Abstract
We characterized naturally occurring pigeon herpesvirus (PiHV; Columbid alphaherpesvirus 1) infection in domestic pigeons in California. We retrieved and analyzed 62 pathology reports produced between 1991 and 2014 at the California Animal Health and Food Safety Laboratory System. In 56 of the cases, the diagnosis of PiHV infection was established based on histopathology, either alone (44 cases) or combined with virus isolation (VI; 8), transmission electron microscopy (TEM; 3), or PCR (1); in the remaining 6 cases, the diagnosis was established based on VI (5 cases) or TEM (1) alone. PiHV infection affected 1 system in 34, 2 in 16, and ≥3 systems in 6 cases; data were not available for the remaining 6 cases. Most commonly affected was the digestive system (55 cases), followed by the respiratory tract (5) and lymphoid system (2). The liver (39 cases), crop (17), and esophagus (14) were the organs affected most commonly. Many affected cells often bore single eosinophilic intranuclear inclusion bodies. PiHV infection was a secondary diagnosis or incidental finding in 35 cases. Most (55) cases had 1 (21), or up to 4 (34), other concurrent infections; the most common concurrent infections were pigeon circoviral infection (26), trichomonosis (24), aspergillosis (11), and colibacillosis (10).
Pigeon herpesvirus (PiHV; Herpesviridae, Mardivirus, Columbid alphaherpesvirus 1)10,11 is closely related to gallid herpesvirus 2 (serotype 1; Gallid alphaherpesvirus 2), the etiologic agent of Marek disease in chickens. 36 The natural hosts of PiHV are columbid birds, including domestic and free-ranging pigeons and doves.17,21,23 Non-columbid birds, including diurnal and nocturnal birds of prey and others, can also be affected.14,22–24,27,36 Several avian species can be carriers of PiHV, and most pigeons worldwide are infected latently by PiHV and can shed PiHV intermittently through feces and nasal secretions.9,20,21,37 Shedding is increased in individuals suffering from stress associated with factors such as breeding, racing, shows, high flock density, nutritional deficiencies, poor hygiene, and concomitant diseases.3,10,13,16,21
PiHV-associated disease, seen mainly in pigeons 2–10-wk-old not protected by maternal antibodies,10,21 usually causes systemic infections resulting in sudden death with no premonitory signs. In older pigeons, PiHV infection usually causes upper respiratory tract infections characterized clinically by sneezing, dyspnea, and unilateral or bilateral conjunctivitis.10,13 Other clinical signs, such as anorexia, depression, vomiting, diarrhea, torticollis, and paralysis, may also be observed.10,13,21,37 Lesions of PiHV-associated disease include necrosis, with diphtheritic membranes in the upper respiratory and digestive tracts, hepatomegaly, and hepatocellular degeneration and/or necrosis. A diagnostic microscopic finding is the presence of amphophilic intranuclear inclusion bodies (INIBs) within epithelial cells of the respiratory and digestive tracts and hepatocytes.2,5,14,24 Because of the finding of INIBs, the term “herpesviral inclusion-body disease” has been used widely to describe PiHV infection in columbid and non-columbid birds.2,5,10,13,20,21,33
PiHV infection, first identified in pigeons from New York State in 1943, 31 was initially termed Smadel disease, 37 and was characterized 2 y later in U.S. Army homing pigeons. 32 Since then, individual cases or outbreaks in domestic and free-ranging pigeons have been reported throughout the world, including Australia,6,23 Belgium,10,21 China,15,39 Hungary, 34 Japan, 38 Morocco, 18 the Netherlands,16,20 New Zealand, 33 Poland, 36 South Africa, 25 Spain, 5 Thailand, 12 the UK,8,9 and throughout the United States.2,13,29,31,32
Although the pathologic findings of naturally occurring PiHV-associated disease in domestic pigeons have been described, we found no analysis of the range of PiHV-affected target organs and commonly detected concurrent infections associated with this viral infection in domestic pigeons in a search of PubMed, CAB Direct, and Web of Science, suggesting that it is likely that no such analysis have been reported to date in pigeons. Our objective was to analyze the clinical and pathologic features of natural PiHV infection in racing, fancy, and meat pigeons submitted to the California Animal Health and Food Safety Laboratory System (CAHFS) of the University of California–Davis for autopsy and diagnostic workup between 1991 and 2014.
