Abstract
Macropodid alphaherpesvirus 2 (MaAHV2) is best described in macropods and has been implicated in outbreaks among captive marsupial populations in Australia. Natural disease caused by herpesviruses has not been reported previously in opossum species, to our knowledge. One Virginia opossum (Didelphis virginiana) and 1 water opossum (Chironectes minimus) were submitted for postmortem examination from a zoo that housed 6 opossums, all of which died within several weeks. Red kangaroos (Macropus rufus) and red-necked wallabies (Macropus rufogriseus) were also present at the facility. Liver samples from both opossums were submitted for transmission electron microscopy and whole-genome sequencing. Microscopically, both opossums had multifocal necrosis in the liver and lung, with intranuclear inclusion bodies within hepatocytes and pneumocytes. Another significant finding in the Virginia opossum was sepsis, with isolation of Streptococcus didelphis from various organs. Ultrastructural analysis of formalin-fixed liver tissue identified herpesviral replication complexes in both opossums; negative-stain electron microscopy of unfixed liver tissue repeatedly yielded a negative result. The herpesvirus had >99% nucleotide identity with MaAHV2. These 2 cases indicate that both opossum species are susceptible to MaAHV2 infection, and the outbreak has implications for mixed-species facilities that house macropods.
Alphaherpesviruses are enveloped DNA viruses with a wide host range and the capacity to establish latent infections. 13 Macropod herpesviruses occur Australia-wide and have been implicated in disease outbreaks among captive marsupial populations in Australia. 23 Two alphaherpesviruses isolated from macropods in Australia, termed macropodid alphaherpesvirus 1 (MaAHV1; Orthoherpesviridae, Simplexvirus macropodidalpha1) and macropodid alphaherpesvirus 2 (MaAHV2; Simplexvirus macropodidalpha2), have been accepted as distinct species by the International Committee on Taxonomy of Viruses (ICTV). 13 Additional novel alphaherpesviruses have been isolated from macropods in Australia, including macropodid alphaherpesvirus 4 (MaAHV4; Simplexvirus macropodidalpha4). 21 In the United States, macropodid gammaherpesvirus 3 (MaGHV3; unclassified Gammaherpesvirinae) was first identified in a group of captive eastern grey kangaroos (Macropus giganteus) with cloacal ulcers, 18 and an unidentified herpesvirus was detected in a red kangaroo (Macropus rufus) with hepatitis. 4 Other gammaherpesviruses, macropodid gammaherpesviruses 5 and 6, have been identified in free-living Australian populations of swamp wallabies (Wallabia bicolor) and quokkas (Setonix brachyurus), respectively.15,19
Opossums are a diverse group of New World marsupials of the family Didelphidae, which are endemic to the Americas. 22 The Virginia opossum (syn. North American opossum; Didelphis virginiana) is a large opossum species that naturally lives in temperate regions of North and Central America, and the water opossum (Chironectes minimus) is a semiaquatic marsupial that inhabits tropical and subtropical forests, ranging from southern Mexico to Uruguay. 22
We retrieved no reports of macropod herpesviral infections in opossums in a search of Google, PubMed, and CAB Direct, using search terms “herpesvirus” and “opossum,” suggesting that macropod herpesviral infection has not been reported in opossum species.
Materials and methods
Animals, sample collection, and histopathology
One Virginia opossum and 1 water opossum were submitted for postmortem examination to the Minnesota Veterinary Diagnostic Laboratory (MVDL; St. Paul, MN, USA) from a zoo that had housed 4 Virginia opossums and 2 water opossums. The 2 water opossums had recently been transferred to the zoo from the facility of an animal vendor; the Virginia opossums had been at the zoo for ~6 mo. The Virginia opossums and the water opossums were housed in separate enclosures with a shared interior mesh divider. Two weeks after the arrival of the water opossums, all 4 Virginia opossums died within several weeks, starting with 2 senior females (>5-y-old) and followed by 2 young males (<2-y-old). The 2 water opossums died ~30 d after moving to the zoo and within a few days of the death of the last Virginia opossum. Macropods comprising 9 red kangaroos and 3 red-necked wallabies (Macropus rufogriseus) were housed at the zoo ~30 m from the opossum enclosure; macropods were not present at the vendor facility during the stay of the water opossums.
