Abstract
We conducted a cross-sectional serologic study at Kampala City abattoir in Uganda on 287 small ruminants (221 goats and 66 sheep) to determine the seroprevalence of brucellosis. The samples were tested using a modified rose bengal test (mRBT) and an indirect ELISA (iELISA). Small ruminant Brucella spp. seropositivity was 18 of 287 (6.3%) by mRBT and 19 of 287 (6.6%) by iELISA. The prevalence of brucellosis by mRBT was non-significantly higher in goats (17 of 221; 7.7%) than in sheep (1 of 66, 1.5%; p = 0.069), and also non-significantly higher by the iELISA in goats (18 of 221; 8.1%) than in sheep (1 of 66, 1.5%; p = 0.057). Brucellosis in slaughtered goats and sheep is a public health hazard to abattoir workers and consumers that calls for control and eradication measures at the farm level, given that testing is not carried out routinely at slaughter points.
One of the animal health and zoonotic concerns in Uganda is brucellosis.8,13 The major zoonotic species are Brucella melitensis, B. abortus, and B. suis, which are transmitted to humans mainly from cattle, sheep, goats, pigs, camels, and wildlife. 14 Brucellosis is a direct zoonosis that is transmitted via fluids or products of diseased animals by direct contact or consumption of contaminated products. Brucella can enter the human body through breaks in the skin, conjunctivae, and respiratory and alimentary tracts. Ingestion of unpasteurized dairy products, such as milk, fresh cheese, cream, ice cream, or yogurt, is the most common transmission pathway for foodborne brucellosis. Meat products often have a low bacterial load, but consumers of raw or undercooked meat delicacies could be at risk. Exposure to brucellosis is an occupational risk for farmers, veterinarians, laboratorians, and abattoir workers. 12
Brucellosis is a public health challenge, yet it is neglected in many parts of the world, especially in low-income countries. 5 Brucellosis is also a significant barrier to international trade. The disease burden in many developed countries is low, but brucellosis is highly endemic in livestock and people in sub-Saharan Africa. 15 This could be as a result of the lack of robust testing capacity, which can lead to misdiagnosis and hence can hamper control efforts. In Uganda, various food safety promotion studies have been done to monitor the burden of brucellosis along animal value chains. At the farm level, the prevalence of brucellosis has been reported to be >30% in cattle 1 and 13–43% in goats7,13; scant data exist on the disease prevalence in sheep. Non-ruminants (such as pigs) have low prevalence rates, thus presenting a minimal risk of acquiring brucellosis from pigs or their products. 4
Animal slaughter sites, which generate most of the meat consumed in Uganda, lack rigorous veterinary public health surveillance and diagnostic systems, which directly impacts strategies for zoonosis prevention and control. The direct-contact nature of abattoir work with carcasses ensures that workers are at risk of exposure to Brucella spp. Studies of the prevalence of brucellosis7,8,13,15 have used various screening tests, such as the rose bengal test (RBT) or ELISA, with fewer trials with molecular and bacterial culture methods. Data evaluating the performance of the tests is scanty in Uganda, which may affect the choice of tests in national disease eradication campaigns, such as the test-and-slaughter approaches that have been effective in the developed world. We investigated the seroprevalence of brucellosis in small ruminants (goats and sheep) slaughtered at an abattoir in Kampala, Uganda, using a modified RBT (mRBT) and an indirect ELISA (iELISA).
Our cross-sectional study was conducted at the Kampala City abattoir, which is one of the most frequently used animal slaughter sites in Kampala, Uganda; the animals slaughtered there originate primarily from Uganda. Research approval was obtained from the institutional review board of the COVAB, Makerere University (SBLS/REC/16/003). Permission to access the slaughterhouse premises during slaughter was obtained from the abattoir authorities before commencement of our study.
