Abstract
A 14-y-old, castrated male, diabetic, domestic longhaired cat was presented for investigation of anemia. General examination revealed widespread cutaneous erythematous macules and patches. Hematology and bone marrow aspiration revealed severe regenerative anemia and marked erythroid hyperplasia, respectively. Low numbers of intermediate-to-large, atypical lymphocytes were observed in the blood smear and bone marrow aspirates. Various imaging modalities demonstrated a diffuse pulmonary bronchial pattern, multifocal mural thickening of the urinary bladder, splenomegaly, and mild tri-cavitary effusion. Skin biopsies and cytologic examination of the pleural effusion demonstrated round-cell neoplasia consistent with lymphoma. Autopsy confirmed disseminated T-cell lymphoma, mostly affecting the urinary bladder, stomach, lymph nodes, and interscapular subcutis and muscles. Angiocentrism and nerve infiltration were present. The cutaneous erythematous patches, characterized by perivascular neoplastic lymphocytic infiltrates and angiodestruction, were a manifestation of the disseminated lymphoma in this cat, similar to the lesions reported in humans affected by angioimmunoblastic T-cell lymphoma.
A 14-y-old, castrated male, domestic longhaired cat weighing 5.5 kg was referred to the Animal Hospital at Murdoch University (Murdoch, WA, Australia) for investigation of anemia. The cat had a 5-d history of lethargy, hyporexia, transient stranguria and hematuria, and a single episode of vomition. Blood tests performed by the