Abstract
Soft tissue sarcomas (STSs) are common well-described cutaneous neoplasms in many domestic species. Few cutaneous neoplasms have been reported in cervids, other than papilloma virus–induced benign cutaneous fibromas (BCFs). Two wild-caught Alaskan moose (Alces alces), housed at different North American zoos, were both presented with lameness and a mass. The gross appearance of each mass varied greatly; case 1 had a distinct, exophytic, ulcerated mass near the right carpal joint similar in appearance to a BCF, and case 2 had a pronounced swelling over the left hip. Based on histologic evaluation, both masses were diagnosed as STS. The clinical presentation of these masses included rapid growth and associated lameness; however, gross appearance was markedly different. Histopathology revealed characteristics typical of STS in other species, including spindle-shaped cells arranged in streaming sheets with ovoid nuclei. STS should be included as a differential diagnosis for moose with a cutaneous or subcutaneous soft tissue mass or swelling.
Soft tissue sarcomas (STSs) are common cutaneous neoplasms, and are described most frequently in dogs and cats.1,2,13 STSs can arise at any anatomic site from mesenchymal tissue and appear grossly as well-demarcated fleshy masses.1,2,9,13,15 Histologic analysis is a mainstay of STS diagnosis; however, features of STSs vary widely, and identification of the cell type of origin is often challenging.5,7,9,10,17 Immunohistochemistry (IHC) has been described as an ancillary diagnostic tool used in conjunction with histologic evaluation to differentiate STSs in dogs and cats.7,17 Neoplasms classified as STS include fibrosarcomas, myxofibrosarcomas, peripheral nerve sheath tumors, perivascular wall tumors, liposarcomas, and rhabdomyosarcomas.1,2,13,15 Although histologic features among these types of STS vary, clinical behavior, including rate of local recurrence, metastasis, and response to chemotherapeutic and radiation therapy, are similar and predicted by degree of differentiation, mitotic counts, and proportion of tumor necrosis.1,2,4,13,15
In the family Cervidae, the cutaneous neoplasm identified most commonly is benign cutaneous fibroma (BCF).11,19,21–23 BCF is a non-encapsulated tumor that has an identified viral etiology.6,19,20,22,23 The causative viral agent is a deltapapillomavirus related to bovine papilloma 1 virus.6,22,23 Histopathologic epidermal changes that differentiate BCF from STS include epidermal hyperplasia with acanthosis, orthokeratotic hyperkeratosis, and formation of rete ridges;11,14,19,20–24 however, these benign fibromas have been erroneously referred to as fibrosarcomas, which is a type of STS.19,22 This misuse of nomenclature is commonly observed in discussions of STS, and can impede accurate diagnosis and management of these cases. 4 Viral-induced fibromas are generally considered benign, small, and have been shown to regress in wild cervids without treatment, whereas STSs do not regress, are locally aggressive, and require complete surgical excision for successful treatment.1,2,6,10,13,15,23 STSs, including fibrosarcomas and myxofibrosarcomas, have been identified in cervids, but are considered rare.5,9,10,22 We describe here the clinical history, presentation, and histopathologic characteristics of STS in 2 Alaskan moose (syn. Eurasian moose; Alces alces).
Case 1 was a 5-y-old wild-caught, intact male Alaskan moose at the Potter Park Zoo (Lansing, MI, USA) that had an intermittent grade 3 of 5 right forelimb lameness and an associated 5-cm diameter, ulcerated, well-circumscribed, exophytic dermal mass on the palmar aspect of the right carpus (Fig. 1A). Given the gross appearance of this mass and previous reports of cutaneous tumors in moose, the mass was presumed to be a BCF. 22 Following rapid growth and ulceration, the mass was surgically excised under sterile field conditions; however, given the location of the mass, excision with wide margins was not achieved.