Blood samples were obtained from 287 small ruminants at the time of slaughter in June and July 2016. We assumed Brucella seropositivity in small ruminants to be ~20% at a 95% CI and a desired precision of 5%, totaling 246 animals. However, given the minimal number of sheep presented for slaughter, more sampling days were added to capture more sheep, thus obtaining 287 animal samples. We chose to sample in June and July to avoid the rainy season (Fig. 1), during which disease outbreaks often occur. From the ~40 animals slaughtered per day, we sampled 10–14 animals by a simple random technique on alternating 2 or 3 random-picked days in a week. During collection, age, sex, and breed of the small ruminants were captured.
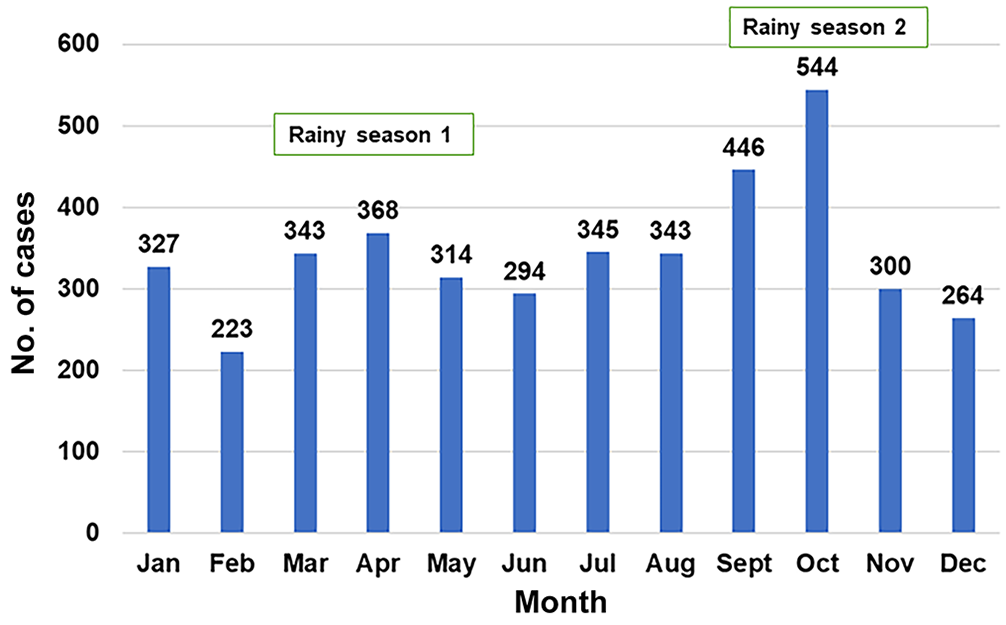
The monthly distribution of cases submitted to the Central Diagnostic Laboratory (Makerere University, Kampala, Uganda) for bacterial disease diagnosis from January 2012 to December 2019. Higher numbers of animal samples (usually reflecting disease outbreaks in the field) are submitted during the rainy seasons of the year.
We sampled 287 small ruminants, 221 (77%) goats and 66 (23%) sheep; 120 (41.8%) were female, 167 (58.2%) were male. Fifty-two (18.1%) were ≤1-y-old, 160 (55.7%) 2–3-y-old, and 75 (26.1%) were ≥4-y-old. The sampled goat breeds included local (Mubende [72], local [42], small East African [21]) and exotic (Boer [38], Black Bengal [16], Saanen [12], Jamunapari [10], Toggenburg [6], Red Sokoto [3], Red Head [1]) breeds. The sampled sheep breeds included local (Red Maasai [32], white sheep [11], local [7], small East African [4], Mubende [2], Sebei [1]) and exotic (California Red [5], Blackhead Persian [4]) breeds.
