old (Table 1).
All autopsied piglets were emaciated and dehydrated, had liquid-to-pasty contents in the cecum, colon, rectum, and perineum, and had distended, thin-walled intestines. In outbreaks B and C, the piglets also had full stomachs.
The most frequent small intestinal histologic lesions were villus atrophy (29 of 34), vacuolation of the epithelial cells at the tips of villi (27 of 34), cell debris in the lamina propria (LP; 20 of 34; Fig. 1), and epithelial attenuation (12 of 34; Table 2). In outbreak D, RV and KoV were detected in 6 samples each. RV RT-PCR detection occurred in 3 of 8 samples, and IHC detection occurred in 5 of 11 (4 of 8 undergoing both tests) samples, with 1 sample positive in both RT-PCR and IHC.
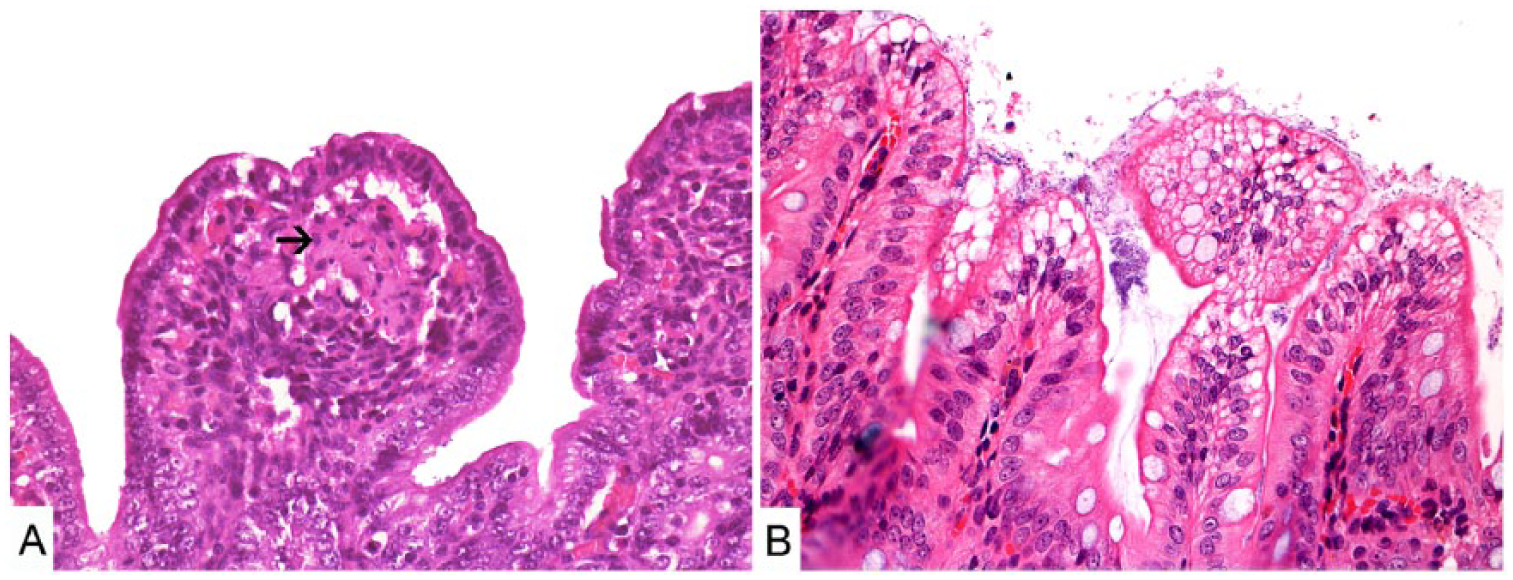
Histologic sections of piglet small intestine.
Lesions observed in the small intestine of piglets autopsied per rotaviral outbreak in southern Brazil, 2011–2013 (

BA = bacteria adherent; C = coccidia; CD in LP = cell debris in lamina propria; CH = crypt hyperplasia; E = edema; EA = epithelial attenuation; EN = enterocyte necrosis; EV = epithelial vacuolation; FE = fibrin exudate; LD = lacteal dilation; LI = lymphocytic infiltrate; MI = mixed infiltrate; NI = neutrophilic infiltrate; VA = villus atrophy.
In outbreak A, only discrete lesions were present in 3 of 11 cases; none of the cases of this outbreak had bacteria adhered to large intestinal mucosa. In outbreaks B and C, large intestinal lesions were similar, with a high proportion of cases with bacteria adherent to the mucosa (6 of 12 cases). In outbreak D, 3 of 11 cases had multiple lesions; large intestines were normal in the other cases of this outbreak. Vacuolation of superficial epithelium and bacteria adherent to the mucosa were observed in 3 of 11 cases each. Neutrophilic infiltrate in the mesentery and neutrophils in the lumen were features observed only in this outbreak (1 of 11 cases).
