Abstract
Senecavirus A (SVA) infection in pigs causes vesicular disease and results in a short viremia and transient shedding of the virus, mainly in oral fluids and feces. Here we describe the consistent prolonged shedding of SVA in the semen of 2 boars, and persistence of SVA within the tonsils and testes of 3 adult boars. Two SVA-infected boars that were identified on a Minnesota sow farm in 2017 shed SVA RNA in semen for >3 mo after an outbreak of vesicular disease had occurred on the farm. SVA was isolated from 1 semen sample collected 9 d after clinical disease began on the farm. The third SVA-infected boar was identified on an Indiana sow farm in 2020. All boars had SVA RNA detected in the testes and tonsils by RT-rtPCR, with lower Ct values obtained for the testes than from the tonsils. All boars had multifocal lymphocytic orchitis with segmental degeneration and atrophy of the germinal epithelium within the seminiferous tubules. One boar also had areas of seminiferous tubule collapse and interstitial fibrosis within the testes. In all boars, in situ hybridization demonstrated the presence of SVA mRNA within cells located basally in the seminiferous tubules of the testes, and within the basal surface epithelial cells, crypt epithelial cells, and subepithelial and parafollicular lymphocytes and histiocytes of the tonsil.
Senecavirus A (SVA; Picornaviridae, Senecavirus) is a picornavirus that infects swine and has been demonstrated to cause a vesicular dermatitis that is indistinguishable from other transboundary vesicular diseases, such as foot-and-mouth disease (FMD). In addition, SVA has been associated with increased neonatal mortality during the early stages of an epidemic. 15 SVA has a worldwide distribution, with infections in swine reported from Canada, the United States, Brazil, China, Colombia, Thailand, and Vietnam. 7 All ages of pigs have been reported to be infected during outbreaks, and although both male and female pigs are described as being affected, documentation of in-depth investigations into SVA infections in boars has been limited.
One of the earliest reports of vesicular disease in a U.S. pig that was associated with concurrent detection of SVA RNA was in a 6-mo-old Chester White intact boar that had been at the Indiana State Fair in 2010. 16 This boar developed dermal and oral vesicles and was euthanized because of progression of clinical signs. Various tissues were examined from this boar, but evaluations of the reproductive tract or semen were not documented. In a 2015 report of SVA-associated disease in a boar, semen was collected from a clinically recovered boar in Iowa ~20 d after SVA-associated disease manifested on the farm, and animals were tested for SVA. 1 The sperm-rich portion of the semen was positive for SVA by reverse-transcription real-time PCR (RT-rtPCR), with a cycle threshold (Ct) value of 34.4. Virus isolation for SVA from this semen sample was negative.
To our knowledge, there are no published reports related to SVA-positive semen causing disease; however, the presence of SVA in boar semen could present a biosecurity risk for the swine industry, given the reliance of many sow farms on externally sourced fresh extended semen for artificial insemination. This risk and overall knowledge gap of the manifestations of SVA infections in boars warrants further investigations of SVA infection in boars and SVA shedding in boar semen. We evaluated semen shedding in boars naturally infected with SVA and determined the tissue distribution of SVA within selected tissues of infected boars, including within the reproductive tract.
Materials and methods
Swine on farm 1, a 2,000-sow farrow-to-wean farm in Minnesota became infected with SVA in early January 2017. The farm experienced a sudden increase in neonatal mortality and diarrhea, which was followed by appearance of dermal vesicles and ulcers in sows and piglets. A foreign animal disease investigation of the site demonstrated that the vesicular fluid was positive for SVA and negative for FMD virus by PCR. Three boars used to elicit signs of estrus in gilts and sows (heat-check boars) were housed on this farm. Boars 1 and 2 developed snout vesicles, but boar 3, which was housed in nose-to-nose contact with the other boars, did not have dermal or oral vesicles observed during the reported time period. To control and eradicate SVA on this farm, the herd veterinarian instituted a load-close-expose protocol. Briefly, the farm received as many replacement gilts as space allowed and then stopped further animal introductions on the farm for 3 mo. The replacement gilts and any gestating sows without observed vesicles were exposed by nose-to-nose contact with boars 1 and 2, or given diluted vesicular fluid by intranasal administration. As part of the monitoring plan on the farm to determine when it would be safe to reopen the farm, testing was performed on samples from boars 1–3. Samples included gel-free semen, serum, feces, and oral swabs, which were obtained at various intervals throughout the herd closure period. Boars 1 and 2 were subsequently euthanized 92 d post-appearance (92 dpa) of the first vesicles on the farm. Postmortem examinations were performed, and tissue samples were collected from these 2 boars.