At least 3 mL of blood were collected by syringe from the jugular vein of each selected animal immediately after slaughter. The blood was transferred into a 5-mL plain Vacutainer tube (Becton, Dickinson). For each sampling day, all samples collected were transported on ice in a cool box to the central diagnostic laboratory at the College of Veterinary Medicine, Animal Resources and Biosecurity (COVAB), Makerere University, Kampala, Uganda. In the laboratory, the samples, which had clotted during transport, were centrifuged at 2,000 × g for 15 min. Sera were aliquoted into sterile cryotubes that were stored at −20°C pending testing.
For each sample, parallel testing was done using a RBT (Rose Bengal rapid slide agglutination antigen kit, RSA-RB; IDvet) and an iELISA (ID Screen brucellosis serum indirect multi-species; IDvet). Known positive and negative internal controls were run with the samples. The standard RBT was modified as recommended for samples of sheep and goats 2 : 25 μL of antigen and 75 μL of serum were mixed on a white tile. While rocking the tile, agglutination that occurred within 4 min was considered to indicate seropositivity. The iELISA kit used detects IgG antibodies to Brucella spp. that express the smooth lipopolysaccharide, such as B. abortus, B. melitensis, and B. suis. Based on the manufacturer’s instructions, S:P values were considered to evaluate a sample as positive if ≥120%. 6
Data were captured in Excel v.2016 (Microsoft) and analyzed with Stata 14 (StataCorp). Descriptive data were presented as calculated frequencies and percentages. The prevalence of brucellosis was calculated as the proportion of samples that tested positive of the total number of samples for the respective species (sheep and goats). The corresponding prevalence CIs were computed as exact binomial 95% CIs. The chi-squared (χ2) test was used to compare the Brucella seropositivity between species and intrinsic variables (age, sex, breed) at a 5% significance level.
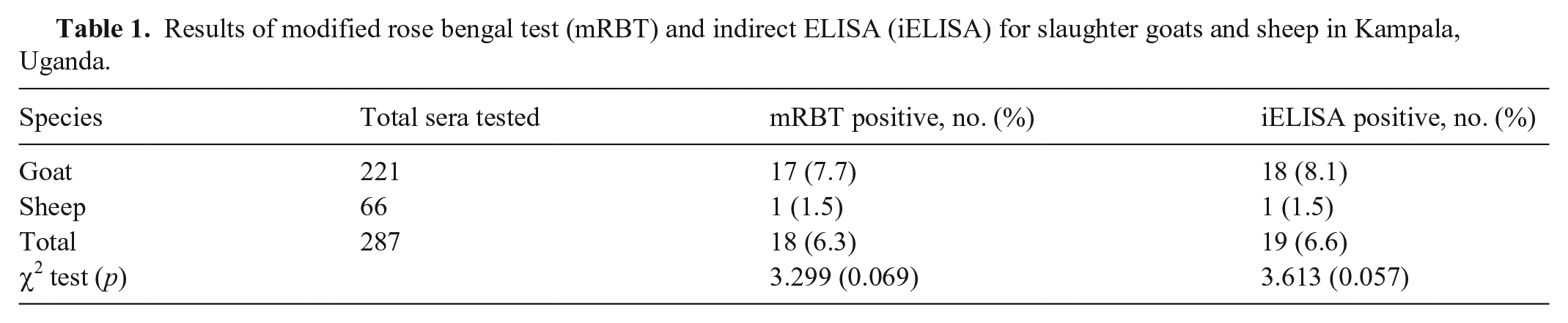
The overall seroprevalence of brucellosis in sheep and goats was 6.3% (18 of 287; 95% CI: 3.8–9.7) by mRBT and 6.6% (19 of 287; 95% CI: 4.0–10.2) by iELISA (Table 1). The prevalence of brucellosis was not significantly higher by mRBT in goats (7.7%; 95% CI: 4.5–12.0) than in sheep (1.5%, 95% CI: 0.0–8.2; p = 0.069), nor with the iELISA in goats (8.1%; 95% CI: 4.9–12.6) than in sheep (1.5%, 95% CI: 0.0–8.2; p = 0.057). Although we found a slight difference in the prevalence of Brucella antibodies, in subsequent analyses, we used the iELISA results based on the test properties (iELISA has higher specificity than the mRBT).10,13,16
Results of modified rose bengal test (mRBT) and indirect ELISA (iELISA) for slaughter goats and sheep in Kampala, Uganda.
