Abstract
Serum protein electrophoresis (SPE) shows the distribution of protein fractions, helping clinicians to characterize some pathologic processes. Information is lacking in the literature about SPE alterations in dogs with chronic hepatitis (CH). Our aim was to describe SPE alterations in canine CH, to compare SPE results to histologic scores, and to study SPE trends during follow-up. We reviewed retrospectively case data from dogs with a histologic diagnosis of CH. Only cases with SPE, CBC, and serum chemistry results available were included. Dogs were divided into subgroups based on histologic necroinflammatory activity (A) and fibrosis (F) scores (groups A0-1 and A>1; groups F<2 and F≥2). We included 26 dogs; 15 had follow-up SPE. The most common SPE alterations at admission were hypoalbuminemia (n = 16), increases in α1-globulins (n = 11), γ-globulins (n = 11), α2-globulins (n = 8), β2-globulins (n = 7), and β1-globulins (n = 6), and decreased albumin:globulin (A:G) ratios (n = 20). Four of 11 dogs had β-γ bridging. Groups with higher A and F scores had higher β2-globulins. Eleven of 15 dogs with a post-treatment SPE had a decrease in γ-globulins and increase in A:G ratio compared to their T0, although there was no statistically significant difference. Although further studies are warranted, SPE may be useful for monitoring canine CH.
Serum protein electrophoresis (SPE) is a useful laboratory technique helping clinicians to characterize serum proteins.4,11 Depending on which protein alterations are observed, SPE can help to identify the nature of the ongoing inflammatory process.4,10,11,14 In human gastroenterology, SPE is thought of as an additional tool in the diagnostic work-up and can be used in monitoring chronic liver diseases.4,11,14 In veterinary medicine, SPE is performed to evaluate the presence of acute-phase protein (APP) responses, as well as polyclonal, oligoclonal, and monoclonal gammopathies.4,11 Dogs with chronic hepatitis (CH) can have decreased albumin (a major APP), 4 increased or decreased α-globulins, and increased β- and γ-globulins,9,10 but the literature lacks recent studies that specifically focus on the electrophoretic pattern of dogs with CH; the only report dates back to 1995. 9 That study also suggested that dogs with liver disease, hypoalbuminemia, and decreased α-globulins had a poor prognosis. 9 Beta-gamma bridging, defined as loss of the depression between the β- and γ-globulin bands, has been suggested to be present in CH in both veterinary and human medicine.4,11,14 In dogs, CH is diagnosed histologically by a combination of hepatocellular apoptosis or necrosis, mononuclear or mixed inflammatory cell infiltrate, with optional regeneration and fibrosis,3,13 and histology is used to establish the severity of the disease by grading of necroinflammation and fibrosis. 13
Given the lack of information regarding SPE in dogs with CH, our main aim was to describe SPE alterations in dogs diagnosed with CH, and to evaluate a relationship between SPE and inflammatory grades and fibrosis stages. In addition, we also aimed to evaluate SPE during the CH follow-up.
We searched the Veterinary Teaching Hospital (VTH) database of the University of Pisa (Pisa, Italy) for records of dogs with CH diagnosed from 2011 to 2021. Routinely in our hospital and according to current guidelines, 13 liver sampling included 3–5 laparoscopic or laparotomic biopsies for each dog. All liver biopsies were reviewed by a single histopathologist (F. Abramo) to confirm the CH diagnosis.7,13 Following WSAVA guidelines, necroinflammatory activity was graded as A0 = absent, A1 = slight, A2 = mild, A3 = moderate, A4 = marked, or A5 = very marked. Fibrosis was staged as 0 = absent, 1 = mild, 2 = moderate, 3 = marked, or 4 = very marked.7,13 Dogs were divided into necroinflammatory activity groups: A0-1 (absent to slight) and A>1 (mild to very marked),5,13 and into fibrosis groups F<2 (absent to mild) and F≥2 (moderate to very marked). 13
Dogs were included if the results of SPE, CBC, and serum chemistry performed within 7 d from liver biopsy were available to review. Dogs were excluded if an infectious disease was suspected or diagnosed. All dogs underwent immunomodulatory prednisolone therapy following the ACVIM consensus statement on canine CH starting at 1 mg/kg q12h and tapered within 2–3 mo, with or without cyclosporine, depending on the clinical decision, and supportive therapy with specific diet, hepatoprotectants (silymarin, S-adenosylmethionine), probiotics, and vitamin supplementation. 15 Dogs that had an abnormal liver copper concentration, also underwent d-penicillamine therapy. 15 A follow-up SPE performed within 3–4 mo after the start of therapy was collected when available. The clinical history and hematobiochemical parameters of dogs at admission and during follow-up were also reviewed. Presence or absence of hyporexia or anorexia, vomiting, attitude and weakness, abdominal free fluid, and/or diarrhea were collected at admission and during follow-up. A dog was considered improved if there was a subjective reduction of admission clinical signs at follow-up.