The second case was identified in 2020 in a heat-check boar on an Indiana sow farm on farm 2, which is in the same farm system as farm 1. Swine on the affected sow farm developed vesicular disease caused by SVA infection in early September 2020. On this sow farm, 9 boars from 4 different barns had initial samples of gel-free semen collected and tested 49 dpa. A second semen collection of 16 boars was performed 127 dpa. One boar (boar 4) was euthanized at 156 dpa, and tonsil and testis were collected and tested.
All animals were under the care of licensed veterinarians, and the farms were certified under the Pork Quality Assurance Plus guidelines (PQA Plus, Pork Checkoff and National Pork Board). The sampling and testing performed were client-requested tests.
Before testing, oral swabs were submerged in 2 mL of 1× PBS, vortexed for 20 s, centrifuged at 3,000 × g for 2 min, and the supernatant was used for testing. Similarly, feces (~4 g) was suspended in 4 mL of 1× PBS, vortexed for 20 s, centrifuged at 3,000 × g for 2 min, and the supernatant was used for testing. Tissue samples were suspended in an equivalent volume of 1× PBS, mechanically homogenized (Stomacher 80 microBiomaster; Seward) for 5 min, centrifuged at 4,000 × g for 30 min, and the supernatant was used for testing. Tissue samples were processed routinely for histopathology and in situ hybridization (ISH). Briefly, samples were fixed in 10% neutral-buffered formalin for 24 h, processed routinely, and embedded in paraffin.
RNA was extracted from swab supernatant, feces supernatant, tissue supernatant, serum, and semen using a magnetic particle processor (MagMAX Express-96 magnetic particle processor; Applied Biosystems) and a commercial extraction kit (Ambion MagMAX-96 viral RNA isolation kit; Life Technologies), following the manufacturer’s directions. The extracted RNA was tested for SVA RNA using a commercial TaqMan RT-rtPCR kit (EZ-SVA; Tetracore), run on a rtPCR machine (7500 real-time PCR system; Applied Biosystems), using the following cycles: RT cycle, 1 cycle at 48°C for 15 min; Taq-activation cycle, 1 cycle at 95°C for 2 min; denaturation cycle, 45 cycles at 95°C for 5 s; annealing and extension cycle, 45 cycles at 60°C for 40 s. Each test plate had a negative extraction control well, positive extraction control well, and each well contained an exogenous internal control. Wells with a Ct value <36.0 were considered positive. Wells with a Ct value ≥36.0 were considered negative.
Virus isolation was performed using the highly permissive human lung carcinoma cell line (NCI-H1299 CRL-5803; American Type Culture Collection [ATCC]) maintained in cell culture medium at 37°C with 5% CO2. The cell culture medium was a commercial medium (RPMI-1640 medium 30-2001; ATCC) supplemented with fetal bovine serum (10% by volume; HyClone), amphotericin B (amphotericin B solubilized powder, 0.00162 mM; Sigma-Aldrich), neomycin (neomycin trisulfate salt hydrate powder, 0.165 mM; Sigma-Aldrich), penicillin (penicillin G potassium salt powder, 764 mM; Sigma-Aldrich), and streptomycin (streptomycin sulfate salt, 0.624 mM; Sigma-Aldrich). Isolation was performed using 24-well plates with 70–80% confluent cellular monolayers. Samples were each inoculated in 2 wells: 1 well received 500 µL of undiluted sample and the second well received 300 µL of sample diluted 1:3 in cell culture medium. Semen samples were diluted 1:20 in cell culture medium prior to cell inoculation because of cellular toxicity when inoculated undiluted. Virus isolation plates were incubated with the inoculated samples for 1 h at 37°C with 5% CO2, washed 3 times with Hank balanced salt solution (HBSS 30-2213; ATCC), and then incubated with 2 mL of cell culture medium at 37°C with 5% CO2 for 7 d. Plates were examined daily with an inverted brightfield microscope (TMS; Nikon) for cytopathic effects (CPE), such as cellular swelling, lysis, or detachment. Two positive control wells, inoculated with a previously confirmed SVA isolate, and 2 non-inoculated negative control wells were included with each virus isolation plate. Virus isolation was confirmed in wells exhibiting CPE by indirect immunofluorescence. The immunofluorescence assay was performed on cytologic preparations placed onto 12-well glass slides, dried, fixed in acetone, incubated with porcine anti-SVA serum for 1 h, rinsed 3 times, incubated with FITC-labeled caprine anti-swine IgG (H+L) antibody (KPL antibody 5230-0297; SeraCare) for 1 h, rinsed 3 times, counterstained with Evans blue for 2 min, and then visualized with a fluorescence microscope (Leitz Laborlux S; Leica). Samples that exhibited immunofluorescence were considered virus-isolation positive. Each immunofluorescence assay slide included one positive control well and one negative control well. Samples not exhibiting CPE or that were negative on immunofluorescence after exhibiting CPE were passaged again, up to 5 times. Samples that did not exhibit CPE on the fifth passage were considered negative.