Both opossums underwent postmortem examinations. Tissue samples were fixed in 10% neutral-buffered formalin, including brain, heart, lungs, liver, spleen, kidneys, stomach, intestine, skeletal muscle, and bone marrow. The formalin-fixed tissues were processed routinely, and 4-µm sections were stained with H&E.
Bacteriology
Unfixed samples of liver, lung, and spleen from the Virginia opossum, and unfixed samples of liver and lung from the water opossum, were submitted for routine aerobic culture. The samples were inoculated on 5% sheep blood agar (SBA; prepared in-house according to the standards provided by CLSI manual Quality Control for Commercially Prepared Microbiological Culture Media 6 ), MacConkey agar (dehydrated culture media: MacConkey agar; BD BBL), Columbia nalidixic acid agar (CNA; prepared plated media: Columbia CNA agar with 5% sheep blood; BD BBL), and brilliant green agar (dehydrated culture media: brilliant green agar; BD BBL) as part of routine aerobic culture. The SBA and CNA plates were incubated at 37°C in 5% CO2 for 18–24 h. The plates were examined for growth, and bacterial colonies were identified (Microflex LT MALDI-TOF; Bruker). The mass spectrometer was calibrated with a range of 3,637.8–16,952.3 Da prior to sample testing using the bacterial test standard (Bruker), per the manufacturer’s recommendations. In addition, pooled tissues and intestine from the Virginia opossum were submitted for Salmonella culture. For Salmonella culture, the tissues were added to Hajna tetrathionate broth and incubated overnight at 40°C. After incubation, the broth was plated to BG and xylose lysine deoxycholate (XLD) agar plates. The plates were incubated overnight and examined for colonies resembling Salmonella species.
Transmission electron microscopy
For detection with direct negative-contrast transmission electron microscopy (TEM), viral particles were extracted, cleaned, concentrated, and contrasted using a modified nebulization method. 3 Briefly, fresh liver samples from both opossums were subjected to bead-mill homogenization (Bead Ruptor 12; Omni International) at 5 m/s velocity for 45 s. The homogenate was suspended in 15 mL of double-distilled water, centrifuged at 2,000 × g for 10 min at 30°C, and filtered with a series of 5.0-, 1.2-, and 0.8-μm syringe filters (Fisher Scientific). The supernatant was then ultracentrifuged at 285,000 × g for 110 min at 5°C (Optima L-80XP ultracentrifuge; Beckman Coulter). A mixed re-suspended virus pellet–1% sodium phosphotungstate contrast solution was micro-aerosolized onto a 200-mesh formvar/carbon-coated electron microscope grid using a glass nebulizer (Electron Microscopy Sciences [EMS]). The viral particles were identified by finding repetitive profiles with consistent dimensions, morphologic features, and structural characteristics that match the dimensions and features accepted and published by the Report of the ICTV. 13
For tissue ultrastructure assessment, 1–2-mm3 cubes of formalin-fixed liver samples from both opossums were post-fixed in Karnovsky fixative and 1% osmium tetroxide in 0.1 M cacodylate buffer. Using an automated processor, tissues were embedded in resin as described elsewhere. 2 Briefly, samples were dehydrated using a 25–100% ethyl alcohol gradient and then infiltrated with resin (Embed 812; EMS). Resin tissue blocks were trimmed and sectioned (UC6 ultramicrotome; Leica). Contrasted thin sections (60–70 nm) were visualized (1400 transmission electron microscope; Jeol). Images were obtained and analyzed (OneView camera system 1095, 16 megapixels with the Gatan microscope suite [GMS3.0]; Gatan).
