Abstract
Adenoviral infections among raptors are best described in falcons and are characterized most commonly by necrotizing hepatitis and splenitis; only one case has been reported in a hawk. Five red-tailed hawks (Buteo jamaicensis) and a broad-winged hawk (Buteo platypterus) had an adenoviral infection based on history, histopathology, negative-stain electron microscopy, and PCR. All birds had acute onset of illness resulting in death; 3 had evidence of a concurrent bacterial infection. Microscopically, all 6 birds had solitary, pale eosinophilic-to-amphophilic, intranuclear inclusion bodies within presumed hematopoietic cells in bone marrow and macrophages in spleen. Five of the 6 birds had similar inclusions within hepatocytes and Kupffer cells. All but one bird had severe bone marrow necrosis. There was moderate splenic necrosis (3 of 6) and mild-to-marked hepatic necrosis (4 of 6). Negative-stain electron microscopy demonstrated adenoviral particles in bone marrow (5 of 6), liver (1 of 5), and/or spleen (1 of 5). PCR was positive for adenovirus in bone marrow (3 of 5), liver (1 of 3), spleen (4 of 6), and/or intestinal contents (2 of 3). Viral DNA polymerase gene sequences clustered within the Siadenovirus genus. There was 99% nucleotide identity to one another and 90% nucleotide identity with the closest related adenovirus (Harris hawk, EU715130). Our case series expands on the limited knowledge of adenoviral infections in hawks. The splenic and hepatic necrosis, and particularly the hitherto unreported bone marrow necrosis, suggest that adenoviral infection is clinically relevant and potentially fatal in hawks.
Keywords
Adenoviruses are nonenveloped DNA viruses of 70–90 nm diameter. The genome is linear double-stranded DNA of 26–45 Kbp. To date, 6 genera belong to the family Adenoviridae: Atadenovirus, Aviadenovirus, Ichtadenovirus, Mastadenovirus, Siadenovirus, and Testadenovirus. 15 Birds can be infected by adenoviruses of the Aviadenovirus, Siadenovirus, and Atadenovirus genera.
Adenovirus in raptors has been best described in falcons as individual cases or outbreaks, most often involving falcon adenovirus 1 (FaAdV1; Falcon aviadenovirus) of the Aviadenovirus genus, although fowl adenovirus (FAdV; Fowl aviadenovirus) 2 and 6 have also been reported.7,26,31,36,37 Falcon species with published adenoviral infection include American kestrels (Falco sparverius), Mauritius kestrels (Falco punctatus), Northern aplomado falcons (Falco femoralis septentrionalis), peregrine falcons (Falco peregrinus), Taita falcons (Falco fasciinucha), hybrid falcons (Falco rusticolus × Falco peregrinus), and gyrfalcons (Falco rusticolus).7,8,26,30,31,34,36,37
In contrast, there have only been rare reports of adenoviral infection in hawks, including a single report of a siadenovirus (raptor adenovirus 1, RAdV1; Raptor siadenovirus) in a Harris hawk (Parabuteo unicinctus), and an aviadenovirus (FAdV4) in black kites (Milvus migrans), which are close relatives of hawks.22,39 Other raptor species with reported adenoviral infection include a Bengal eagle owl (syn. rock eagle owl; Bubo bengalensis) and a Verreaux eagle owl (Bubo lacteus) with RAdV1; and tropical screech owls (Megascops choliba), involving either a novel hitherto unnamed aviadenovirus or a novel atadenovirus species.16,20,39 Lesions associated with adenoviruses in raptors involve hepatic necrosis most consistently, with intranuclear inclusion bodies (INIBs). Other reported lesions include splenitis, nephritis, and, less commonly, lymphoid depletion of the bursa of Fabricius, ventriculitis, and proventriculitis.
Our objective was to describe the gross and histologic lesions, electron microscopy (EM) findings, as well as PCR and sequencing findings of adenoviral infection in hawks from The Raptor Center (TRC; University of Minnesota, St. Paul, MN, USA), with distinct involvement of the bone marrow.
Materials and methods
Animals, sample collection, and histopathology
We selected retrospectively 5 red-tailed hawks (Buteo jamaicensis) and 1 broad-winged hawk (Buteo platypterus) that had been submitted for autopsy by TRC (Table 1). Autopsies were performed by 1 of 2 board-certified veterinary pathologists (A. Wünschmann, A.G. Armién) between September 2014 and August 2019 and fulfilled all of the following criteria: 1) similar clinical history of being rehabilitated at TRC after initial admission for traumatic injuries, 2) developed acute rapidly progressing clinical signs leading to death or necessitating euthanasia within days of onset, 3) histologically detectable INIBs in the liver, spleen, and/or bone marrow, 4) detection of adenoviral particles in liver, spleen, bone marrow, and/or intestinal contents by negative-contrast EM, and 5) positive for adenovirus by PCR in the liver, spleen, bone marrow, and/or intestinal contents. In addition, we included 2 red-tailed hawks housed at TRC as control birds, and undertook the same procedures (autopsy, histopathology, negative-stain EM, and PCR) as the study subjects.
Species, sex, age, date of admission to The Raptor Center (TRC) of the University of Minnesota, duration of human care in our study, reason for admission to TRC, and CBC findings performed within 24 h of death.