The presence of anti-SVA antibodies was assessed by an indirect immunofluorescent antibody (IFA) test, as described previously. 5 Briefly, wells on a 96-well plate of fixed NCI-H1299 cells infected previously with SVA were incubated for 1 h with 2-fold dilutions of serum from 1:40–1:320, rinsed, incubated with FITC-labeled caprine anti-swine IgG antibody for 1 h, rinsed, and then visualized with an inverted fluorescence microscope (Axiovert 40 CFL; Zeiss). Samples with wells exhibiting immunofluorescence at ≥1:40 dilutions were considered seropositive.
For histopathology, formalin-fixed, paraffin-embedded (FFPE) tissues were sectioned at 3 µm, mounted on glass slides, stained with H&E using an autostainer (Autostainer XL; Leica), and examined (by MJ Sturos and FA Vannucci) using a brightfield microscope (BX45; Olympus). Images were obtained from tissues exhibiting lesions using digitized slides (EasyScan Pro; Motic) and viewed on a digital slide viewer (ImageScope; Aperio). Tissues examined from boars 1 and 2 included heart, lung, liver, spleen, kidney, intestine, tonsil, testis, epididymis, ductus deferens, spermatic cord, vesicular gland, prostate gland, bulbourethral gland, penis, and prepuce. Tissues examined from boar 4 included testis and tonsil.
ISH for SVA mRNA was performed on sections of tonsil and testis, as described previously. 13 Briefly, FFPE tissue sections were mounted on glass slides, deparaffinized, treated with hydrogen peroxide, boiled in buffer, incubated with protease, and then incubated with a commercial ISH probe targeting the SVA VP1 mRNA and signal amplifiers that exhibit red coloring (RNAscope; Advanced Cell Diagnostics), counterstained with hematoxylin, and visualized by a veterinary pathologist by brightfield microscopy. Images were obtained as described for the histopathology images.