Next-generation sequencing analysis
In both cases, total nucleic acid was extracted from 200 mL of the resuspended EM pellet (MagMax Pathogen RNA/DNA kit; Thermo Fisher) following the recommendations of the manufacturer. Extracted DNA was purified (AMPure XP beads; Beckman Coulter) and used as input to a library generated with a ligation sequencing kit (SQK-LSK109; Oxford Nanopore Technologies [ONT]). First, DNA was end-repaired (NEBNext Ultra II DNA end prep and repair kit; New England Biolabs [NEB]), purified using AMPure XP beads in a ratio of 1:1 volume of beads per sample, and then eluted in 30 µL of nuclease-free water. Sequencing adapters (AMX) were ligated to the DNA (NEBNext Quick T4 DNA ligase; NEB) by incubation at room temperature for 10 min. The adapter-ligated DNA library was purified with AMPure XP beads in a ratio of 1:2.5 volume of beads per sample, followed by 2 washes with S Fragment buffer (ONT) and elution in 7 µL of elution buffer. The library was loaded onto a flowcell (R9.4.1; Flongle) and run on a sequencer (GridION Mk1; ONT). Prior to starting the run, the sequencing kit SQK-LSK109 and the “super accuracy” basecaller were selected in the MinKNOW data analysis software (v.22.08.9). After sequencing for 24 h, FASTQ files containing “pass” reads (Q-score ≥ 10) were loaded into Geneious Prime (v.2022.1.1; Dotmatics) and mapped to a reference alphaherpesvirus 2 genome (GenBank MT900475) using the MiniMap2 plugin. A consensus sequence with a coverage of ≥90X across the entire genome was submitted to GenBank (OP684796). A multiple sequence alignment of the DNA polymerase gene was produced using the MUSCLE algorithm in Geneious Prime (v. 2022.1.1). Phylogenetic analysis using the neighbor-joining method was performed, and bootstrap values were calculated using 1,000 pseudo-replicates. All publicly available full-length DNA polymerase gene sequences from macropodid herpesviruses, including MaAHV1–3 were used in the phylogenetic analysis. In addition, several herpesviruses including a human, a chimpanzee, and a bat-associated herpesvirus were included as out-groups.
Results
Clinical findings
The Virginia opossum was an overweight, 8-mo-old, intact male that developed hindlimb paralysis ~24 h before death. The water opossum was an overweight, 6-mo-old, intact male that died without premonitory signs. Both animals were submitted fresh (unfrozen) in a state of good postmortem preservation.
Macroscopic findings
The Virginia opossum had a moderately enlarged, brown liver. The myocardium was diffusely pale with subtle brown discoloration in the left ventricular papillary muscle. The lungs were diffusely reddened, wet, and heavy with dark-red, well-demarcated, up to 5-mm foci, accounting for ~5% of the total lung surface area. The skin of the face had a few pinpoint crusts. The subcutis and periarticular soft tissues of the right stifle joint were slightly expanded and red-brown. The medial iliac lymph nodes were reddened. The thoracic cavity contained a small amount of clear, watery fluid. The water opossum had only mild hydrothorax.
