Abstract
Acute and, particularly, chronic copper exposures, along with defects in hepatic copper metabolism, altered excretion of copper, and/or nutritional imbalances between copper and other trace elements, can lead to hepatic accumulation of copper and primary copper toxicosis. There is interspecies variation in susceptibility to copper toxicosis, with sheep being the species most likely to develop this condition. Adult dairy goats and Boer crosses are generally considered resistant to chronic copper toxicosis, especially the hemolytic stage of this disease. The current report is rather unusual in that it describes instances of naturally occurring copper toxicosis with hemolysis and hemoglobinuric nephrosis in 3 adult Boer goats. In 2 of these goats, a possible source of excessive dietary copper was investigated but not definitively identified. In the third goat, the etiologic factors associated with the copper toxicosis were not determined. It appears that mature Boer goats are susceptible to the hemolytic stage of chronic copper toxicosis, which was not observed in a recent, large-scale copper intoxication involving lactating dairy goats. Copper analyses on both liver and kidney samples were necessary to confirm the diagnosis of copper toxicosis in all 3 goats. All feedstuffs associated with instances of copper toxicosis should be analyzed for iron, molybdenum, sulphur, and zinc as well as copper to determine what nutritional factors are contributing to the pathogenesis of this disease. Consideration also should be given to the ingestion of hepatotoxic plants and other toxic exposures, which could predispose an animal to secondary chronic copper toxicosis.
Two forms of primary copper toxicosis, acute and chronic, are generally described in the veterinary toxicology literature and refer to the duration of copper exposure, as opposed to the onset of clinical signs. 13,17 Although the distinction between these 2 forms of primary copper intoxication remains somewhat arbitrary, acute copper toxicosis is thought to be less common than chronic copper toxicosis 17,21 and is most often associated with the rapid onset of severe gastroenteritis, characterized by abdominal pain, diarrhea, emesis, anorexia, dehydration, and shock, following ingestion or parenteral administration of excessive copper in anthelmintics, footbaths, mineral supplements, or improperly formulated rations. 16,17,19,21,24 If animals survive the initial clinical signs, death associated with hepatic necrosis, hemolysis, and hemoglobinuria, all of which are commonly associated with chronic copper toxicosis, can occur within a few days of exposure. 16,18,19,21,24
Defects in hepatic metabolism and altered biliary and urinary excretion of copper (frequently related to high dietary intake of copper) and/or nutritional imbalances between copper and other trace elements can all result in massive hepatic stores of copper over time and lead to what has been referred to as chronic copper toxicosis, based on the suspected duration of copper exposure. 7,13,17,21 Hepatic accumulation of copper eventually causes hepatocellular damage during the prehemolytic stage of chronic copper toxicosis. The prehemolytic stage, which is frequently subclinical, is followed by what often appears to be a sudden onset of acute hepatocellular degeneration and necrosis associated with copper-induced oxidative damage. 7,13,14,24,26 Although chronic primary copper toxicosis generally occurs after multiple exposures to copper over a prolonged period of time ranging from weeks to even months, cases of copper intoxication in calves with hepatic and, in some instances, renal changes typical of chronic copper toxicosis have been reported several weeks after a single administration of time-released boluses containing copper oxide wires as a neonate. 8,25 In addition, exposure to hepatotoxicants, including toxic plants, can also predispose animals to hepatic copper accumulation over time and cause what has been referred to as secondary chronic copper toxicosis. 12,15,21
In contrast to acute copper intoxication, which is initially characterized by gastroenteritis, the prehemolytic stage of chronic copper toxicosis often presents clinically as a hepatopathy 8,13,25,26 and, especially in the case of sheep, remains subclinical in the prehemolytic stage until large quantities of copper are suddenly released from hepatic stores. A stressful event, such as transportation, pregnancy, lactation, strenuous exercise and/or handling, or disease and malnutrition, can apparently trigger the breakdown of copper-containing lysosomes, resulting in severe hepatocellular disease and release of copper from the liver. 2,7,19,21,26 As can happen in acute copper toxicosis, the sudden increase in blood copper concentrations causes formation of methemoglobin and Heinz bodies as well as lipid peroxidation of erythrocyte membranes and intravascular hemolysis of damaged erythrocytes 7,17 Severe, acute anemia with subsequent hemoglobinuric nephrosis frequently develops, 7,13,18,21 and affected animals usually exhibit a sudden onset of depression, anorexia, thirst, rumen stasis, weakness, recumbency, hemoglobinuria, icterus and, potentially, death within 1–2 days. 7,13,17–19,21,24