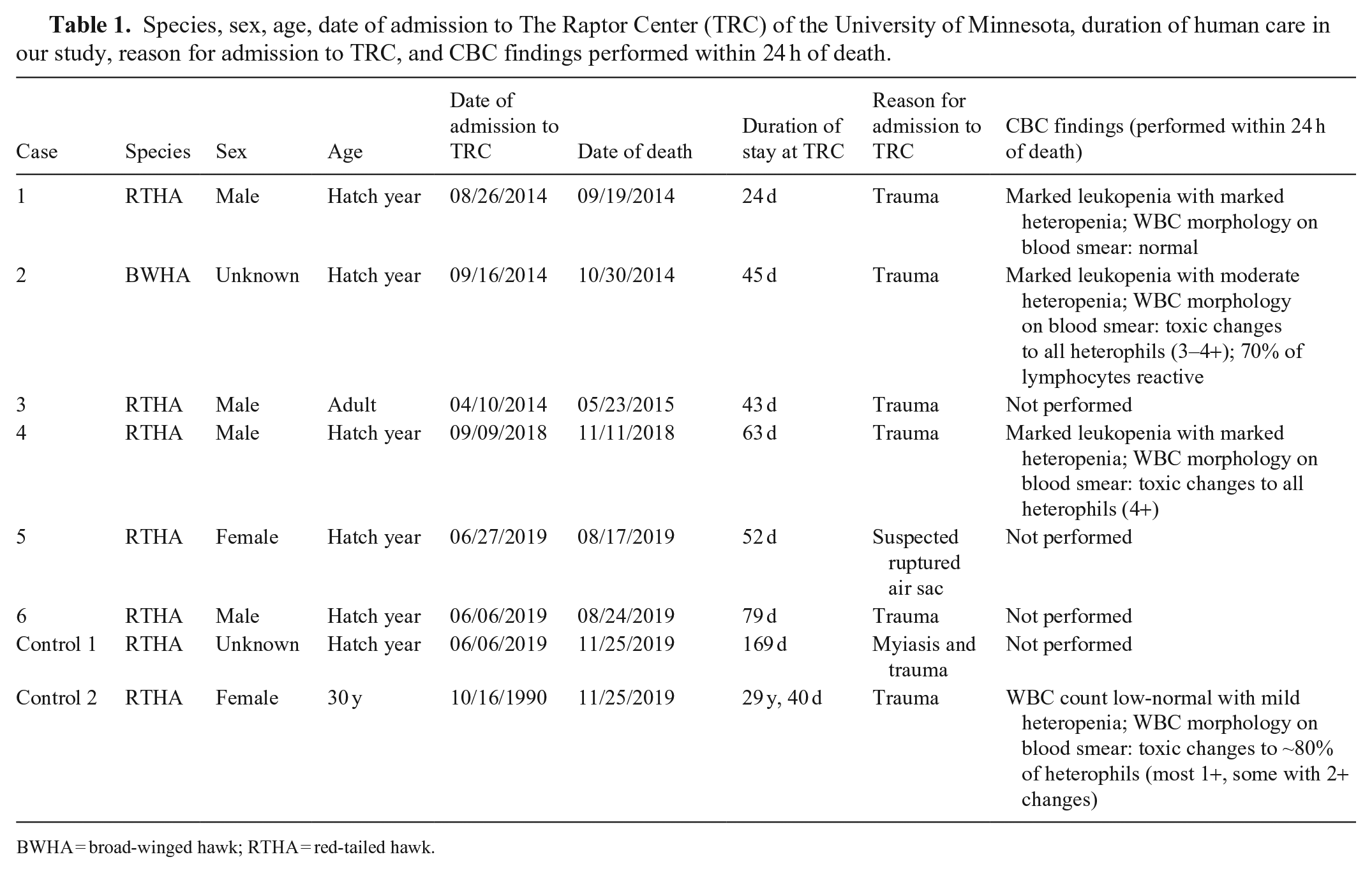
BWHA = broad-winged hawk; RTHA = red-tailed hawk.
Blood samples were collected from 3 cases (1, 2, 4), and 1 control bird within 24 h of death and submitted for a CBC and blood smear examination (Table 1; Suppl. Tables 1, 2). The CBC results were compared to RIs in each of the hawk species.3,14
Tissue samples were fixed in 10% neutral-buffered formalin, including tibiotarsal bone marrow, liver, spleen, brain, heart, lungs, kidneys (6 cases and both control cases for these tissues), ventriculus (5 cases), proventriculus (5 cases and 1 control case), intestine (4 cases and both control cases), and the bursa of Fabricius (2 cases). The formalin-fixed tissues were processed routinely, and 4-µm sections were stained with H&E. In case 5, additional 4-µm sections were stained with Gram, periodic acid–Schiff (PAS), Grocott methenamine silver (GMS), and acid-fast stains. Histologic lesions, such as necrosis, were graded as none, mild, moderate, or marked on the basis of estimated percentage of affected tissue. The estimated number of viral INIBs was graded as none, rare, or occasional. An anticoagulant panel (brodifacoum, bromadiolone, chlorophacinone) was performed on unfixed liver from case 2 by the Michigan State University Veterinary Diagnostic Laboratory (Lansing, MI, USA).
Negative-contrast EM was performed using unfixed bone marrow (all cases including the control cases), liver, spleen, and intestinal contents (cases 2–6 and both control cases) to examine for viral particles as described previously. 2 The samples were placed in a 20-mL tube, and double-distilled water was added to 15 mL final volume. After 10 min at room temperature, 1 mL of the tissue suspension was filtered with 0.45-µm syringe filters (Fisher Scientific). Fifty µL of filtered suspension or supernatant was transferred to Airfuge tubes (Beckman Coulter) with formvar-coated 200-mesh copper grids (Electron Microscopy Sciences) and centrifuged at 30 psi (Beckman Coulter) for 10 min. Grids were washed and stained with 1% phosphotungstic acid (Electron Microscopy Sciences) for 1 min. An ultrastructural examination was performed using formalin-fixed bone marrow and liver from case 5. For this purpose, 1-mm3 fragments of the formalin-fixed bone marrow and liver were post-fixed with 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer. Samples were processed as described elsewhere. 2 A camera and software (AMT capture engine v.7.00; Advanced Microscopy Techniques) was used to take images, and the image analysis was carried out using ImageJ (NIHR public domain).
PCR and sequencing
DNA was extracted (DNeasy blood & tissue kit; Qiagen) from unfixed spleen from case 1; unfixed spleen and bone marrow from cases 2 and 3; unfixed spleen, liver, bone marrow, and intestine from cases 4–6 and both control cases; and formalin-fixed, paraffin-embedded (FFPE) tissues, which included bone marrow, kidney, and skeletal muscle, from case 1. An adenovirus PCR amplifying DNA polymerase gene (~1,200 bp) was performed using methods described previously. 13 HotStarTaq master mix kit (Qiagen) was used for DNA amplification, in a total volume of 25 μL containing 2.5 μL of DNA, 10 μmol each of forward and reverse primers, and 12.5 μL of master mix (deoxynucleoside triphosphate, MgCl, DNA polymerase). The PCR conditions were: initial denaturation at 95°C for 15 min followed by 35 cycles of denaturation at 94°C for 30 s; annealing at 54°C for 45 s; extension at 72°C for 60 s; and a final extension step at 72°C for 10 min. The PCR products were visualized by UV transillumination of electrophoresed 1.2% gel stained with ethidium bromide. Purified PCR products (QIAquick PCR purification kit; Qiagen) were sequenced at the University of Minnesota Genomics Center by Sanger dideoxy sequencing using both forward and reverse primers, and then assembled by Sequencher v.5.1 (http://www.genecodes.com) and Geneious Prime 2020.0.5 (Biomatters). 19 The obtained nucleotide (nt) sequences were then compared with other GenBank sequences using BLAST (https://www.ncbi.nlm.nih.gov/BLAST/).