Microscopic findings
Histologically, the liver of both opossums had randomly scattered areas of hepatocellular necrosis (Fig. 1A) as well as mild lymphoplasmacytic periportal hepatitis. Frequently, hepatocytes within these areas contained a 3–7-µm eosinophilic intranuclear inclusion body (INIB). The lungs of both cases, but to a greater degree the water opossum, had areas of alveolar necrosis with associated neutrophilic inflammation and hemorrhage (Fig. 1B). Infrequently, sloughed pneumocytes had INIBs as described in the liver. Within the lungs of the Virginia opossum, alveolar lumina contained infrequent aggregates of cocci.
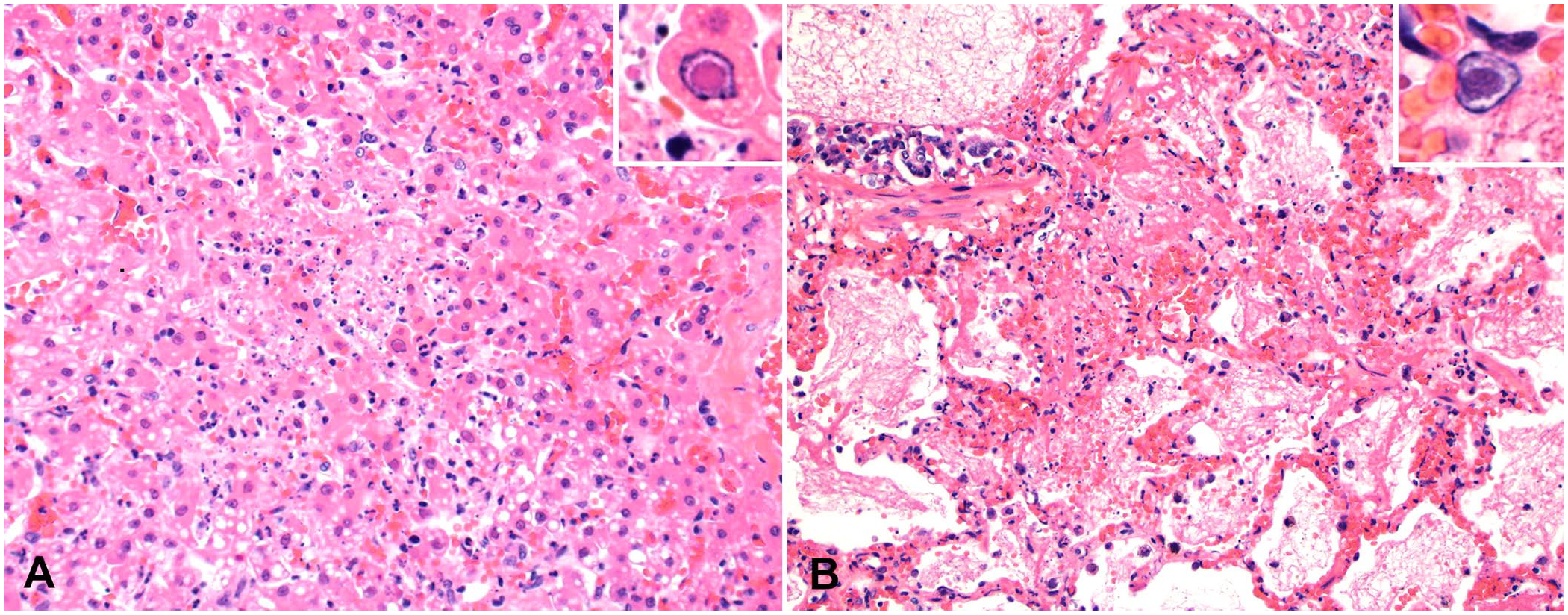
In addition to the lesions attributable to herpesviral infection, the Virginia opossum had lesions caused by a suspected bite wound on the right hindlimb with subsequent septicemia. Within the muscle and soft tissues of the right stifle joint were bundles of degenerate skeletal myofibers with associated neutrophilic and histiocytic inflammation. The muscle tissue was hemorrhagic, with fibrin and infrequent aggregates of cocci. The heart had areas of lytic myocardial necrosis, with fibrin, degenerate neutrophils, and aggregates of cocci. Within the kidneys, the interstitium was infiltrated by infrequent clusters of non-degenerate and degenerate neutrophils admixed with small numbers of cocci. The cortex, medulla, and sinuses of the medial iliac lymph node were expanded by large numbers of degenerate neutrophils and fewer macrophages, admixed with fibrin, cellular debris, and large numbers of cocci. The haired skin of the intermandibular region had an ulcer with superficial granulation tissue formation, deeper areas of lytic necrosis within the dermis, a few degenerate neutrophils, and aggregates of cocci.
Bacteriology
Beta-hemolytic Streptococcus didelphis was isolated by aerobic culture in 4+ numbers from the liver, spleen, and lungs of the Virginia opossum; there was no significant growth from the intestine by aerobic culture or from the pooled tissue by Salmonella culture. There was no significant growth from the lung or liver of the water opossum by aerobic culture.
Transmission electron microscopy
Virus screening using direct negative-contrast TEM on frozen then thawed hepatic tissue yielded very few equivocal and fragmented viral particles. At the ultrastructural level, hepatocyte inclusions that were observed on light microscopy were intranuclear herpesviral replication and assembly complexes in both cases (Fig. 2). Intranuclear replication complexes were composed of variably dense, amorphous-to-filamentous viroplasm with immature and maturing, 90–100-nm capsids (Fig. 3A). Primary enveloped virions were present within the perinuclear space (Fig. 3A). Nucleocapsids within the cytoplasm acquired tegument proteins at electron-dense deposits, forming mature virions within vesicles (secondary envelopment; Fig. 3B). The intercellular space contained numerous free, mature, enveloped virions.