Total serum protein (TSP), albumin and total globulin (Liasys; Assel), and SPE (Pretty; Interlab) were performed for each dog at the Laboratory of Clinical Pathology of the VTH. SPE was routinely performed using split beta agarose gel, with an automated platform using a specific kit (SRE602K; Interlab). SPE results were displayed on the manufacturer’s software (Elfolab; Interlab), and each fraction was separated into 6 bands: albumin, α1- and α2-globulins, β1- and β2-globulins, and γ-globulins. Each SPE fraction was considered decreased if their absolute value was below the RI. Increases in the γ-region (hypergammaglobulinemia) were classified as polyclonal gammopathy if there was a wide-based γ-globulin elevation, or as monoclonal gammopathy if the electrophoretic trace had a narrow, spike-like peak.4,14
Continuous variables were non-normally distributed, reported as median and range, and were compared between groups using the Mann–Whitney U-test. Continuous data were compared between A and F subgroups using the Mann–Whitney U-test. Gamma-globulins and the albumin:globulin (A:G) ratio were compared between admission (T0) and follow-up using the Wilcoxon matched-pairs signed-rank test. Statistical analysis was performed using statistical software (Prism, v.6; GraphPad), and significance was taken as p ≤ 0.05.
We included 26 dogs; median age was 9 y (range: 1–14 y). Nine dogs were intact males, 2 were castrated males, 6 were intact females, and 9 were spayed females. Included were 7 mixed-breed dogs, 4 Cocker Spaniels, 3 Labrador Retrievers, 2 Jack Russell Terriers, 1 American Staffordshire Terrier, 1 Poodle, 1 Standard Poodle, 1 Bernese Mountain Dog, 1 French Bulldog, 1 Dobermann Pinscher, 1 German Wirehaired Pointer, 1 Golden Retriever, 1 German Shepherd, and 1 Miniature Pinscher.
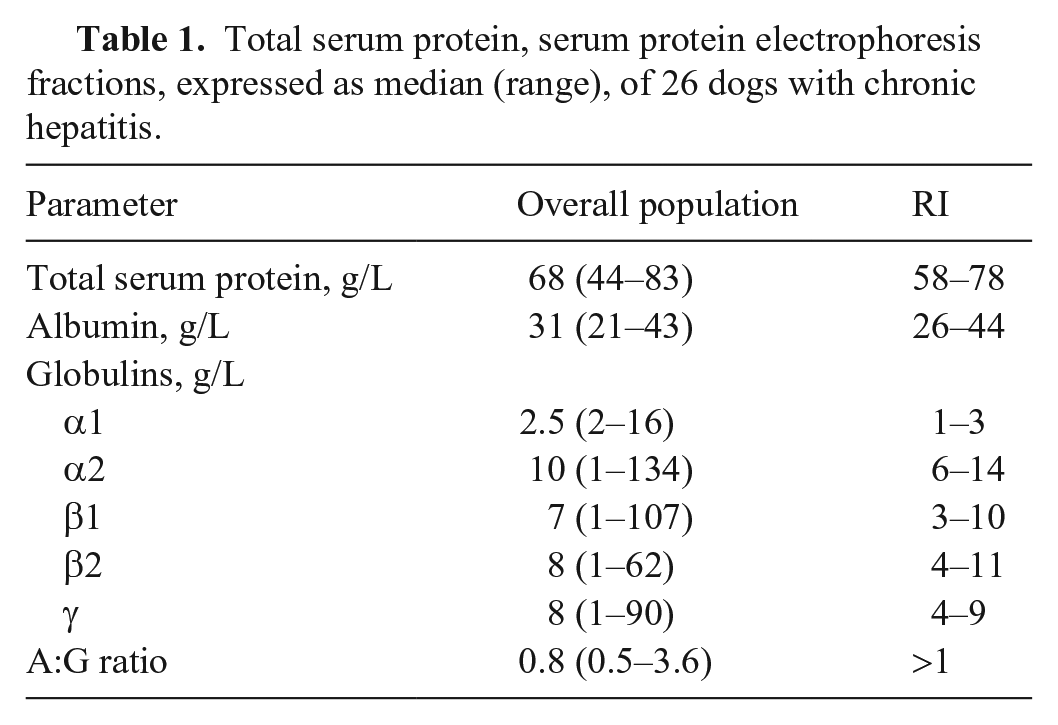
TSP was decreased in 8 of 26 dogs, increased in 2 dogs, and was in the RI in 16 dogs. The most common SPE alterations were decreased A:G ratio (n = 20) followed by hypoalbuminemia (n = 16), increased α1- and γ-globulins (both n = 11) and α2-globulins (n = 8), and increased β2-globulins (n = 7) and β1-globulins (n = 6; Table 1). Four dogs had β-γ bridging (Fig. 1). No dogs had a monoclonal gammopathy.