Materials and methods
Archival case retrieval
The archives of the Davis, San Bernardino, Tulare, and Turlock laboratories of CAHFS were searched for domestic pigeon necropsy submissions with a diagnosis of PiHV infection, received from January 1, 1991 to June 30, 2014. Cases from the Fresno Laboratory of CAHFS included submissions between January 1, 1991 and September 30, 2008, when that laboratory was closed. Pathology reports of all cases retrieved were reviewed, and we included cases with a diagnosis compatible with PiHV infection, either as the primary or secondary diagnosis. Data retrieved from all CAHFS pathology reports included: number of pigeons submitted, breed or production type (fancy, meat, or racing), county of origin, year of submission, methods by which the diagnosis of PiHV infection was based, gross and microscopic findings, and presence of concurrent infections.
The diagnostic differentiation between primary and secondary PiHV infection was made primarily by the pathologist in charge of each case studied and was also reviewed and confirmed by the authors of this paper. Ancillary tests were performed on all birds subjected to postmortem examination following CAHFS standard operating and routine procedures, unless otherwise specified.
Histology
Postmortem examination was performed in all cases, and samples of the oral cavity wall, tongue, pharynx/larynx, esophagus, crop, proventriculus, gizzard, small and large intestine, cloaca, liver, pancreas, conjunctiva, nasal cavity wall, sinuses, trachea, heart, lungs, air sacs, bone marrow, brain, eyes, ears, kidneys, skin, thymus, spleen, cecal tonsils, bursa of Fabricius, adrenal glands, testis, ovaries, and/or thyroid glands were collected and fixed by immersion in 10% neutral-buffered formalin (pH 7.2) for 24–72 h. All tissues were processed by standard histologic techniques to produce 4-µm, H&E-stained sections.
Bacteriology, virology, and parasitology
Samples of heart, trachea, lung, liver, spleen, and/or bone marrow were collected aseptically in most cases, inoculated onto MacConkey and 5% sheep blood Columbia agar plates (Hardy), and incubated aerobically or in a 5–10% CO2 atmosphere at 37°C for 24–48 h. Respiratory tract samples were inoculated aseptically onto modified Frey and Ortmeyer liquid and solid media (Hardy) and incubated in a 5–10% CO2 atmosphere at 35–37°C for 7 d. DNA extracts of liver samples were subjected to real-time PCR with primers designed to detect a fragment of the Salmonella-specific invA gene. 7 RNA extracts from oropharyngeal and cloacal swab samples were subjected to reverse-transcription real-time PCR (RT-rtPCR) to detect the avian influenza A virus matrix gene and pigeon paramyxovirus 1 matrix and fusion genes according to the national testing standard operating procedures published by the National Animal Health Laboratory Network (NAHLN). Small and large intestinal contents and fecal samples were analyzed by scraping and direct microscopic observation, and fecal flotation examination, respectively.
Results
A primary or secondary diagnosis of PiHV infection was made in 62 domestic pigeon cases, involving 97 birds during the 23.5-y study period. Of these, 21 cases (25 birds), 11 cases (14), 3 cases (3), 10 cases (10), and 17 cases (45) were submitted to the Davis, San Bernardino, Tulare, Fresno, and Turlock branches of CAHFS, respectively. PiHV infection was the primary infection in 27 and a secondary infection in 35 of 62 cases. In 18 and 13 of 31 cases in which age was specified, birds were >10-wk-old or 2–10-wk-old, respectively. Birds belonged to the racing, meat, or fancy breed or production type in 8, 13, and 4 of the 25 cases in which the breed or production type was specified, respectively.
The diagnosis of PiHV infection was based on histopathology, either alone (44 cases), or by histopathology combined with VI (8), TEM (3), or PCR (1). In a few cases, the diagnosis was based on VI (5 cases) or TEM (1 case) alone.
Grossly, the liver was the organ affected most frequently (38 cases), and in most cases had randomly distributed, pinpoint, pale necrotic foci (Fig. 1A). Most spleens had similar gross findings in our case series. Other gross lesions included hyperemic mucosa with or without randomly distributed necrotic foci in the oral cavity wall, pharynx, and larynx; caseous plaques covering mucosal surfaces of the pharynx, larynx, crop, and esophagus; focal-to-multifocal hyperemic esophageal and intestinal mucosa; variably sized, randomly distributed, pale nodules in lungs; and fibrinous exudate and white plaques in air sacs.