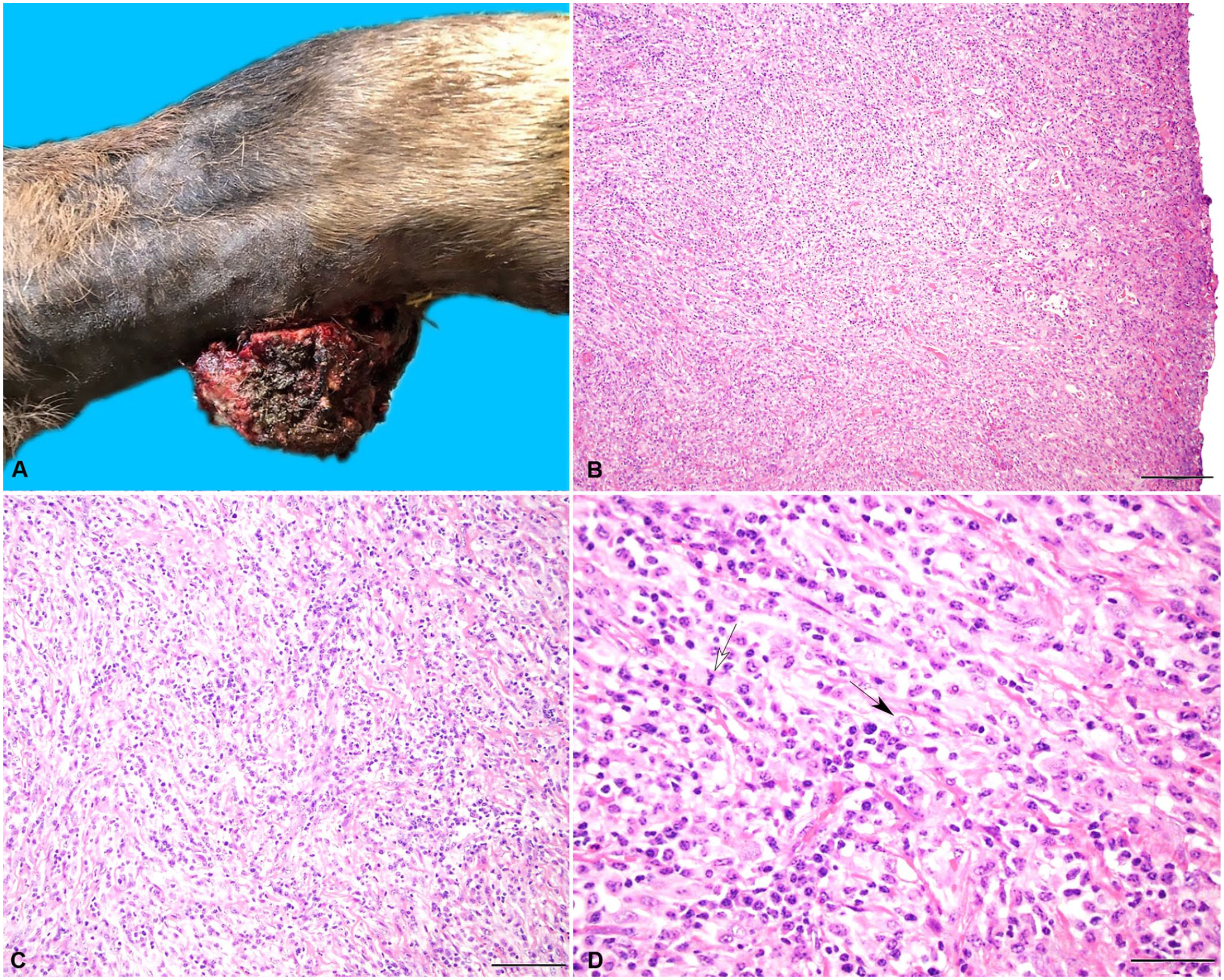
The soft tissue sarcoma (STS) from case 1, a zoo-housed 5-y-old moose, was present on the palmar aspect of the right carpus and contains streams and bundles of spindle cells on a collagenous stroma.
The dermal mass was submitted for histologic evaluation at Michigan State University Veterinary Diagnostic Laboratory (MSU-VDL; Lansing, MI, USA). Histologic analysis revealed neoplastic spindle cells arranged in streams and bundles on a collagenous stroma (Fig. 1B, 1C). Neoplastic cells had indistinct cell borders, contained variable amounts of eosinophilic cytoplasm, and had ovoid nuclei. The nuclei contained finely stippled to vesiculate chromatin and 2–6 nucleoli. A few multinucleate giant cells were observed, with marked anisokaryosis and frequent karyomegaly, and there were 14 mitoses in 10 consecutive 400× (2.37 mm 2 ) fields (Fig. 1D). The neoplastic features extended to the surgical margins. The mass was infiltrated by a large number of lymphocytes and neutrophils throughout, and the overlying epidermis was ulcerated extensively, with large numbers of degenerate neutrophils and cellular debris.