Total serum protein, serum protein electrophoresis fractions, expressed as median (range), of 26 dogs with chronic hepatitis.
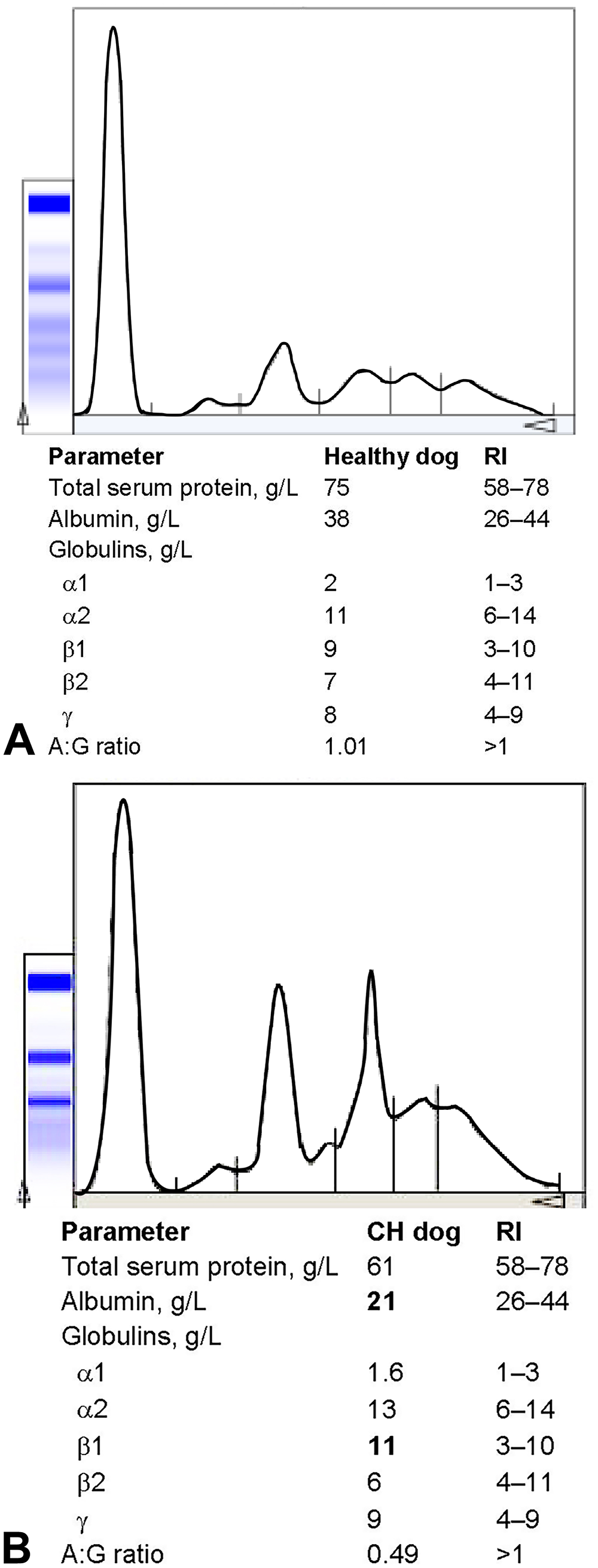
Serum protein electrophoresis.