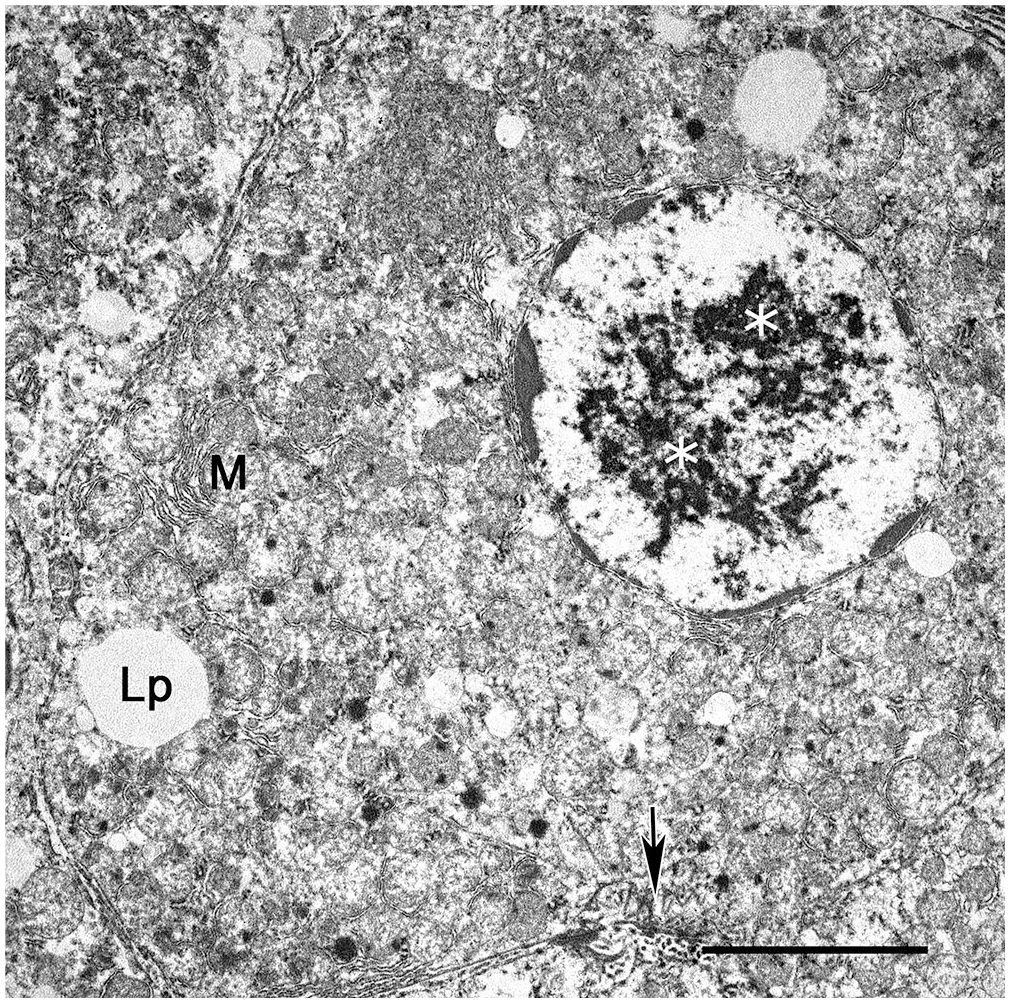
Hepatocytes infected with macropodid alphaherpesvirus 2 in a water opossum. Herpesviral replication and assembly complexes within the nucleus and cytoplasm of a hepatocyte (white asterisks). The chromatin is clustered at the periphery of the nucleus. Lipid droplets (Lp), mitochondria (M), biliary canaliculi (arrow). Bar = 5 μm. Transmission electron microscopy.
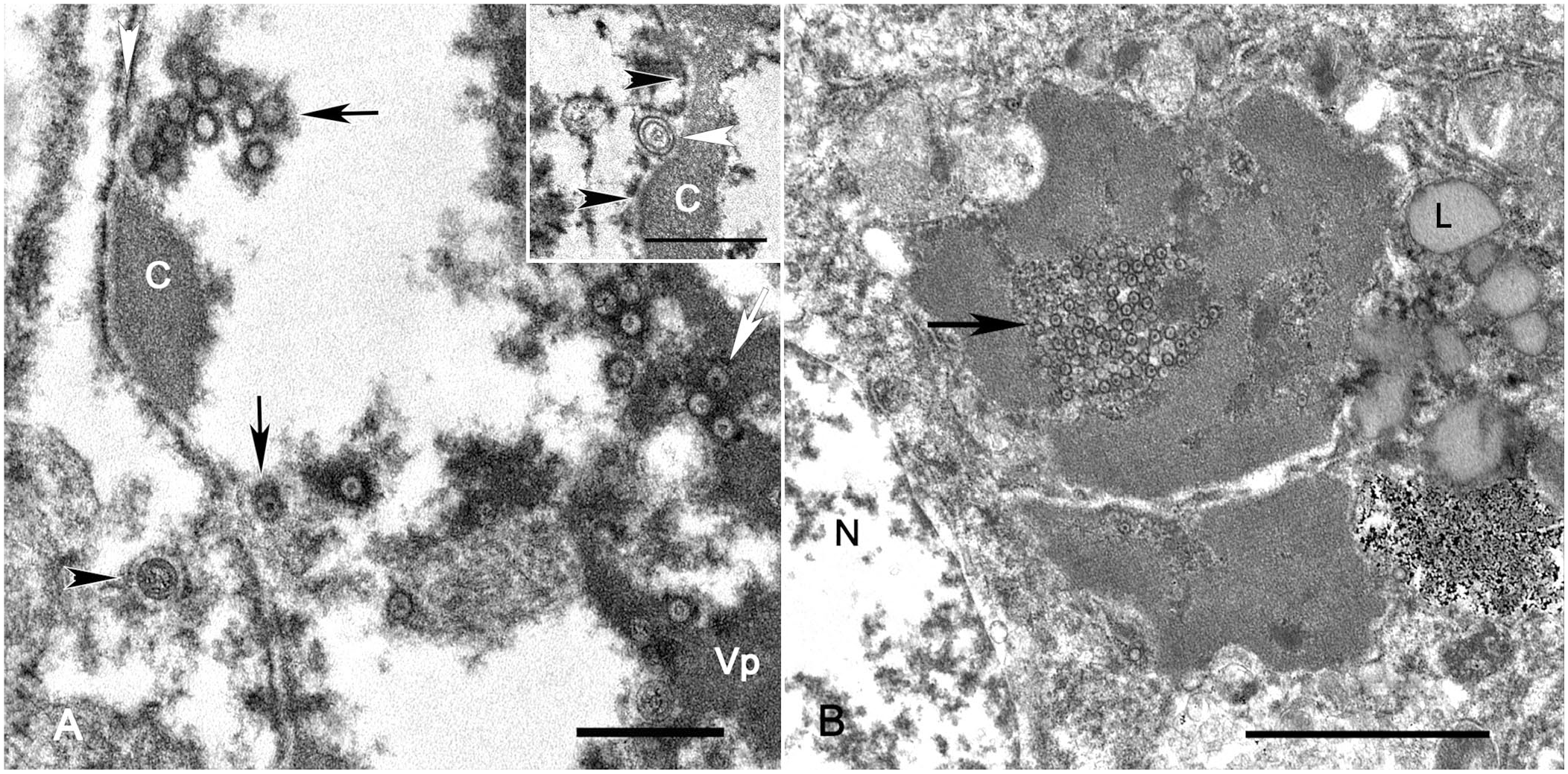
Hepatocytes infected with macropodid alphaherpesvirus 2 in a water opossum. Transmission electron microscopy.
Next-generation sequencing analysis
The entire genome of the water opossum herpesvirus was sequenced at >90X coverage; only 50% of the Virginia opossum herpesvirus was recovered at a low depth of coverage (<5%). The sequences of the herpesvirus from the Virginia opossum were 100% identical to the water opossum herpesviral sequence. Only 2 other full-length herpesviral genomes in GenBank had close similarity to the water opossum genome. One was a MaAHV2 strain (MT900475) isolated from a black forest wallaby in Australia (99.9% nucleotide homology). The other was a MaAHV4 strain (MT900474) isolated from an eastern grey kangaroo in Australia (93.4% nucleotide homology). Across the conserved DNA polymerase gene, the next most similar alphaherpesviral strains in GenBank were a MaAHV1 strain (79.4% nucleotide homology) followed by a bat-associated alphaherpesvirus (66.9% nucleotide homology), a chimpanzee alphaherpesvirus 1 (66.8% nucleotide homology), and a human alphaherpesvirus 2 strain (66.3% nucleotide homology; Fig. 4).
