Abstract
Bcl-2, an anti-apoptotic protein, is commonly overexpressed in follicular lymphomas in humans. This is usually the result of a chromosomal translocation that transposes the Bcl-2 gene into the immunoglobulin gene locus. The immunohistochemical assessment of this overexpression can be used as a tool for the differentiation of follicular lymphoma and follicular hyperplasia. In cats, little information about the expression of Bcl-2 in follicular lymphoma exists. We investigated 18 follicular lymphomas histologically and immunohistochemically for the expression of Bcl-2, CD3, CD45R, and feline leukemia virus. Clonality was assessed by PCR for antigen receptor gene rearrangements. Although the histology resembled that of their human counterparts, diffuse expression of Bcl-2 within the follicles of the feline lymphomas, as seen in human cases, was not present. Only single cells within the follicles, comparable to the reactive controls, were positive for Bcl-2 expression. The mean survival time of 4.6 y confirmed the indolent character of the tumor. None of the clinical parameters assessed were statistically significant predictors of survival. Furthermore, a statistically significant difference in survival of animals with or without anti-neoplastic therapy was also not demonstrable.
Keywords
Introduction
In humans, follicular lymphoma is the second most common form of non-Hodgkin lymphoma.4,8,12,33 Follicular lymphoma affects adults typically, with a median age in the sixth decade of life. 33 The majority (~85–90%) of these tumors express the Bcl-2 (“B-cell lymphoma/leukemia 2”) 35 protein. 18 In many cases, this is associated with the chromosomal translocation t(14;18)(q32,q21)10,43 that juxtaposes the locus of the immunoglobulin heavy chain (IGH) on chromosome 14 and the Bcl2 locus on chromosome 18 30 resulting in an overexpression of Bcl–2. This overexpression promotes the survival of cells in follicular lymphomas. 24 Localized to the outer membrane of mitochondria, Bcl-2 inhibits apoptosis by dimerization with pro-apoptotic proteins such as BAX (“Bcl-2 associated X, apoptosis regulator”). 28 This inhibits the release of other pro-apoptotic proteins such as cytochrome C,17,42 and thereby interrupts the apoptotic cascade. Physiologically, Bcl-2 expression in lymphoid tissues is restricted to nonproliferative mature lymphocytes in T- and B-cell areas of spleen, lymph nodes, and tonsils. Surviving T-cells in the thymic medulla and small mature lymphocytes in primary follicles of lymphatic tissues also show Bcl-2 expression. 5 In contrast, proliferative zones of spleen, lymph nodes, and tonsils lack the expression of Bcl-2, except for single nonproliferating positive cells.13,23,29 A few non-lymphoid tissues with high rates of apoptosis also express Bcl-2, such as glandular epithelium regulated by hormones or growth factors, complex differentiated epithelia with long-lived stem cells (e.g., skin, intestine), and long-lived post-mitotic cells such as neurons. 13
In cats, a species with a high incidence of hematopoietic malignancies, follicular lymphomas are rare. 38 Six follicular lymphomas were reported in 602 lymphomas of cats (0.9%). 37 Such tumors are usually regarded as indolent, but information about disease progression in cats is sparse. Furthermore, little information is available about the role of Bcl-2 expression in feline lymphomas,7,14,22 with no explicit focus on follicular lymphomas. Our aim in this retrospective study was to test the hypothesis that expression of Bcl-2 plays a significant role in feline follicular lymphomas similar to its role in human follicular lymphomas, and that immunohistochemistry of Bcl-2 expression may aid in the diagnosis of follicular lymphomas. Finally, we aimed to provide descriptive data of a collection of these rare tumors.
Materials and methods
Patients, samples, and tumor grading
Eighteen feline follicular lymphomas were examined; 4 samples from cats with follicular hyperplasias served as non-neoplastic controls (Table 1). Formalin-fixed, paraffin-embedded (FFPE) tissue samples were obtained from the archives of the diagnostic laboratory of the Institut fuer Veterinaer-Pathologie of the Justus-Liebig-University Giessen (Giessen, Germany), the Institut fuer Veterinaer-Pathologie of the University Leipzig (Leipzig, Germany), and the Fachpraxis fuer Tierpathologie (Munich, Germany).
Characteristics of cats and lymph node biopsy location.