According to the necroinflammatory grading scheme, 10 liver biopsies were placed in group A0-1, and 16 in group A>1. Beta2-globulins were significantly higher in group A>1 compared to group A0-1 (p = 0.016). According to fibrosis staging, 16 samples were included in group F<2, and the remaining 10 in group F≥2. Beta2-globulins were significantly higher in group F≥2 compared to group F<2 (p = 0.02). No other differences in SPE fractions were detected between A and F subgroups. No differences in absolute concentration of SPE fractions were highlighted between the subgroup classes within the A and F groups.
Fifteen dogs had follow-up SPE performed 3–4 mo after the first visit. Based on medical records, 4 of 15 dogs were classified as not improved, whereas the remaining 9 had improved. Twelve of 15 dogs had a decrease in γ-globulins (Fig. 2A) and an increase in the A:G ratio (Fig. 2B), although, there were no significant differences (all p > 0.05). The 3 dogs that did not have a decrease in γ-globulins did not have improvement in clinical parameters. In addition, 5 of 6 dogs that had increased β2-globulins at admission had a decrease at the follow-up SPE.
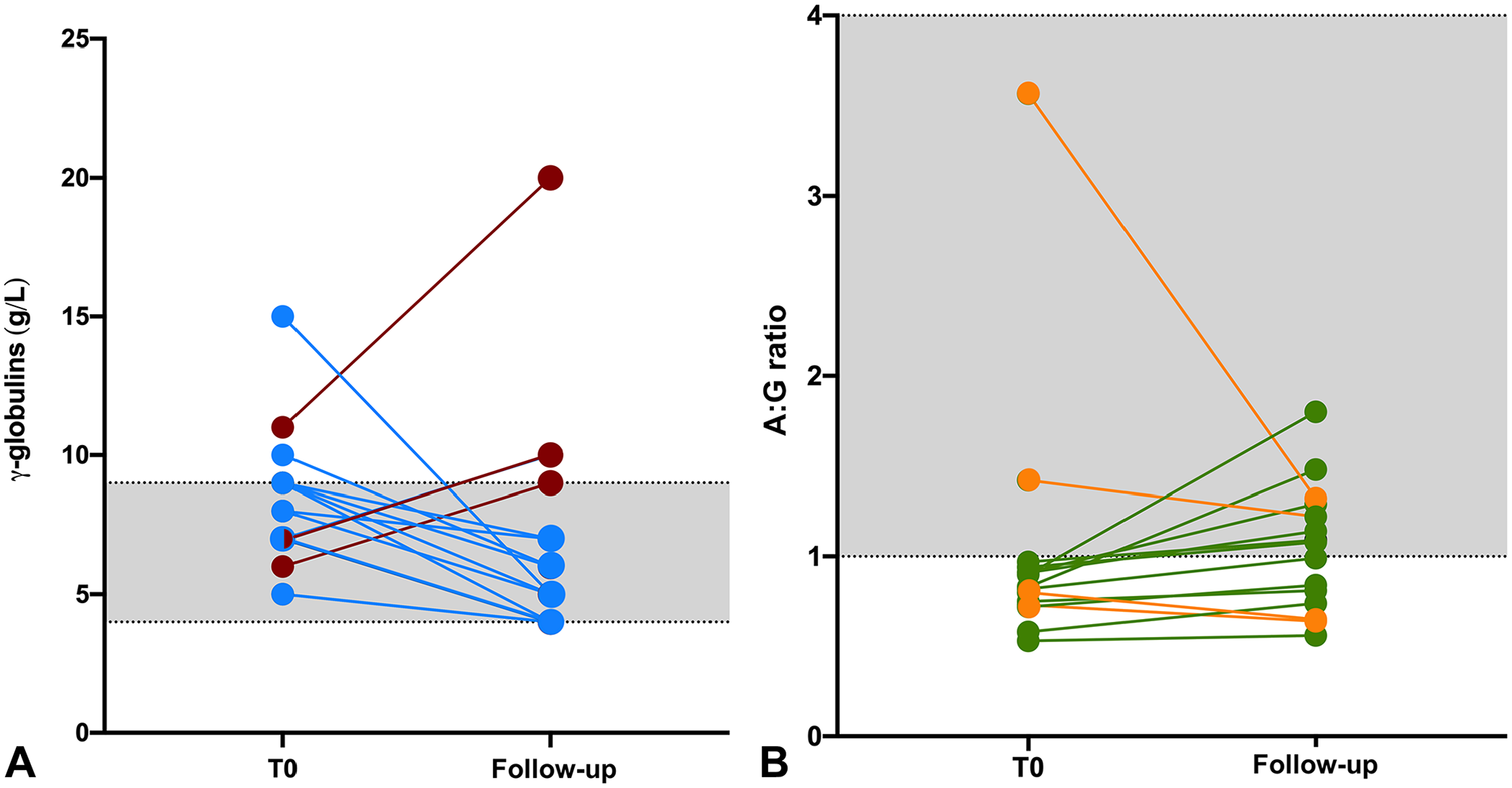
γ-globulins and albumin:globulin (A:G) ratio at admission (T0) and in follow-up serum protein electrophoresis (SPE; performed within 3–4 mo after the start of therapy) of 15 of 26 dogs with chronic hepatitis.