Sequence alignment and phylogenetic analysis
To examine genetic relationships among FAdV and other avian adenovirus isolates, we determined partial nucleotide sequences of the DNA polymerase gene. Sequencing was performed on 5 of the 6 cases that were adenovirus PCR positive. Sequencing could not be performed on case 6 despite positivity to adenovirus DNA polymerase gene PCR because of inadequate sample quality. The phylogenetic analysis was performed on 857-nt curated sequences and compared with the published sequences of raptor adenovirus and other siadenoviruses available in GenBank. Geneious Prime 2020.0.5 was used for sequence alignment and percent identity analysis. The sequences were aligned using ClustalW, 35 and a phylogenetic tree was constructed in MEGA 7.0. 23 The phylogenetic tree was constructed by comparing 857 nt of the DNA polymerase gene using the maximum-likelihood method based on the Hasegawa–Kishino–Yano model with 1,000 bootstraps.
Bacteriology
Unfixed liver, kidney, lung, and intestine samples from case 1; liver and intestines from cases 2 and 3; liver from cases 4–6; and intestine from control case 2 were submitted for routine aerobic culture. The samples were inoculated on 5% sheep blood agar (SBA), MacConkey agar, Columbia nalidixic acid agar (CAN), and brilliant green agar (BGA) as part of routine aerobic culture. The SBA and CAN plates were incubated at 37°C in oxygen for 18–24 h. The plates were examined for growth, and bacterial colonies were identified using a Microflex LT MALDI-TOF (Bruker). The mass spectrometer was calibrated with a range of 3,637.8–16,952.3 Da prior to sample testing using the bacterial test standard (Bruker), per the manufacturer’s recommendations. In addition, pooled tissues from cases 1 and 2; liver from cases 3 and 4; and intestine from control case 2 were submitted for salmonella culture. For salmonella culture, the tissues were added to Hajna tetrathionate broth and incubated overnight at 40°C. After incubation, the broth was plated to BGA and xylose–lysine–deoxycholate agar plates. The plates were incubated overnight and examined for colonies resembling Salmonella spp.
Virus isolation
Homogenized bone marrow and spleen tissues of cases 1 and 2 were inoculated into the allantoic fluid and yolk sac of 9–11-d-old chicken embryos using a standard protocol. 33 A total of 3 passages, incubated for 5 d at 37°C, were examined. The allantoic fluid was analyzed by a hemagglutination test using turkey erythrocytes.
Results
Clinical findings
The 8 birds (including both control birds) were admitted to TRC with traumatic injuries. All birds were found within 30–145 km (20–90 mi) of TRC, but from different counties. All birds were healing well from their injuries or surgeries and were being prepared for release, excluding control bird 2, which was an educational bird at TRC. The raptors were predominantly fed mice and rats, occasionally supplemented with day-old chicks. All affected hawks spent at least 24 d for rehabilitation at TRC before the deaths occurred. Three deaths occurred in the fall, 2 occurred in late summer, and 1 in late spring (Table 1). All affected hawks either died without showing clinical signs or died after a short course of clinical disease that lasted for <1–4 d. Clinical signs included a short period of weight loss, depression, regurgitation, bright green mutes (i.e., excreta including feces, urate, and urine), and inappetence. One control bird was found dead in its enclosure without premonitory clinical signs. The other control had been an educational bird for >29 y, had progressively worsening orthopedic problems, was inappetent and regurgitating, and was euthanized for quality-of-life reasons.
Clinical pathology findings
The 3 sampled affected birds had marked leukopenia with moderate-to-marked heteropenia. In cases 1 and 4, a CBC performed 2 wk to 2 mo prior had been within normal limits. Cases 2 and 4 had toxic changes in the heterophils, and case 2 had reactive lymphocytes. Control bird 2 had a low-normal WBC count with mild heteropenia, and there were mild toxic changes in the heterophils.
Macroscopic findings
The birds were in a good nutritional state to overweight. All 6 affected birds had mildly to moderately enlarged spleens, 3 of which also had white-to-beige pinpoint foci. Four birds had mildly to moderately enlarged livers and 2 birds had mottled red bone marrows. The mucosa of the ventriculus was reddened in 2 affected birds. Additional findings included a mildly prominent keel, blood between liver lobe and sternum, and a reddened right lung lobe in case 1; and generalized pallor with marked hemorrhage along the right humerus in case 2. The anticoagulant panel on case 2 was negative. Control case 1 had widespread deposition of urate crystals in the kidneys and on the liver and heart. Control case 2 had degenerative lesions of the stifle joints. Neither of the control cases had changes in liver, spleen, bone marrow, intestine, or ventriculus, excluding the widespread urate deposition in control case 1.
Microscopic findings
Histologically, marked multifocal necrosis of the tibiotarsal bone marrow was present in 5 of the 6 affected birds (Fig. 1A), and was characterized by replacement of adipose tissue by eosinophilic, granular debris and/or eosinophilic, homogeneous, proteinaceous material. Hematopoietic precursor cells were hypereosinophilic and shrunken, with pyknotic or karyorrhectic nuclei. Most of the affected hematopoietic precursor cells appeared to be of the myeloid lineage. The sixth affected hawk had marked myeloid hyperplasia and erythroid hypoplasia without evidence of bone marrow necrosis. In this bird, the bone marrow was ~80% cellular, with ~90% of the hematopoietic precursor cells of myeloid origin, including small-to-moderate numbers of immature granulocytic precursor cells, and with only rare, scattered erythroid hematopoietic precursor cells.