referring veterinarian showed mild regenerative anemia and a markedly prolonged activated partial thromboplastin time (APTT; Suppl. Table 1). One dose of an antiemetic (maropitant citrate 1 mg/kg SC, Cerenia; Zoetis) was given prior to referral. The cat had a history of recurrent lower urinary tract disease, and diabetes mellitus managed with twice daily SC injections of glargine insulin for 3 y. Feline leukemia virus (FeLV) vaccination had been administered 13 mo prior.
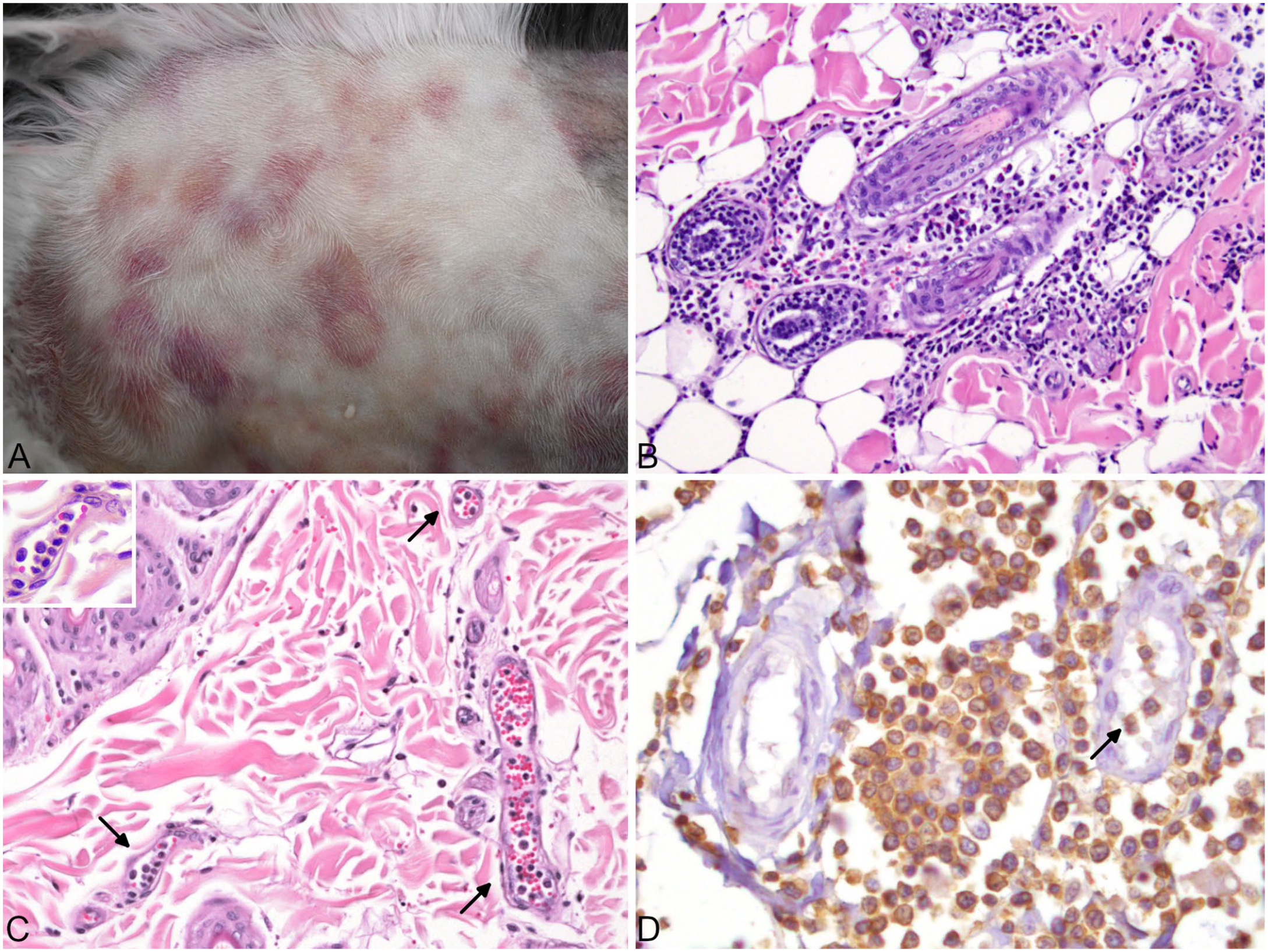
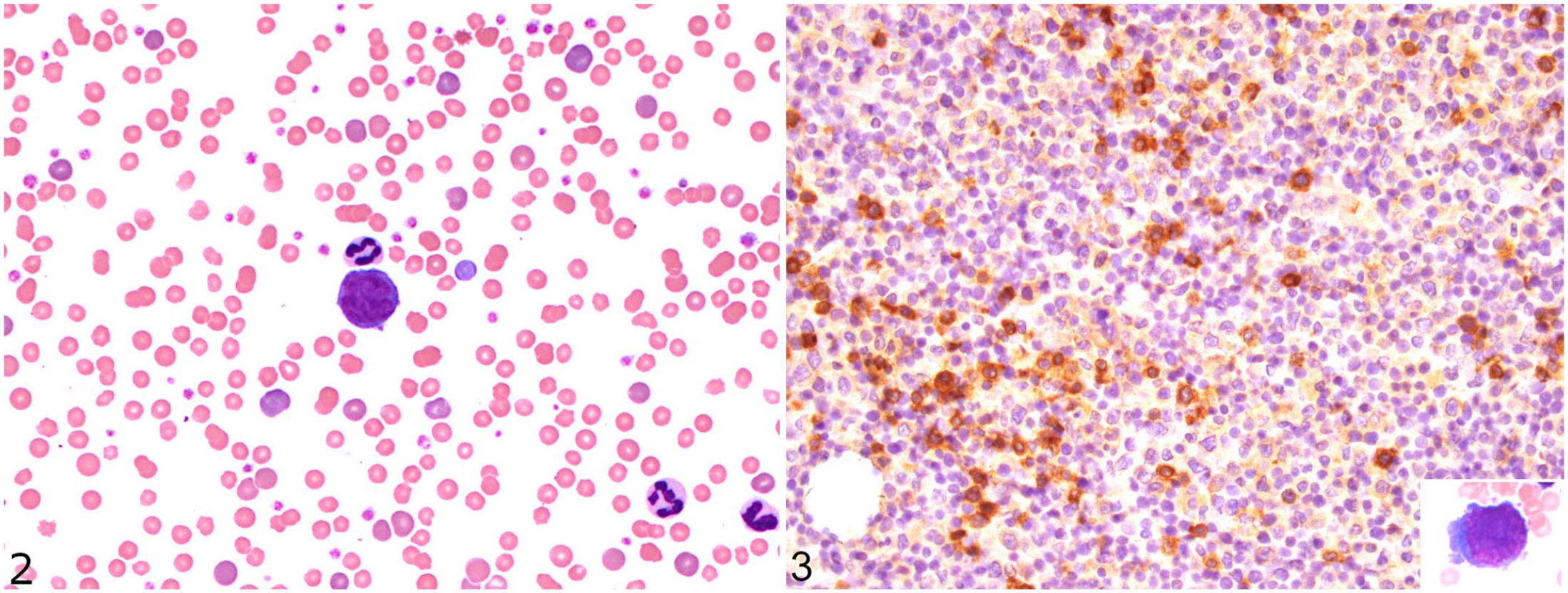
The physical examination revealed bilateral prescapular lymphadenomegaly; generalized cutaneous erythematous macules and patches were evident after clipping the fur (Fig. 1A). Some patches had an annular appearance. Cutaneous or subcutaneous nodules were not detected. The initial diagnostic evaluation included a CBC, molecular testing for hemotropic mycoplasmas, rotational thromboelastometry (ROTEM), serum biochemistry profile, urinalysis, and diagnostic imaging. Hematology (Suppl. Table 1) confirmed the finding of the referring veterinarian of a highly regenerative anemia. The leukon was characterized by mild leukocytosis as a result of mature neutrophilia and monocytosis, with mild lymphopenia and mild basophilia. Blood smear evaluation revealed low numbers of intermediate-to-large, atypical, and rarely blastic, lymphoid cells (Fig. 2). Hemotropic parasites were not seen, and PCR testing was negative for
Cutaneous metastases of disseminated T-cell lymphoma in a 14-y-old cat.
