Abstract
South American camelids (SACs) have become increasingly popular as livestock and companion animals in the Midwestern United States. With increased ownership, postmortem evaluations and samples available for diagnostic assessment are being submitted more frequently to veterinary diagnostic laboratories. We searched archived pathology records at the Wisconsin Veterinary Diagnostic Laboratory (WVDL) between 2009–2019 for SAC cases. Postmortem records from 166 alpacas and 27 llamas were included, with an average of 1.42 diagnoses per animal. The overall average diagnostic rate was 79.8%. Abortion and neonatal cases (fetus to 1-wk-old) had the lowest diagnostic rate (33.3%) and was the only group with a diagnostic rate statistically significantly lower than the average. The most common diagnoses were gastrointestinal parasitism and suboptimal nutritional status or inanition; 78% of cases diagnosed with suboptimal nutritional status were also parasitized. The gastrointestinal parasites identified most frequently were Eimeria sp. and strongyles, especially Nematodirus sp. Our findings may aid allocation of diagnostic resources to better serve regional SAC populations and provide a framework for practitioners who send samples to laboratories for analysis. Submission of whole bodies, euthanized animals, fresh tissue, and inclusion of placental tissues in abortion cases are recommended for optimal diagnostic outcome.
South American camelids (SACs) have become increasingly popular in the USA in recent decades. In 2017, in the state of Wisconsin, there were an estimated 3,532 alpacas (Vicugna pacos) at 300 operations and 1,649 llamas (Lama glama) at 339 operations. 17 Geographically, Wisconsin is largely composed of prairie and wetlands, which are starkly different habitats compared to the mountain ranges from which these species originated. A different spectrum of diseases may be expected in SACs raised in this part of the continent than in their region of origin. No population-level reports of diseases in SACs from Wisconsin or the Upper Midwest USA were available in the current literature. Additionally, certain factors of postmortem specimen submission may influence diagnostic success.
Archived postmortem reports recovered from the laboratory information management system (LIMS) at the Wisconsin Veterinary Diagnostic Laboratory (WVDL; Madison, WI) were searched from January 1, 2009, to December 31, 2019, for llama and alpaca cases (the database was also searched for vicuna and guanaco cases, but none was found). We included records of 193 SAC postmortem examinations. Information recorded from each case, when available, included the type of submission (whole body postmortem examination, or fresh and fixed tissues from a field examination), signalment (sex, age, species), city and state of origin, date of submission, whether autolysis was noted in the postmortem report (Y/N), whether the animal was euthanized (Y/N), diagnosis(es) (as reported by the primary pathologist of each case), and specific infectious agent(s), when available. Some animals had >1 diagnosis or >1 infectious agent. If indicated that multiple causes led to death or euthanasia, 1–3 diagnoses and infectious agents were included for each animal. Cases were classified as “undiagnosed” if this term or similar terms (inconclusive, indeterminant, unknown, unsure, etc.) were used in the reported final diagnosis.
Submissions were divided into the following age categories (based on standard ages of weaning): abortion, stillborn, and neonate (fetus up to 1-wk-old), cria (1-wk to 6-mo-old, or indicated as “cria” on the submission), juvenile (6-mo to 2-y-old), adult (2–10-y-old, or indicated as “adult”), and aged adult (≥10-y-old). Submission dates were divided into seasonal categories: spring (March–May), summer (June–August), autumn (September–November), and winter (December–February). Reports included gross and microscopic examination of whole bodies or various tissues received from postmortem examinations conducted in the field. Microbiology tests, including bacterial and fungal cultures, fecal and ingesta parasitology procedures, PCR testing, and trace mineral analyses, were elected by the primary pathologist for each case at the time of the postmortem examination. Percentages and averages were calculated in Excel (v.16.59; Microsoft). Odds ratios and CIs were determined by a 2-sided Fisher exact test using STATA (StataCorp).
We identified 193 cases (166 alpacas and 27 llamas) by search criteria from the LIMS at WVDL (Table 1); 81% of submissions originated within Wisconsin. Most submissions were whole-body postmortem examinations (54.4%). Sixty-two percent (62%) of submissions were female, 28.5% were intact males, and 2.6% were castrated males (no spayed females were identified, remaining submissions did not designate sex). Winter was the most common season for submissions (67; 34.7%), followed by autumn (52; 26.9%), spring (38; 19.7%), and summer (36; 18.7%). Adults were the age category submitted most frequently (85; 44.0%), followed by juveniles (35; 18.1%), crias (30; 15.5%), abortions and neonates (27; 14.0%), and aged adults (15; 7.8%); age was not designated for 1 case. Autolysis was noted in 70 reports (36.3%). Eighteen animals (9.3%) were euthanized by intravenous barbiturate injection, and 152 (78.8%) had died naturally; the manner of death was not indicated for the remaining submissions.