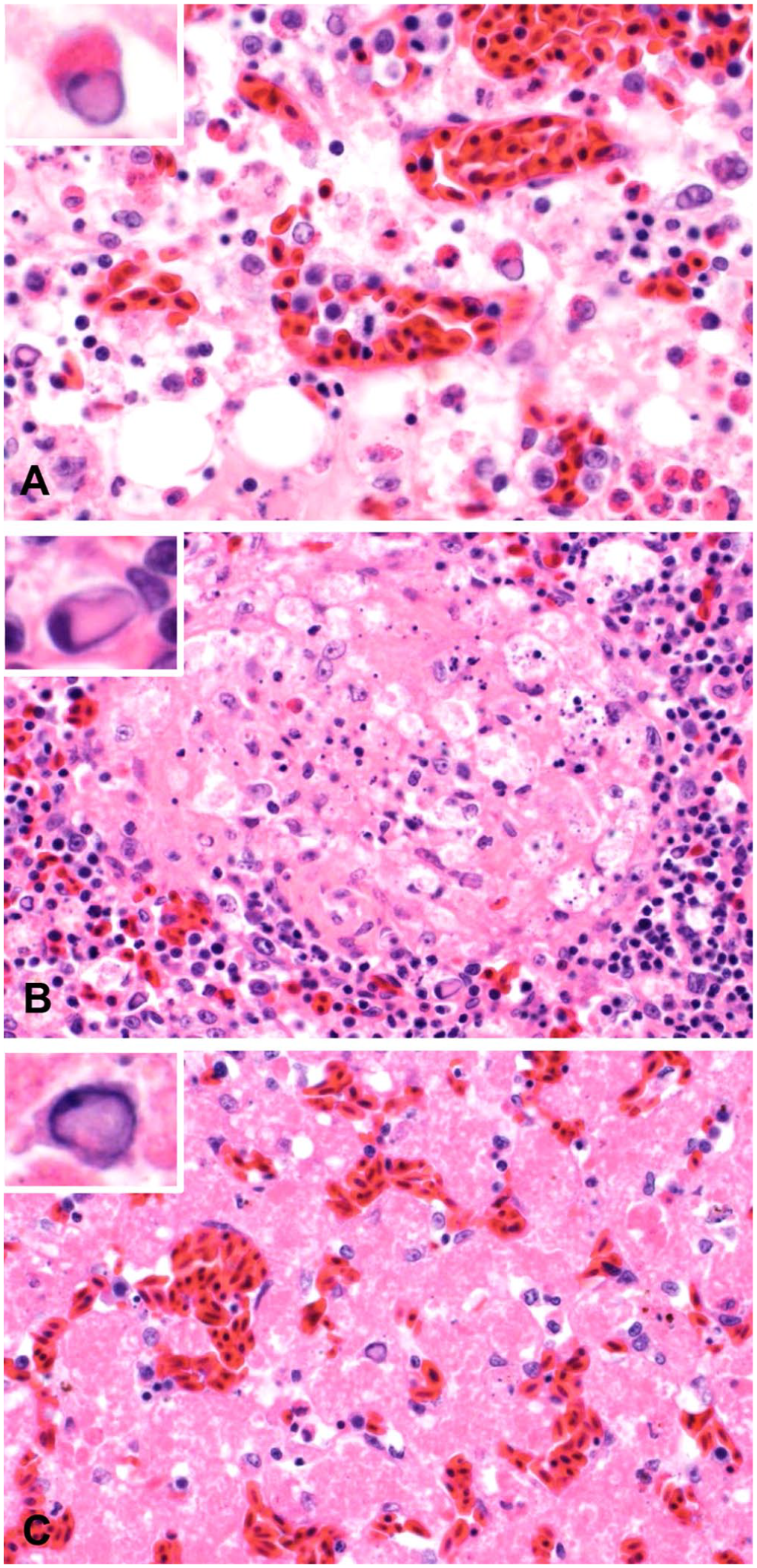
Bone marrow, spleen, and liver necrosis in red-tailed hawks. H&E.
The spleen of 3 of the affected 6 hawks had multifocal necrosis (Fig. 1B), predominantly located near sheathed capillaries and/or within the white pulp. The necrotic foci were characterized by eosinophilic granular debris, admixed with pyknotic or karyorrhectic nuclei. In addition, lymphocytolysis was widespread in the periarteriolar lymphatic sheath and follicles of the white pulp. Of the remaining spleens without splenic necrosis, 2 had prominent reticular cells and increased numbers of histiocytes in the red pulp, which was interpreted as red pulp hyperplasia. In the other spleen, the ellipsoids were surrounded by 4–6 layers of foamy histiocytes, interpreted as splenic histiocytosis.
Multifocal, mild-to-marked necrosis was present in the livers of 4 of the affected hawks (Fig. 1C). The necrosis varied from rare individual cell necrosis to variably sized clusters of hepatocytes. The necrotic hepatocytes were hypereosinophilic and shrunken, with absence of nuclei or had pyknotic nuclei. The hepatic cords were collapsed in severely affected areas. In 1 of the 6 livers, hepatocytes contained discrete, optically clear, cytoplasmic vacuoles (consistent with lipid) multifocally.
The 2 bursas of Fabricius examined histologically had marked lymphoid depletion with lymphocytolysis, and 1 had epithelial necrosis with replacement of the epithelium by eosinophilic, granular debris and pyknotic nuclei. Another hawk had mild necrotizing ventriculitis, characterized by small foci of loss of the superficial epithelium and replacement of the epithelium by a small amount of granular eosinophilic necrotic debris. A small number of degenerate inflammatory cells, presumably predominantly heterophils, were scattered within the superficial ventricular epithelium and deep lamina propria. Beneath the affected areas, many small-caliber blood vessels of the lamina propria were occluded by thrombi.
Case 3 had colonies of intravascular coccobacilli in multiple organs, and case 4 had intravascular and extravascular colonies of large cocci in multiple organs, indicating septicemia. The large cocci in case 4 were gram-negative and acid-fast positive. The organisms did not stain with GMS stain and were negative with the PAS reaction. Case 1 had coccobacilli within the bone marrow. Two of the 6 affected hawks had mild-to-moderate pulmonary hemorrhage, one of which also had a focally extensive area of pleuritis overlying an area of hemorrhage, characterized by moderate infiltrates of histiocytes and heterophils admixed with fibrin deposition and granular necrotic debris. The same hawk had marked hemorrhage surrounding the right humerus.