All sample processing and testing was performed at the University of Minnesota Veterinary Diagnostic Laboratory (St. Paul, MN, USA), an AAVLD-accredited laboratory, using validated tests and standard operating procedures.
Results
For farm 1, SVA was consistently detected in the semen of 2 boars by PCR (Table 1). Oral swabs and fecal swabs collected from boar 1 on 35, 55, 63, and 70 dpa and from boar 2 on 70 dpa were negative by PCR. One serum sample was collected from boar 1 on 55 dpa and was negative by PCR. On postmortem examination, there were no significant gross lesions in boars 1 or 2. Several tissues tested by PCR were positive (Table 2). For farm 2, semen from 4 of the 9 boars tested at 49 dpa was PCR-positive, with Ct values of 32.3, 33.1, 35.4, and 35.6. Semen samples collected at 127 dpa were all negative. Tonsil and testis from boar 4 were both positive (Table 2).
Senecavirus A (SVA) RT-rtPCR results for boar semen collected over time from heat-check boars on farm 1, a sow farm with swine infected with SVA.
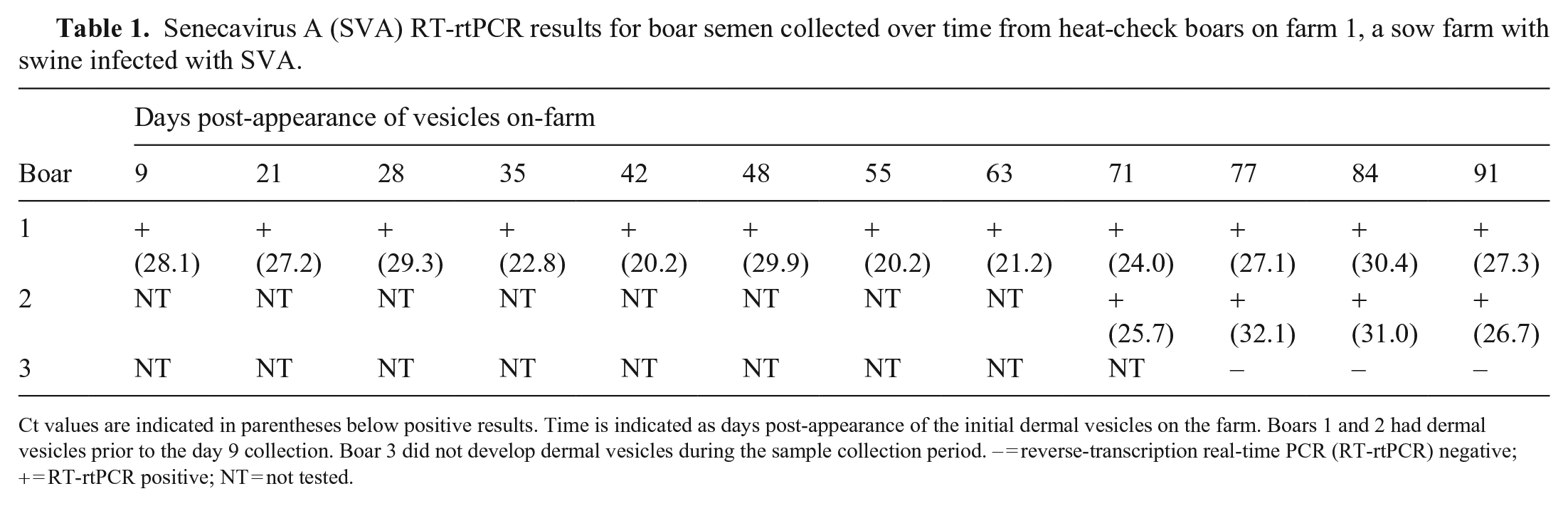
Ct values are indicated in parentheses below positive results. Time is indicated as days post-appearance of the initial dermal vesicles on the farm. Boars 1 and 2 had dermal vesicles prior to the day 9 collection. Boar 3 did not develop dermal vesicles during the sample collection period. – = reverse-transcription real-time PCR (RT-rtPCR) negative; + = RT-rtPCR positive; NT = not tested.
Senecavirus A (SVA) RT-rtPCR results for selected tissues from boars with SVA-positive semen.
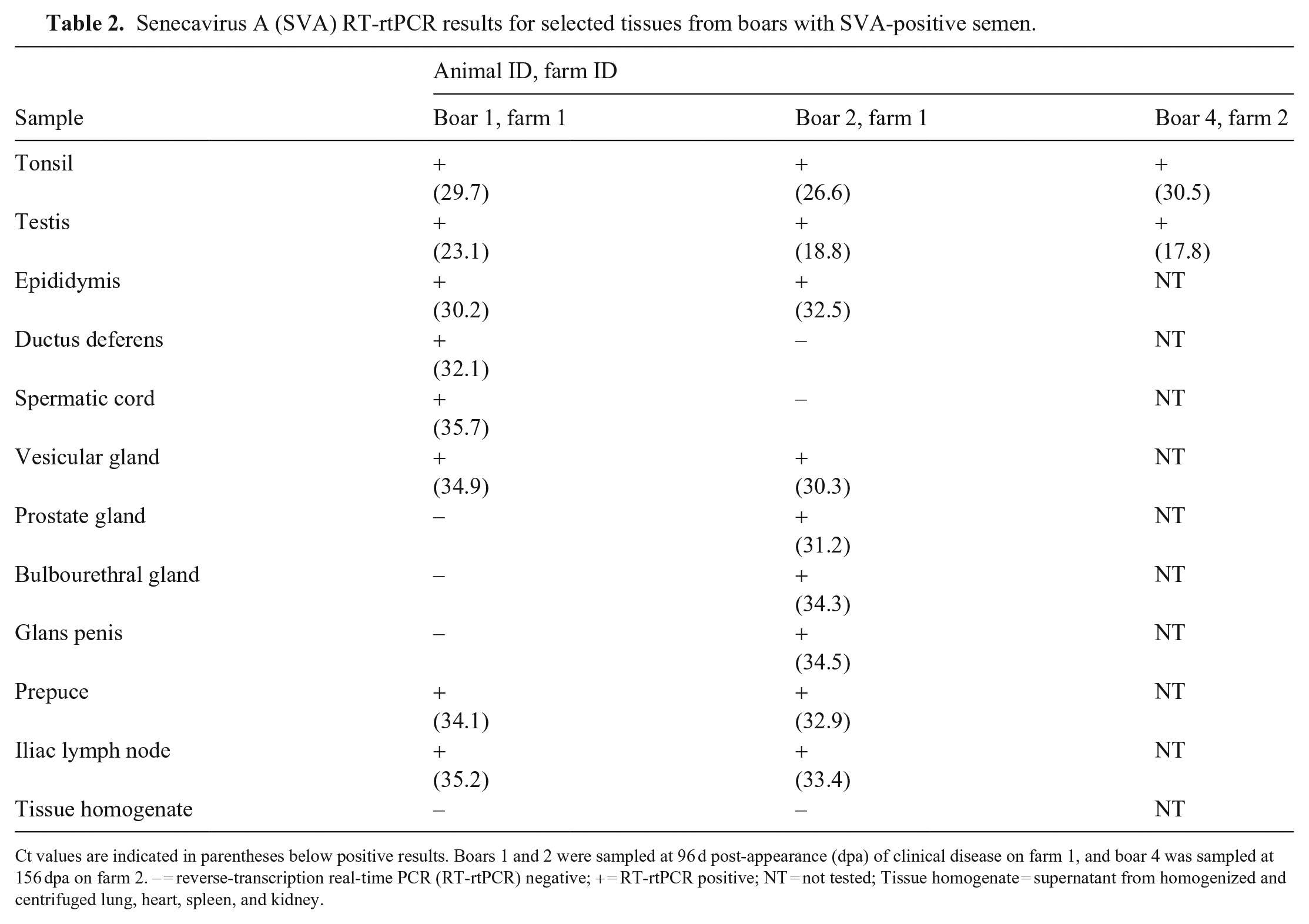
Ct values are indicated in parentheses below positive results. Boars 1 and 2 were sampled at 96 d post-appearance (dpa) of clinical disease on farm 1, and boar 4 was sampled at 156 dpa on farm 2. – = reverse-transcription real-time PCR (RT-rtPCR) negative; + = RT-rtPCR positive; NT = not tested; Tissue homogenate = supernatant from homogenized and centrifuged lung, heart, spleen, and kidney.