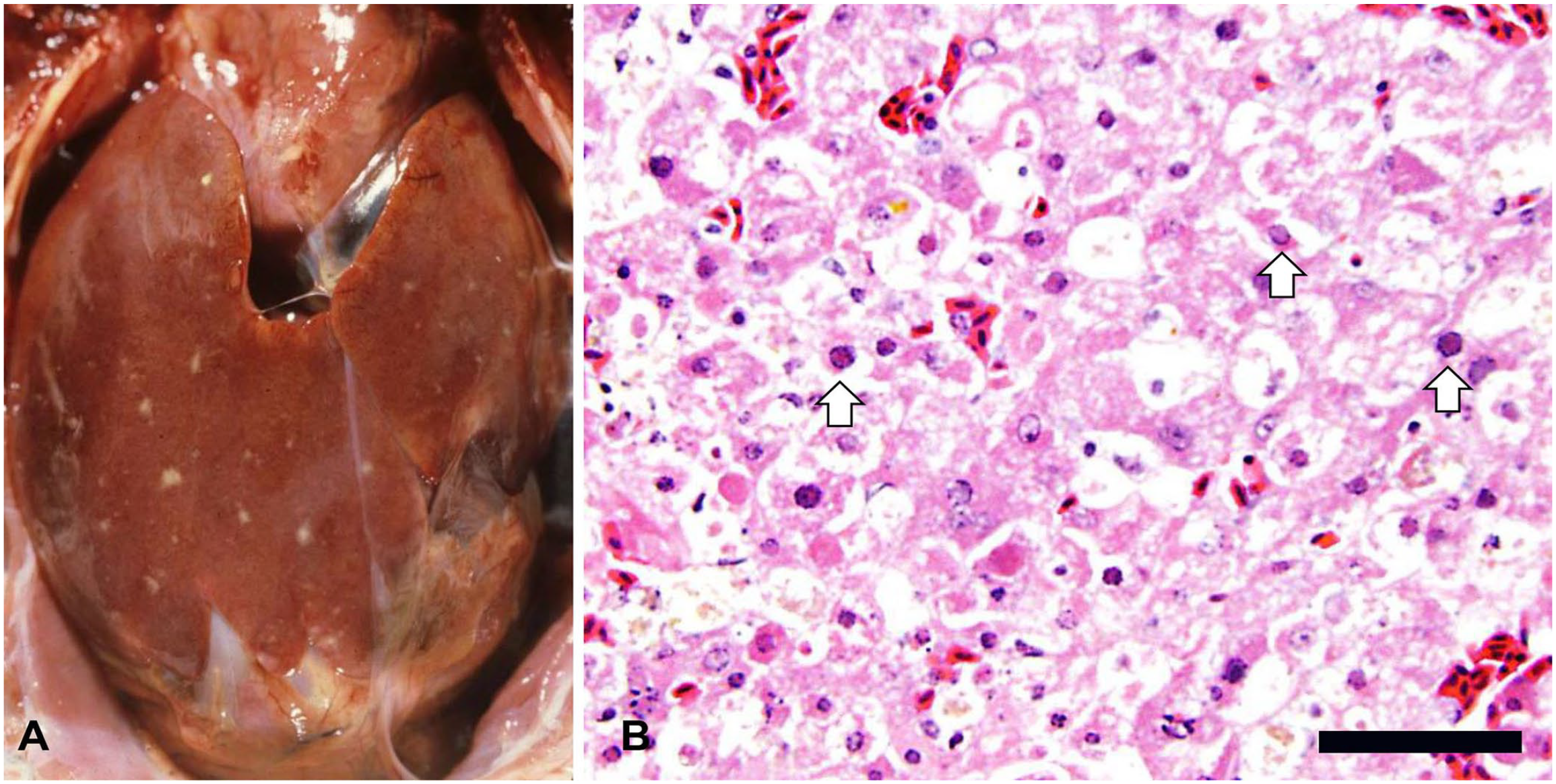
Liver of a pigeon with pigeon herpesviral infection.
