Abstract
An 8-wk-old, male, mixed-breed puppy was adopted from a rescue organization. From the time of adoption, the puppy suffered episodes of illness affecting various organ systems, which resolved with supportive therapy but relapsed once medical therapy was discontinued. Review of the hematologic data revealed cyclic fluctuations in circulating blood cells. Cyclicity was most prominent in neutrophils, with recurrent severe neutropenia. Neutropenic episodes lasted 5–6 d, with regular cycles of 11–14 d between nadir neutrophil counts. Genetic testing determined that the patient was homozygous mutant for the frameshift mutation in the adaptor protein complex 3 β-subunit (AP3B1) gene, originally identified in gray collies with cyclic hematopoiesis (CH). Pedigree information was not available, but the patient’s features were phenotypically distinct from those of collies. We describe here a case of the AP3B1 mutation in a mixed-breed dog that did not resemble a collie, undescribed previously, to our knowledge. Our findings indicate that the AP3B1 mutation and CH are present within the general canine population and are not restricted to collies.
Keywords
An 8-wk-old, male, mixed-breed puppy was adopted from a rescue organization in Ontario, Canada. After adoption, the puppy had intermittent medical problems, and was presented for veterinary assessment at a primary care veterinary clinic within the first week of adoption. At that time, he was lethargic, pyrexic (40.0°C), and nonambulatory with right hock joint effusion, bilateral carpal hyperextension, and pain. The patient was prescribed gabapentin and acetaminophen (Tylenol; Johnson & Johnson) for pain, and amoxicillin–clavulanic acid (Clavaseptin; Vetoquinol) for potential septic arthritis. Joint effusion resolved, but the carpi remained painful on flexion. A diagnosis of metaphyseal osteopathy was made based on clinical signs and supporting radiographic findings, and the patient was started on oral prednisone on day 6 following presentation. Neutropenia was identified on a CBC 15 d following initial presentation.
