Abstract
Perivascular wall tumors (PWTs) are common well-known canine mesenchymal tumors. The term PWT has not yet been applied to cats; only 2 cases of feline soft tissue hemangiopericytomas (HEPs) are available. In human medicine, sinonasal HEP-like tumor/glomangiopericytoma (SHPCL/GP) and intranasal solitary fibrous tumor (SFT) are well-known mesenchymal tumors with staghorn vasculature and low malignant potential; however, these entities have not been described in small animals. We describe here the pathologic and immunohistochemical features of 2 cases of feline intranasal mesenchymal tumors consistent with PWTs and resembling human SHPCL/GP (case 1), and human intranasal SFT (case 2). Both cats developed intranasal, unilateral, polypoid, expansile neoplasms with a mostly patternless growth of spindle cells, minimal stroma, and prominent staghorn vessels. The stroma was PAS negative, which excludes a glomus tumor. Immunohistochemistry identified diffuse vimentin and PDGFRβ expression. Case 1 was α-SMA positive (as is human SHPCL/GP); case 2 was negative (as is human intranasal SFT). Both tumors were incompletely excised, leading to recurrence in case 1. Case 2 was lost to follow up. To our knowledge, intranasal PWTs have not been reported previously in cats. The frequency of the lesions is not known, but awareness of these entities may assist in their recognition and better characterization in the future.
Keywords
Primary intranasal tumors comprise 1–8.4% of feline tumors6,13; ~57% are non-epithelial, with lymphomas representing 28.5% of all cases 14 ; 43% are carcinomas. 14 Primary feline intranasal mesenchymal tumors are considered rare, with fibrosarcoma being the most frequent and representing 8% of all cases. 14 However, primary intranasal mesenchymal perivascular wall tumors (PWTs) have not been described in cats, to our knowledge; the term PWT has not yet been applied to cats in general, and only 2 cases of putative soft tissue hemangiopericytoma (HEP) have been reported.3,4 In veterinary medicine, HEP was once a diagnostic term erroneously applied to canine soft tissue tumors with prominent perivascular whorling. However, most of these tumors did not have the microscopic growth characteristic of human HEPs, and were subsequently renamed correctly as PWTs.1,16 PWTs are neoplasms derived from vascular mural cells, with the exclusion of endothelium.1,16 HEP represents one of the rarest PWT subtypes, with histomorphologic features of sheets of uniform spindle cells, minimal-to-absent intervening stroma, and typical thin-walled, branching vessels (staghorn pattern).1,16
In people, sinonasal HEP-like tumor/glomangiopericytoma (SHPCL/GP)8,17 and solitary fibrous tumor (SFT)7,18 are well-known perivascular- (SHPCL/GP) and fibroblast- (SFT) derived entities included in the group of sinonasal mesenchymal tumors with borderline-to-low malignant potential.
We describe here 2 cases of primary feline intranasal mesenchymal tumors with prominent staghorn vasculature consistent with intranasal PWTs. Their histomorphology resembled reported cases of feline soft tissue HEP,3,4 and human cases of SHPCL/GP (case 1)8,17 and SFT (case 2).7,18
Case 1 was an 8-y-old spayed female domestic shorthair cat that had developed sneezing, unilateral left epistaxis, left eyelid chemosis, and ocular serous discharge, and was examined by a private oncology specialist. Clinical examination revealed unilateral left partial nasal obstruction caused by an intranasal polypoid mass. Involvement of nasal sinuses was not investigated. Differential diagnoses included foreign-body reaction, nasal polyp, and neoplasia. Initially, the owner refused nasal biopsy and invasive surgery. Doxycycline (50 mg, q24h PO, for 10 d) and prednisolone (5 mg, q24h PO, for 10 d, and then maintenance dose of 2.5 mg q24h PO) were administered. After initial improvement for 3 wk, clinical signs worsened progressively, and the cat was presented because of open-mouth breathing. With the owner’s consent, palliative surgical excision was performed.
Case 2 was a 19-y-old castrated male domestic shorthair cat with an intranasal polypoid mass and clinical signs of eyelid chemosis, ocular serous discharge, and intermittent epistaxis for >1 y, examined by a private oncology specialist from a second veterinary clinic. No information regarding therapy was retrieved. Endoscopic pinch biopsies were performed.