Positive IHC staining occurred in 21 of the 34 cases (Fig. 2). Outbreaks A, B, C, and D had 8, 5, 3, and 5 cases stain positively using IHC, respectively (Table 3). Marked IHC staining was observed in 5 cases of outbreak A and 2 cases from outbreak B. Outbreaks C and D had no cases with marked staining (Table 3). The CPF averages of the positive slides in outbreaks A, B, C, and D were 7.4, 1.3, 0.7, and 1.2, respectively. The patterns of labeling differed in outbreaks A and D from outbreaks B and C. In outbreaks A and D, multiple granules occupied the entire cytoplasm, whereas in outbreaks B and C, strongly stained oval-to-round structures were in close proximity to the nucleus. Samples tested with the isotype control antibody did not have any labeling. There was no IHC staining in the crypt cells or in the enterocytes of the cecum and colon.
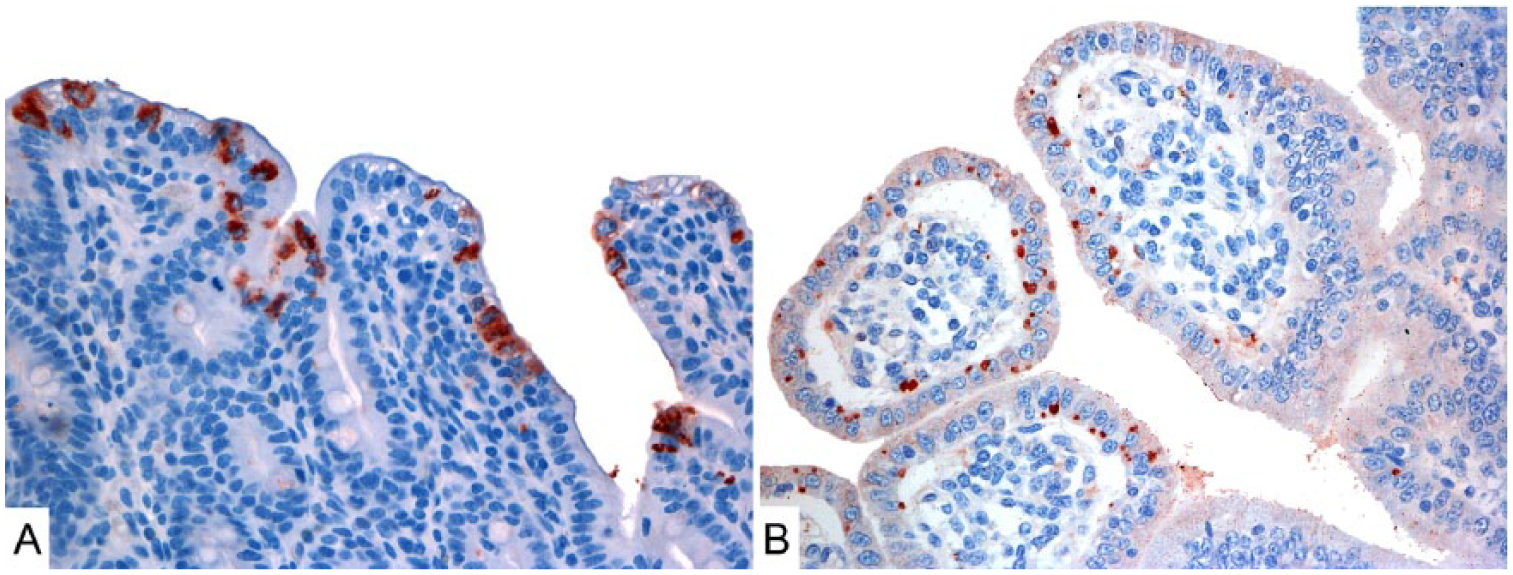
IHC-stained histologic sections of porcine small intestines from outbreaks A and C.