In all affected cases, the nuclei of occasional presumed hematopoietic precursor cells (bone marrow), and reticular cells and macrophages (spleen), were expanded by 4.5–9-µm, pale-eosinophilic to amphophilic INIBs. The chromatin of affected nuclei was marginated. Similar INIBs were seen in a few hepatocytes and Kupffer cells in 5 of the 6 affected cases. In most cases, these INIBs were most frequent in the bone marrow, followed by the spleen, with only rare INIBs in the liver (Table 2). In addition, rare, similar INIBs were seen within leukocytes within the ventriculus of the one hawk with histologic evidence of ventriculitis, and within intravascular leukocytes in the lung and kidney of another affected case. Differential identities for the INIBs included adenovirus, herpesvirus, polyomavirus, parvovirus, or nonviral pseudoinclusions.
Histologic lesions and results of PCR and electron microscopy (EM).
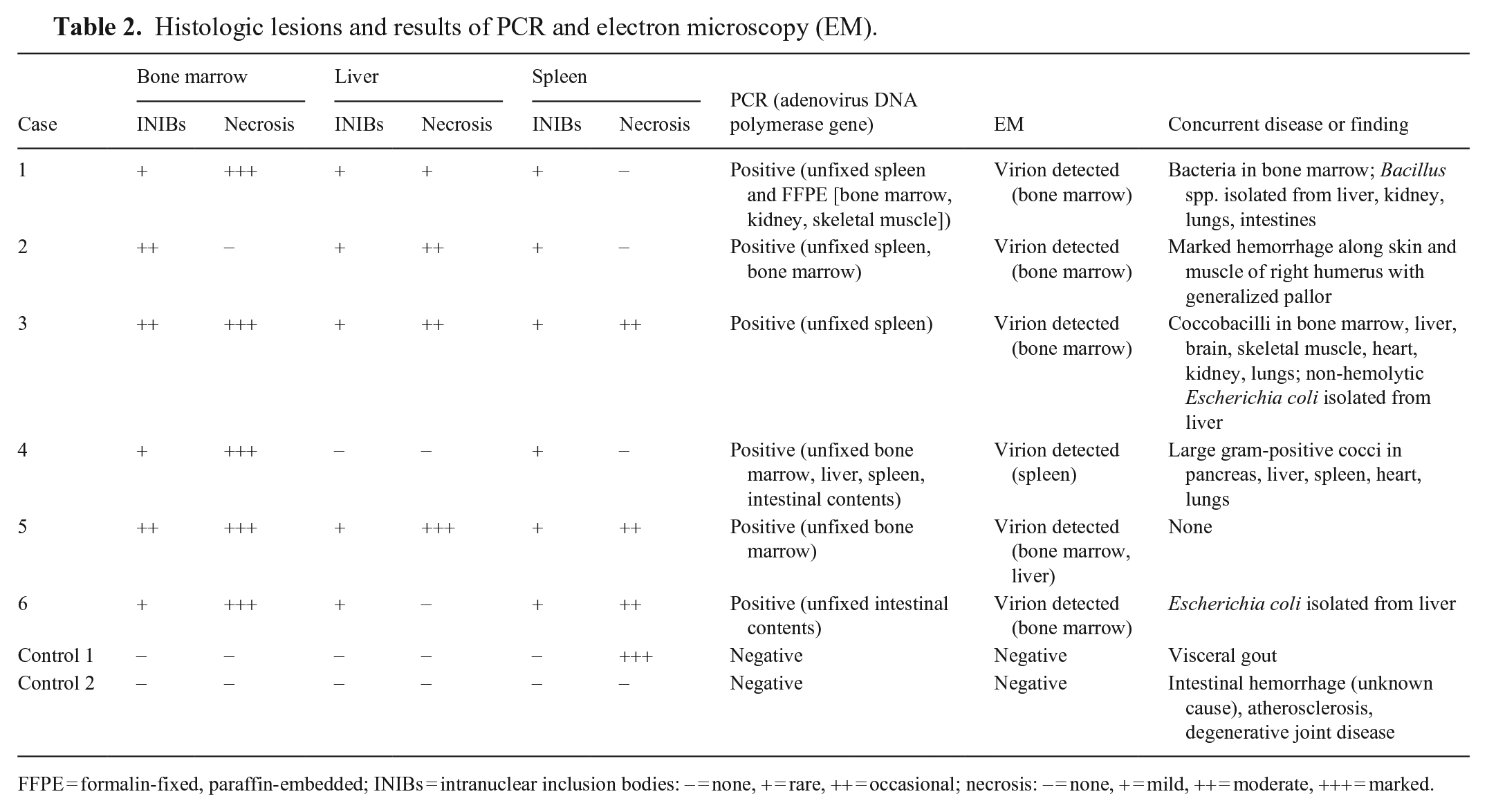
FFPE = formalin-fixed, paraffin-embedded; INIBs = intranuclear inclusion bodies: − = none, + = rare, ++ = occasional; necrosis: − = none, + = mild, ++ = moderate, +++ = marked.
Control case 1 had marked deposition of urate crystals, with gouty tophi in various organs, including the kidney, spleen, lungs, bone marrow, liver, pancreas, and heart. Urate deposition was associated with marked necrosis in the spleen and mild necrosis in the myocardium. Control case 2 had mild aortic tunica intima degeneration and mineralization, and hemorrhage within the small intestine. INIBs were not identified in any of the examined tissues of the control cases.
Transmission electron microscopy
On negative-contrast EM, nonenveloped, icosahedral, hexagonal viral particles were found in the bone marrow in 5 of the 6 (Fig. 2A), the spleen in 1 of the 5, and liver of 1 of the 5 examined affected cases (Table 2). Viral particles were sparse. The viral particles were 82.4–95.4 nm in diameter (n = 9; SD = 4.8). Vertex fibers were not observed. No viral particles were identified on negative contrast in the intestinal contents of the submitted cases, nor in any of the tissues from the control cases. Plastic-embedded tissues from the bone marrow and liver from case 5 were examined by transmission EM. Infrequently, bone marrow reticular cells and hepatocytes had evidence of intranuclear virus replication and assembly complexes that were composed of immature and mature virions arranged in loose aggregates (Fig. 2B). Granular-to-fibrillary viroplasm and assembled particles distended the nucleus, and replaced and displaced the chromatin to the nuclear margin. Paracrystalline arrays were not observed. Associated degenerative changes in the infected cells included swelling of the cisternal system, fat and glycogen depletion, and chromatin lysis. The mature virions were 72.7–92.1 nm in diameter (n = 21; SD = 4.8) in the plastic-embedded material. These EM findings were most consistent with adenoviral particles.
