Abstract
We evaluated the effect of different storage conditions on the concentration of serum total proteins and their electrophoretic fractions in sheep. Blood samples were collected from 12 female Comisana sheep, and each serum obtained was divided into 4 aliquots. The first aliquot was analyzed immediately after separation (T0); the second was refrigerated at 4°C and analyzed after 24 h (T1), 48 h (T2), and 72 h (T3); the third was stored at −20°C immediately after centrifugation and analyzed after 2 days (T4); and the last aliquot was stored at −20°C after 24-h refrigeration and analyzed after 1 wk (T5). The concentrations of serum total proteins and their electrophoretic fractions (albumin, α-, β1-, β2-, and γ-globulins) was determined. A statistically significant (p < 0.05) effect of storage conditions on all serum electrophoretic protein fractions and on the albumin-to-globulin (A/G) ratio was found. The values of albumin, α-globulins, and A/G ratio increased at T4 with respect to T0. The levels of α-globulins were higher at T1 than T0; the β1-globulins levels changed at T3, whereas the β2- and γ-globulins values as well as the A/G ratio changed at T2 and T3. At T4, we found an increase in the serum concentrations of albumin, α1-globulins, and on the A/G ratio, and a decrease of γ fraction. At T5, the concentration of α-globulins was higher than at T0. Hence, we recommend electrophoretic analysis of fresh ovine serum samples or of samples refrigerated for no more than 24 h to obtain accurate data.
Clinical biochemistry investigations play an essential role in laboratory testing and provide important information for clinicians in the diagnosis, prognosis, screening, and monitoring of diseases. Many medical decisions are made based on laboratory data. 12 This information is useful only if laboratory results are accurate, relevant, and accepted by clinicians so that they can be used properly to guide clinical decision making. 15 The accuracy of test results is affected by many factors, primarily preanalytic variation, including sample collection and handling as well as biological factors including age, sex, pregnancy, and nutritional status of an animal. 8 In order to determine nonphysiologic changes in patients, preanalytic and analytic variation must be minimized in order to not have misleading effects on the results. According to standard blood-handling guidelines, plasma or serum should be separated from cells as soon as possible after clot formation is completed. Serum is the preferred sample in clinical laboratories for most routine biochemical investigations. 8 If it is not possible to examine samples immediately after centrifugation, samples must be stored properly. Inadequate storage of biological samples represents a potential source of preanalytic errors and may significantly affect the concentration of many biochemical parameters.7,9
Changes in serum protein electrophoretic fractions after storage of serum under different conditions have been studied in humans, cattle, and horses.2,4,6,16 However, the effect of sample storage on the concentration of serum proteins and their electrophoretic fractions is poorly documented in sheep.
Protein electrophoresis is the current reference method for the fractionation of serum proteins in clinical biochemistry. 10 Serum proteins are separated according to their molecular size and charge and then divided into 4 basic fractions, namely albumin, α-, β-, and γ-globulins.3,5 Identification and quantification of protein fractions assists in identifying animals with dysproteinemias and protein pattern abnormalities.1,14 We determined the effect of different storage conditions on the concentrations of serum total proteins and their electrophoretic fractions in sheep in order to define the useful length and temperature for serum storage.
Our study was carried out in September on a farm in northeastern Sicily. All animals were kept under natural photoperiod and ambient temperature, which on sampling days was 19–22°C, with a mean of 20°C, and a mean relative humidity of 76%. The farm produces sheep milk and lambs. The 12 clinically healthy Comisana ewes studied (3–5 y of age, mean body weight of 50.0 ± 3.0 kg) were free from internal and external parasites. The sheep were treated for endoparasites twice a year. Their health status was evaluated based on rectal temperature, heart rate, respiratory profile, appetite, fecal consistency, and hematologic profile. No disease events were reported during the study period.
Our study was performed during the official routine veterinary inspection of the farm. Protocols of animal husbandry and experimentation were reviewed and approved in accordance with the standards recommended by the Guide for the Care and Use of Laboratory Animals and Directive 2010/63/EU for animal experiments.
Animals were housed in a barn with an outdoor pen and were fed a balanced ration. The ewes were grazed daily on an improved natural pasture from 10:00 to 18:00 h.
Ten milliliters of blood were drawn from each animal by direct jugular venipuncture into vacuum tubes containing clot activator (Terumo, Tokyo, Japan). Blood samples were collected from each animal by the same operator before feeding. Blood samples were allowed to clot for 20 min at room temperature and centrifuged at 2,325 × g for 15 min. The serum obtained, neither hemolyzed nor lipemic, was fractionated into 4 aliquots. One aliquot was analyzed immediately after centrifugation (T0). The second aliquot was refrigerated at 4°C and analyzed after 24 h (T1), 48 h (T2), and 72 h (T3). The third aliquot was stored at −20°C immediately after centrifugation and analyzed after 2 days (T4), and the last aliquot was refrigerated at 4°C for 24 h, then stored at −20°C, and analyzed after 1 wk (T5).