Staining intensities of IHC per rotaviral outbreak in piglets from southern Brazil, 2011–2013 (
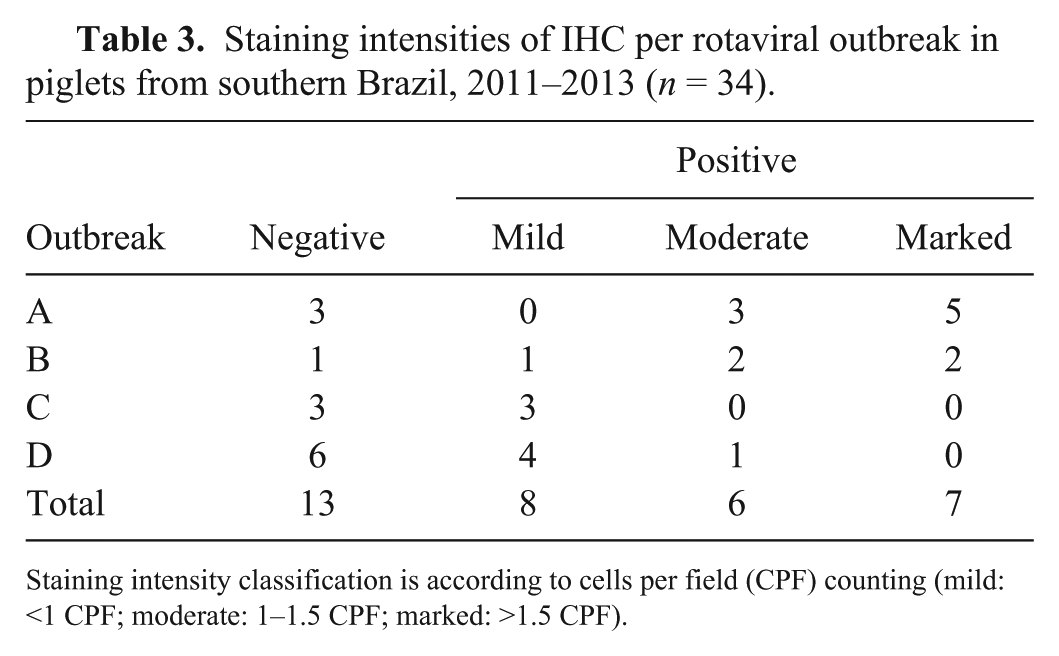
Staining intensity classification is according to cells per field (CPF) counting (mild: <1 CPF; moderate: 1–1.5 CPF; marked: >1.5 CPF).
RV was detected by molecular assays in 20 of 30 cases (Table 4).
Rotavirus (RV) infection cases in autopsies of piglets with single and multiple RV types detected per outbreak using molecular assays.
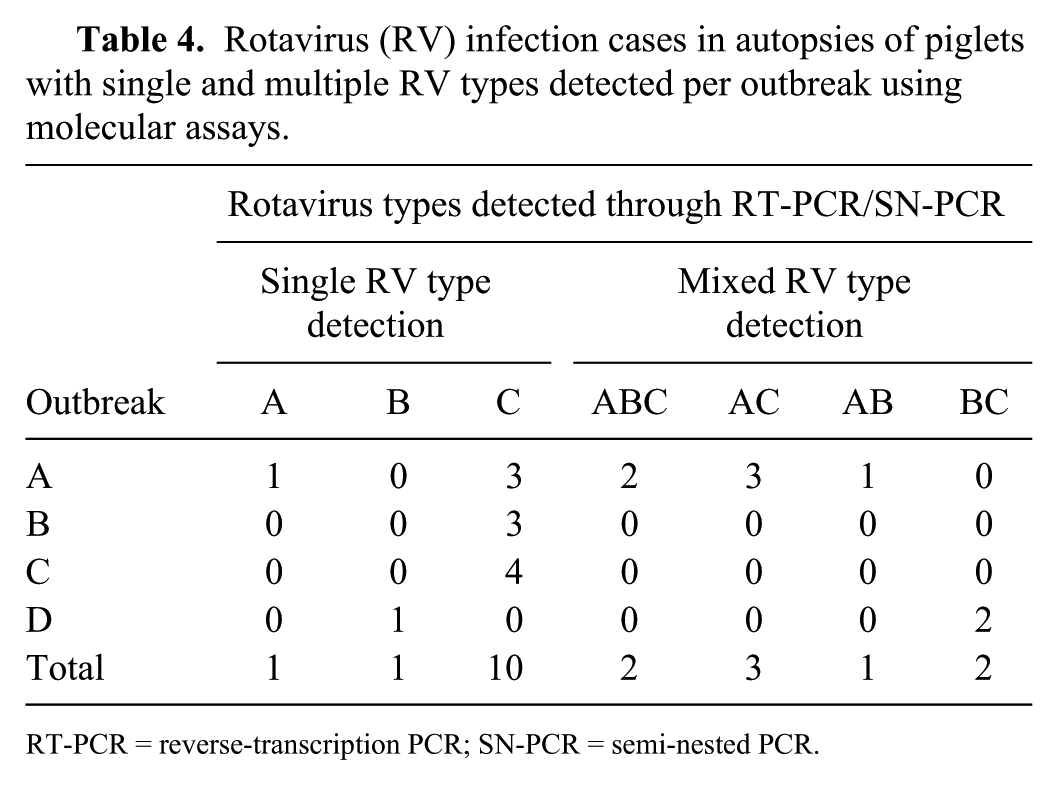
RT-PCR = reverse-transcription PCR; SN-PCR = semi-nested PCR.