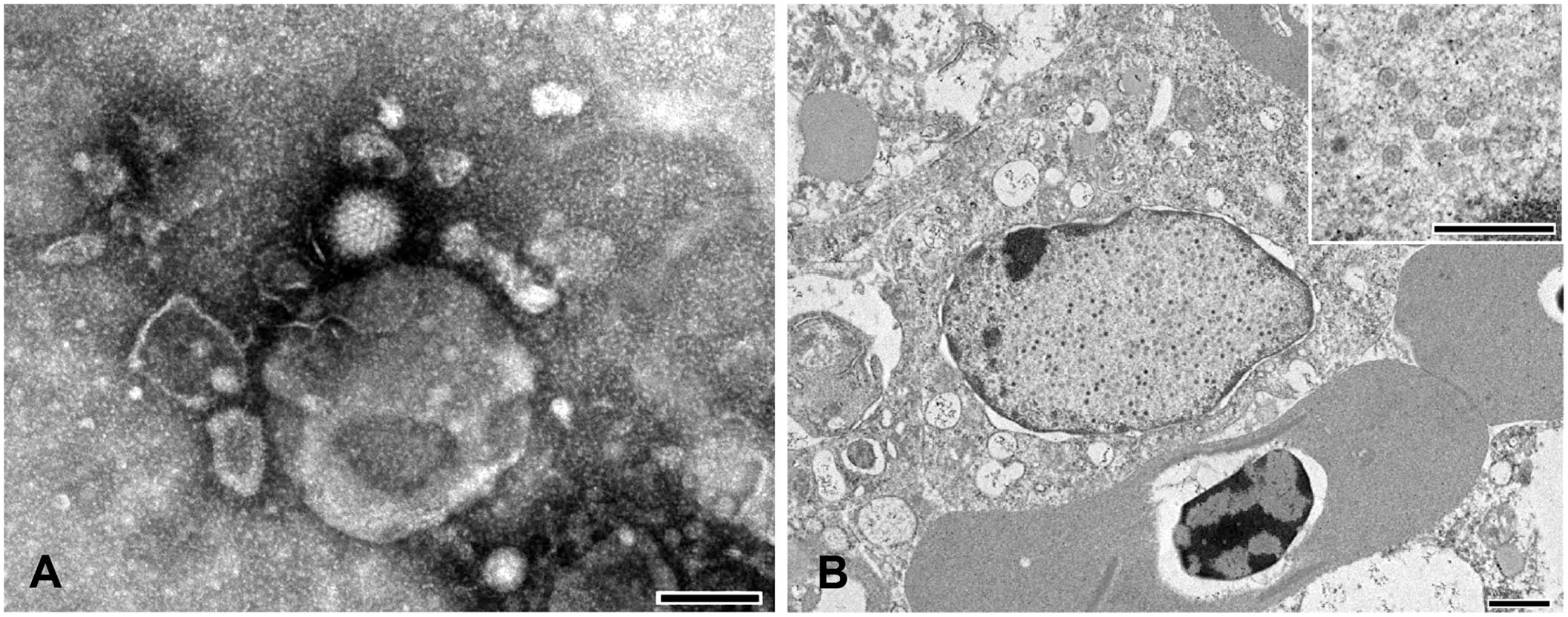
Electron microscopy of a red-tailed hawk.
Bacteriology
Bacillus spp. were cultured in large numbers (4+) from various organs in case 1; moderate numbers (3+) of Escherichia coli were cultured from the liver of case 3. Large gram-positive coccoid bacteria that were not further identifiable were isolated in large numbers (4+) from the liver of case 4. Escherichia spp. were isolated from the liver of case 6 in small numbers (2+), but bacteria were not seen histologically; hence, the cultured bacteria were considered to be postmortem contamination. The remaining aerobic culture results yielded no growth or no significant growth, and salmonella culture did not yield any growth of Salmonella species in any of the submitted cases.
Virology
Viral isolation by egg inoculation was unsuccessful in both cases 1 and 2.
PCR and sequence analysis
Based on the EM features, adenoviral infection was strongly suspected, and PCR was selected to confirm the presence of adenovirus. The bone marrow from 3 of 5 affected hawks, liver from 1 of 3, spleen from 4 of 6, and intestinal contents from 2 of 3 cases were positive for adenovirus by PCR assay (Table 2). The FFPE specimen from case 1 was also positive for adenovirus by PCR assay. All tissues from the control cases were negative by PCR. The nucleotide sequences (GenBank OM522337–OM522342) were blasted in NCBI to confirm that these are adenovirus sequences. All study sequences were >99% identical to one another and showed highest nucleotide identity (90%) with RAdV1 (EU715130) sequenced from a Harris hawk examined in the United Kingdom (Fig. 3; Table 3). Based on phylogenetic analysis, our sequences clustered as one group within the siadenoviruses. The sequences have common ancestors with adenoviruses from raptors, turkeys, south polar skuas, psittacines, budgerigars, and other siadenoviruses.

Phylogenetic tree constructed by comparing 857 nucleotides of the DNA polymerase gene of siadenoviruses using the maximum-likelihood method based on the Hasegawa–Kishino–Yano model with 1,000 bootstraps.
The percent nucleotide identity of the study sequences compared with the various avian adenoviruses.