The organs affected microscopically by PiHV infection included, in decreasing order of prevalence, the liver, crop, esophagus, spleen, intestine, lungs, trachea, oral cavity wall, pharynx and/or larynx and sinuses, conjunctiva, nasal cavity wall, pancreas and tongue, bone marrow, ear, kidneys and proventriculus, adrenal glands, air sacs, bursa of Fabricius, brain, cloaca, eye, gonads (testis and ovary), heart, skin, thymus, and thyroid gland (Fig. 2). No microscopic changes were observed in the other organs examined. Most birds had ≥2 organs affected (Fig. 3). The hepatic changes ranged from individual hepatocellular degeneration and necrosis (Fig. 1B) to focally extensive or multiple randomly distributed foci of coagulative necrosis. Areas of necrosis frequently had hepatocytes replaced by proteinaceous exudate and delimited by congested sinusoids. Affected hepatocytes often had single eosinophilic or variably basophilic INIBs that marginated chromatin and nucleoli. Microscopic changes observed in other organs ranged from individual epithelial or parenchymal cells undergoing degeneration or necrosis to randomly distributed, variably extensive areas of necrosis. Within these areas, epithelial or parenchymal cells often bore eosinophilic INIBs similar to those described in the liver, with or without associated mononuclear or mixed inflammatory cell infiltration of the lamina propria or neighboring parenchyma. Detection of eosinophilic INIBs in areas of necrosis of one or more organ was the single histologic criterion on which the histologic diagnosis of PiHV infection was based.
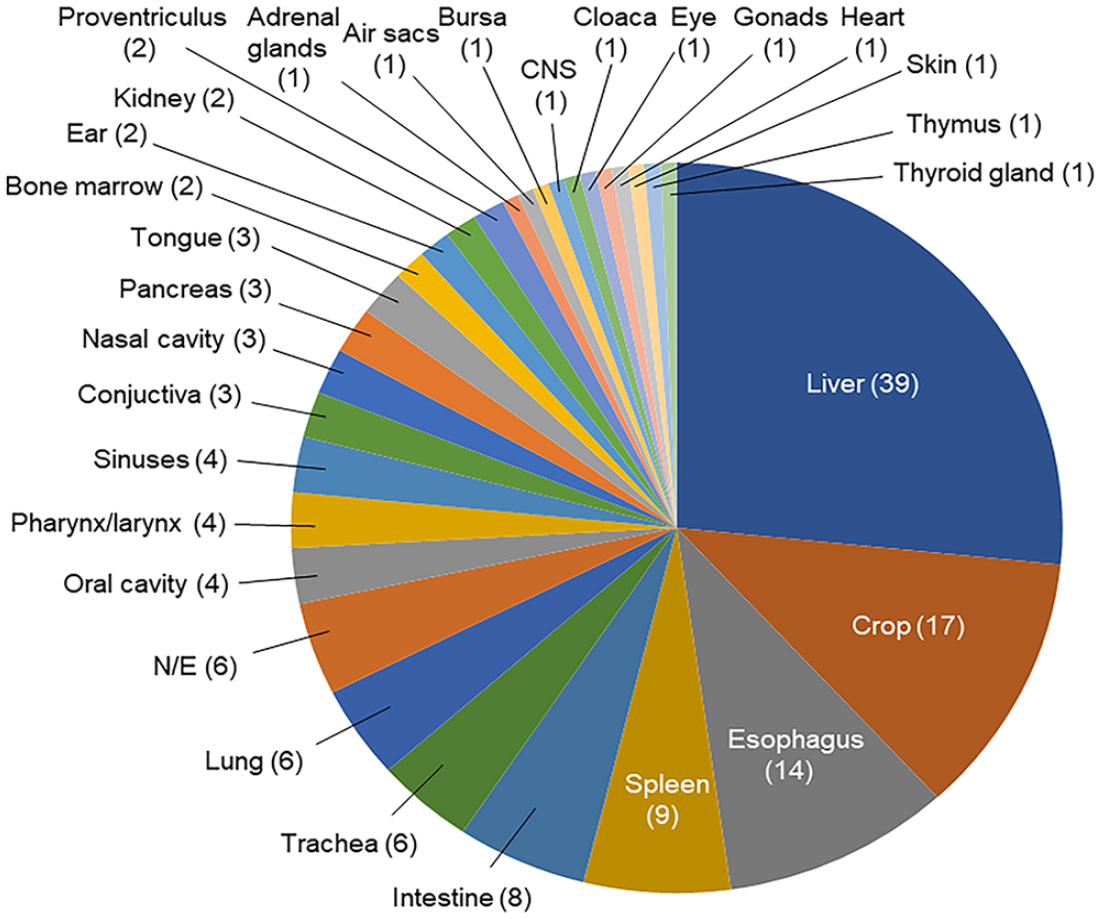
Organs affected by pigeon herpesviral infection in 62 cases diagnosed at the California Animal Health and Food Safety Laboratory System. N/E = no information was available on the systems affected.