South American camelid cases received at WVDL, 2009–2019, including species, state of origin, type of submission, sex, season of submission, age category, whether autolysis was noted in the postmortem examination report, and whether the animal was euthanized. Diagnostic rate, odds of finding a diagnosis(es) compared to average, and CIs as calculated by Fisher exact test are listed.
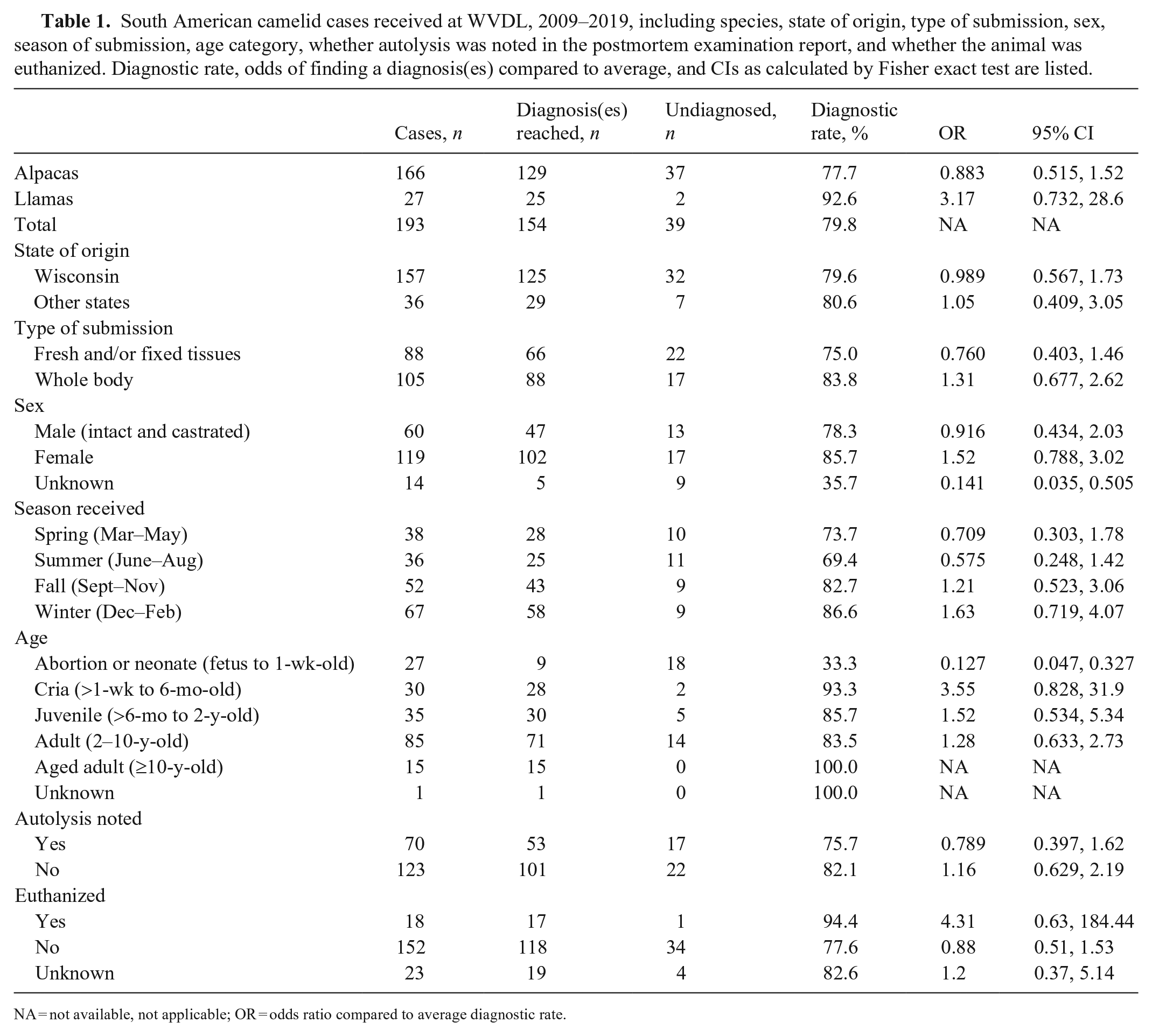
NA = not available, not applicable; OR = odds ratio compared to average diagnostic rate.