It is thought that sheep are predisposed to chronic copper toxicosis because of their reduced biliary and urinary excretion of copper, 17,24 the distribution of zinc-and copper-binding proteins in the liver, 21 and the relatively small difference between the copper concentrations reported to be adequate for sheep rations (5–10 mg/kg, 7–11 mg/kg, or 10–20 mg/kg on a dry matter basis, depending on the reference) and those dietary copper concentrations considered to be potentially toxic (>15, 20, or 30 mg/kg on a dry matter basis). 13,14,18 In contrast, cattle, horses, swine, and poultry tend to be more resistant to copper accumulation and chronic copper toxicosis, with maximum tolerable dry matter concentrations of dietary copper being approximately 50 mg/kg for cattle and horses, 250 mg/kg for swine, and 300–500 mg/kg for poultry. 13 In a previous study, ponies were even reported to tolerate dietary copper concentrations approaching 800 mg/kg for 6 months. 20 However, histopathologic examinations of the livers were not apparently performed, and it is extremely important to recognize that copper bioavailability and dietary concentrations of molybdenum and sulphur also play important roles in the pathogenesis of chronic copper toxicosis. 3,6,13
Goats, especially adults, are generally considered to be resistant to chronic copper toxicosis 7,14,21,23 and have been shown to tolerate dietary concentrations of copper (10–23 mg/kg or ≤40 mg/kg, depending on the reference) associated with chronic copper toxicosis in sheep. 14,18,22 However, as with sheep, susceptibility to chronic copper toxicosis in goats varies with breed. 3,14,21 There are a limited number of reports regarding chronic copper intoxication in goats, 1,5,21–23 and there is no uniformly accepted maximum tolerable dietary concentration of copper reported for goats. 14,18 Hemolytic crisis with hemoglobinuria has been reported in goats in conjunction with acute and experimental copper exposures, 1,18,21,23,28 especially those involving preruminant juveniles, 21 but these findings appear to be of much less common occurrence in adults of this species than in sheep. 5,21 Hemolysis and hemoglobinuric nephrosis were suspected in preruminant Angora goats chronically exposed to a milk replacer intended for calves and experimentally induced chronic caprine copper toxicoses, where goats received in excess of 20 mg of copper sulfate per kg of body weight per day. 1 It should also be noted that, in one of those experiments, 20 mg of copper sulfate per kg of body weight was given twice daily, and copper-induced hemolytic crisis was observed in only 1 of 3 goats. 21,23 In contrast, a thoroughly investigated instance of naturally occurring chronic copper toxicosis in lactating dairy goats was recently reported with different clinical and pathological findings. 5 Although a definitive source of excessive copper was identified in the incident, the occurrence of hepatopathies in the complete absence of hemolytic crises or hemoglobin-induced nephroses was considered notable. 5 Although some references have suggested that meat goats, especially adults, appear to be more resistant to copper intoxication than other ruminants, especially young calves and sheep, 14,29 the Veterinary Medical Diagnostic Laboratory (VMDL) at the University of Missouri (Columbia, MO) has received several anecdotal but unconfirmed reports of suspected copper intoxication in Boer goats over the last several years. A recently published abstract reported copper toxicosis with hemolysis in 3 Boer goat kids fed a high copper-containing mineral mix (ACVP Annual Meeting 2008, Vet Pathol 45:740, Abstract 35).
The present report is rather unusual in that it describes naturally occurring field incidents of copper toxicosis with hemolysis and hemoglobinuric nephrosis in 3 adult Boer goats. A review of these cases provides both gross and histopathologic findings likely to be observed with chronic copper toxicosis in this breed of goat, information regarding the susceptibility of Boer goats to the hemolytic stage of chronic copper toxicosis, and additional factors worthy of consideration when investigating field cases of chronic copper toxicosis in goats.