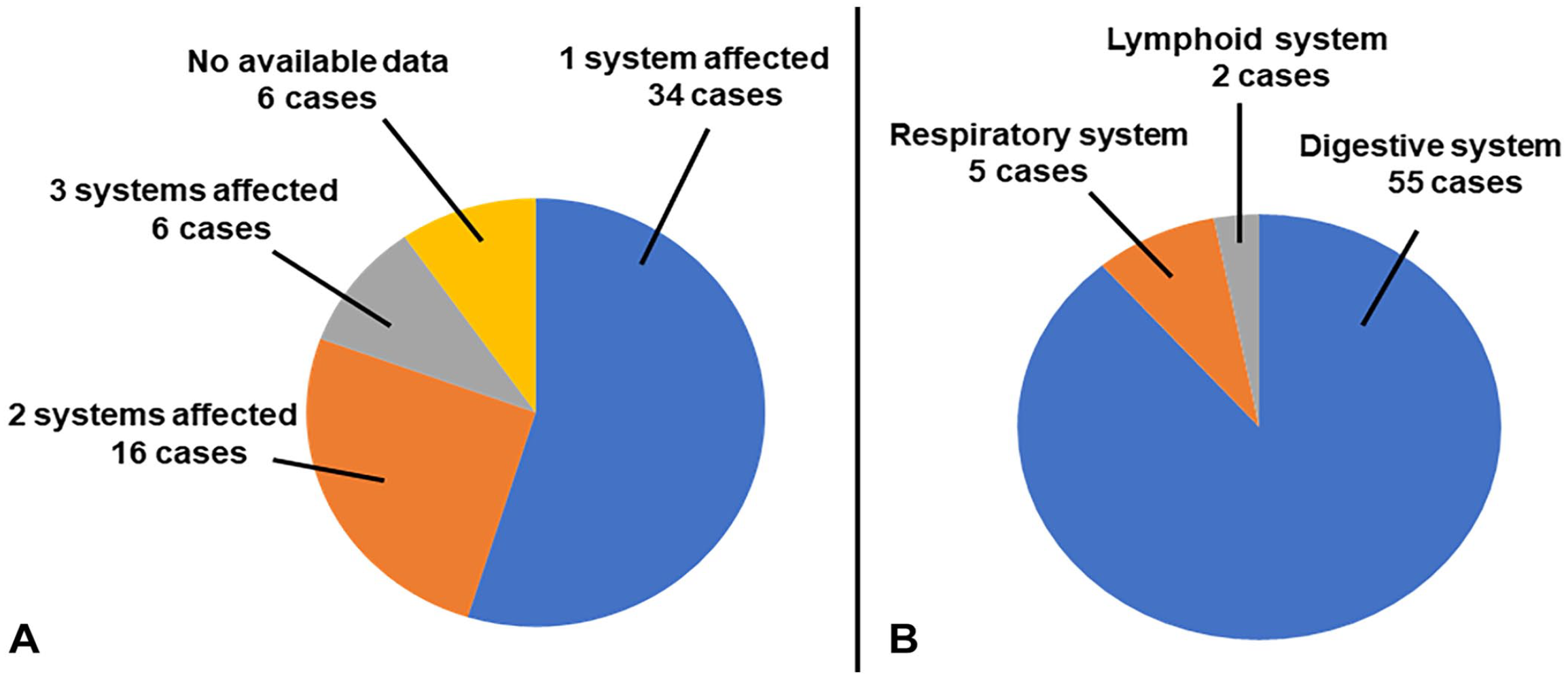
Systems affected by pigeon herpesviral infection in 62 cases diagnosed at the California Animal Health and Food Safety Laboratory System.
Lesions of PiHV infection were seen in 1 system in 34 of 62 cases, 2 systems in 16 cases, and ≥3 systems in 6 cases. In 6 cases, no information was available about the systems affected. In the cases in which only 1 system was affected, the digestive system was affected in 30 of 34 cases, whereas the respiratory and lymphoid systems were affected in 3 cases and 1 case, respectively (Fig. 3). The liver was affected in all cases in which only the digestive system was affected.