IHC markers, including vimentin, smooth muscle actin (SMA), laminin, and desmin, were used to evaluate fibroblast, muscle, perimyocyte, or peripheral nerve sheath tumor differentiation.7,17 Marker CD204 was used to evaluate histiocytic sarcoma differentiation.7,17 IHC revealed diffuse cytoplasmic staining for vimentin and rare foci of cytoplasmic staining for SMA and S100, which confirmed the histologic suspicion of a STS and indicated fibrosarcoma as the top differential. 17 Neoplastic cells were negative for CD204, desmin, and laminin. PCR, using primers for generic papillomaviruses E1 and L1m, did not detect papilloma virus.3,12,16 Mixed bacterial growth was identified by aerobic and anaerobic culture, including Morganella morganii, Trueperella pyogenes, Streptococcus dysgalactiae, Clostridium cadaveris, Bacteroides fragilis, and Lactobacillus spp. Based on the histopathologic findings of neoplastic spindle cells, supportive IHC results, and lack of viral etiology, this mass was diagnosed as a STS. Given that neoplastic cells extended to the deep margin, indicating incomplete surgical excision, a single electrochemotherapy treatment was performed under general anesthesia 21 d after surgery. Carboplatin was injected within and along the margins of the mass followed by electrical pulse therapy.
Forty days following mass removal, the moose was found recumbent with marked hypoglycemia (1.72 mmol/L; RI: 5.3–10.1 mmol/L) and hypocalcemia (1.62 mmol/L; RI: 2.4–3.0 mmol/l). 18 Despite supportive medical care, the moose showed no improvement, and was euthanized. The body was submitted for postmortem examination to the MSU-VDL. The moose was markedly underconditioned (body condition score 2 of 9) with minimal fat stores and serous atrophy of fat. Erythema and blunting of rumen papillae consistent with chronic ruminal acidosis was noted. Based on the severity of lesions, emaciation secondary to chronic ruminal acidosis was considered a major contributing factor in morbidity and mortality. There was no gross or histologic evidence of local recurrence or metastasis of the STS at the surgical site. We could not determine the contribution of electrochemotherapy to the absence of either local recurrence or metastasis in this case, given that long-term monitoring was not possible. Long-term monitoring of electrochemotherapy as an adjunctive treatment for cervid STS is necessary to determine the effectiveness of this treatment.