Virus isolation was successful after 2 passages in cell culture from 1 semen sample from boar 1, collected on 9 dpa. All other PCR-positive semen samples from boars 1 and 2 were negative after 5 passages. Virus isolation was also negative for all PCR-positive tissue samples that were collected during the postmortem examination of boars 1 and 2. Testis from boar 4 was negative after 3 passages. Virus isolation was not performed for the negative semen samples from boar 3 from farm 1 or any of the semen samples from farm 2.
Sera collected at 72 dpa from boars 1 and 2 from farm 1 were positive for SVA-specific IgG by indirect IFA at ≥1:80; serum from boar 3 from farm 1 was negative (<1:40). Sera were not tested from the boars at farm 2.
On histopathology, there were areas of normal testicular tissue (Figs. 1, 2); however, there was segmental degeneration and atrophy of the germinal epithelium within the seminiferous tubules and multifocal lymphocytic orchitis in the testes of all 3 boars that had postmortem examinations performed (Figs. 3–6). Maturation was disordered in degenerate germinal epithelial cells, and the germ cells were multinucleated in some areas and sloughing into the tubular lumina (Figs. 5, 6). Severely affected seminiferous tubules had complete atrophy or loss of the germinal epithelium, were lined only by Sertoli cells, and had empty dilated tubular lumina (Fig. 5). The interstitium surrounding the degenerate and atrophic tubules was infiltrated by lymphocytes (Figs. 3–6). Boar 4 also had lobules with collapse of segments of the seminiferous tubules that were devoid of both germinal epithelial cells and Sertoli cells. In these lobules, there was often interstitial fibrosis (Figs. 3, 4).