Excised tissues from both cats were fixed in 10% neutral-buffered formalin and processed routinely for histopathology (H&E), histochemistry (periodic acid–Schiff [PAS] stain), and immunohistochemistry (IHC). Tumor sections were immunolabeled for vimentin, α–smooth muscle actin (α-SMA), platelet-derived growth factor receptor β (PDGFRβ), factor VIII–related antigen (FVIIIRA), nerve growth factor receptor (NGFR), and S100. Case 1 was additionally immunolabeled for desmin, calponin, and β-catenin (Table 1). IHC detection was performed using an avidin–biotin complex (ABC), and the reaction was developed using 3,3′-diaminobenzidine (DAB).
Primary antibodies used for the identification of intranasal perivascular wall tumors in 2 cats.
α-SMA = α–smooth muscle actin; FVIIIRA = factor VIII–related antigen; NGFR = nerve growth factor receptor; PDGFRβ = platelet-derived growth factor receptor β.
Microscopically, under a multifocally eroded-to-ulcerated mucosa, a non-demarcated, non-encapsulated, expansive neoplasm was observed in both cats. The neoplasms replaced deep nasal tissues and were composed of patternless sheets and short bundles of uniform spindle cells with minimal-to-absent intervening stroma in association with numerous, variably sized, thin-walled, frequently ramified (branched), and often dilated blood vessels (staghorn pattern; Figs. 1–4). Intervening newly deposited stroma was mostly absent, but multifocal areas of edematous matrix were present, often in close association with staghorn vessels (Figs. 1–4). The neoplasm in case 1 was separated from the overlying mucosa by a band of uninvolved connective tissue of the preexisting nasal lamina propria (Grenz zone; Fig. 2). Neoplastic cells had indistinct borders, moderate amounts of eosinophilic cytoplasm, oval nuclei with finely granular