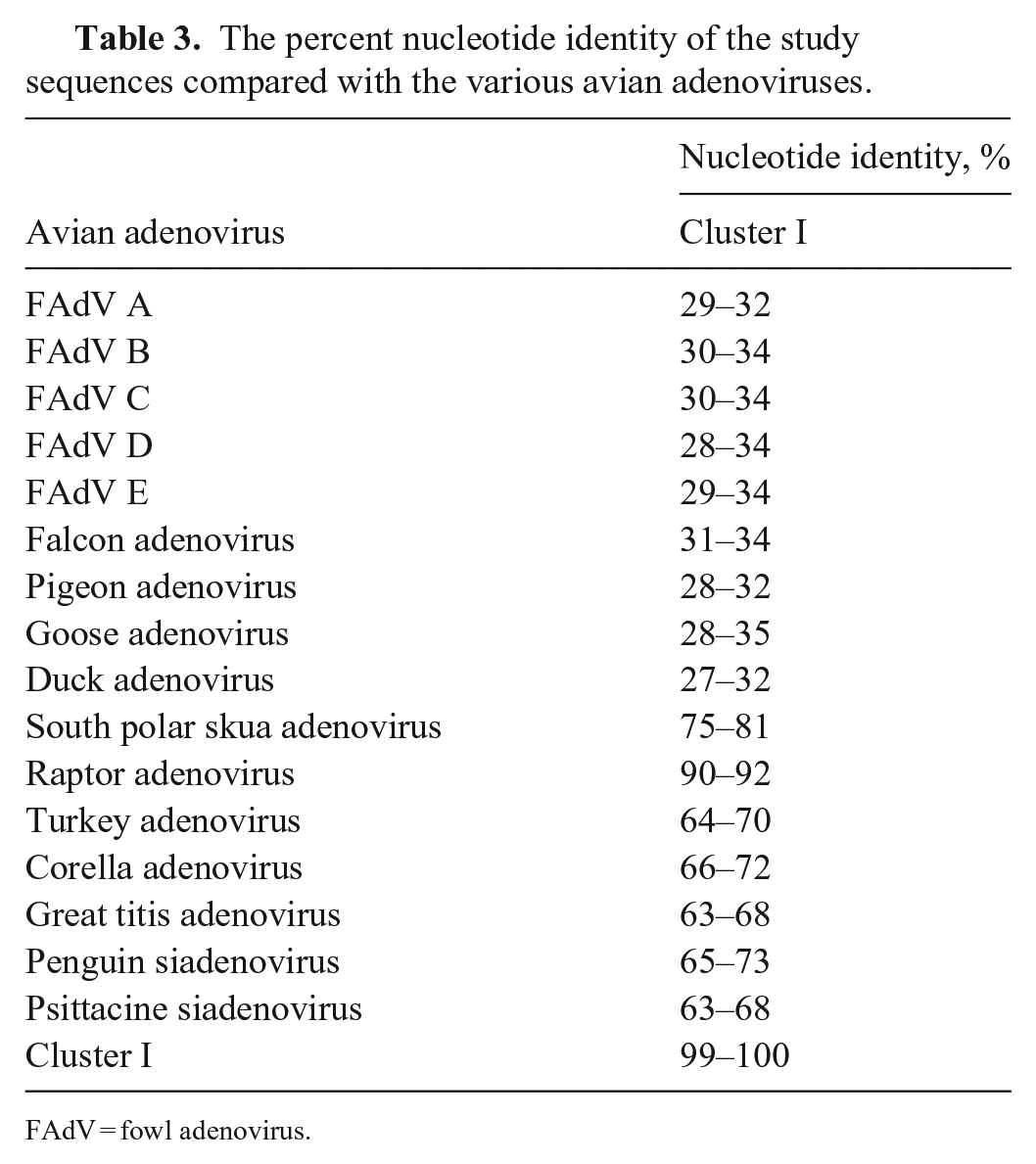
FAdV = fowl adenovirus.
Discussion
Our diagnoses of adenoviral infections were based on histologic features, EM, and the presence of adenoviral DNA by PCR assay in all cases. Virus isolation in the 2 attempted cases was unsuccessful, likely because of the unavailability of suitable cell lines. For example, FaAdV1 requires a specific cell line (peregrine falcon embryo fibroblasts) for successful isolation, and isolation for RAdV1 has been unsuccessful given that it requires permissive cell lines.21,27 The identified virus in our cases belongs to the genus Siadenovirus, and hence is different from FaAdV1 of the Aviadenovirus genus.
A viral infection, such as adenovirus, herpesvirus, polyomavirus, or parvovirus, was suspected initially based on the presence of INIBs in cells of the liver, spleen, and bone marrow, although the presence of nonviral pseudoinclusions could not be excluded by light microscopy alone. In most of the published raptor cases, the adenoviral inclusion bodies were typically described as large, basophilic to occasionally eosinophilic inclusion bodies that occupied the entire nucleus, or were surrounded by a clear halo,7,8,20,26,30,31,34,36,37,39 which differed from the INIBs observed in our hawks. In addition, in the published raptor cases, the INIBs were reported most commonly, and often seen in large numbers, in the liver.7,8,20,36 In our case series, in most of the birds, the liver had rare INIBs compared to the bone marrow. Given the distribution and appearance of these INIBs, a definitive identification of adenovirus could not be reached confidently on light microscopy.
EM can aid in understanding viral structures and assembly, and this knowledge can be used to identify the virus family. 28 The transmission EM findings confirmed that the INIBs were composed of viral particles. Despite the lack of paracrystalline arrays, adenovirus was strongly suspected based on the size, lack of outer envelope, structural features, and location within the host cells. Polyomaviruses and parvoviruses are smaller (40–45 nm and 23–28 nm, respectively), and thus were excluded.6,25 Herpesviral particles are typically larger (150–200 nm), and virus assembly takes place within both the nucleus and cytoplasm.11,24,28 Although nonenveloped herpesviral particles can be found within the nucleus, enveloped herpesviral particles are seen in the nuclear envelope and within the cytosol.24,28 Electron-dense viral tegument proteins and various membrane vesicles are also seen within the cytosol with herpesviral infection, which were absent in our study.24,28 In contrast, adenoviruses are nonenveloped, 70–90 nm, and virus assembly occurs within the nucleus, although viral particles can also be found within endosomes and sporadically within the cytosol.12,17 Hence adenovirus was considered most likely, and this was confirmed by an adenoviral PCR assay. The morphogenesis of adenoviruses is incompletely understood, 5 and further investigation is required to determine whether the morphogenesis may differ depending on the adenoviral species and/or strains, and/or host species, and/or host cell type, which may explain the reduced numbers of virions assembled, and resulting lack of paracrystalline arrays in our study. For example, variation at the ultrastructural level has been reported in viral particle distribution and arrangement between different FAdV strains. 1 In other viruses, such as certain flaviviruses, variation in the virus maturation process, as reflected by variation in viral particle numbers, has been reported with cell lines from different host species, and with different strains of the same virus (e.g., dengue virus). 32
The polymerase gene is the most conserved gene among adenoviruses and hence was our principal target for the molecular analysis.21,39 The study sequences were 99–100% identical, indicating that a similar strain was circulating over the period of 2014–2019. The study sequences are divergent from previously related sequences, with 90% identity with the most closely related RAdV1 sequence. Further analysis of other genes, such as the hexon gene, or whole-genome sequencing, would be necessary to characterize the viruses in more detail.