Between mid-August and mid-October of 2007, 3 adult Boer goats were necropsied at the VMDL for what appeared to be death following a relatively short period of clinical disease. Two of these goats were presented to the Food Animal Clinic of the Veterinary Medical Teaching Hospital (VMTH; University of Missouri, Columbia, MO) for inappetence and lethargy. Goats 1 and 2 were intact females, approximately 1 year of age, and weighed 43.1 and 52 kg, respectively. They were from a group of 9 goats having access to cattle pasture. Upon presentation, goat 1 was hypothermic and exhibiting opisthotonos, dorsomedial strabismus, bradycardia, and shallow breathing. Goat 2 was presented for a suspected urinary tract infection after the owner noted red urine the previous week. The goat was moribund, anuric, and severely anemic (hematocrit of 10%, reference [ref.] interval: 23–36%) with normal plasma protein (6.8g/dl, 6.0–7.5 g/dl) at the time of admission to the VMTH. Aspartate aminotransferase activity (430 U/l, ref. interval: 46–161 U/l), gamma-glutamyltransferase activity (241 U/l, ref. interval: 34–65 U/l), and total bilirubin concentration (0.8 mg/dl, ref. interval: 0–0.1 mg/dl), as well as urea nitrogen (325 mg/dl, ref. interval: 10–20 mg/dl) and creatinine (24.5 mg/dl, ref. interval: 1.0–1.8 mg/dl), were markedly elevated. Goat 1 was euthanized, and goat 2 died in the clinic later that night. Goat 3 was an intact male, approximately 2.5 years of age, from another goatherd consisting of 27 individuals, and he was presented to another veterinary clinic with a history of diarrhea/soft feces and icterus, prior to his death. All 3 goats were submitted to the VMDL for postmortem examinations.
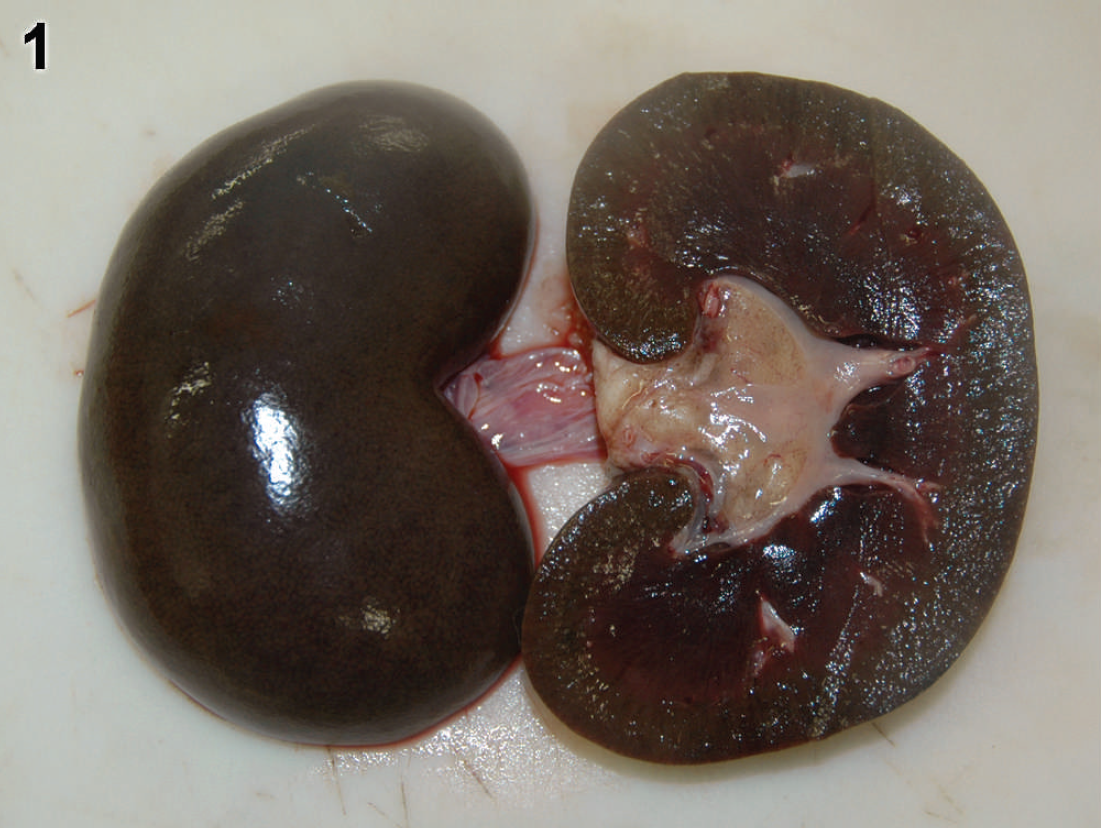
Kidney from adult Boer goat 2 with a “gunmetal”-colored appearance of both the subcapsular and cut surfaces characteristic of chronic copper toxicosis.