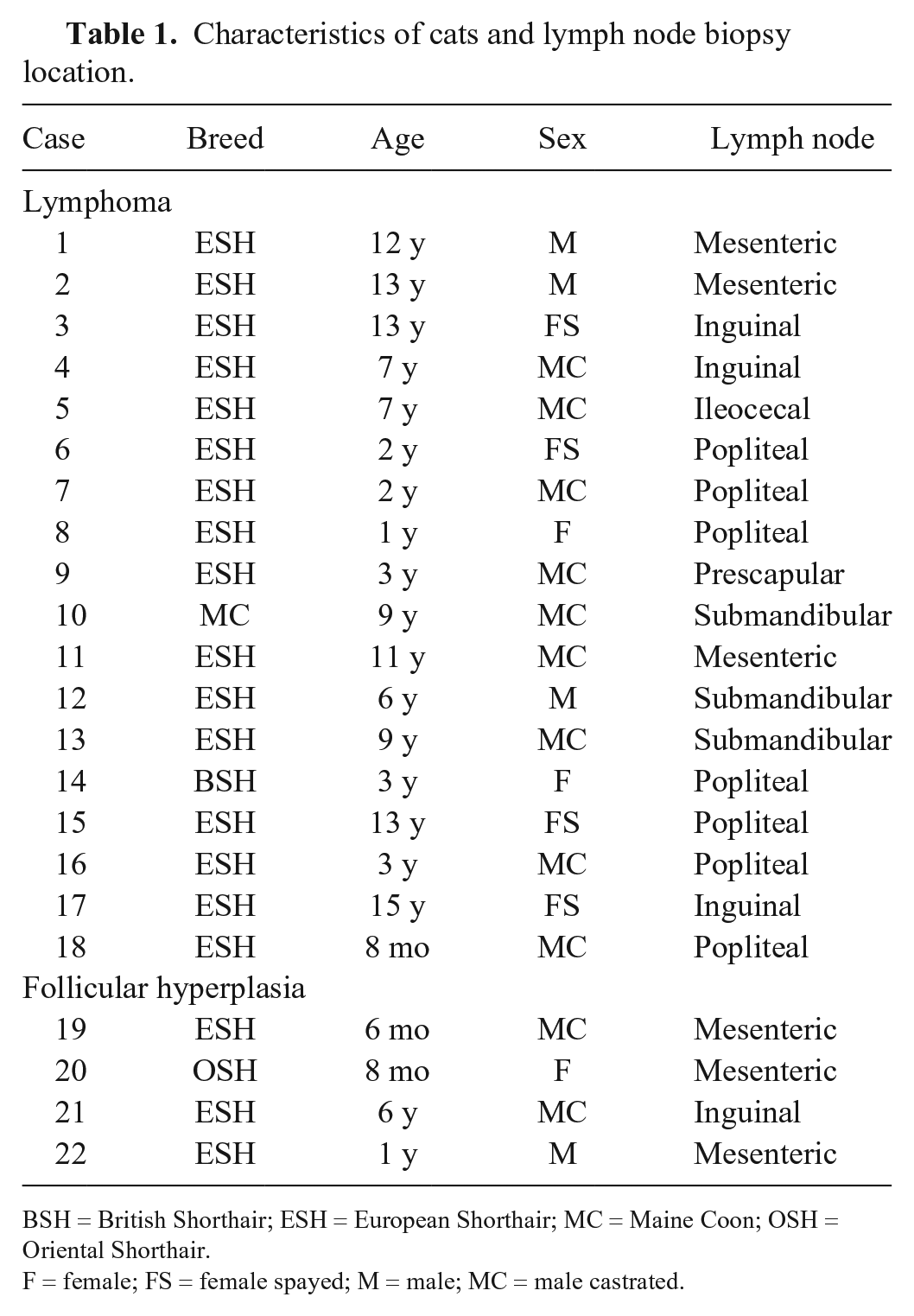
BSH = British Shorthair; ESH = European Shorthair; MC = Maine Coon; OSH = Oriental Shorthair.
F = female; FS = female spayed; M = male; MC = male castrated.
Four samples (1 follicular lymphoma grade 1, 1 follicular lymphoma grade 2, and 2 follicular hyperplasias) of human patients served as controls in all methods. Unstained slides of these samples were kindly provided by the NCT (National Center for Tumor Diseases, Heidelberg, Germany) tissue bank.
Follicular lymphomas were diagnosed according to the WHO classification schemes33,38 in H&E-stained slides. Given that follicular lymphoma is difficult to diagnose (differentiation from follicular hyperplasias), only cases diagnosed concordantly by at least 2 pathologists were included. Ambiguous cases were excluded.
According to the WHO schemes,33,38 we graded lymphomas by determining the proportion of large cells (centroblasts) and small cleaved cells (centrocytes). Follicles in 10 high-power fields were evaluated (randomly selected, without focus on those with the most numerous large cells) 33 : grade I = 0–5 centroblasts; grade II = 6–15 centroblasts; and grade III = >15 centroblasts.
We assigned the lymphomas to one of the following growth patterns: “follicular” (>75% follicular areas), “follicular and diffuse” (25–75% follicular areas), and “focally follicular” (<25% follicular areas, but still visible). 33 We did not apply the “diffuse” category (0% follicular) because diagnosis of diffuse large B-cell lymphomas in human samples is only possible by determination of the immunophenotype of germinal center cells and/or presence of the t(14;18) translocation, and therefore not applicable for cats.