Four cases in relatively advanced autolysis underwent histologic and IHC analysis; however, they were not included in the enteric viral assay, therefore the RV survey through both RT-PCR and IHC were performed concomitantly in only 30 of 34 cases. Detection of RV by either RT-PCR or IHC occurred in all cases tested with both techniques in outbreak A (10 of 10 cases), 5 of 6 cases from outbreak B, 5 of 6 cases from outbreak C, and 6 of 8 cases tested in outbreak D. Agreement between RT-PCR and IHC detection occurred in 17 of 30 cases tested with both techniques.
Detection results of SaV, NoV, KoV, PEDV, and TGEV per outbreak are summarized in Table 5.
Detection of other enteric viruses in rotavirus-associated diarrhea cases affecting piglets in southern Brazil, 2011–2013.

KoV = kobuvirus; NoV = norovirus; PEDV = porcine epidemic diarrhea virus; SaV = sapovirus; TGEV = transmissible gastroenteritis virus; dash (–) = not detected.
Bacterial isolation and microscopic observation in rotavirus-associated diarrhea cases affecting piglets occurring in southern Brazil, 2011–2013.
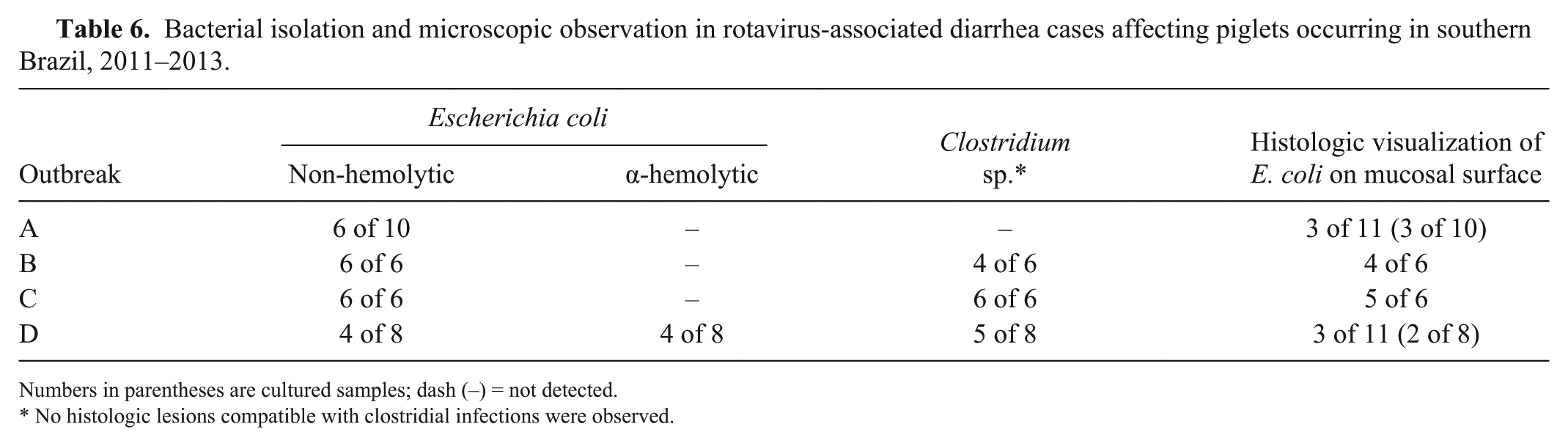
Numbers in parentheses are cultured samples; dash (–) = not detected.
No histologic lesions compatible with clostridial infections were observed.