chromatin, and indistinct nucleoli. Anisocytosis and anisokaryosis were mild (Fig. 3). Mitotic figures were absent in 10 contiguous high-power fields (ocular field number 22 mm, 40× objective corresponding to a standard area of 2.37 mm2). Lymphatic invasion was not noted in either neoplasm. Multifocal, moderate lymphocytic, and less commonly neutrophilic, inflammation was variably present throughout both neoplasms (Fig. 3) Additionally, case 1 contained multifocal, moderate, eosinophilic inflammation. Both tumors were also characterized by variably sized, multifocal, necrotic and/or hemorrhagic areas (<20% of both samples). The 2 lesions had been excised incompletely and extended to surgical margins. The sample from case 1 derived from a palliative, incomplete excisional surgery, and complete tangential section of the biopsy revealed that the tumor extended to cut borders, as did the tumor in the sample from case 2, which consisted of endoscopic pinch biopsies.
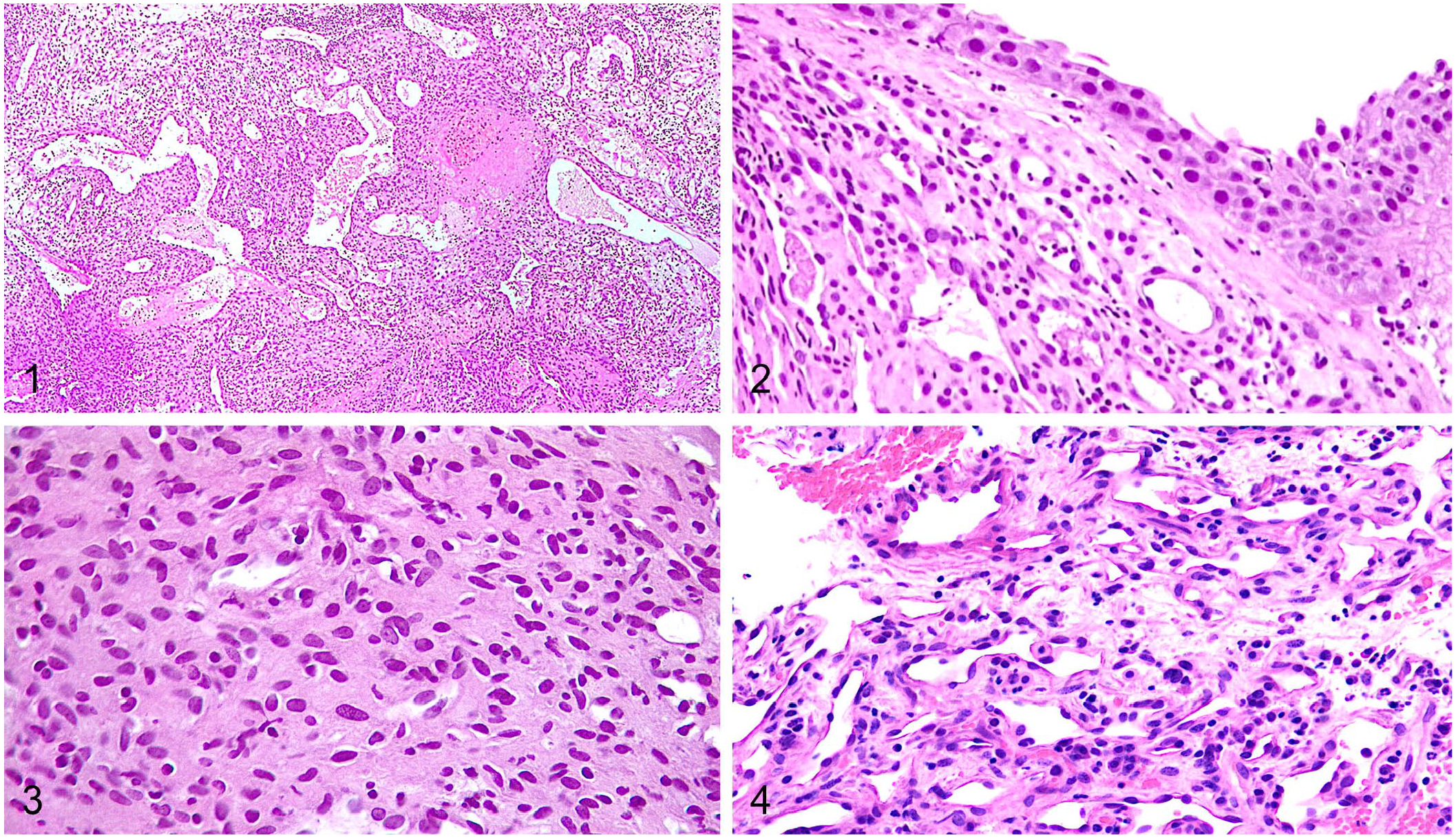
Histologic features of primary intranasal perivascular wall tumors (PWTs) in 2 cats. H&E.
The 2 cases were PAS negative, excluding the presence of newly deposited basement membrane material encircling tumor cells, and thus discounting a diagnosis of glomus tumor.5,19 In both tumors, 100% of neoplastic cells expressed intracytoplasmic vimentin and PDGFRβ intensely (Fig. 5) and were FVIIIRA, NGFR, and S100 negative. Approximately 90% of neoplastic cells of case 1 were positive for intracytoplasmic α-SMA (Fig. 6) and were diffusely negative for desmin, calponin, and β-catenin. Case 2 did not express α-SMA; thus, no additional myoid markers were investigated. Negativity for FVIIIRA, NGFR, and S100 ruled out endothelial (angiomas, angiosarcomas) and nerve sheath tumor derivations, respectively.