Over the next 5 mo, the patient experienced intermittent episodes of illness consisting of pyrexia, granulomatous nodular dermatitis and panniculitis, oral ulceration, vomiting, and urinary tract infections. Referral to the Ontario Veterinary College followed, where the patient was noted to have enlarged mandibular, superficial cervical, and popliteal lymph nodes. Cytologic evaluation of lymph node aspirates revealed suppurative inflammation without identifiable microorganisms. Throughout 8 episodes of illness, the patient’s condition improved with antimicrobial therapy, analgesia, and fluid therapy, but relapsed after therapy was discontinued. Review of hematologic data revealed cyclic neutropenia that coincided with episodes of clinical illnesses. Neutropenic episodes lasted for 5–6 d, with 11–14 d between nadir neutrophil counts (Fig. 1).
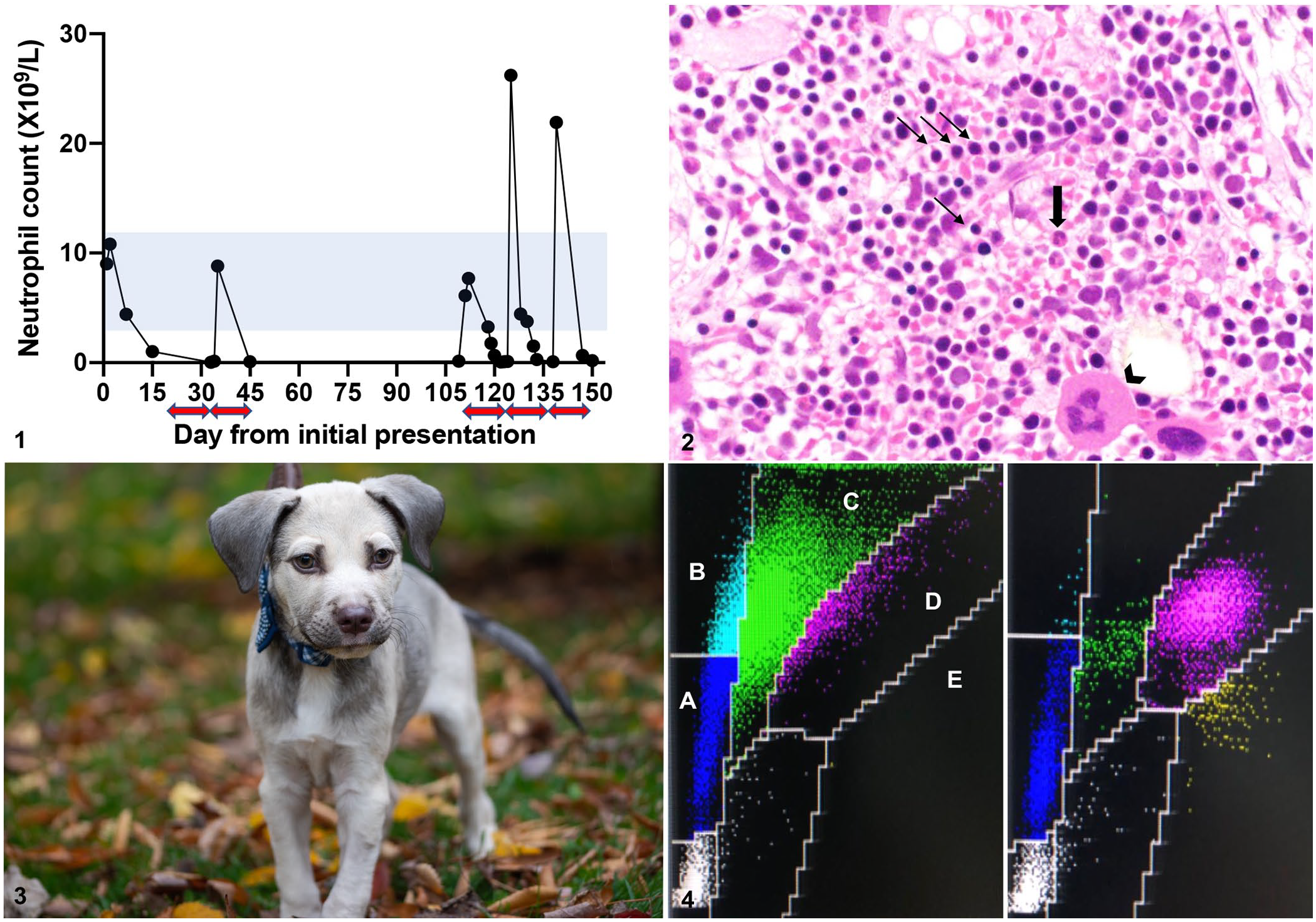
Cyclic hematopoiesis (CH) in a mixed-breed dog.
A bone marrow aspirate and core biopsy were collected during a neutropenic episode at 120 d following initial presentation; granulocytic hypoplasia was marked (Fig. 2). The number and morphology of erythrocytic and megakaryocytic precursors were unremarkable. The cyclic nature of neutropenia was suggestive of canine cyclic hematopoiesis (CH), but the patient had physical features different from the gray collie phenotype expected with the disease. Based on appearance, a presumptive cross between a Labrador Retriever and a Mastiff was theorized, but the ancestral breeds were unknown and pedigree information was not available. The patient’s coat color was a mixture of gray, brown, and white, which was suggestive of hypopigmentation (Fig. 3). DNA testing for breed ancestry was not performed; hence, collie ancestry could not be confirmed.