T-cell lymphoma in a 14-y-old cat.
Initial treatment of the cat included intravenous fluid therapy with compound sodium lactate (Baxter) at 2.2 mL/kg/h. On day 2 of hospitalization, the PCV had decreased to 0.16 L/L (RI: 0.30–0.45 L/L). The cat was blood typed as A (LabTest BT A+B; Alvedia) and transfused a major cross-match–compatible unit of type A feline packed RBCs without complication. The post-transfusion PCV was 0.19 L/L. Given the high degree of suspicion for lymphoma or leukemia in light of circulating atypical lymphocytes, bone marrow aspirates from the proximal humerus and skin biopsies were obtained under general anesthesia. Bone marrow cytology revealed a decreased myeloid:erythroid ratio (0.19) as a result of erythroid hyperplasia, and occasional atypical intermediate-to-large lymphocytes (Fig. 3; Suppl. Table 3). The low percentage of atypical lymphocytes rendered leukemia unlikely. Given that the cat did not show evidence of hemorrhage, immune-mediated hemolytic anemia (IMHA) was deemed the most likely cause of the severe, regenerative anemia, and immunosuppressive treatment was commenced with cyclosporine 4.5 mg/kg PO q12h (Neoral; Novartis).
Despite treatment, the clinical condition of the cat deteriorated with worsening of the pleural effusion and development of pericardial effusion. Ultrasound-guided thoracocentesis was performed. The cytospin of the pleural fluid was highly cellular (54.5 × 109/L) with a protein concentration of 24 g/L and specific gravity of 1.020. A differential cell count revealed 96% lymphocytes, 3% macrophages, and 1% neutrophils. The lymphocytic population was heterogeneous, with frequent small intracytoplasmic purple granules, increased atypical mitotic figures, and bi- and multinucleation highly suggestive of lymphoma (Suppl. Fig. 2). Histologic examination of biopsies from the cutaneous erythematous patches revealed perivascular and periadnexal infiltrates of round cells, with frequent intravascular tumor cells (Fig. 1B, 1C). Epitheliotropism was absent. A presumptive diagnosis of disseminated lymphoma was made, and, given the guarded prognosis, the cat was euthanized.
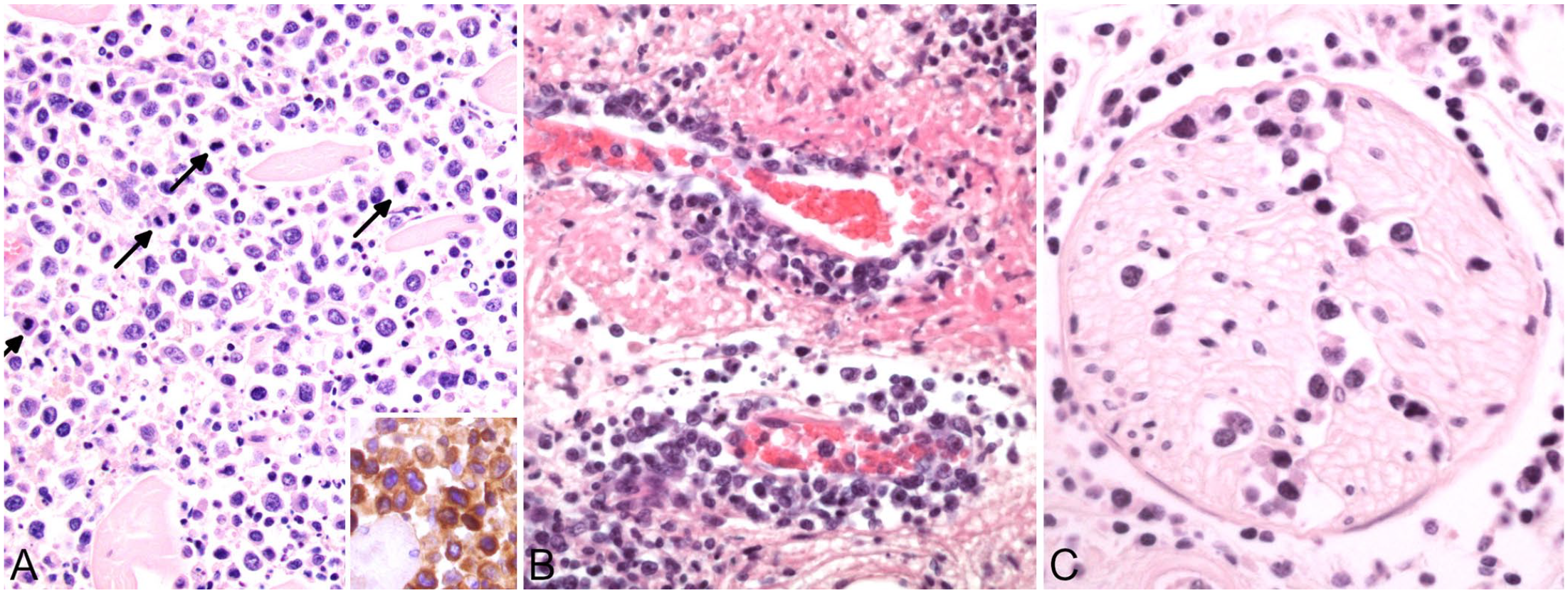