The serum total protein concentration was determined by the biuret method using a commercial kit by means of an automated ultraviolet–visible spectrophotometer (Model Slim, SEAC, Florence, Italy). Electrophoresis was performed using an automated system (Sel Vet 24, SELEO Engineering, Naples, Italy) according to the manufacturer’s procedures. For each sample, 25 μL of serum were applied to numbered sample wells in an acetate cellulose film. Each holder accommodated up to 24 samples. Films were electrophoresed for 28 min at 180 V. After electrophoresis, films were simultaneously fixed using an automated system, stained with red stain acid solution (Ponceau S 0.2%, trichloroacetic acid 3%) for 10 min, and then dried at 37°C. After de-staining in acetic acid and drying completely for 15 min, films were scanned on a densitometer, and electrophoretic curves plus related quantitative specific protein concentrations for each sample were displayed, using computer software (Sel Vet 24, SELEO).
For assay precision, all serum samples were tested in duplicate by the same operator, and the reproducibility between runs was assessed by testing the same sheep serum sample at 4 different times within 24 h. The intra-assay coefficient of variation was <6% for all measured parameters. All samples were analyzed by the same operator, who determined the lines separating fractions in the densitometer tracing. Relative protein concentrations within each fraction were determined as the optical absorbance percentage, and absolute concentrations (g/L) were calculated using the total protein concentration. The major protein fractions were divided from cathode to anode, as albumin, α-, β1-, β2-, and γ-globulins. 13
Statistical analysis of each parameter was based on the average values obtained. Data were normally distributed (p < 0.05, Kolmogorov–Smirnov test). One-way repeated-measures analysis of variance, followed by Dunnett post hoc comparison test, was applied to determine the statistical effect of storage conditions on studied parameters. Values of p < 0.05 were considered statistically significant. Data were analyzed using statistical software (LDT 5, GraphPad Software, Dan Diego, CA).