Case 2 was a 9-mo-old, wild-caught, intact male Alaskan moose at the Columbus Zoo and Aquarium (Columbus, OH, USA) that was presented because of lameness and a rapidly growing 30 × 30 cm, firm, soft tissue swelling over the point of the left hip (Fig. 2A). Ultrasound of the swelling revealed pockets of fluid and solid material, later identified as blood clots. An incision was made on the ventral margin of the swelling and copious amounts of serum, clotted blood, and several chicken-fat clots were flushed from the space underlying the swelling. Culture of the fluid revealed no bacterial growth. Pelvic radiographs were unremarkable. On surgical exploration, a well-circumscribed tan-to-white mass, with a blood-filled central cavity, was discovered near the femoral head. Neoplasia was suspected, and given the non-resectable nature of the mass, the moose was euthanized.
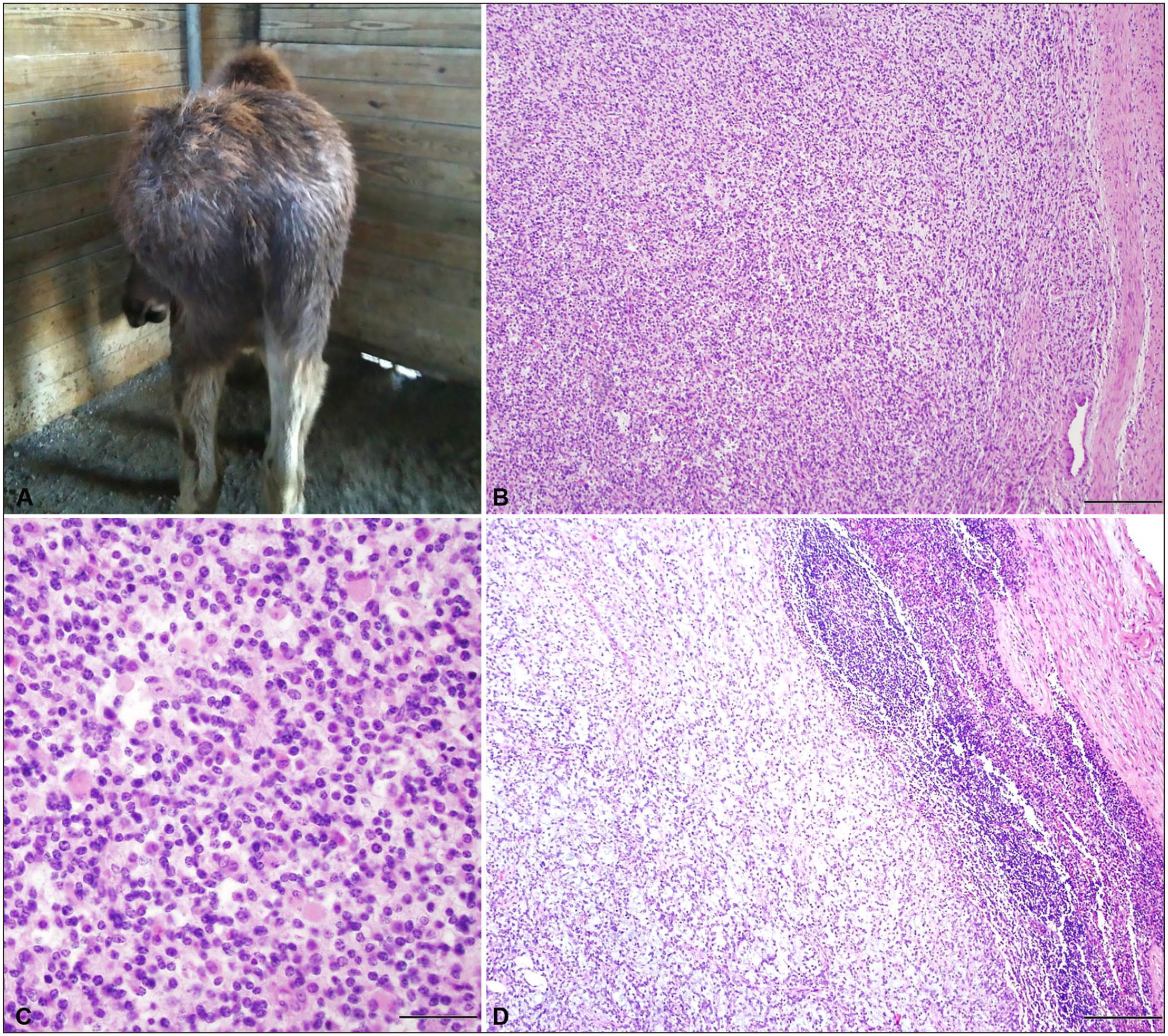
The soft tissue sarcoma (STS) from case 2, a zoo-housed 9-mo-old moose, was present on the left hip, contained spindle cells on a fibrovascular stroma, and had metastasized to the sublumbar lymph node.
Postmortem examination was performed at the Department of Veterinary Biosciences at the Ohio State University College of Veterinary Medicine (OSU-CVM; Columbus, OH, USA). A 25 × 14 × 21 cm, well-demarcated mass with distinct lobules extended deep within and between the middle and deep gluteal muscles, and the left sublumbar lymph node was enlarged. On cut section, the lymph node contained white homogeneous foci that bulged on cut section, suggestive of metastasis. Histologic evaluation of the mass revealed spindle-shaped neoplastic cells arranged in streams, occasionally whirling around small blood vessels, supported by a fine fibrovascular stroma (Fig. 2B). The neoplastic cells had indistinct borders and wispy-to-flocculent cytoplasm. Occasional neoplastic cells contained moderate amounts of eosinophilic cytoplasm with eccentric nuclei (Fig. 2C). Neoplastic cell nuclei were ovoid and contained finely stippled to vesiculate chromatin and often several punctate nucleoli. Approximately 40% of the neoplasm contained pale basophilic stippled material separating neoplastic cells. Anisocytosis and anisokaryosis were mild. Mitoses averaged 30 in 10 consecutive 400× (2.37-mm 2 ) fields. The cortical architecture of the left sublumbar lymph node was compressed by large metastatic nodules composed of neoplastic spindle cells as described (Fig. 2D).