The predominant findings at autopsy were widespread cutaneous erythematous macules and patches, red plaque-like lesions (up to 3-mm thick) affecting ~75% of the bladder mucosa, splenomegaly, generalized mild-to-moderate lymphadenomegaly, left atrial and hepatic pallor, ulcerative gastric lesions, and white-to-red poorly demarcated foci in the muscles of the interscapular region and the left thigh. Other notable gross findings included suffusions within the subcutis and mesentery, and the presence of small volumes of orange-stained fluid within the thoracic and pericardial cavities. Histologic evaluation revealed large numbers of neoplastic round cells infiltrating the urinary bladder, subcutis and striated muscles (especially in the interscapular region; Fig. 4A), heart, and stomach. Also affected were lymph nodes, visceral adipose tissue, lung (with peribronchial-vascular and, to a lesser extent, subpleural patterns; Suppl. Figs. 3, 4), pancreas, skin, spleen, liver, kidney, duodenum (intravascular tumor cells only), uvea, and bone marrow (Fig. 3). Brain and adrenal glands were not infiltrated by neoplastic cells. Histologic evaluation of the pancreas also showed exocrine nodular hyperplasia, which is a common incidental finding in aged cats, and lesions in the islets of Langerhans characterized by amyloidosis (confirmed by Congo red staining) and loss of cells consistent with diabetes mellitus.
Interscapular region in a 14-y-old cat with disseminated T-cell lymphoma.
The neoplastic round cells were small-to-large lymphocytes with marked atypia, including anisocytosis, anisokaryosis, irregular nuclei, bi- and trinucleation, and atypical mitotic figures. The number of mitoses per 0.237 mm2 (hpf) was highly variable depending on the tissue: 0–3 in the skin, 9 in the stomach, and up to 20 in the urinary bladder and the interscapular muscles. Angiocentrism (Suppl. Fig. 4), angiodestruction (Fig. 4B), and nerve infiltration (Fig. 4C) were present in several tissues, especially in the subcutaneous and visceral adipose tissue and striated muscles. Intravascular tumor cells were widespread. Immunohistochemical staining was performed manually (Suppl. Table 4). Neoplastic cells were strongly positive for the T-cell marker CD3 and negative for the B-cell marker CD20, supporting the diagnosis of T-cell lymphoma (Fig. 1D; Fig. 4A inset).