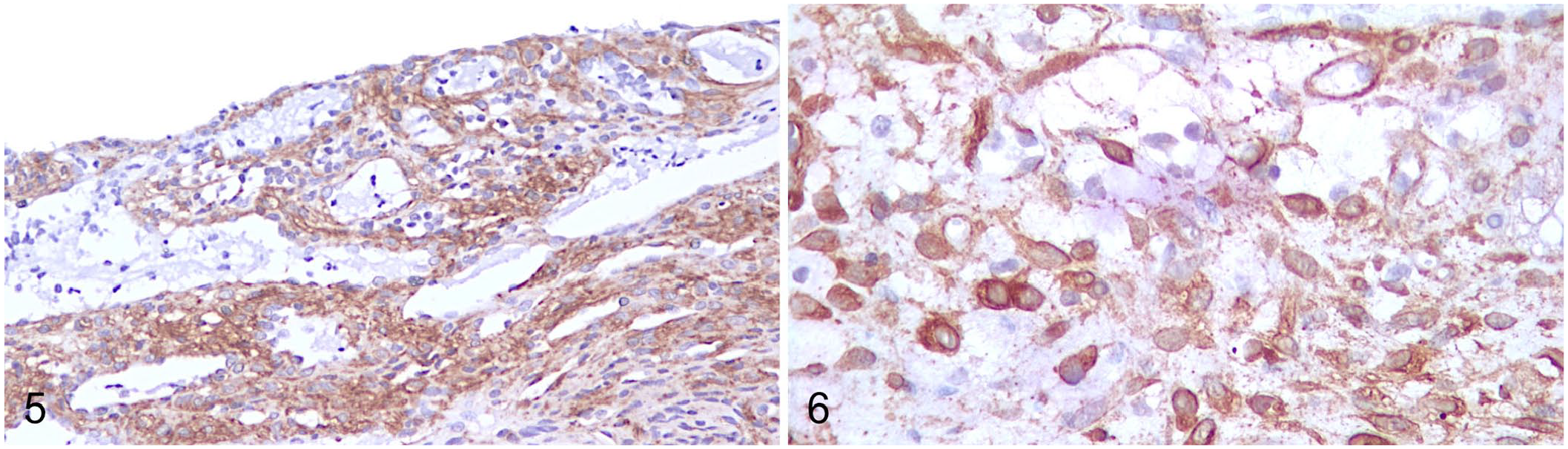
Immunohistochemical features of primary intranasal perivascular wall tumors (PWTs) in 2 cats.
Morphologically and phenotypically, the tumors were consistent with a diagnosis of PWTs. 16 Morphology was similar to that of the 2 cases of feline HEP.3,4 Additionally, case 1 resembled more closely human SHPCL/GP8,17; case 2 paralleled descriptions of human intranasal SFT.7,18
Four months after surgery and continuous corticosteroid therapy, the lesion of case 1 recurred and completely occluded the left nasal cavity with septal deviation and epistaxis, thus the cat was euthanized at the owner’s request. A postmortem examination was not performed. Case 2 was lost to follow up.
Both intranasal feline tumors described here had morphologic and phenotypic features consistent with the diagnosis of intranasal PWTs and resembled reported cases of feline soft tissue HEP3,4 (Fig. 1; Table 2). The 2 feline HEPs reported previously were diagnosed based on the staghorn vasculature and immunophenotype.3,4 Neoplastic cells in both reports expressed vimentin and focal S100, and were negative for cytokeratin, desmin, glial fibrillary acidic protein (GFAP), and neuron-specific enolase (NSE).3,4 Both intranasal feline PWTs described here paralleled those findings, except for their negativity to S100 (Table 2).
Histopathologic and immunohistochemical findings of intranasal perivascular wall tumors (PWTs) compared to feline subcutaneous hemangiopericytoma (HEP), canine subcutaneous HEP, human sinonasal HEP-like tumor/glomangiopericytoma (SHPCL/GP), and human intranasal solitary fibrous tumor (SFT).