A venous blood sample in EDTA was submitted to Paw Print Genetics (Neogen) for genetic testing of the 1-bp deletion in the adaptor protein complex 3 β-subunit (AP3B1) gene at chr3:28,663,129-28,663,130 (CanFam3) associated with cyclic neutropenia in collies. 2 The patient was confirmed to be a homozygous mutant via 2 independent genotyping methods using the SeqStudio genetic analyzer (Thermo Fisher) and the MassARRAY system (Agena Bioscience), confirming a diagnosis of canine CH. Of note, this sample was initially tested through another commercial veterinary genetic testing laboratory and reported to be heterozygous for the mutation. Because various factors, including testing methods, mutation types, polymorphisms, and sample quality, can impact genetic testing, and not all laboratories follow the same standards, clinicians should interpret genetic test results in conjunction with the clinical presentation, and laboratories should be consulted in case of discrepant results.3,18 Depending on the type of assay, up to 23% of results for detection of canine CH may be erroneous. 3
In dogs, CH, also known as cyclic neutropenia and gray collie syndrome, was first reported in gray collies in 1967, and has since been synonymous with this breed variant, in which it is an autosomal recessive condition.9,13 Affected collies have abnormal coat pigmentation ranging from dark pewter gray to silver, with cyclic fluctuations in hematopoietic cells and humoral regulators of hematopoiesis. 20 The most notable feature is cycling of the peripheral neutrophil count, leading to regular, 3–4-d periods of neutropenia, with 8–14 d reported between nadir neutrophil counts, which are often near zero.5,13 Cycle lengths vary slightly depending on the individual, but they last 11–12 d in most affected dogs. 5 Neutropenic episodes are linked to increased susceptibility to infections, with death being frequent within the first few months of life.6,13 Additionally, neutrophils of affected dogs have impaired metabolism and bactericidal functions, including abnormally increased post-phagocytic C1-glucose oxidation, decreased iodination of bacteria, and lower levels of intracellular myeloperoxidase. 4
Infections are opportunistic and may affect the gastrointestinal tract, lungs, kidneys, bones, and skin. 6 Gonadal hypoplasia can be noted, along with ill-thrift.6,20 Pyrexia is a frequent clinical sign, and dogs that survive beyond 30-wk-old often have evidence of amyloidosis within the spleen, liver, kidneys, and gastrointestinal tract. 6 Neutropenia is associated with marked depletion of neutrophil precursors in the bone marrow, implying a defect early in the maturation process. 13 Recovery from neutropenia is usually followed by transient neutrophilia, a dip, and a second peak in neutrophil count before the subsequent neutropenic episode. 5 Blood monocytes, eosinophils, reticulocytes, and platelets also follow distinct cycles of similar length. 5 Eosinophil and monocyte counts increase during neutropenia. 5 Monocytosis is noted as neutropenia resolves, and lasts for several days. 13 Reticulocyte counts increase during neutropenia, followed by an increase in mature erythrocyte density. 5
The nadir platelet count occurs when the neutrophil counts are normal, with the platelet count increasing as neutrophils fall into the neutropenic period, and decreasing as the neutrophil count increases. 5 Thrombocytopenia does not occur, but platelet function is defective in affected dogs, with markedly decreased serotonin and adenine nucleotides within granule storage pools, and impaired platelet aggregation following stimulation with collagen, platelet-activating factor, 12-O-tetradecanoylphorbol-13-acetate, and thrombin. 12 In our patient, similar cycles within circulating monocytes, platelets, and reticulocytes were noted (Suppl. Figs. 1, 2; Suppl. Table 1). Circulating neutrophils had decreased peroxidase content when analyzed by an Advia 2120i automated hematology analyzer (Siemens; Fig. 4). In our patient, platelet function testing through Plateletworks (Helena Laboratories) indicated decreased platelet aggregation following the addition of ADP (7% aggregation at 10 min, RI: >70% aggregation). However, platelet function testing was within the RI when tested with the Innovance platelet function analyzer 200 system (Siemens), using the collagen/ADP, collagen/epinephrine cartridges, and P2Y cartridges. These negative results were suspected to be the result of the tests not being sufficiently sensitive to detect the platelet function defect. Thromboelastography (Haemonetics) identified a hypercoagulable state as a result of an increased coagulation index (4.5; RI: −3 to +3) secondary to increased maximum amplitude (78.2 mm; RI: 51–69 mm). The hypercoagulable state was attributed to chronic corticosteroid therapy.