Immunohistochemistry
Four-μm paraffin sections were deparaffinized in xylene and rehydrated through graded alcohols. Endogenous peroxidases were inhibited by incubation (30 min) in methanol–hydrogen peroxide. Slides were labeled with antibodies against Bcl-2, CD45R (B-cell marker),9,16,26,27 CD3 (T-cell marker), and feline leukemia virus (FeLV; Table 2). Chromogenic methods were either ABC (Avidin-biotin complex; Linaris Biologische Produkte, Dossenheim, Germany) for Bcl-2 and CD45R, or PAP (Peroxidase-antiperoxidase; Dako/Agilent Technologies, Santa Clara, CA) for CD3, as well as horseradish peroxidase–conjugated rabbit anti-mouse IgGs (Dako/Agilent Technologies) for FeLV. All feline tissues were stained with each antibody described above. The human control tissues were stained with anti–Bcl-2 antibodies only. For each antibody, a positive control was included (Bcl-2: feline tonsil; CD45R/CD3: feline lymph node; FeLV: lymph node of a FeLV-infected cat). For negative controls, primary antibodies were replaced by non-reacting antibodies.
Primary antibodies used for immunohistochemistry of feline lymph nodes with either follicular lymphoma or follicular hyperplasia.

TRS = Target retrieval solution (Agilent Technologies, Santa Clara, CA); TUF = Target unmasking fluid (Thermo Fisher Scientific, Dreieich, Germany). Suppliers: Leica Biosystems Newcastle, Newcastle Upon Tyne, UK; Abcam, Cambridge, UK; LINARIS Biologische Produkte, Dossenheim, Germany; Agilent Technologies, Santa Clara, CA; Custom Monoclonals International, Sacramento, CA.
Clinical data
If contact was possible, the referring veterinarian and/or owner of the patient were asked to provide information about the patient’s state (dead, alive), cause of death, time of death, lymph node enlargement at time of biopsy (solitary, multiple/regional, generalized), lymph node enlargement after biopsy, and therapy.
The Kaplan–Meier estimator was used to estimate survival functions with regard to tumor grade, lymph node enlargement at time of biopsy, and therapy (therapy with possible anti-neoplastic effect or no such therapy). Cats still living at the end of the observation period and cats that died of reasons obviously not related to lymphoma (e.g., trauma) were censored. Cats with no or only partial clinical data available were not included in the respective calculations. Log-rank (Mantel–Cox) test and, if applicable, Gehan–Breslow–Wilcoxon test were used to test for significant differences between survival curves. The significance threshold was set at 0.05. Statistical analyses were performed with Prism v.5.0 (GraphPad Software, San Diego, CA).
PCR for antigen receptor rearrangement
DNA was extracted (QIAamp DNA FFPE tissue kit; Qiagen, Hilden, Germany) as recommended by the manufacturer. DNA concentration was measured photometrically at room temperature (NanoDrop 2000; Thermo Scientific, Waltham, MA) after homogenization of the samples at 63°C. All samples were stored at −20°C. DNA was amplified using a previously published primer system. 41 Each reaction was run in duplicate and, in each run, a non-template control as well as a clonal and a polyclonal control were included.
Capillary electrophoretic analysis of the PCR for antigen receptor rearrangement (PARR) amplicons was performed (ABI Prism 310 genetic analyzer; Life Technologies, Darmstadt, Germany). PCR products were used either pure or diluted (1:100 in water). The pure or diluted product was denatured (Hi-Di formamide; Life Technologies) mixed with size standard (GeneScan 500 LIZ dye size standard; Life Technologies) at 95°C for 5 min, followed by 10-min incubation on ice. Injection was performed at 15,000 V for 5 s. Temperature during electrophoresis was 60°C. The sample was considered clonal if 1 or 2 discernible peaks were visible and reproducible in the replicates. Pseudoclonality was considered when peaks were not reproducible, and a polyclonal pattern was defined as multiple peaks of PCR products spread over the expected size range, ideally in a Gaussian curve-like distribution.
Results
The median age of the affected cats was 7 y (range: 8 mo to 15 y). Most cats were European Shorthair (16 of 18), with 2 other breeds represented (1 Maine Coon, 1 British Shorthair). Most affected animals were male (12 of 18); 9 were castrated, 3 were intact. Of the 6 female cats, 4 were spayed, 2 were intact.
All cases had almost complete effacement of the lymph node architecture by the expansions of neoplastic lymphocytes (Fig. 1). Neoplastic follicles were relatively evenly distributed within cortex and medulla. Some cases (8 of 18) had prominent interfollicular areas such that the follicles appeared less densely packed. In 11 of 18 tumors, follicles were quite uniform in size and shape. However, 7 of 18 tumors had more variably sized and shaped follicles. In all tumors, only a faded-to-absent mantle zone was visible at the periphery of neoplastic follicles. Diminished or lost polarity was evident in all cases, but in some follicles (particularly in cortical locations), polarity was still detectable. Tingible body macrophages were present in all follicles in variable numbers associated with and proportional to variable numbers of apoptotic cells. Mitoses were also present in all follicles, mostly randomly distributed within the follicles. However, in follicles with retained polarity, mitoses were mainly visible in the darker zones. In 3 cases, interfollicular fibrosis was present.