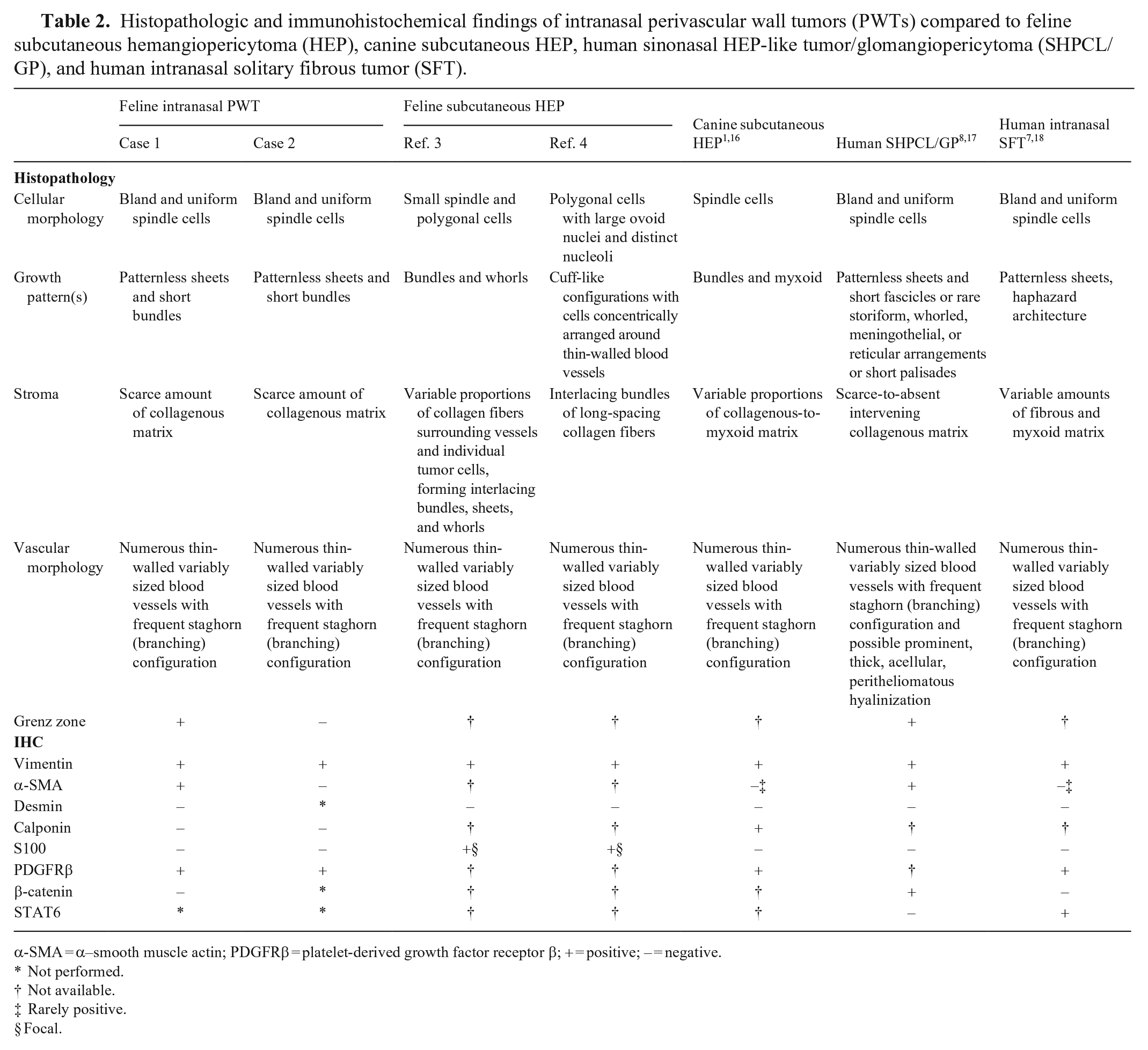
α-SMA = α–smooth muscle actin; PDGFRβ = platelet-derived growth factor receptor β; + = positive; – = negative.
Not performed.
Not available.
Rarely positive.
Focal.
In human and veterinary medicine, confusion regarding the term HEP is ongoing.1,15,16 In veterinary medicine, the term HEP was used to define all canine soft tissue tumors characterized by perivascular whorling; however, this feature is not a typical pattern of HEP and therefore, these tumors have been renamed as PWTs.1,16 Currently, PWTs are considered tumors in a morphologic and phenotypic continuum recapitulating that of vascular mural cells.1,16 Although regarded as a rare PWT subtype, HEP is still included among PWTs, and it is considered to have a pericytic origin in dogs.1,16 In humans, the term HEP has been applied historically to neoplasms characterized by monotonous cellularity and with staghorn vessels. 15 However, the morphologic features once considered typical of HEP can be variably found in other tumors, including myopericytoma and glomangiopericytoma. 9 Furthermore, the pericytic origin of soft tissue HEP in humans has been questioned and favors a fibroblastic derivation. 12 Given that HEP shares many histologic, immunophenotypic, and cytogenetic features with SFT, the consensus is that these 2 entities are part of the same spectrum of lesions. 12 Taking into account the aforementioned considerations in dogs and humans, to simplify the nomenclature, we chose to identify the reported tumors as “feline intranasal PWTs.” We hope that the identification of additional cases in the future will allow for a better characterization of this neoplasms in cats.