Discussion
None of the farms studied had vaccinated against RV-A, hence piglets may have been more susceptible to RV-A infection. Herds from outbreak A had been vaccinated against enterotoxigenic
Villus atrophy, epithelial vacuolation, epithelial attenuation, and cell debris in the LP were the most common microscopic alterations observed in our study. These lesions are frequently described in rotaviral enteritis, with vacuolation of enterocytes observed 1–7 d post RV infection as well as a high level of apoptosis of enterocytes during RV infection. 4 Villus atrophy can be observed in piglets by 36 hpi, with recovery starting after 90 hpi. 8
Rotavirus was detected through either IHC or RT-PCR in all samples of outbreak A, and there were a few cases with isolation and observation of
Histologic observation plus bacterial isolation were the criteria used to diagnose colibacillosis in our study. In outbreak B, bacteria compatible with
Outbreak C had 5 cases positive (in IHC and/or RT-PCR) for RV, and 5 cases with
Outbreak D had lesions in the small intestine similar to those in the first outbreak; the large intestine had cases in which bacteria were observed adhered to small and/or large intestine. In outbreak D, RV was detected by either RT-PCR or IHC in 6 of 8 samples.
Two samples from outbreak D and 1 from outbreak C were positive only for KoV, did not have bacteria adherent to the intestinal mucosa, were not positive for RV antigens in the IHC, and had microscopic lesions in the small intestine. These cases had moderate-to-marked villus atrophy and epithelial vacuolation, and mild presence of cell debris in the LP. Porcine KoV is endemic in many farms and is distributed worldwide. It is associated with diarrhea and has caused viremia in pigs. 30 The most likely scenario is that these lesions were caused by RV because the lesions can occur in intestinal segments where there is no virus. 8 Moreover, no reference gene controls were added to the enteric virus assay, therefore some of the 10 RV-negative samples in our study could be attributed to the presence of inhibitors during RNA extraction or PCR procedures. However, villus atrophy and epithelial vacuolation are caused by several intestinal viruses,6,7,16,30 and it is possible that KoV caused these lesions.
Outbreak D also had some cases with colitis. Submucosal congestion, neutrophilic infiltrate, and mesenteric edema were observed in 2 cases each. Inflammatory lesions in the digestive system were observed in a 2015 reproduction of KoV infection in piglets 35 ; therefore, our findings could be related to KoV detection in these cases.
All samples were negative for TGEV and PEDV. Worldwide, these viruses cause outbreaks of diarrhea and should always be included in the differentials for diagnosis of diarrhea in pigs.19,20 However, these viruses have not been reported to occur in Brazil.
IHC staining revealed RV antigens in 21 cases. Outbreak A had the most intense staining, with 5 of the 8 positive cases staining markedly. Sample selection could have affected the sensitivity of this detection method, given that we collected sections of several portions of jejunum and ileum instead of a standardized selection of only distal jejunum and ileum, where RV is found in higher titers. 8
RV-A, RV-B, and RV-C were detected in outbreak A. The higher intensity of IHC labeling observed can be attributed to the major presence of RV-A in this outbreak. Most of the piglets affected in this outbreak were <1-wk-old, which reinforces the positive relationship between young age and RV infection. 12
Samples from outbreaks A and D that were RT-PCR negative for RV-A but positive for RV-C, or for RV-B and RV-C, exhibited a regular whole-cytoplasm pattern of IHC staining, identical to the staining observed in RV-A RT-PCR–positive samples present in outbreak A. VP6 possesses high antigenic and genetic variation within the same RV species and between different RV species. 34 Nonetheless, some studies show evidence of a high homology between RV-A and RV-C. 5 Samples from outbreaks B and C, in which only RV-C was detected by RT-PCR in autopsy samples, had less intense IHC labeling, appearing as isolated round spots positioned next to the nucleus, suggestive of viroplasms. Viroplasms are electron-dense structures filled with VP6 antigen that are formed inside infected cells during RV replication. 10 It is likely that the IHC labeling was concentrated in viroplasms, resulting in positive results even with a low level of complementarity with the antibody used. However, because VP6 sequences were not assessed in our study, conclusions about the phenotypic differences in the VP6 among RV-C from outbreaks B and C and from outbreaks A and D cannot be drawn from the IHC staining differences observed among outbreaks.
RV-C is described as an important cause of diarrheal outbreaks in pigs. 26 To date, with improvement in sensitivity of detection techniques, the apparent prevalence of RV-C detection has increased.24,26 Our study reinforces these facts, given that RV-C was detected in 17 of 30 samples, 10 of which were single species detections. In Italy, a study showed that RV-C was the most prevalent enteric virus and was detected in 100% of the pig farms studied; nevertheless, single detection of RV-C was found in only 3 of 102 fecal samples analyzed in that study. 25 Our study agrees with these results, given that the outbreaks that were primarily affected by non–RV-A also had lesions related to other agents detected concurrently.
Footnotes
Acknowledgements
We thank our colleagues and professors for their technical support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Financial support was provided by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