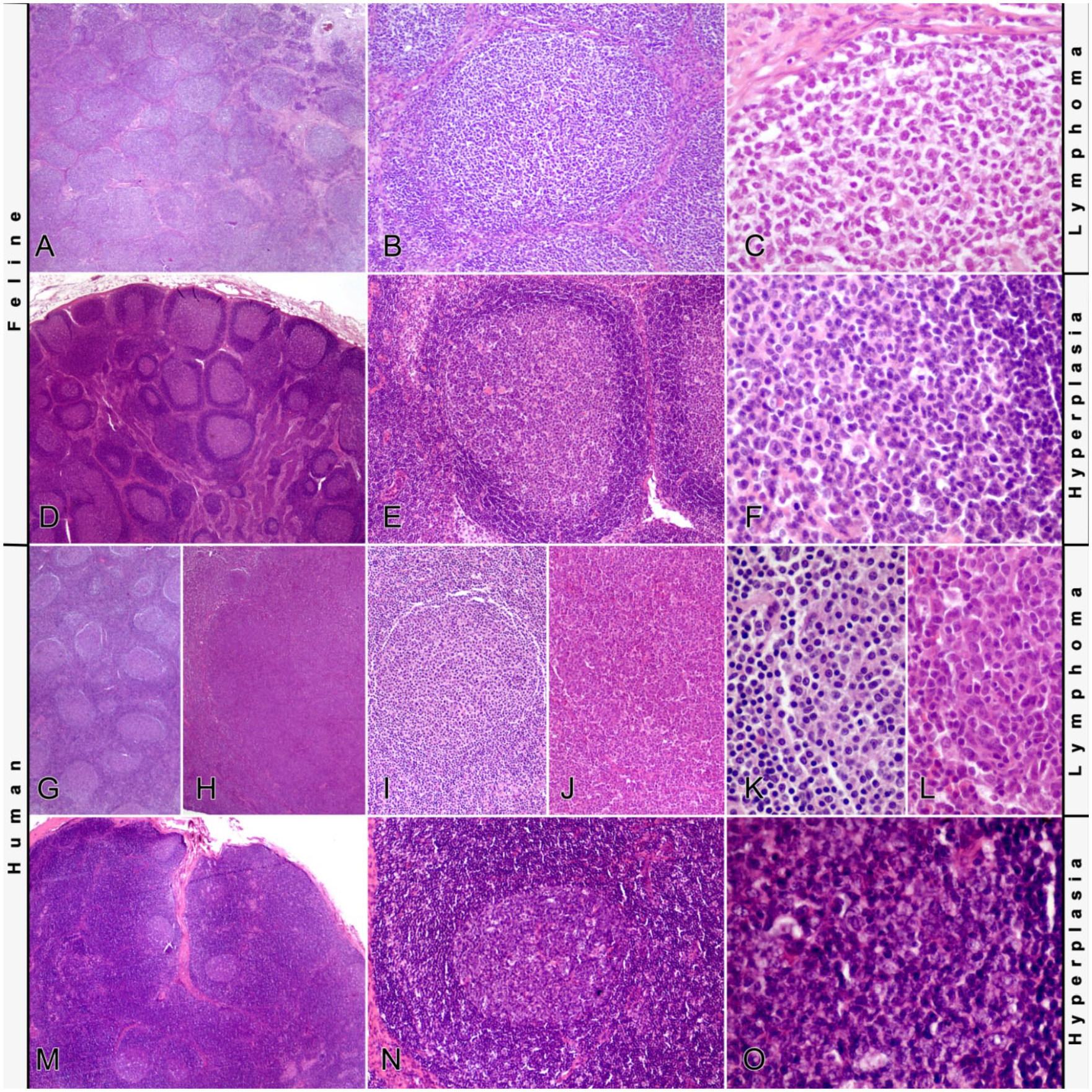
Histology of follicular lymphomas and follicular hyperplasia.
Two lymphomas were classified as grade I, 8 lymphomas as grade II, and 7 of the samples as grade III. One of the samples was too small to investigate 10 follicles; because only 8 follicles could be investigated and the average number of centroblasts per follicle was 5.3, which is at the threshold between grade I and II, this tumor was graded as grade I-II (as proposed by the WHO scheme 33 in humans). Seventeen tumors had a pure follicular pattern. Only case 2 had a follicular and diffuse pattern, with 70% follicular area.
All follicles were positive for CD45R and negative for CD3. All samples were negative for the FeLV-specific antigens gp70 and p27. The polyclonal antibody against Bcl-2 (Table 2) produced no specific reaction in the tested feline tissues. In contrast, the monoclonal antibody (bcl-2/100/D5, Table 2) stained the feline tonsil as expected. Therefore, the monoclonal antibody was applied subsequently.