Most domestic pigeon cases primarily or secondarily affected by PiHV infection had either 1 (n = 21) or up to 4 (n = 34) concurrent viral, bacterial, fungal, or parasitic infections. These concurrent infections included, in descending order of prevalence, pigeon circovirus (PiCV; Circoviridae, Circovirus) infection, trichomonosis, aspergillosis, colibacillosis, salmonellosis, internal parasitic infections, pigeon pox virus infection, chlamydiosis, pigeon paramyxovirus infection, external parasitic infections, mycoplasmosis, staphylococcosis, crop candidiasis, coccidiosis, Spironucleus spp. infection, adenoviral infection, Pasteurella spp. infection, and Haemoproteus spp. infection (Fig. 4).
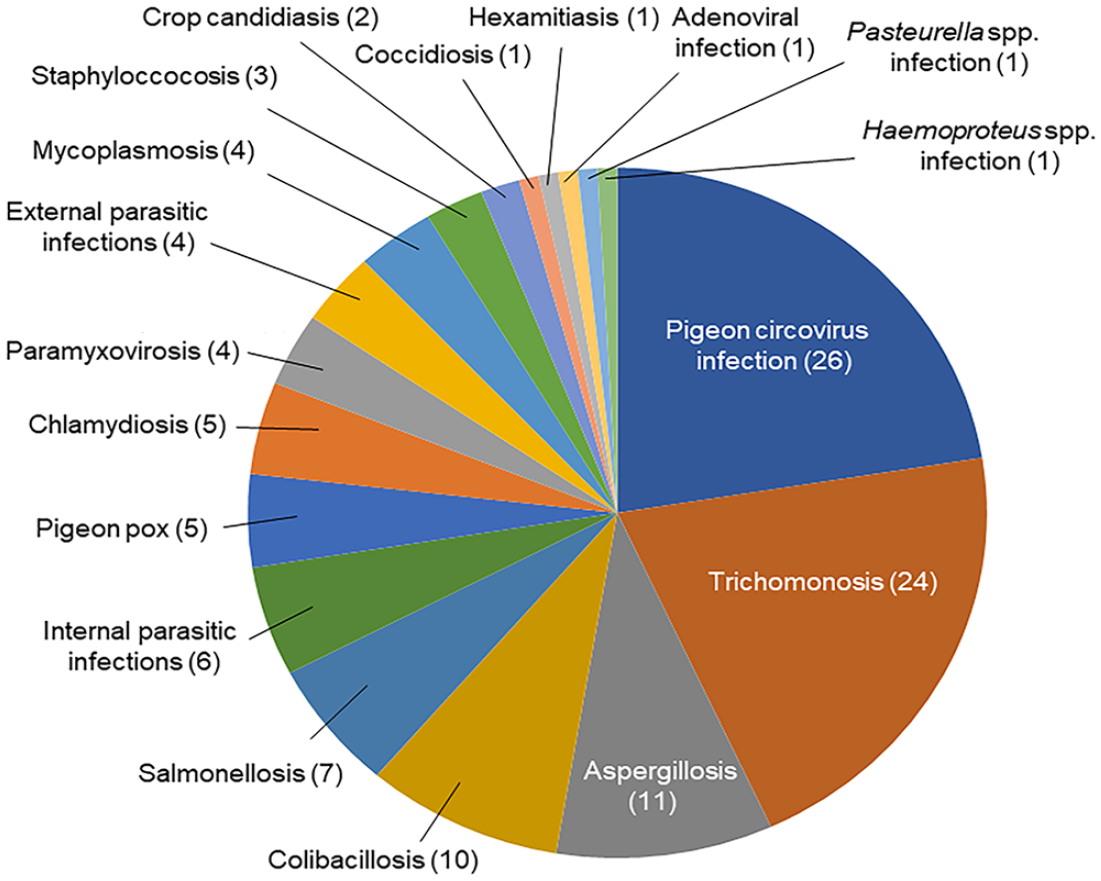
Concurrent infections associated with pigeon herpesviral infection in 62 cases diagnosed at the California Animal Health and Food Safety Laboratory System.