In humans, 2 intranasal mesenchymal neoplasms are associated with prominent staghorn vessels: SHPCL/GP8,17 and sinonasal SFT.7,18 The 2 feline intranasal tumors described here had clinical presentations and gross aspects paralleling descriptions of those 2 human entities. Indeed, human SHPCL/GP and sinonasal SFT are nearly always unilateral, expansive, and polypoid, and can extend into paranasal sinuses (this last feature was not investigated in our cases).7,8,17,18 Tumor growth commonly causes nasal obstruction and epistaxis.7,8,17,18 Microscopic features in our cases also paralleled those described in humans, including moderate cellularity, expansive nodular growth, spindle-to-oval neoplastic cells with no specific pattern associated with numerous staghorn vessels, and the presence of a Grenz zone in case 17,8,17,18 (Figs. 1–4; Table 2). SHPCL/GP is characterized by scarce-to-absent intervening stroma8,17; SFT usually has variable amounts of fibrous and myxoid stroma.7,18 Additionally, eosinophilic inflammation, as present in case 1, is also reported frequently in human SHPCL/GP.8,17
Neoplastic cells of SHPCL/GP display a myoid phenotype, with diffuse reactivity for α-SMA but lacking significant expression of desmin.8,17 Additionally, given CTNNB1 missense mutations, neoplastic cells express aberrant nuclear β-catenin. 10 The SFT cells have an immunohistochemical-specific reaction with STAT6 (nuclear) and CD34, but do not express actins, S100, desmin, or β-catenin.7,18 In our cats, only case 1 expressed α-SMA, and was contemporarily negative for desmin and calponin, most closely paralleling the myoid phenotype reported in human SHPCL/GP.8,17 Case 1 was β-catenin negative suggesting that no mutation was related to tumor development in this cat. 10 Case 2 was α-SMA negative, thus additional myoid markers such as desmin were not assessed; this phenotype is similar to that of human SFT7,18 (Table 2). No additional conclusions could be drawn, given that α-SMA expression has not been investigated in feline soft tissue HEP,3,4 although its expression is reported as variable in canine PWTs. 16
In both of our intranasal feline tumor cases, diffuse PDGFRβ expression was observed (Fig. 5). PDGFRβ is a tyrosine-protein kinase that plays an essential role in the regulation of blood vessel development and is considered a marker of perivascular origin. 11 PDGFRβ is expressed by neoplastic cells in canine PWTs (including HEP) 2 and also in human intranasal SFT. 12
Differential diagnoses for case 1 included other tumors with myoid phenotype, such as angioleiomyoma, myopericytoma, and glomus tumor. However, cell morphology, growth patterns, and desmin negativity excluded angioleiomyoma and myopericytoma. Glomus tumors are rare in cats, with only 2 cases described: 1 in the head 5 and 1 in a digit. 19 Feline glomus tumors occur as nodular, discrete, expansive neoplasms composed of sheets and bundles of round-to-polygonal-to-spindle cells surrounding small and medium blood vessels.5,19 In the digital glomus tumor, individual neoplastic cells were encircled by PAS-positive basal lamina, a feature considered diagnostic for glomus tumors in all animal species. 19 Also, neoplastic glomus cells expressed vimentin and α-SMA, and were S100, desmin, and von Willebrand factor negative.5,19 Despite some morphologic similarities, PAS negativity and staghorn vessels excluded the diagnosis of glomus tumor in our 2 cases.
Most human SHPCL/GP and intranasal SFT are considered indolent tumors with an excellent prognosis following complete surgical excision and with possible recurrence after incomplete excision.7,8,17,18 In cat 1, surgical excision was incomplete and the tumor recurred, deviating the nasal septum, occluding the left nasal cavity, and thus leading to euthanasia after 4 mo. Because neoplastic cells in our case expressed diffuse PDGFRβ, tyrosine kinase inhibitors should be investigated as a possible therapy for these tumor types, with the aim to reduce the relapse rate and prolong the time to relapse after incomplete surgical excision of this tumor type in cats.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to research, authorship, and/or publication of this article.
Funding
The authors received no outside financial support for the research, authorship, and/or publication of this article.