In immunohistochemistry with the monoclonal antibody (Fig. 2), primary follicles stained for Bcl-2 as did mature lymphocytes within the mantle zones and in extrafollicular areas (Fig. 2A–F). Centers of neoplastic follicles were predominantly negative for Bcl-2 expression (as were secondary follicles in non-neoplastic tissues), with the exception of single cells sometimes arranged in small nests or cords, often originating from the mantle zone but also isolated within the follicle center. Intensity of Bcl-2 staining of the positive cells varied from intense to weak. Follicles without polarity showed randomly distributed positive cells; positive cells in polarized follicles were more prominent within the lighter zone. A difference in number and staining intensity of these cells between lymphomas and follicular hyperplasias was not appreciated. Rarely, single diffuse Bcl-2–positive follicle centers were observed both within lymphomas and follicular hyperplasias.
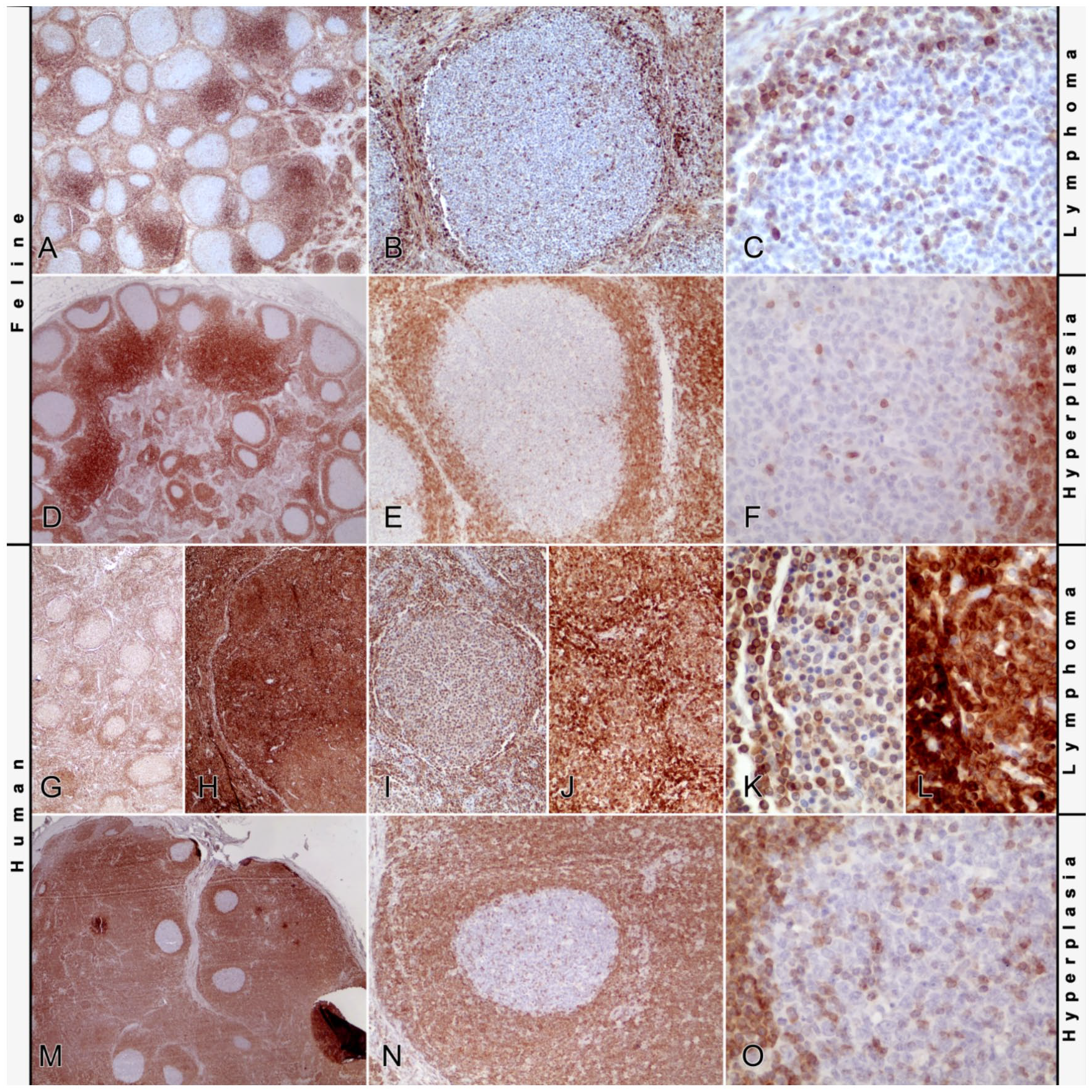
Bcl-2 immunohistochemistry of follicular lymphomas and follicular hyperplasia.
As expected, staining of the human tissues as reaction controls (Fig. 2G–O) resulted in positive cells within the mantle zones and interfollicular areas and negative secondary follicles with single positive cells in the follicular hyperplasia cases. Follicles of the human follicular lymphomas stained diffusely positive, with the grade 2 lymphoma showing more intense staining than the grade 1 lymphoma.