In human patients that develop CH and liver cirrhosis, a decrease of albumin and α2-globulins and a polyclonal increase of γ-globulins is often seen, and β-γ bridging may be present. 14 The most common SPE alterations reported in dogs with CH are decreased albumin and increased α- and γ-globulin concentrations.9,10 Hypoalbuminemia was the most common alteration in SPE of dogs in our study. Given that the liver is the site of albumin synthesis, liver diseases, such as CH, may cause downregulation of albumin synthesis and a shift to globulin production, resulting in hypoalbuminemia.2,4,11 In our population, the median albumin concentration was lower than the median concentration of globulins; hence, A:G ratios were low.
Increases in α1- and α2-globulins were common in cases of CH as well, affecting 11 dogs. The α region is composed of some APPs that can be elevated in dogs with both systemic inflammation and hepatic disease. 9 In our study population, the increase of APPs could be associated with chronic-active hepatitis or with acute hepatitis. 4
Eleven of 26 dogs in our study had hypergammaglobulinemia. Elevations in the γ-region in dogs are described as very common in inflammation, and in CH in particular.4,9–11 Chronic inflammation caused by chronic antigenic stimulation can lead to the production of immunoglobulins, which migrate in the γ-globulin region and produce a polyclonal peak. 4 All dogs included in our study that had increased γ-globulins had a polyclonal peak. In humans, the immune-mediated etiology of CH has been widely studied, 8 and it has been proposed also in dogs; however, there is no test to confirm this hypothesis. 15
Beta-globulins were elevated in 13 dogs, and their increase can be caused by both systemic inflammation and hepatic disease.4,12 According to human and veterinary diagnostic pathology textbooks, β-γ bridging is considered “virtually pathognomonic” for chronic-active hepatitis or hepatic cirrhosis.4,11,14 A 2010 veterinary study that included 11 dogs, 11 cats, and 3 horses showed that β-γ bridging did not have a strong predictive value for hepatic disease in dogs, cats, or horses, concluding that β-γ bridging should not be considered pathognomonic for hepatic disease in those species. 1 Our finding of 4 of 11 dogs with β-γ bridging is in line with work confirming that β-γ bridging may occur in dogs with CH, but is not pathognomonic. 1 Hypoalbuminemia and elevations in β- and γ-globulins as indicators of inflammation2,4,10,12 are supported by the γ-globulin and A:G behavior in the follow-up SPE in our cases. In fact, most of improved dogs had decreases in γ-globulins and/or an increase in A:G ratio in the follow-up SPE.
The histologic diagnosis of CH must include histologic scoring. 13 Given that there is no universally accepted scheme for canine CH histologic scoring, we used a modified Ishak histologic scheme, 14 as used in other studies.6,13,15 Beta2-globulins were higher in dogs with a necroinflammatory score >1 and fibrosis scores ≥2. The main proteins migrating in the β2-globulin region include complement and immunoglobulins, which could be the reason that dogs with elevated A and F scores had elevated β2-globulins. This evidence, in addition to γ-globulins and A:G ratio changes on follow-up SPE, encourage the use of SPE in monitoring CH, given that it is a less invasive method than liver biopsy. Nevertheless, other studies are necessary to validate the diagnostic and prognostic utility of SPE.
Our study has some limitations. The necessity of liver histology to reach a diagnosis of CH caused a restricted number of dogs to be included. Liver biopsy must be conducted following current guidelines, and biopsy cannot be applied to each dog with liver disease given the risks connected to the procedure. 15 Another limitation is the lack of comparison between SPE and other inflammatory markers (e.g., C-reactive protein), the production of which might also be affected by liver disease in CH dogs.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