Diagnoses were achieved for 77.7% of alpaca cases and 92.6% of llama cases (79.8% average; Table 2). Of those 154 animals, 37.7% had concurrent disease processes, with an average of 1.42 diagnoses per animal. Gastrointestinal parasitism was the most common diagnosis (18.6% of total diagnoses) followed by suboptimal nutritional status or inanition (12.2% of total diagnoses). Suboptimal nutritional status or inanition was defined as poor body condition, serous atrophy of fat, or as otherwise indicated by the primary pathologist at the time of postmortem evaluation. Most (78%) cases diagnosed with suboptimal nutritional status were also diagnosed with parasitism. Parasitism was diagnosed more frequently in juvenile (31.7%) and adult (34.1%) SACs than in crias and aged adults. Cases of parasitism and suboptimal nutritional status were more likely (46.3% and 48.1%, respectively) to be received during autumn. Septicemia, pneumonia, verminous hepatitis, and neural larva migrans were diagnosed more frequently during winter.
Diagnoses found in South American camelid postmortem examination reports at WVDL, 2009–2019, listed by organ system. Up to 3 diagnoses were included for each animal.

Suspected ionophores, vitamin E/selenium deficiency, plant toxicosis.
Several factors were associated with diagnostic rates less than the average, including fresh and fixed tissue submissions, male SAC submissions, submissions received in spring and summer, abortion and neonatal submissions, submissions with autolysis noted in the report, and submissions in which the animal had died naturally. Abortion and neonatal cases had the lowest diagnostic rate (33.3%) and were 0.127 times as likely to have a diagnosis as the average SAC case (p < 0.0001) compared by a 2-sided Fisher exact test. If sex was not listed, the diagnostic rate was significantly lower compared to average (p = 0.0008). Llamas (species category) and crias (age category) were significantly more likely to have a diagnosis compared to the average diagnostic rate (p = 0.08 and p = 0.05, respectively).
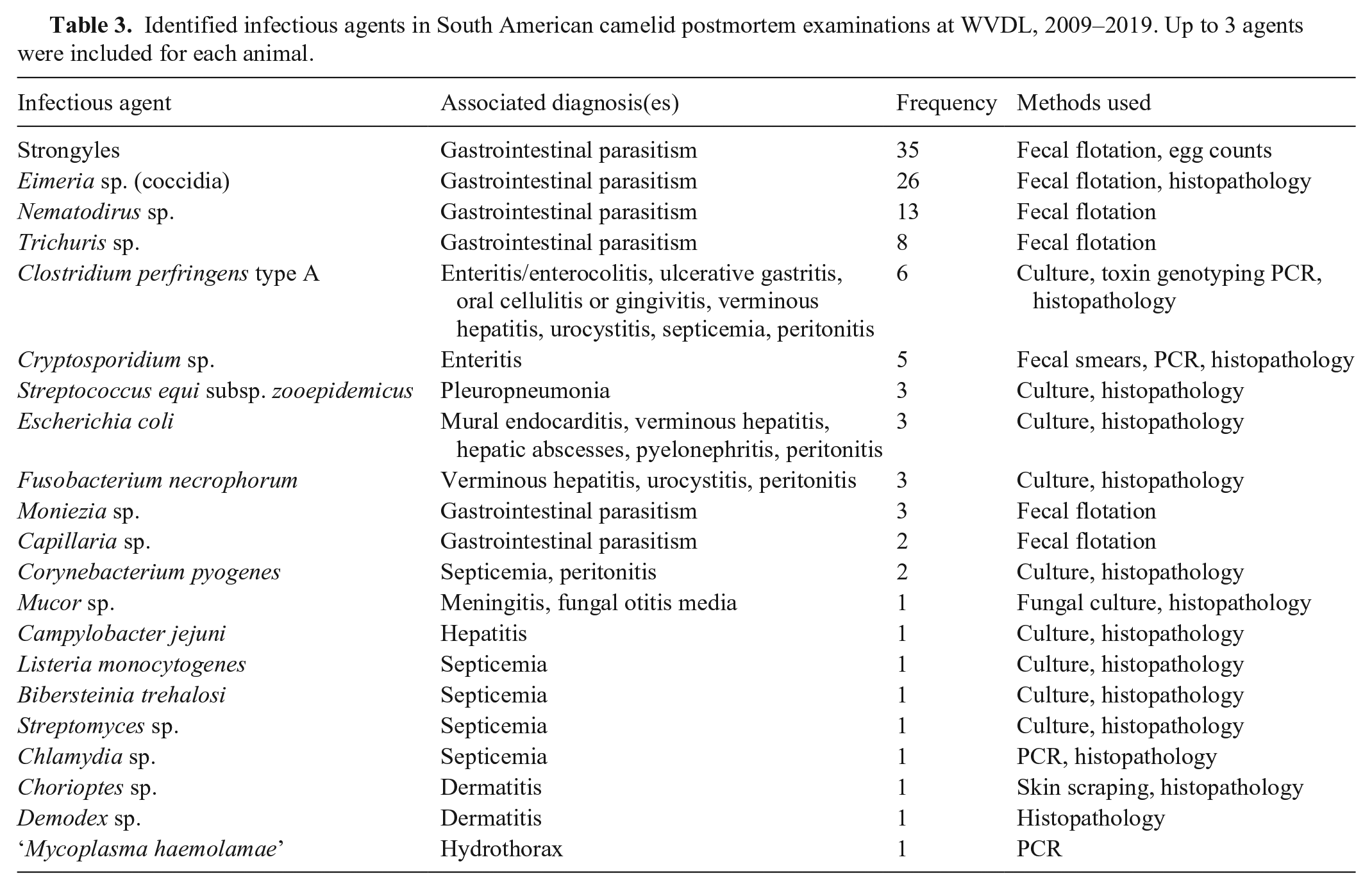
Infectious agents were identified in 61 cases (Table 3). The gastrointestinal parasites identified most frequently by fecal flotation and/or egg counts were Eimeria sp. and strongyles, especially Nematodirus sp. (counted separately from other strongyle eggs on parasitic examinations in reports). Clostridial infection (diagnosed by culture and/or toxin genotyping) was associated with numerous morphologic diagnoses in 6 animals, including enteritis or enterocolitis, ulcerative gastritis, oral cellulitis or gingivitis, verminous hepatitis, urocystitis, septicemia, and peritonitis. Escherichia coli and Fusobacterium necrophorum were also associated