Popliteal lymph nodes were the most common biopsy site (7 of 18) followed by mesenteric (4 of 18), inguinal (3 of 18), submandibular (3 of 18), and prescapular (1 of 18) lymph nodes. Information about the enlargement of lymph nodes at the time of the biopsy was available for 16 cats with lymphoma. Eight cats had only single enlarged lymph nodes, 4 cats had multiple (but regionally) enlarged lymph nodes, and 4 cats had generalized lymph node enlargement. Of the animals with hyperplastic lymph nodes, 3 had multiple enlarged lymph nodes, 1 had enlargement of a solitary lymph node only. For 14 cats, information about lymph node enlargement in the further course of the disease after the biopsy was available. Seven animals had further enlarged lymph nodes in the course of the disease. Of these 7 cats, 2 animals retained the generalized lymphadenomegaly they had at the time of biopsy, and 1 developed generalized enlargement of the lymph nodes after a single enlarged node at the time of biopsy. In 2 animals with multiple enlarged intra-abdominal lymph nodes at the time of biopsy, these enlarged lymph nodes were still detectable until death of the animals. One animal developed generalized lymphadenomegaly after chemotherapy at irregular intervals, which subsided after treatment at regular intervals. Another cat had a period of recurrent general enlargement of the lymph nodes that eventually stopped. Enlarged lymph nodes were not detectable 6 y after biopsy and until death of the animal. For 7 animals, enlargement of further lymph nodes was not reported. However, these cases were not closely monitored by the referring veterinarians. No enlargement of further nodes was reported post biopsy in cats in the hyperplasia group.
Seven cats were treated for lymphoma. In 1 animal, the COP (cyclophosphamide, vincristine [Oncovin], prednisone) protocol was applied, 1 cat was treated with chlorambucil and prednisone, and 5 cats were treated with prednisone alone. In most of these cases, the therapy was supplemented with antibiotics and/or analgesic drugs. Nine animals were only treated with antibiotics and/or analgesic drugs. No information about therapy was available for 2 cats.
The status at the end of the study was known for 14 cats. These cats were included in the Kaplan–Meier estimator. Eleven animals either died spontaneously (3 of 14) or were euthanized (8 of 14). Of the cats that died spontaneously, 1 was killed in a car accident (censored in the Kaplan–Meier estimator), and 2 were found dead. Most commonly reported reasons for euthanasia were anorexia, cachexia, and/or poor general condition. Three animals were alive at the end of the study (censored in the Kaplan–Meier estimator). The median survival time was 4.58 y.
Median survival of cats administered anti-neoplastic therapy was 5.79 y compared to 1.49 y for cats without antitumor treatment (Fig. 3A). The 1-y, 2-y, and 5-y survival rates were 100%, 86%, and 69% for the animals with therapy and 57%, 29%, and 29% for the animals without antitumor therapy. With log-rank (Mantel–Cox) and Gehan–Breslow–Wilcoxon tests, there was no statistically significant difference between the 2 survival curves (p = 0.401 and p = 0.105).
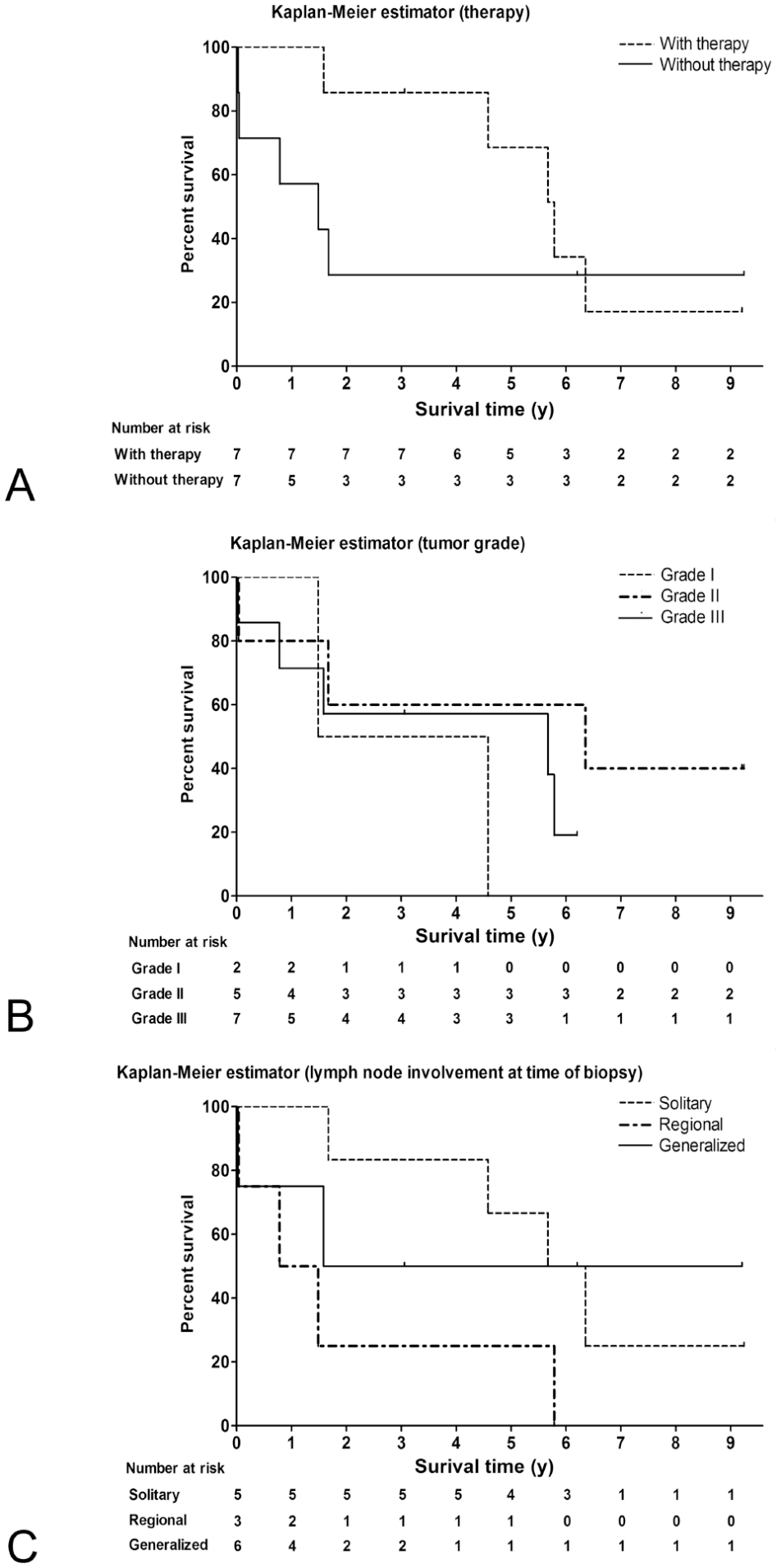
Kaplan–Meier survival curves of animals with follicular lymphoma.
Median survival of cats with tumor grade I was 3.03 y, with grade II 6.35 y, and with grade III 5.52 y. The 1-y, 2-y, and 5-y survival rates for grade I tumors were 100%, 50%, and 0%. For grade II tumors, the rates were 80%, 60%, and 60%. For animals with grade III tumors, the rates were 71%, 57%, and 57%. There was no statistically significant difference between the curves by using the log-rank (Mantel–Cox) test (p = 0.398).
Median survival of cats with solitary lymph node enlargement was 6.01 y, with regional enlargement 1.13 y, and generalized lymph node enlargement 5.42 y (Fig. 3C). The 1-y, 2-y, and 5-y survival rates for cats with single enlarged lymph nodes were 100%, 84%, and 67%, and with regional enlargement 50%, 25%, and 25%, respectively. For cats with generalized lymph node enlargement, the rates were 75%, 50%, and 50%. There was no statistically significant difference between the curves by using the log-rank (Mantel–Cox) test (p = 0.168). The influence of lymph node enlargement after biopsy was not calculated because information was insufficient for statistical analysis.