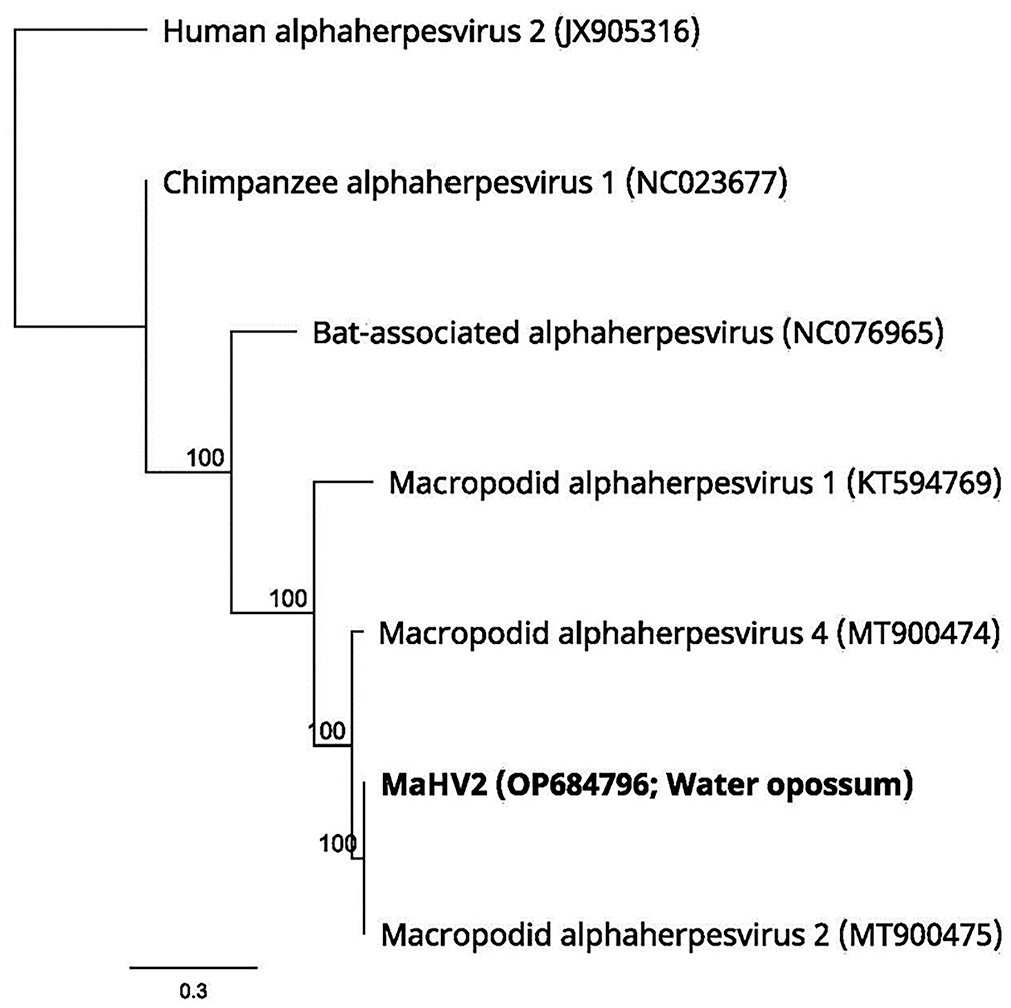
Phylogenetic tree. The sequence of the water opossum herpesvirus is 99.9% identical to macropodid alphaherpesvirus 2. The numbers at the nodes represent bootstrap confidence values for 1,000 replicates based on the neighbor-joining tree building method. The scale bar = 0.3 substitutions per nucleotide position.
Discussion
Our diagnosis of MaAHV2 infection in the water opossum is based on histologic features, electron microscopy, and whole-genome sequencing. In the Virginia opossum, only short fragments of viral DNA were amplified and sequenced. The employed genome sequencing platform is sensitive to certain pitfalls, and our failure to sequence the entire genome may have resulted from an insufficient viral load in the sample or suboptimal preservation of the tissue. However, the amplified fragments were identical to the MaAHV2 genome sequences observed in the water opossum. Therefore, and because the animals were housed together and had similar hepatic lesions with INIBs in liver and lungs, both animals were likely infected with the same herpesvirus (MaAHV2).
From the diagnostic perspective, it is unusual that, despite viral particles seen in TEM samples as well as the numerous hepatocellular INIBs seen on light microscopy, only minimal and equivocal herpesvirus particles were detected with direct negative-contrast TEM. Considering the known high sensitivity of TEM, this discrepancy, which is unusual in our experience at the California Animal Health & Food Safety Laboratory System (CAHFS) electron microscopy laboratory, hints at a possible limitation in the preparation protocol. This shortfall may have failed to account for an intrinsic tissue factor, such as nanoparticles (proteins) that agglutinate with virions during ultracentrifugation, impeding or obscuring the detection of viral particles through negative-contrast TEM.