Liver from adult Boer goat 1 with chronic copper toxicity. Note centrilobular, vacuolar degeneration and neighboring hepatocytes and Kupffer cells containing intracytoplasmic granules (arrows) on hematoxylin and eosin (

Kidney from adult Boer goat 2 with chronic copper toxicity. Note renal tubules dilated with hemoglobin casts and tubular necrosis. Hematoxylin and eosin stain. Bar = 50 μm.
At necropsy, there was no evidence of gastroenteritis in any of the 3 goats, and the livers of goats 1 and 2 were mildly yellow, while the liver of goat 3 was bright yellow-orange with soft texture and rounded margins. The gallbladders were distended with bile in goats 1 and 3, and goat 3 had extremely icteric mucous membranes, and subcutaneous and adipose tissues. Goats 1 and 2 had pale pink mucous membranes, and all 3 goats had diffusely dark brown to black (“gunmetal”) colored kidneys (Fig. 1). The urinary bladder of goats 2 and 3 contained small quantities of dark red-brown urine, while the urinary bladder was empty in goat 1.
Copper concentrations in collected biological samples (mg/kg).*
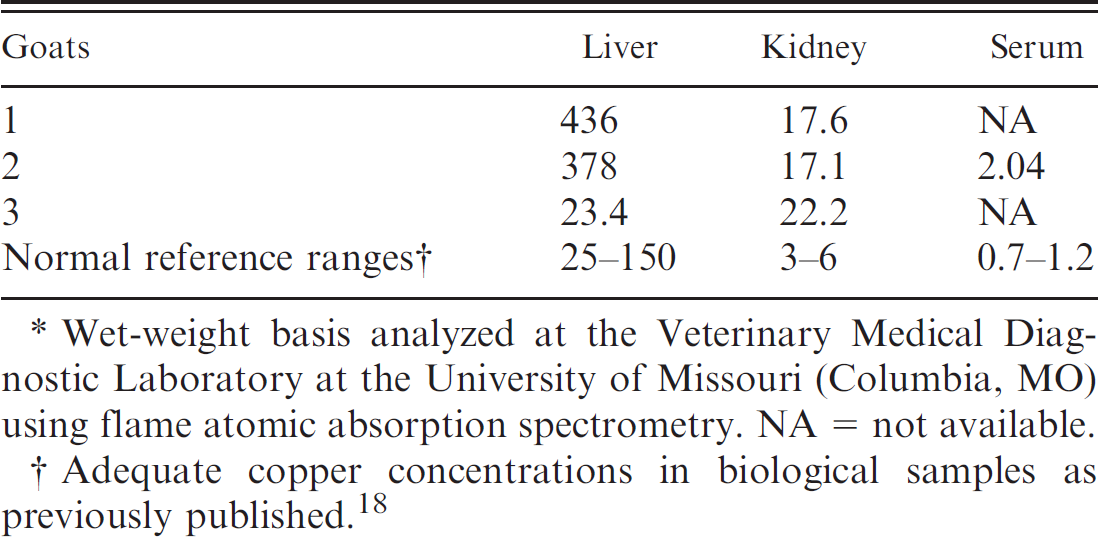
Wet-weight basis analyzed at the Veterinary Medical Diagnostic Laboratory at the University of Missouri (Columbia, MO) using flame atomic absorption spectrometry. NA = not available.