The causative genetic mutation in gray collies was identified in the AP3B1 gene, wherein insertion of an adenine in exon 21 causes a frameshift mutation and premature termination. 2 This defect results in impaired intracellular trafficking and misdirection of proteins to membranes rather than granules. 2 Consequently, dogs with CH have markedly reduced mature neutrophil elastase and instead accumulate enzymatically inactive neutrophil elastase precursors.2,15 In vitro studies showed that neutrophil elastase antagonizes the actions of granulocyte colony-stimulating factor (G-CSF), suggesting a role in providing negative feedback on granulopoiesis. 7 The importance of neutrophil elastase in hematopoiesis is also supported by its role in human CH. In humans, CH is caused by mutations in the ELANE (previously ELA2) gene, which encodes for neutrophil elastase. 14 Almost all cases of CH in humans are autosomal dominant. 9 The disease in humans has neutrophil cycles of 21 d, a number correctly predicted by allometric scaling. 16 In contrast to dogs, mutations in AP3B1 in humans lead to Hermansky-Pudlak syndrome type 2, a disorder that includes partial albinism, platelet granule deficiencies, and congenital, but not cyclic, neutropenia.2,9 Mutations in AP3B1 also impair trafficking of tyrosinase in melanocytes and proteins to platelets, which explains hypopigmentation and defective platelet function in affected humans and dogs. 1
Overall, CH in dogs and humans are closely related but not identical diseases. Various distinct mutations in ELANE have been identified in CH of humans, but not all mutations lead to cyclicity or neutropenia, suggesting that the roles of neutrophil elastase and the exact reason for cyclicity remain incompletely elucidated. 9 Disturbance of a feedback loop, as hypothesized for neutrophil elastase, remains a prominent theory. 9 Other hypotheses include the mislocalization and the unfolded protein theories, whereby alterations in protein subcellular location impair regulatory functions, and mutations lead to protein misfolding with a subsequent stress response and apoptosis, respectively. 9 However, not all neutrophil elastase mutations impact subcellular localization, and not all mutations consistently evoke the unfolding protein response. 9
Lithium carbonate and endotoxin were explored initially as therapeutic options, and although capable of abrogating hematopoietic cell cycling, these are no longer considered therapeutic options, likely because of adverse effects. 8 Elimination of cycling was noted in dogs treated with either recombinant canine G-CSF or human G-CSF, but development of antibodies against human G-CSF resulted in disease recurrence in dogs, which limits its use. 17 Allogenic bone marrow transplantation can cure both the hematopoietic dysfunction and hypopigmentation, but is limited because of cost and difficulties in finding a compatible donor. 20 Interestingly, bone marrow transplants from diseased gray collies to clinically normal dogs can transfer the disease. 19
We have documented here a case of CH in a mixed-breed dog phenotypically distinct from a collie, with a homozygous AP3B1 gene mutation. Reports of CH in non-collie dogs are sparse and include a colony of collie and collie-Beagle mixed-breed dogs purposely bred for studying this disease, and a single Bassett Hound.10,11 The Bassett Hound had neither the coat color dilution nor the AP3B1 gene mutation, suggesting that additional mutations may result in similar fluctuations in hematopoietic cells. 11 Therapy following diagnosis in the dog in our report included prophylactic antibiotics and prednisone to preempt the effects of neutropenia. Episodes of clinical illness and neutropenia were treated with appropriate supportive care, as required. Treatment with recombinant human G-CSF was considered but not pursued given both unavailability and concerns for development of antibodies.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387221115179 – Supplemental material for Cyclic hematopoiesis in a mixed-breed dog: case report and brief review
Supplemental material, sj-pdf-1-vdi-10.1177_10406387221115179 for Cyclic hematopoiesis in a mixed-breed dog: case report and brief review by Gary K. C. Lee, Courtney Barbosa, Gitte Andersen, Christina J. Ramirez, Matthew Kornya, Anthony Abrams-Ogg, Katherine Morrison, Gabriella Diamantino, R. Darren Wood, Janet Beeler-Marfisi, Fernanda Ampuero, Laetitia Tatiersky and Dorothee Bienzle in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
Christina J. Ramirez is an employee of Paw Print Genetics, Neogen, which provides genetic testing for inherited disorders to breeders, owners, and veterinarians. The other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