A significant effect of storage conditions was found on all serum electrophoretic protein fractions and on the albumin-to-globulin (A/G) ratio (p < 0.05; Table 1, Fig. 1). A higher concentration of α-globulins was found at T1 than T0 (p < 0.05). A decrease of β2- and γ-globulins was found at T2 (p < 0.05). The greatest numbers of parameters were altered at T3 and at T4. In particular, Dunnett posttest showed that at T3 the concentration of β1-, β2-, and γ-globulins was lower (p < 0.05) and the A/G ratio was significantly higher compared to T0 (p < 0.05). An increase in the serum levels of albumin, α-globulins, and of A/G ratios (p < 0.05), and a decrease of γ fraction were found at T4 compared with T0. At T5, the concentration of α-globulins was significantly higher compared with T0 (p < 0.05).
Serum total proteins and their electrophoretic fractions measured in fresh serum (T0), 24-h refrigerated serum (T1), 48-h refrigerated serum (T2), 72-h refrigerated serum (T3), serum stored at −20°C immediately after centrifugation and analyzed after 2 days (T4), and serum stored at −20°C after 24-h refrigeration and analyzed after 7 days (T5) obtained from 12 Comisana ewes.*
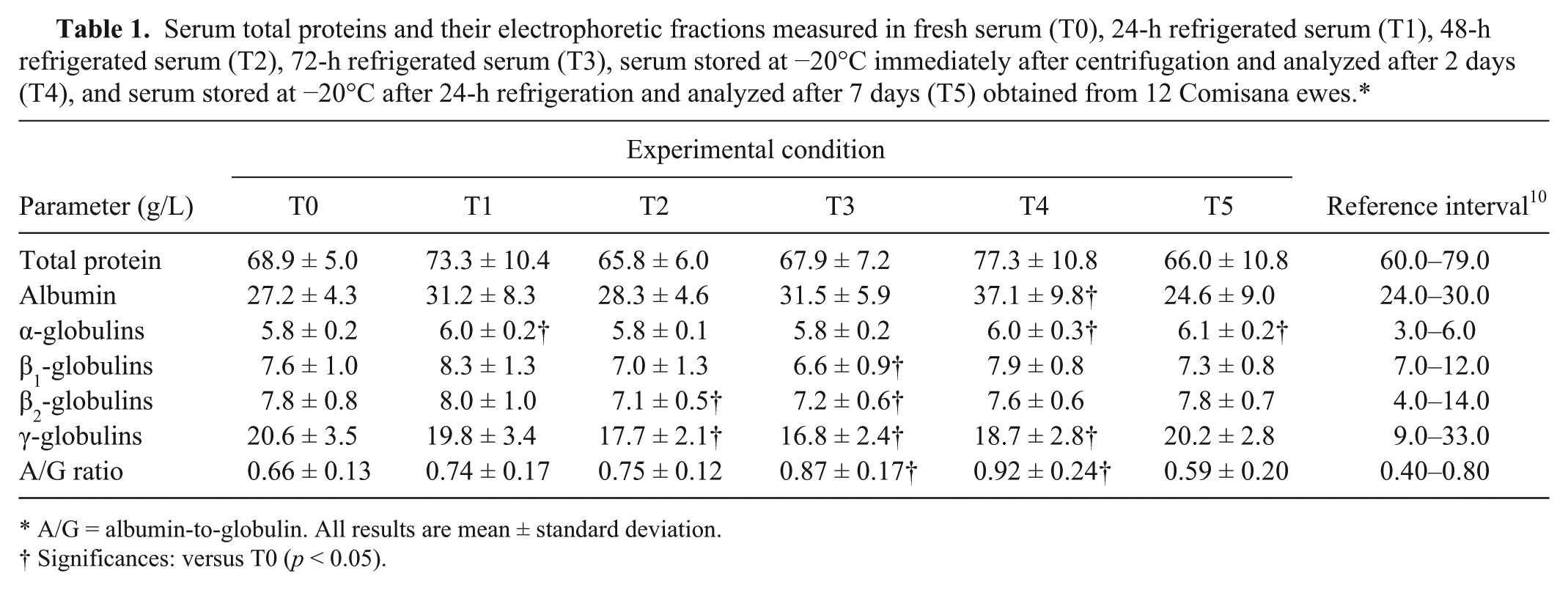
A/G = albumin-to-globulin. All results are mean ± standard deviation.
Significances: versus T0 (p < 0.05).
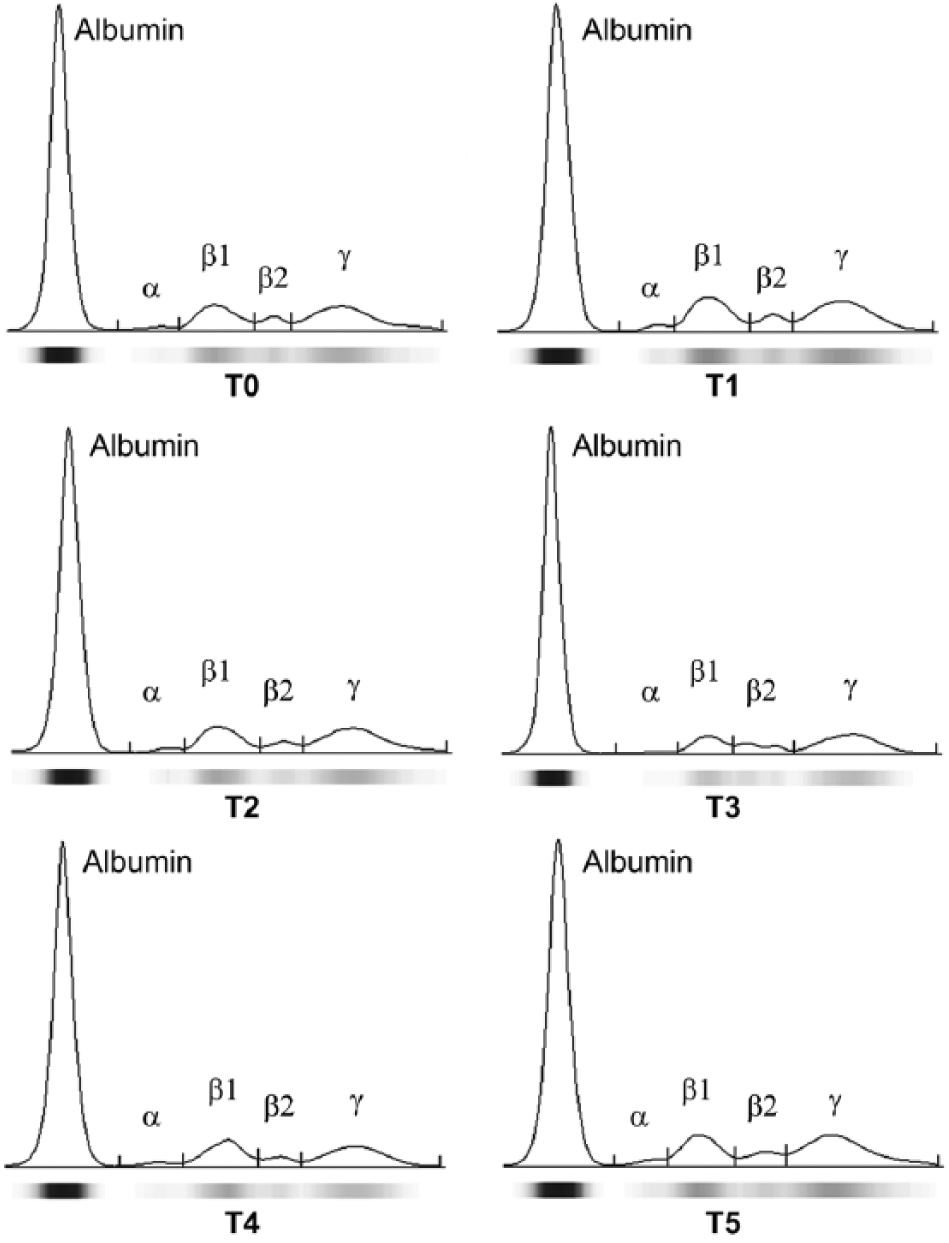
Representative protein electrophoretograms of Comisana ewes obtained from fresh serum (T0), 24-h refrigerated serum (T1), 48-h refrigerated serum (T2), 72-h refrigerated serum (T3), serum stored at −20°C immediately after centrifugation and analyzed after 2 days (T4), and serum stored at −20°C after 24-h refrigeration and analyzed after 7 days (T5).
Total protein values were not statistically significantly influenced by storage conditions in our study. The concentration of total proteins in sheep serum has been found to increase until the second week of storage at −20°C and then gradually decreases to the 26th week of storage. 11 A higher value of serum total protein has been reported in the equine species after short-term freezing compared with the value measured in fresh serum. 2
The results obtained in our study of ovine sera showed that all electrophoretic protein fractions, as well as the A/G ratio, were influenced by storage time and temperature. However, only the changes found in albumin and A/G values were clinically significant given that their values measured at T3 and T4 were outside the reference interval adopted in our laboratory. 