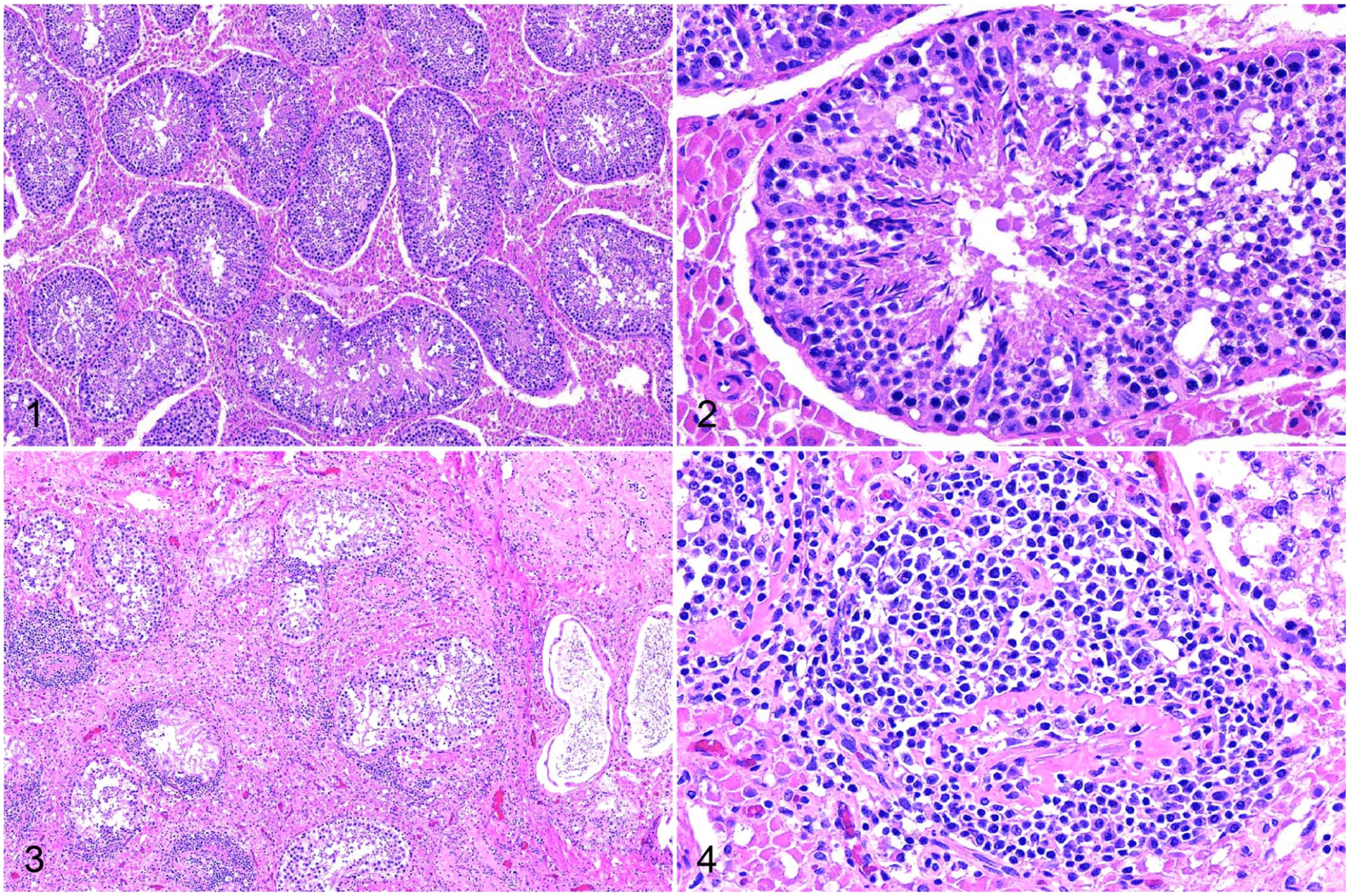
Senecavirus A infection of testes in a domestic pig.
Senecavirus A (SVA) infection of testes in a domestic pig.
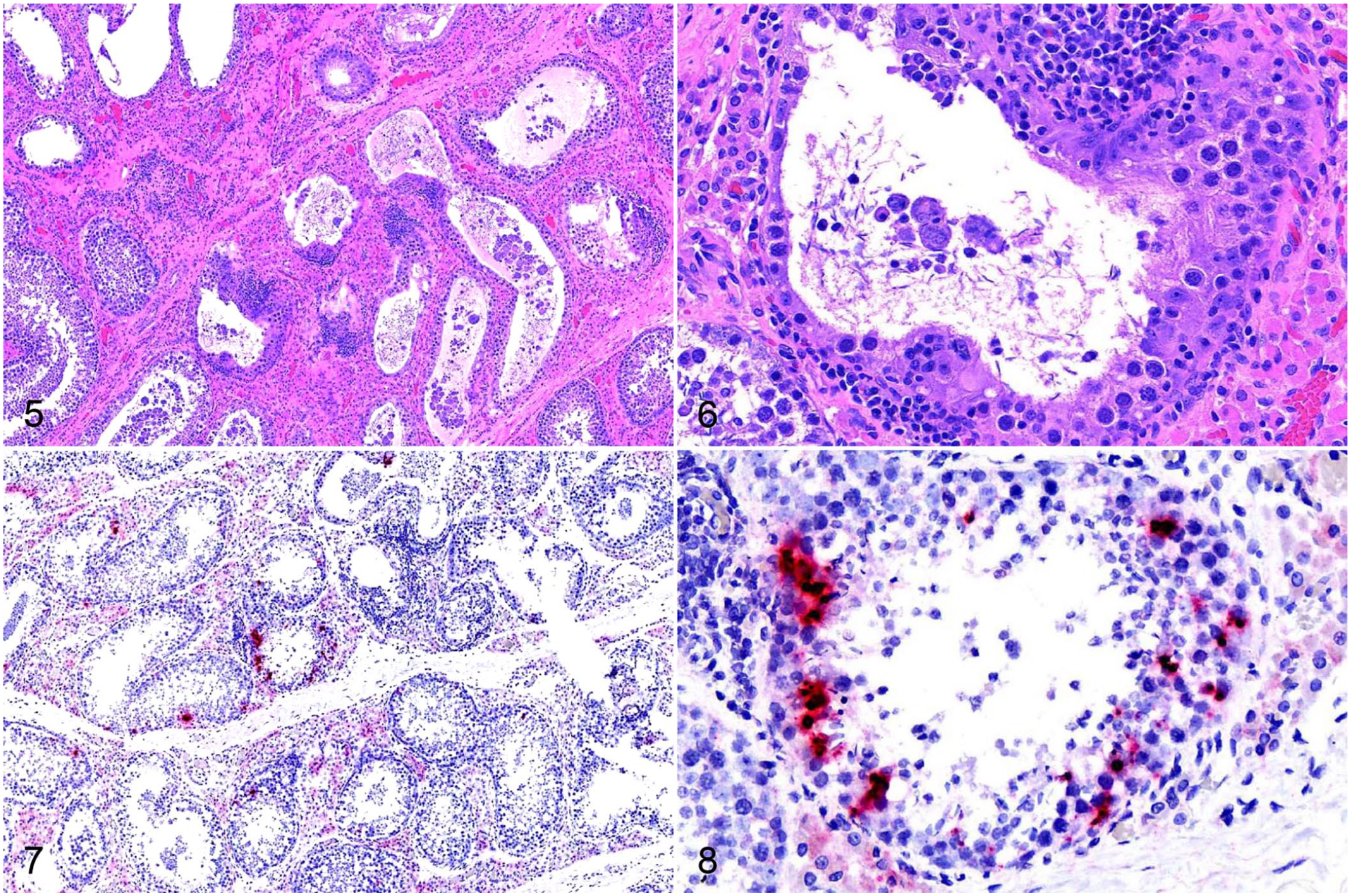
Within the testis, there was clumped-to-punctate red signal for SVA RNA within and adjacent to the seminiferous tubules (Figs. 7, 8). The signal in these areas was predominantly within the cytoplasm of cells located basally at intervals within the seminiferous tubules, interpreted most likely to be Sertoli cells, and occurred in both degenerate and non-degenerate segments of the tubules. Rarely, punctate-to-clumped positive SVA ISH signal was within cytoplasm of lymphocytes infiltrating the interstitium around degenerate tubules.
The palatine tonsils of all boars had numerous secondary follicles with prominent germinal centers, consistent with follicular lymphoid hyperplasia (Fig. 9). Within the tonsils, there was punctate and clumped cytoplasmic signal, predominantly in the lymphocytes and histiocytes within the parafollicular and subepithelial areas, as well as within the cytoplasm of some crypt epithelial cells and basilar surface epithelial cells (Figs. 10, 11).
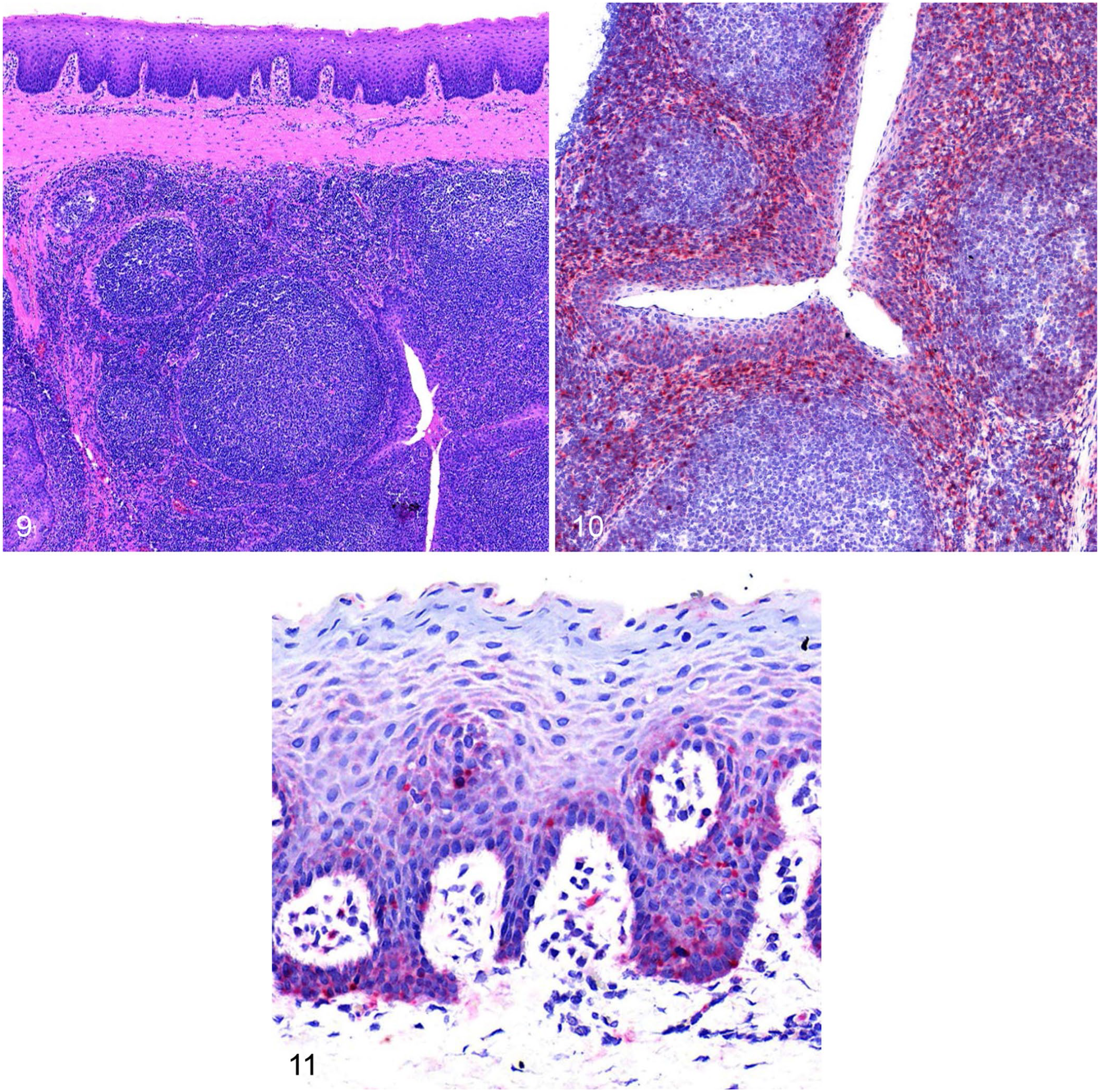
Senecavirus A (SVA) infection of palatine tonsil in boar 2.
Discussion