Lymphoma is one of the most common tumors in cats, and can develop in any tissue or organ. To our knowledge, disseminated T-cell lymphoma has not been reported previously in a cat with a combination of cutaneous erythematous patches and presumptive IMHA. In cats, regenerative anemia can be caused by infectious diseases, exposure to drugs or toxins, genetic disorders, or immune reactions against RBCs. 10 Although IMHA is an infrequent cause of feline anemia, 11 we suspected secondary IMHA associated with lymphoma in our patient. This association has been described in 2 young sibling cats with disseminated T-cell lymphoma, 7 and was mentioned in a few cases in 2 retrospective studies.10,15 Other putative triggers of secondary IMHA, such as feline hemotropic mycoplasmas or toxic-metabolic causes, were excluded. 1 Although we did not test for FeLV, the cat had received an FeLV vaccine 13 mo before. Given the rapid clinical deterioration of the cat and the presumptive diagnosis of lymphoma, neither Coombs nor saline agglutination tests were performed; despite low sensitivity, these tests may have been useful in assessing an immune-mediated pathogenesis.6,10 Flow cytometry would have been another useful test to consider. In humans, IMHA is a recognized complication of lymphoma, and may involve the emergence of forbidden clones, formation of neoantigens, and immune dysregulation. 2 The prolonged APTT and INTEM clotting time indicated an abnormality in the intrinsic coagulation cascade, or may have been spurious. Factor XII deficiency was considered most likely given that it is reported in cats, is associated with abnormalities in thromboelastography similar to those observed in our patient, and is not associated with spontaneous bleeding. 3 Nonetheless, a factor XII assay was not performed and thus factor XII deficiency could not be confirmed. Factor VIII or IX deficiency were considered unlikely given that these conditions are associated with spontaneous bleeding, which was not noted in our cat’s history. Other causes of coagulopathy, including disseminated intravascular coagulation, were considered less likely given that all other ROTEM parameters were within RIs.
Widespread erythematous non-scaling cutaneous patches without reported pruritus was a prominent abnormality in our case. The main clinical differential diagnosis was cutaneous neoplastic infiltration, especially a cutaneous epitheliotropic T-cell lymphoma, 5 and vasculitis. Skin biopsies revealed perivascular infiltration by round cells, with frequent intravascular tumor cells. Immunohistochemical evaluation confirmed the lack of epitheliotropism, and demonstrated strong expression for CD3. Based on the histologic pattern of the neoplastic cells in the dermis compared to those in other tissues, such as the interscapular musculature, the cutaneous erythematous patches were considered a manifestation of disseminated T-cell lymphoma.
A possible association of feline lymphoma with chronic inflammation has been discussed in the literature. Cutaneous non-epitheliotropic T-cell lymphomas have been reported at the site of a previous traumatic fracture corrected with a metal implant, 9 and at injection sites. 14 Cutaneous lymphoma arising at the site of insulin injection has been described in humans. 16 Injection site-associated disseminated subcutaneous lymphoma may have been present in our case. The cat received twice-daily SC injections of glargine insulin in the shoulder area for 3 y, and the interscapular subcutis and muscles were severely infiltrated by neoplastic cells. A massive neoplastic infiltrate was also present in the urinary bladder, another site of chronic inflammation based on our patient’s history of recurrent lower urinary tract disease. Given the widespread dissemination of the lymphoma, it is impossible to say with certainty which tissue was first affected and if neoplastic infiltrates originated in a single or multiple sites. Given the immunohistochemical results (i.e., strong positivity of the neoplastic cells for CD3 and total lack of immunoreactivity for CD20 in all evaluated tissues) and financial constraints, PCR for antigen receptor rearrangement (PARR) analysis was not performed. PARR testing would have been interesting to assess if clonal rearrangement was similar in the most severely affected tissues and the skin. Furthermore, expression of FeLV p27 and gp70 antigens by immunohistochemistry and detection of the provirus by PCR were not studied in this annually vaccinated cat. 17 Given that FeLV vaccination does not always protect from infection, 8 we cannot exclude a contribution of FeLV to the development of lymphoma.
Interestingly, our cat shared several clinical features of angioimmunoblastic T-cell lymphoma, a rare and aggressive subtype of peripheral T-cell lymphoma in humans.4,13 Clinicopathologic similarities include the presence of: cutaneous maculopatches, extranodal disease (including the spleen and bone marrow), and anemia with a positive Coombs test in approximately one-third of human cases. 13 Skin biopsies of human patients reveal perivascular lymphocytic infiltrates and/or angiodestruction; both findings were visible in our case. A definitive diagnosis requires immunohistochemical study with a panel of markers of T follicular helper cells, such as C-X-C motif chemokine ligand 13, programmed cell death protein 1, inducible T-cell co-stimulator, CD10, and B-cell lymphoma 6 protein. 12 These markers are available routinely only in humans to date. In our cat, flow cytometric immunophenotyping could have been useful to go further in the classification of the disseminated T-cell lymphoma.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387221133543 – Supplemental material for Disseminated T-cell lymphoma with non-epitheliotropic cutaneous involvement in a cat with erythematous patches and regenerative anemia
Supplemental material, sj-pdf-1-vdi-10.1177_10406387221133543 for Disseminated T-cell lymphoma with non-epitheliotropic cutaneous involvement in a cat with erythematous patches and regenerative anemia by Cynthia Robveille, Mark W. Kim, Jason Stayt, Claire R. Sharp and Kathrin F. A. Langner in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Brendan Groves for the histologic processing of the samples, and the owners for permission to perform the autopsy.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