Discussion
The finding of eosinophilic INIBs in affected organs is diagnostic of PiHV infection. 2 In our retrospective study, the liver and upper and lower digestive tracts were the organs affected most commonly microscopically; other systems, including both the upper and lower respiratory tracts, were affected much less frequently. These findings contrast with previous descriptions of naturally occurring PiHV infections in which the upper respiratory and digestive tracts were the systems affected most frequently in pigeons.10,11 Birds >10-wk-old and birds of the meat production type were more frequently represented in our study; however, conclusions or correlations cannot be drawn on these parameters given the limited number of cases in which these data were specified.
The 4 most common concurrent coinfections in PiHV-infected domestic pigeons in our retrospective study were, in descending order, PiCV infection, trichomonosis, aspergillosis, and colibacillosis. PiCV is one of the major immunosuppressive pathogens of domestic pigeons, particularly affecting pigeons <1-y-old. 35 PiCV causes severe lymphoid depletion of the bursa of Fabricius and other lymphoid tissues, resulting in immunosuppression.1,9,35 Thus, PiCV predisposes to a plethora of secondary infections, including the young pigeon disease syndrome (YPDS) of juvenile pigeons reported in Europe. 26 In our case series, PiCV infection may have predisposed birds to concurrent infections with up to 4 pathogens in at least 26 cases. In addition, it is noteworthy that, in our series, there were 4 cases in 2–10-wk-old pigeons with concurrent infections mimicking YPDS.
Trichomonosis was the second most common concurrent disease in our case series. Trichomonosis, or canker, is a disease of the avian upper digestive tract caused by the flagellate Trichomonas gallinae.3,10 Pigeons are the primary host of this protozoal infection that principally affects squabs and young birds, with high mortality rates, but rarely adults.3,10 Grossly, white or yellow caseous plaques or caseous and necrotic nodules are noted in the oropharynx, esophagus, and crop mucosa, blocking the passage of food. 2 In our series, only 4 of 24 cases of PiHV-infected adult pigeons had the typical gross lesions of trichomonosis. The presence of immunosuppressive agents (such as PiCV) or nitroimidazole resistance 28 may have facilitated the pathogenicity of this protozoal agent in these birds.
Aspergillosis, the third most common concurrent infection in our case series, is an opportunistic fungal disease, mainly caused by Aspergillus fumigatus, that affects a wide range of avian species. 4 Grossly, firm, pale to yellow-or-white lung nodules or air sac plaques are seen. 2 Similarly, Aspergillus spp. affected the respiratory system in our series, and appeared to be involved in PiHV infection together with other viral and bacterial pathogens that cause serious respiratory problems in domestic pigeons. 10
Colibacillosis usually causes fibrinous pneumonia and airsacculitis and may also affect several organs of the celomic cavity; it has also been associated with coinfections. 10 Colibacillosis was the fourth most common concurrent infection in our series and was noted in 10 cases. 10
Although mycoplasma species affecting pigeons, including Mycoplasma columborale, M. columbinum, and M. columbinasale, were identified as highly prevalent primary or secondary respiratory pathogens of pigeons in several locations in Europe,16,19,30 including in PiHV-infected racing pigeon lofts from the Netherlands, 16 we found only a few such cases in PiHV-infected domestic pigeon cases in California. Similarly, other major pathogens of domestic pigeons, including Salmonella spp. and pigeon paramyxovirus, were also observed in small numbers in our study, indicating an apparent lack of association between these pathogens and PiHV infection. Affected skin, urogenital system, endocrine system, lymphoid system, bone marrow, and CNS had many eosinophilic INIBs with or without associated necrotic foci and inflammatory cell responses. These microscopic findings in systems other than the digestive or respiratory systems were uncommon prior to our report and highlight the wide range of potential target tissues in systemic infections by this viral pathogen, with or without concurrent infections.
Early detection of the possibly underdiagnosed or misdiagnosed PiHV infection in nestlings and adult carriers can help prevent decreased growth and mortalities, and associated economic losses, in the squab industry, or prevent low racing performance and spread of this infection among racing birds. Finally, our study highlighted the role of postmortem gross and histologic examinations as key laboratory tools for the diagnosis of PiHV infection even in the presence of various concurrent infections.
Footnotes
Acknowledgements
We thank Lucy Gomes for her help in the accession search, and all pathologists and technical staff of the CAHFS who performed the postmortem examinations and ancillary tests, respectively, on these cases.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding for our work was provided by the California Animal Health and Food Safety Laboratory System, University of California–Davis.