Adequate copper concentrations in biological samples as previously published. 18
On histopathologic examination, no significant microscopic lesions were observed in sections of the gastrointestinal tract from any of the 3 goats. There was moderate, centrilobular, vacuolar degeneration and necrosis of hepatocytes in the livers from goats 1 and 2 (Fig. 2a), while the hepatocellular degeneration and necrosis in goat 3 were much more diffuse, with individualization of the hepatocytes. In all 3 goats, mild to moderate numbers of Kupffer cells and hepatocytes in the centrilobular areas contained intracytoplasmic yellow–gray granules, which stained positive with rhodanine, a copper stain (Fig. 2b), and there was evidence of bile stasis and extramedullary hematopoiesis. The kidneys of all 3 of the goats had large numbers of cortical and medullary renal tubules dilated with hemoglobin casts as well as some protein casts and acute necrosis of proximal convoluted epithelium (Fig. 3). Goat 1 had multiple, scattered renal tubules containing numerous hyaline droplets in the renal epithelial cells and scattered aggregates of extramedullary hematopoiesis, while both goats 1 and 2 had moderate to large numbers of renal tubules with brown intracytoplasmic granules, which stained positive with rhodanine.
Copper, iron, molybdenum, sulfur, and zinc concentrations in feed samples (mg/kg).*
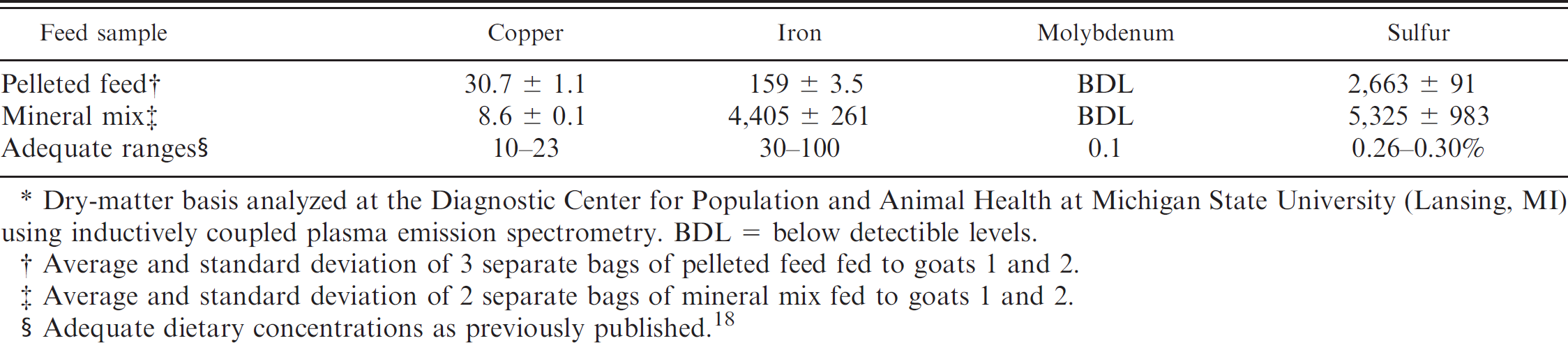
Dry-matter basis analyzed at the Diagnostic Center for Population and Animal Health at Michigan State University (Lansing, MI) using inductively coupled plasma emission spectrometry. BDL = below detectible levels.
Average and standard deviation of 3 separate bags of pelleted feed fed to goats 1 and 2.
Average and standard deviation of 2 separate bags of mineral mix fed to goats 1 and 2.