In goats, Brucella seropositivity was significantly higher in females (12.6%) than in males (5.2%, p = 0.049; Table 2). Seropositivity was significantly higher in goats ≥4-y-old (15.5%) than in those <4-y-old (5.5%, p = 0.017; Table 2). The local breeds of goats and sheep were found to be more often positive, but non-significantly (10.3% and 2.2%), than the exotic breeds (4.7% and 0.0%; Table 2). There were no significant differences in the seropositivity of sheep to brucellosis in relation to their sex, age, or breed (p > 0.05; Table 2).
Seroprevalence of Brucella antibodies among goats and sheep by sex, age group, and breed-wise classification.
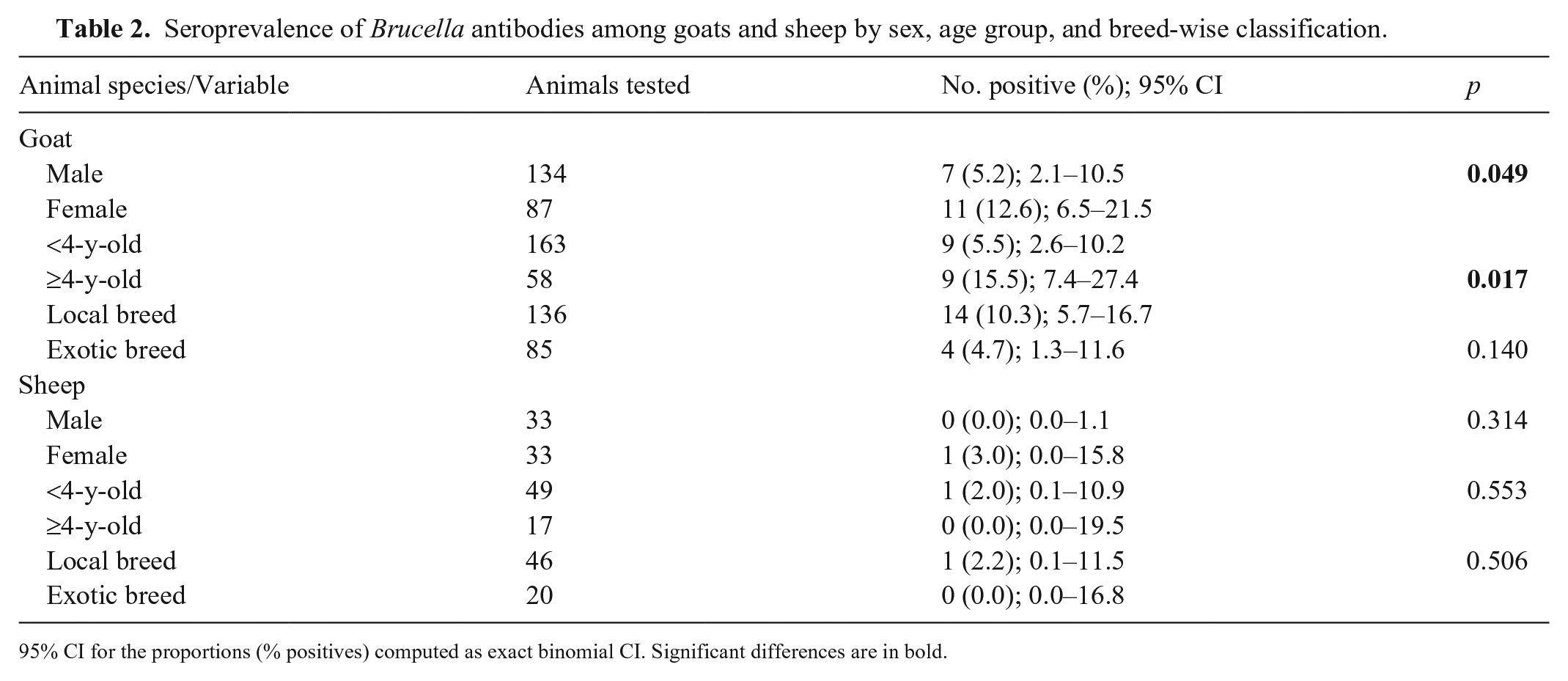
95% CI for the proportions (% positives) computed as exact binomial CI. Significant differences are in bold.