PARR was performed on all 18 samples. None of the reactions were clonal. Twelve samples had in at least one of the primer combinations of either single peaks or double peaks, or a peak discernible from the polyclonal background, that were not reproducible in the repeated reaction. These reactions were regarded as pseudoclonal. Six samples had a polyclonal pattern in all reactions.
Discussion
The histology of the feline follicular lymphomas in our study resembled the histology of their human counterpart, which is characterized by densely packed follicles effacing normal nodal architecture. Neoplastic follicles are often poorly defined, with attenuated or absent mantle zones and loss of polarity of the follicles (distribution of centrocytes and centroblasts in different zones within reactive germinal centers). 38 However, in some tumors, some follicles maintained polarity. Given that these follicles were primarily located at the margin of the node, they were regarded as remaining reactive follicles. In humans, this phenomenon is known as “partial follicular lymphoma.” 1 Tumor grades were diagnosed according to the WHO scheme. 38 However, grading of this type of lymphoma is challenging. A critical fact is that grading is based on subjective estimation of the cellular composition of randomly selected follicular centers. 25 Even more, the differentiation of centroblasts and centrocytes can be difficult, given that centroblasts develop into centrocytes, which hampers a clear-cut differentiation of these cells. 20 Regarding the survival data, a statistically significant difference between the different grades could not be demonstrated. In humans, the prognostic value for survival of tumor grade is under discussion. There are suggestions to reduce the number of grades because there may not be an important clinical difference between grades 1 and 2. Human grade 3 follicular lymphomas on the other hand seem to be associated with a clinically relevant worse outcome, with additional differences between grades 3A and 3B. 33
In human follicular lymphomas, examination of Bcl-2 expression routinely aids in the differentiation of follicular lymphoma from follicular hyperplasia,11,29 although absence of the expression does not exclude the diagnosis of follicular lymphoma. 33 One goal of our study was to investigate the utility of Bcl-2 expression for the differentiation of follicular lymphoma from follicular hyperplasia in cats. For the immunohistochemical detection of Bcl-2, a monoclonal antibody previously reported to be reactive with feline tissue7,14,19,21,32 was used. Staining with this antibody has shown topographic distribution in feline tissues analogous to that described in human tissues. 22 Furthermore, the feline Bcl-2 protein sequence (UniProt accession: M3X1R9, www.uniprot.org) shows 93% (14 of 15 amino acids) homology with the synthetic peptide used to create the anti-human Bcl-2 antibody (clone BCL-2/100/D5; Leica Biosystems Newcastle, Newcastle Upon Tyne, UK), making specific staining of the feline Bcl-2 highly likely. Primary follicles in our study stained positive for Bcl–2. These follicles are composed of recirculating mature B-cells, which physiologically express Bcl–2. 5 In contrast, germinal centers of follicular hyperplasia and follicular lymphoma expressed virtually no Bcl-2, with the exception of single interspersed positive cells. This is regarded as the normal reaction within germinal centers. The positive cells were more frequent in the light zone of polarized follicles in both groups, supporting the theory that polarization marks remaining reactive follicles with normal Bcl-2 positive cells in the selection and differentiation site of the follicle. However, some follicles showed diffuse positivity for Bcl-2 within germinal centers. The relevance of these follicles and the reason for Bcl-2 expression in their germinal centers remains unclear, but given that they were visible in both groups, an association with the neoplastic process is unlikely.