Adequate dietary concentrations as previously published. 18
Based on the history and clinical signs, as well as the gross necropsy and clinical pathology results, chronic copper toxicosis was suspected in the 3 goats. Diagnosis was corroborated by the observed histopathologic findings, all of which were consistent with the pathogenesis of chronic copper toxicosis as well as the absence of severe gastroenteritis, which would have suggested a larger and more acute copper exposure. Liver damage is evidenced in live animals by elevated plasma liver enzyme activities and total bilirubin concentration, as noted in goat 2. The centrilobular to panlobular hepatic degeneration and necrosis with rhodanine-stained granules described in the 3 goats are typical of copper toxicosis 5,13,25,26 and can be exacerbated by the hypoxia associated with hemolytic anemia. 24 The bile stasis observed histologically in all 3 cases was most likely a sequelum to hemolysis, anorexia, and/or illness, but also could have contributed to the excessive hepatic copper accumulation because of impaired biliary excretion of copper. 24 Icterus, a common clinical sign of copper toxicosis in ruminants, 26 was only observed in goat 3. Extramedullary hematopoiesis was noted in the livers of all 3 goats and the kidney from goat 1 and can be observed in animals after a hemolytic crisis. 7
The decreased hematocrit in goat 2 and the contents of the urinary bladders of both goats 2 and 3 were consistent with hemolysis, and all 3 of the goats were diagnosed with hemoglobinuric nephrosis, a sequelum to hemolysis that is a common finding in chronic copper toxicosis in sheep and, to a lesser extent, other domestic animals. 13,18,21 In addition to hemoglobin, which can contribute to renal failure by mechanisms still subject to discussion, several other factors likely play a role in renal disease in the 3 goats. 4,11 High levels of proteinaceous-free hemoglobin being processed by renal tubular epithelium could explain the hyaline droplets observed in the kidney sections from goat 1. The hypoxia caused by the severe acute anemia and copper-induced oxidative damage could have also played a role in the pathogenesis of the acute tubular necrosis observed in the 3 goats.
The tentative diagnosis of copper toxicosis was confirmed by the results of the analyses summarized in Table 1, which were performed by flame atomic absorption spectrometry. Liver, kidney, and serum copper concentrations greater than 230 mg/kg, 12 mg/kg, and 1.2 mg/kg, on wet weight bases, respectively, have been reported to be consistent with copper toxicosis in goats. 5,18 Elevated serum or plasma copper concentrations similar to those in goat 2 have been reported to be a poor indicator of the prehemolytic stage of chronic copper toxicosis 6,19,29 ; however, dairy goats showing clinical signs of copper toxicosis with severe hepatic disease had very high serum copper concentrations in the absence of hemolysis. 5 Liver and kidney copper concentrations, considered together, are thought to be the most reliable diagnostic indicators of copper toxicosis in deceased animals, 6,7,13 but it has been suggested that liver copper concentrations in sheep are a better indicator of chronic copper toxicosis than those in the kidney. 9,10 The superiority of liver copper concentrations for the diagnosis of chronic copper toxicosis in goats might be true in some instances, such as the antemortem diagnosis of the prehemolytic stage of chronic copper toxicosis or in deceased animals that did not exhibit hemolysis, but the role of copper toxicosis in the demise of goat 3, which was in the hemolytic stage of the disease, would have been overlooked if the kidney sample from this animal had not been analyzed for copper.
There was no history of administration of copper-containing hematinics, copper oxide–containing boluses, exposure to swine or poultry litter, copper-containing footbaths, copper plumbing, or hepatoxic plants or chemicals. Ideally, all components of the diet should be analyzed for trace element content in instances of suspected copper toxicosis, and a careful assessment should be made of the proportions of those components in the diet. However, there were logistical and financial constraints on the number of possible sources of copper and/or mineral imbalances that could be investigated in the 3 goats, and only several samples of the pelleted ration and mineral mix fed to goats 1 and 2 were analyzed for minerals using inductively coupled plasma emission spectrometry at the Diagnostic Center for Population and Animal Health at Michigan State University (Lansing, MI). The forage fed to goats 1 and 2 was not analyzed for minerals; however, pasture grasses and hay generally have low copper concentrations, depending on soil copper content. No results of mineral analyses for feedstuffs associated with goat 3 were available for review, and the origin of copper in its case of chronic copper toxicosis was never determined.
The results of the analyses for the most relevant minerals associated with the pathogenesis of chronic copper toxicosis are summarized in Table 2. Samples from the 3 bags of pelleted feed were very consistent in their mineral content. The samples of mineral mix appeared to be more heterogeneous in mineral content than the pelleted feed, but the concentration of copper in this component of the diet, unlike that in many mineral mixes formulated for cattle, was relatively low. The mean copper concentration of approximately 31 mg/kg in the pelleted feed samples submitted was not uncommonly high for concentrates formulated for goats (adequate ranges, depending on the reference: 10–23 or ≤40 mg/kg on a dry matter basis), 14,18 especially if this feed was fed in an appropriate manner.