with numerous disease manifestations. Streptococcus equi subsp. zooepidemicus was identified in all 3 cases of pleuropneumonia. Despite extensive culture and PCR testing, no infectious causes of abortion were identified. Placentas were included in 20 of 23 abortion submissions. The tissues from all 23 abortion cases were cultured, and PCR testing was performed for the following infectious agents: bovine viral diarrhea virus (23 cases; Pestivirus), Neospora caninum (22), Leptospira sp. (21), Chlamydia sp. (18), Coxiella burnetii (18), equine herpesvirus 1 (9; Equid alphaherpesvirus 1), bovine herpesvirus 1 (8; Bovine alphaherpesvirus 1), and Toxoplasma sp. (6). All test results for the aforementioned pathogens were negative.
Identified infectious agents in South American camelid postmortem examinations at WVDL, 2009–2019. Up to 3 agents were included for each animal.
Studies have indicated that postmortem examination of ruminants is an impactful diagnostic tool for clinical medicine. 19 A large study of SAC mortality in England and Wales 15 reported a much lower overall diagnostic rate (34.2%) than we found; the submission of whole carcasses (compared to non-carcass tissue submissions) was a significant positive factor in determining a diagnosis and multiple diagnoses. Similarly, in cattle, sheep, and goats, the greatest influences on reaching a diagnosis(es) were the way in which the animal died (assisted or unassisted [i.e., euthanasia or natural death]), the type of autopsy examination (partial or complete), and the age of the animal (abortions were not considered in that study). 19
There are few approved methods of euthanasia in camelids recognized by the American Veterinary Medical Association, including barbiturate and barbituric acid derivative overdoses, and physical methods, including gunshot and penetrating captive bolt followed by adjunctive procedures.2,9 From our study and our experiences, the limited available methods of euthanasia, lack of euthanasia training for SACs, and increased regulation of chemical euthanasia agents, may prevent or deter clinical practitioners from performing this necessary procedure. Further studies and research into unapproved adjunctive procedures (intrathecal lidocaine administration, intravenous potassium chloride administration, etc.) for camelids after unconsciousness is determined may provide alternative euthanasia methods for SACs that are accessible, cost-effective, and easier for the practitioner to perform once trained. A suspected lack of medical attention prior to death as well as lack of comfort utilizing available euthanasia methods by attending or submitting practitioners may be contributing factors to the predominance of natural deaths in our submissions.