Overall, the seroprevalence of brucellosis in small ruminants was 6.3% by mRBT and 6.6% by iELISA. Along the livestock value chain in Uganda, to our knowledge, Brucella infection has not been reported in slaughtered small ruminants; most studies report farm prevalence rates.7,13 In addition to education, we advocate for farmers to embrace vaccination, which is very uncommon, to reduce the current rates. Another eradication approach could be a test-and-slaughter policy, which however is suggested only when the prevalence of infected animals is <2%. 11 The >6% prevalence in Uganda’s small ruminants may make this policy impossible given the limited government resources for compensation of farmers.
Our small ruminant prevalence rates were slightly lower than rates obtained by others.13,16 This could be explained by spatiotemporal dynamics, such as the origin of samples from different geographic locations, years, and seasons. The prevalence indicated by the mRBT and ELISA results revealed a higher prevalence in goats than in sheep, which agrees with one study, 9 but not another. 16 Noteworthy, slightly more positive cases were detected by the iELISA than the mRBT, a finding that differed from several studies.6,13 In most of the latter studies, as well as our study, the iELISA kit used is a qualitative test that detects only IgG, whereas the RBT kit detects IgM, IgG, and IgA qualitatively. 3 Also, the basic RBT is extremely sensitive but heterospecific 16 ; infection caused by bacteria such as Vibrio cholerae, Yersinia enterocolitica 0:9, Pasteurella spp., Salmonella spp., or some other members of the Brucellaceae family could give more false-positive results in RBT than in the iELISA, a test that may eliminate heterospecific agglutination. The difference in our study results and those reported elsewhere6,13 could be the result of the difference in the method of the RBT used; we based our results on a mRBT, which is more accurate than the basic RBT when testing small ruminants.
A limitation of our study is that we did not examine the occupational risk to abattoir workers of contracting brucellosis, nor did we examine the exposure of the human population to meat from this site. Vaccination could contribute to false-positive results, hence future studies could consider profiling positives further to distinguish seropositivity that resulted from exposure, active infection, or vaccination; our definition of seroprevalence of brucellosis included any source of exposure, natural or vaccination. Also, a high percentage of the goats go through custom slaughter, and we did not test any of those goats.
The mRBT is more economical that the iELISA, has sufficient sensitivity and specificity, and could be adopted in mass testing schemes. At the point of slaughter, positivity could be backed up by clinical signs (weight loss, lameness in goats) and gross lesions (lymphadenitis, orchitis) seen in animals during antemortem and postmortem inspection. In addition, the same test can support surveillance in at-risk abattoir staff to monitor the zoonotic infection rate in exposed humans. Brucellosis is endemic in Uganda, and it impacts the health of humans and animals, ultimately affecting the national economy. 15 Our results support the need for One Health brucellosis control efforts by public health departments in Uganda. 17
Footnotes
Acknowledgements
We thank the Kampala Capital City Authority and the abattoir staff, particularly the veterinary inspector who supported our sample collection. We also thank the Central Diagnostic Laboratory (Makerere University, Kampala, Uganda) where the serologic testing was done.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding was provided by internal funds from whichever affiliated organization provided the funds.