Factors influencing copper absorption from the gastrointestinal tract can play as important a role in the development of chronic copper toxicosis as the concentration of copper in the diet. 14 Feed sources differ widely in copper bioavailability, and copper is frequently more bioavailable in processed feedstuffs than in fresh plant material. 27 Deficiencies in molybdenum, sulphur, and iron and, to a lesser extent, calcium and zinc, can all contribute to the development of chronic copper toxicosis in domestic animals. 13,14,17,21 Low dietary molybdenum and/or sulphur concentrations reduce the formation of insoluble copper complexes in the gastrointestinal tract as well as limit the excretion of copper in the urine and bile. 7,9,17 With respect to sheep, some references have stated that dietary copper to molybdenum ratios should be >2:1 and <20:1; however, others have suggested that chronic copper toxicosis in sheep can occur with normal copper concentrations when copper to molybdenum ratios in the diet exceed 10:1, or, in some instances, 6:1. 6,7,18,24
The mean concentration of molybdenum in the submitted samples of pelleted feed was <1 mg/kg, with the concentrations of iron, sulphur, calcium, or zinc (results not shown) in these pellets being more than adequate. The estimated copper to molybdenum ratio in the pellets fed to goats 1 and 2 was >30:1, suggesting that the elevated ratio between copper and molybdenum in this portion of the diet most likely played a greater role in the development of chronic copper toxicosis in goats 1 and 2 than excessive consumption of copper, especially if this pelleted feed was the primary constituent of the diet. Unfortunately, it was difficult to obtain specific information regarding how much of the pelleted feed was consumed by goats 1 and 2, and the copper status of the other goats in the herd consuming this particular feed was not evaluated. Without this additional data, it was not possible to conclude that the elevated copper to molybdenum ratio in the pelleted feed was the sole “cause” of chronic copper intoxication in goats 1 and 2.
The current cases are some of the first reported, naturally occurring, as opposed to experimentally induced, cases of primary chronic copper toxicosis in adult Boer goats. These instances of chronic copper intoxication were rather unusual in that there was evidence of hemolysis and hemoglobinuric nephrosis in all of the apparently affected animals. Hemolytic crisis, including hemoglobinuria and hemoglobinuric nephrosis, have been previously described as a relatively uncommon occurrence in adult goats with primary copper toxicosis and have generally been observed in conjunction with naturally occurring acute toxicoses or field cases of chronic copper toxicoses involving preruminant juveniles as well as experimentally induced copper intoxications. 1,18,21,23,28 It appears that mature Boer goats are susceptible to the hemolytic stage of chronic copper toxicosis, while this particular stage of chronic copper intoxication was not observed in a recent, large-scale copper intoxication in lactating dairy goats. 5 Copper analyses on both liver and kidney samples were necessary to definitively confirm the diagnosis of copper toxicosis in all 3 goats. The antemortem copper status of apparently clinically normal goats in the herd should be assessed by measuring the copper concentration in liver biopsies from a representative number of animals, and the presence or absence of a hepatopathy should be determined in these animals to identify individuals in the prehemolytic stage of copper intoxication. All feedstuffs should be analyzed for iron, molybdenum, sulphur, and zinc as well as copper to determine what nutritional factors are contributing to the pathogenesis of the disease in a specific instance, and consideration should be given to the ingestion of certain hepatotoxic plants and other potential toxic exposures, which could lead to the development of secondary chronic copper toxicosis. 12,15,21
Acknowledgements. The authors thank Charles Coatney at the Veterinary Medical Diagnostic Laboratory (VMDL; University of Missouri, Columbia, MO) for performing the tissue copper analyses and the Diagnostic Center for Population and Animal Health at Michigan State University (Lansing, MI) for performing the inductively coupled plasma emission spectrometry analyses on the submitted feed sample. The authors also would like to thank the Histology Laboratory at the VMDL for preparing the tissue sections and Howard Wilson, at the University of Missouri, for his assistance with the figures.