IHC, including for S100, CD204, laminin, desmin, and SMA, was performed at MSU-VDL to further differentiate the neoplastic cells. IHC performed at the Department of Veterinary Biosciences at OSU-CVM revealed strong cytoplasmic staining of almost all neoplastic cells for vimentin, desmin, and SMA. These results confirm the histopathologic finding of a STS, and may indicate differentials of fibrosarcoma, leiomyosarcoma, or rhabdomyosarcoma. 17 Rhabdomyosarcomas are a more common neoplastic process in children and young animals; given the young age of the moose, cellular morphology, and desmin immunohistochemical labeling, this was the top differential. PCR using primers for generic papillomaviruses E1 and L1m did not detect papillomavirus.3,12,16
The gross, clinical, and histologic features of STS in these moose closely resemble STS described in other species.2,5,9,10,13,14 STS is a broad categorization for neoplasms that arise from mesenchymal soft tissues; determining the precise cell of origin can be challenging. As a result, establishing unique IHC expression profiles to classify STS is challenging, making interpretation of IHC results difficult in animals. 17 Additionally, little information on the use of IHC markers in nondomestic species exists, and the usefulness of IHC in these cases has not been established. 17 Our IHC results confirmed the histopathologic findings of STS in both cases; however, these results did not definitively differentiate the type of STS present. 17 More research is necessary to establish IHC use and interpretation guidelines in cervid STSs to allow identification of the cell type of origin in these masses.
Both of our STS moose cases were differentiated from BCF based on the lack of papillomavirus detection, the subcutaneous nature of the mass in case 1, and cell morphology and metastases in case 2. STS and BCF have significantly different clinical outcomes.1,2,10,15 BCF are reported to resolve without treatment in many cases, whereas the treatment of choice for STS is surgical resection with wide margins.1,2,6,15,19 STS should be considered a differential diagnosis for any soft tissue swelling or mass causing lameness in moose and should be differentiated histologically from BCF in order to pursue proper treatment and improve case prognosis.
Treatment and management of STSs have been well documented in domestic canids and felids.1,2 STSs have a good prognosis following complete surgical excision with margins.1,2,4,15 If complete resection is unsuccessful, adjunctive treatments such as local radiotherapy, cryotherapy, and intralesional chemotherapy are recommended.2,15 There are reports of the gross and histologic features of STS, including fibrosarcoma and myxofibrosarcoma in cervids; however, the prognoses of these cases were poor, resulting in euthanasia of the individuals with no discussion of treatment or management.5,9,10
Treatment and management were attempted in case 1, including surgical excision of the mass and adjunctive electrochemotherapy, which was well-tolerated. Unfortunately, the moose’s health declined severely several weeks after receiving chemotherapy, therefore long-term monitoring for metastasis and recurrence was not possible; however, local recurrence was not noted on autopsy.
The STS in case 1 extended to the surgical margins, and both masses underwent rapid growth, similar to previous reports of STS.2,13 Although seen most commonly on the limbs, as in case 1, STS can arise from any anatomic location.1,2,4 STSs are generally reported in middle-aged-to-adult animals similar to case 1, whereas the moose in case 2 was young.4,5,9,10,13 The gross presentation of the STS in these cases is markedly different; the STS in case 1 was an ulcerated, exophytic cutaneous mass; the STS in case 2 was an externally visible soft tissue swelling overlying a hematoma. The ulcerated, exophytic mass presentation of the STS in case 1 was most consistent with previously described BCF in moose, leading to BCF being the leading differential for this case.21,22 The generic papillomavirus PCR assays run in both cases did not detect papillomavirus, which is inconsistent with reports of BCF in cervids.6,19,22 The lameness described in both cases has also been associated with STS in other species.2,13
Histologic characteristics of STS are well-described, but highly variable depending on the classification of the STS.4,5,8,9,10 However, several defining features of STS have been described, including spindle-shaped cells, with indistinct cytoplasmic borders arranged in interwoven streaming sheets, and ovoid nuclei. These histologic features were present in both cases. Multinucleate giant cells, as seen in case 1, have also been described in some STSs. 8 The histopathologic findings of the STS in these cases, with supportive evidence from IHC and lack of detection of papillomavirus, allowed definitive diagnosis of STS in both cases.5,9,10
Footnotes
Acknowledgements
We thank the pathologists, autopsy technicians, and staff from the Michigan State University Veterinary Diagnostic Laboratory and the Ohio State University Clinical Diagnostic Laboratories who were involved in the autopsies and histologic evaluation described here. We also thank the animal management and veterinary staff at both Potter Park Zoo and the Columbus Zoo and Aquarium for their contributions to these cases and providing information for our study. Dr. Tu Chun Hsu is a Molecular Pathology Fellow in the NIH Comparative Biomedical Scientist Training Program supported by the National Cancer Institute in partnership with Michigan State University.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