We attributed the death of the Virginia opossum to S. didelphis septicemia with concurrent MaAHV2 infection. S. didelphis was first reported in 2000 from skin, spleen, liver, and lung of 9 deceased Virginia opossums with necrotic skin lesions and septicemia. 16 We attributed the death of the water opossum to the MaAHV2 infection in the absence of any significant comorbidity. Although the remaining 3 Virginia opossums and single water opossum in the zoo collection were not submitted for postmortem examination, we suspect that these animals also died at least in part because of a MaAHV2 infection, given the temporal and spatial clustering of their deaths.
Natural herpesviral infections (macropod herpesvirus or otherwise) have not been documented in opossum species, although in a 1963 study the experimental inoculation of suid alphaherpesvirus 1 (pseudorabies virus) in 2 Virginia opossums resulted in convulsions and the death of both opossums. Histologic examination revealed necrotic areas with INIBs in various organs of these opossums. 20 Similar to other herpesviruses, macropod herpesviruses have been shown to infect non-host species. In a 2015 study of free-living and captive Australian marsupials, the seroprevalence of MaAHV1 or MaAHV2 antibodies in the non-macropod Tasmanian devils (Sarcophilus harrisii) and common wombats (Vombatus ursinus) was 1 of 21 (5%) and 10 of 15 (67%), respectively, although the clinical implications of these findings remain uncertain. 19 In a 2004 study, the experimental inoculation of brushtail possums (Trichosurus vulpecula) with MaAHV1 led to transient infection; however, latent and systemic infection was not achieved. 24 Compared to Australian marsupials, opossums are non-sympatric species with macropods; hence, they may be seen as naïve hosts evolutionarily and therefore potentially more susceptible to disease following infection.
Clinical herpesviral disease in macropods is considered rare, and is typically seen in captive animals. Clinical signs in macropods include sudden death, fever, conjunctivitis, and vesicles and ulcers in the oral mucosa, cloaca, and penis. 23 There have been several reports of macropod herpesviral disease outbreaks in captive macropod populations5,8,10; however, outbreaks of macropod herpesviral infections in captive non-macropod species have not been reported. Although herpesviruses are generally deemed species-specific, limited instances of alphaherpesviral transmission between segregated species in zoologic settings have been reported.1,9,11 The water opossums in our case reportedly did not have contact with macropods at the animal vendor facility prior to arriving at the zoo, which housed red kangaroos and red-necked wallabies. These macropods, which were not tested for MaAHV2, were kept at a distance of ~30 m from the opossums, with which they had no direct contact. Although alphaherpesviruses are enveloped and therefore thought to be fairly unstable in the environment, indirect transmission via aerosols and fomites has been demonstrated. For example, equid alphaherpesviruses 1 and 4 (EqAHV1, EqAHV4) are transmissible via aerosols, and bovine alphaherpesvirus 1 can be spread between calves up to 4 m apart via aerosol transmission.7,14 Furthermore, EqAHV1 persists in the environment for up to 48 h on materials such as leather, fabric, wood shavings, wheat straw, and polystyrene, 17 and remains infectious in water for up to 21 d under optimal conditions. 7 In our 2 cases, the precise mode of exposure to MaAHV2 remains speculative.
Alternatively, it is possible that the Virginia opossum’s physiologic stress in captivity coupled with septicemia triggered the recrudescence of a latent MaAHV2 infection. Nothing is known of the ability of herpesviruses to produce latent infections in opossums. However, akin to herpesviruses in other species, macropod herpesviruses have been reported to occasionally cause disease outbreaks either due to reinfection from an exogenous source or recrudescence of latent infections, particularly during times of stress or immunosuppression. 12 Consequently, it remains a possibility that a reactivated herpesviral infection, in either the septicemic opossum or another opossum, due to transport-related stress or environmental factors, might have initiated subsequent infections among the remaining opossums.
Our cases reveal the susceptibility of both Virginia opossums and water opossums to MaAHV2 infection and the potential for fatal disease outcomes, and highlight the importance of vigilant monitoring when different animal species from distinct habitats coexist in the close confines of multi-species facilities. Given the broader spectrum of species vulnerable to MaAHV2 beyond macropods, and the unknown distribution of macropodid herpesviruses outside Australia, we recommend that institutions exercise caution when introducing non-macropod species to facilities that house macropods.
Footnotes
Acknowledgements
We thank the University of Minnesota Veterinary Diagnostic Laboratory Necropsy, Histology, Molecular, Bacteriology, Virology and Electron Microscopy sections, Dr. Albert Canturri for performing the water opossum postmortem examination, and Christina Heard for the electron microscopy preparations.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