The absence of Bcl-2 expression in most neoplastic follicles may have occurred for several reasons. It is most likely that Bcl-2 does not play the same important role in lymphomagenesis of feline follicular lymphomas as it does in human follicular lymphomas. Given that in human follicular lymphomas the translocation of the Bcl-2 gene into the immunoglobulin heavy chain locus is a key event in the pathogenesis of the tumor, in cats a comparable translocation is probably not involved in the initiation and progression of the tumor. Another reason might be a result of the difficulties in morphologically differentiating follicular lymphoma and follicular hyperplasia. Although we applied the published criteria for the diagnosis of follicular lymphomas and excluded ambiguous cases were excluded, the risk of misdiagnosing follicular hyperplasia as follicular lymphoma in our study remains. Given that follicular lymphoma in humans is a disease of older patients, this risk of misdiagnosis is strengthened by the fact that several of our cases were young cats. On the other hand, given that Bcl-2 expression does not seem to play a significant role in feline follicular lymphomas, pathogenesis and therefore also epidemiology of this disease might be different in cats compared to humans.
Assessment of clonality did not help in this regard, given that none of the lymphomas showed a clonal population. On the other hand, a negative result does not exclude the diagnosis of lymphoma because molecular clonality PCR, although helpful in many instances, has decreased sensitivity in germinal center and post germinal center B-cell lymphomas (including follicular lymphoma) as a result of alterations of the primer binding sites by somatic hypermutation.15,39,41 This phenomenon occurs in normal germinal centers as part of affinity maturation, and is recapitulated in lymphomas originating in this location. Furthermore, a high polyclonal background caused by residual non-neoplastic lymphocytes in incompletely effaced lymph nodes can also make these lymph nodes a difficult target for clonality analysis. 41 However, given that most lymph nodes in our study were almost completely infiltrated by neoplastic cells, this possible reason for lack of clonality identification is unlikely.
The median age of the cats with follicular lymphoma in our study was 7 y. This is comparable to the 5–9.5 y median age of cats with lymphoma in the post-FeLV era.34,36,40 The most affected breed in our study as well as in the previous studies34,36,40 was the European Shorthair, which is probably not the result of higher susceptibility of this breed to lymphomas but rather the higher popularity of the breed. In humans, a slight predisposition of females for follicular lymphoma exists. 4 In our study, most animals were male, consistent with the mild predominance of male cats in other lymphoma studies.6,31,34,36,40
The median survival time of 4.58 y of the cats with lymphoma in our study is consistent with the classification of follicular lymphomas as “indolent” lymphomas. Unfortunately, none of the investigated parameters in our study (i.e., tumor grade, involved lymph nodes) was useful as a predictor of survival. The same applies to the application of anti-tumor therapy. Although the median survival time of cats that were treated was ~4 times longer than of those that were not treated, statistical significance was not reached. However, this might be the consequence of the small number of animals investigated. In humans, asymptomatic patients with follicular lymphoma are treated following the “watch-and-wait” principle,2,3 with anti-tumor therapy only if the patient becomes symptomatic. A median survival time with the “watch-and-wait” principle was 6.7 y compared to 5.9 y with chemotherapy (chlorambucil). 3
The diagnosis of follicular lymphomas in cats is challenging. Bcl-2 expression as frequently seen in human follicular lymphomas was not seen in our series of feline follicular lymphomas. Therefore, this helpful diagnostic tool in human cases is probably not applicable to feline lymphomas. Furthermore, useful predictors of survival time could not be determined from the clinical data, which, at least in part, may be to the result of the small number of available cases.
Footnotes
Acknowledgements
We thank Dr. Volker Vervölgyi for support in statistical analysis, Dr. Wolf von Bomhard for providing samples, and all of the referring veterinarians and owners for providing information about the patients.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