Adenoviruses are thought to be transmitted primarily by the fecal-oral route,8,10,37 although both oral and respiratory transmission have been suggested for siadenoviruses.18,39 However, there has been no clear pathogenesis or consistent source of viral INIBs in the lungs or gastrointestinal tract.18,39 The source of the adenoviruses could not be determined in our case series. In previous cases involving raptors, various sources have been suggested depending on the adenoviral genera involved, including cross-species transmission through feed or via contact with asymptomatic carriers.7,8,21,22,26,31,37 In cases involving FaAdV1 outbreaks, feed was considered an unlikely source because these adenoviruses were distant phylogenetically from FAdVs.7,31,37 Instead, asymptomatic peregrine falcons were suggested to be the carriers or source of the FaAdV1, given the high seroprevalence in both wild and captive peregrine falcons within a breeding facility in an outbreak compared to other raptors, despite many of them being asymptomatic, suggesting that the virus was endemic in this species. 27 Another study looked at the prevalence of antibodies against FAdV (formerly FAdV group I) in raptors of Berlin and Germany, and found only common buzzards to be positive. 10
The seroprevalence of adenoviruses in the raptors admitted to TRC is unknown. All of the hawks in our study were admitted for trauma and did not have any indication of a viral infection at intake. It is possible that the hawks acquired the infection at TRC. Alternatively, the hawks may have been asymptomatic carriers at intake, and the stress associated with being injured and in captivity resulted in disease development. TRC occasionally feeds day-old chicks to their raptors. These chicks were not tested for virus, but the chicks are considered to be an unlikely source given that the study sequences were distant phylogenetically from FAdVs. We detected adenoviral particles in the intestinal contents of 2 subject birds by PCR, indicating possible shedding through the alimentary system. Adenoviruses are relatively resistant to inactivation and can persist in the environment, hence environment and fomites could not be excluded as possible sources.4,9 In our case series, most of the affected birds were hatch years (<~6-mo-old), suggesting a possible role of an immature immune system. However, in the reported raptor cases, affected age groups varied from nestlings to adults.7,8,20,30,31,34,36,37,39
Whether siadenoviruses are primary pathogens, a contributing disease factor (e.g., further to immunosuppression), or merely an incidental finding, has been a matter of debate. 18 Disease manifestation is likely largely dependent on environmental and host factors, as well as coinfections. 18 Most natural infections of adenoviruses in birds are subclinical or have mild, transient signs, and significant disease is often only seen with coinfection with other viral or bacterial pathogens, with toxin exposure, or in immunocompromised individuals.31,39 The clinical significance of the adenoviral infection in our hawks is uncertain because the clinical signs and histologic lesions varied to some degree. Similar to the reported cases of adenoviral infection in raptors, most of our affected birds had hepatic and/or splenic necrosis associated with INIBs. In addition, most had bone marrow necrosis, all had INIBs within the bone marrow, and INIBs were more numerous in the bone marrow in 3 of 6 cases than in other tissues. The marked leukopenia and moderate-to-marked heteropenia along with toxic changes to the heterophils seen in the hawks within 24 h of death was likely secondary to bone marrow necrosis. Not all animals with bone marrow necrosis developed evidence of sepsis, but cases 1, 3, and 4 had marked bone marrow necrosis and developed sepsis. Therefore, we speculate that the bone marrow necrosis impaired the birds’ immune response. In the birds with sepsis, it was difficult to determine if the necrosis was the result of adenoviral infection, sepsis, or both. However, in case 5, there was no evidence of concurrent bacterial disease; hence, at least in this bird, adenoviral infection was considered to be the cause of the hepatic, bone marrow, and splenic necrosis, and subsequent death. In addition, although E. coli was cultured from the liver of case 6, the animal did not have any histologic lesions supportive of bacterial hepatitis, and hence the adenoviral infection likely caused its bone marrow and splenic necrosis.
Case 2 had massive hemorrhage into the soft tissue along the right humerus. Exposure to anticoagulant rodenticide from ingestion of poisoned rodents is common in raptors 38 and was considered as a cause of the hemorrhage in this case; however, the anticoagulant panel was negative. Neither the cause of this hemorrhage nor the correlation with the adenoviral infection is known, especially given that significant hemorrhage was not detected in any other birds with adenoviral infection in our study.
In the reported raptor cases, either bone marrow was not examined or did not have lesions. In raptors, herpesviral infection is considered to be the most common viral cause of bone marrow necrosis. 38 In a case series involving a novel siadenovirus in Sulawesi tortoises, bone marrow necrosis and INIBs within the myeloid and stromal cells were common. 29 The authors speculated that the virus may be immunosuppressive; however, interestingly, although many of the tortoises in that study had coinfections with other pathogens, of the 20 tortoises with bone marrow necrosis, none had concurrent infections. These authors suggested immunosuppression might have involved multiple factors, such as malnutrition, stress, illness, and possibly adenoviral infection.
Our case series expands on the number of documented cases of hawks with siadenoviruses, suggesting that hawks may be more susceptible to adenoviruses than thought previously, and that siadenoviral infection can be clinically significant in hawk species. Histologic examination of bone marrow is important given that bone marrow necrosis was a common finding, and often the most severe lesion in affected hawks. EM was helpful in confirming the virus as adenovirus. However, given the scarcity of viral particles detectable on EM, PCR assay is recommended to confirm adenoviral infection in hawks with suggestive histologic lesions.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387221105240 – Supplemental material for Adenoviral infection in 5 red-tailed hawks and a broad-winged hawk
Supplemental material, sj-pdf-1-vdi-10.1177_10406387221105240 for Adenoviral infection in 5 red-tailed hawks and a broad-winged hawk by Emma H. Torii, Arno Wünschmann, Anibal G. Armién, Sunil K. Mor, Emma Chalupsky, Rahul Kumar and Michelle Willette in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the staff of the University of Minnesota Veterinary Diagnostic Laboratory Necropsy, Histology, Molecular, Bacteriology, Virology and Electron Microscopy sections. We thank Dean Muldoon for the electron microscopy preparations.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