10 According to studies carried out on human 8 and bovine 16 species, serum albumin concentration showed no significant change in serum refrigerated at 4°C. However, the values of albumin measured in serum stored at −20°C (T4) were higher compared with the albumin values obtained from fresh serum (T0). It is possible that there was loss of water from serum samples during freezing or thawing. This result agrees with previous studies carried out on humans 6 and equine 2 and bovine 16 species. Our findings suggest that the α-globulin fraction is relatively stable in serum refrigerated at 4°C. However, a significantly higher concentration of α-globulins was measured in serum stored at −20°C (T4 and T5) compared with values obtained from fresh serum (T0). Similar results were reported by studies carried out on equine and bovine species.2,16
The serum concentration of β1-globulins was statistically significantly lower after 72 h of refrigeration compared with fresh serum samples. The concentration of β2-globulins showed a statistically significant decrease after 48 and 72 h of refrigeration compared to fresh serum samples. The C3 factor of complement, which is located in the β fraction, has a short biological half-life. Therefore, it is the most labile component of this fraction, and its decay may account for the decreased concentration of β-globulins on the electropherograms. According to our results, both β-globulins are stable in serum samples stored at −20°C, both for 2 and 7 days and at 4°C for 24 h. Similar results were reported for human and bovine serum,4,16 whereas a study carried out on equine sera showed significantly higher values of β1- and β2-globulin fractions after freezing of serum. 2
A statistically significant effect of storage temperature and time was found on γ-globulins, which had lower values in serum samples stored at 4°C for 48 h (T2) and 72 h (T3) and in serum samples stored at −20°C for 2 days (T4), compared with values measured in fresh serum samples. The changes found in this serum protein fraction could suggest γ-globulin denaturation caused by excessive handling. 17 The modification of albumin and globulin fraction values obtained in refrigerated and frozen serum samples could explain the higher values of A/G ratio found in serum refrigerated at 4°C for 72 h and after freezing of fresh serum compared to the A/G ratio values obtained in fresh samples. The results obtained in our study confirmed that stability of serum protein electrophoretic fractions under different storage conditions is an important preanalytic factor. In particular, fresh serum samples or samples refrigerated for no more than 24 h are recommended for electrophoretic analysis in sheep in order to obtain accurate data, which can help clinicians to differentiate between sick and healthy animals.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