Regional studies from Sweden, Canada, and the United Kingdom of population-level postmortem diagnoses from diagnostic laboratory records4,12 and producer surveys7,8 have found diagnoses and incidences similar to our findings in the Midwest United States. A guide for diagnostic sampling of SAC postmortem cases is also available. 3 Parasitism and gastroenteritis were frequently identified as the primary cause of illness or death. Wasting or emaciation were frequently reported clinical findings. Parasitism was more common in older animals in our dataset than in other studies focused on neonatal SAC mortality; specifically, rates of coccidia-associated diarrhea in crias were much lower in our dataset.6,13,20 In a 2021 publication about Old World camelids of North America, 11 camels diagnosed with intestinal parasitism had anorexia, weight loss, and diarrhea combined with hypoproteinemia, eosinophilia, and hyponatremia. It was also suggested that clinicians should consider the potential for regional variations in species of gastrointestinal parasites of camels. 11
Suboptimal nutritional status in SACs is frequently remarked upon clinically, often attributed to their unique metabolism. SACs are more susceptible to hepatic lipidosis, vitamin D deficiency, and zinc deficiency than ruminant species. 16 Young animals, and late pregnant and lactating females, are often the first in a herd to be affected. Protein-energy imbalance is more likely than inanition or an overall caloric deficit in most cases if the animal retains its appetite, as opposed to cases of infectious, parasitic, or dental disease in which hyporexia is often noted. 16
Previous studies also indicate low diagnostic success rates in abortion cases.4,7,12,15 Although other factors resulted in lower-than-average diagnostic rates, abortion was the only significant factor that affected diagnostic rates. Abortion cases were received more frequently during the spring and summer (81.4% of abortions received during these 6 mo), which is likely why these seasons had lower than average diagnostic rates; however, seasonal rates were not significantly different from the average of 79.8%. Known causes of abortion in camelids include infections with Leptospira sp., Toxoplasma gondii, Chlamydia sp., Listeria sp., and Streptococcus sp. 14 Abortion rates in SACs as a result of infectious causes can vary dramatically with region and facility,4,7,8,12 but despite evaluation by culture, PCR, and histopathology, no infectious abortions were identified in our dataset.
Neonatal SAC mortality, which was also not observed in significant numbers in our dataset, has been attributed primarily to diarrhea associated with failure of passive transfer and exposure to E. coli, rotavirus, coronavirus, Cryptosporidium sp., Giardia sp., Eimeria sp., or Salmonella sp.6,14,20 Pre-weaning mortality in crias has been shown to be low despite numerous infectious diseases that affect this age group, but difficult birth has been identified as an important risk factor affecting mortality. 13 The gestational age of a cria, which is highly variable in SACs, has also been indicated as a factor influencing survival for crias born before 330 d gestation. 10 Crias are predisposed to metabolic conditions, such as hyperosmolarity, hypernatremia, and hyperglycemia, which can develop quickly as a response to stress and inadequate water intake. 5 Many of these syndromes are clinically defined and treatable, perhaps indicating why neonatal crias were under-represented in our postmortem study.
Neoplasia was diagnosed in 5.2% of the submissions in our study (Table 2), which is within the range reported in other regions (3.0–8.8%).1,12,15,18 The most common neoplasm was lymphoma (4 of 8 total neoplasms diagnosed), which is more frequently identified in young SACs. The age range for lymphoma cases in our study was 4–13 y, which is older than age ranges identified in other retrospective studies.1,18 The mean age of SACs diagnosed with neoplasia in our dataset was 9.0 ± 5.7 y, and all animals identified with neoplasia were alpacas.
Limitations of our study include its retrospective format, relatively small sample size (especially for llamas), variations in tissue submission, and variation in sampling and testing of submissions by the primary pathologists at the time of postmortem examination. Additionally, cases received at the WVDL were likely a biased subset of the total Wisconsin SAC population, potentially excluding cases in which the cost of a postmortem examination was prohibitive, cases that were too autolyzed for submission, and cases that were diagnosed clinically or by gross postmortem examination only. SACs appeared on the Wisconsin Census of Agriculture starting only in 2017, making it difficult to assess mortality trends relative to the state population for more than a few years. Our recommendations for practitioners include submitting whole bodies, euthanizing animals, submitting fresh tissues when possible, and including fetal and placental tissues in abortion cases for optimal diagnostic outcome.
Footnotes
Acknowledgements
A special thanks to WVDL pathologists and staff, past and present, who contributed autopsy reports to the archives. Thanks also to David LaBeause for his aid in searching the electronic archives.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