To our knowledge, the isolation of SVA from the semen of a naturally infected boar and the prolonged shedding of SVA RNA in boar semen, as well as the persistence of SVA RNA in the testis and tonsil of adult boars for up to 156 d after the occurrence of SVA-associated disease on the farms, have not been reported previously in pigs. These findings are vital for farmers and veterinarians when designing health intervention programs to effectively manage and potentially eliminate SVA transmission and persistence in the U.S. and global swine populations.
Although an earlier report documented the potential for SVA RNA to be shed in boar semen, 1 we have confirmed that the shedding of SVA in semen was not an isolated event but occurred on several farms that were spatially and temporally distant from each other. Additionally, for 2 of these boars, persistent shedding of SVA RNA was detected in the semen samples collected at regular intervals from the time of first test (9 dpa) until the time of euthanasia (96 dpa). This prolonged shedding in semen is much longer than the length of viremia as well as the time of shedding reported in oral or nasal secretions and feces of experimentally infected animals. In most experimental reports, viremia lasts 1–2 wk, and shedding in oral or nasal sections or in feces typically ceases 3–4 wk after infection, with peak viremia and shedding occurring in the first week.2,8,11 In contrast, the lowest Ct values in semen, corresponding to the highest amount of RNA present, were detected 6 and 8 wk after the appearance of clinical signs on the farm. Notably, semen shedding continued after the resolution of the skin vesicles and even while anti-SVA serum antibodies were present. Given these findings, the absence of shedding of SVA in semen in previously infected boars needs to be assessed by direct testing of the semen and cannot be extrapolated from the absence of vesicles or the presence of anti-SVA serum antibodies.
Interestingly, although RNA was detected persistently in semen by PCR, infectious virus was isolated on only the first sampling from one of the boars. In these investigations, infectivity was assessed by inoculations of diluted semen onto cell cultures. This is one of the limitations of our study because semen, particularly undiluted semen, commonly causes nonspecific toxicity and cell death in cultured cells. Dilution of the semen was necessary to maintain cell culture viability, but dilution likely resulted in reduced sensitivity of the procedure to detect infectious virus. It may be beneficial for future investigations into the infectivity of SVA in semen to include bioassays in pigs; however, this is not a test method currently in routine use at most veterinary diagnostic laboratories. There are several other potential reasons for the failure to isolate infectious virus from the remaining semen samples. One is that, if intact virions are being shed from the testis, they may become opsonized and neutralized by antibodies present in seminal plasma or are acted upon by other anti-viral factors, such as defensins or enzymes that abrogate their infectivity. 3 Another consideration is that the RNA detected may be from fragmented noninfectious virus or subgenomic nucleic acid that includes the target segment of the PCR assay.
The primary source of the SVA RNA being shed in the semen is not definitive, but seems most likely to have originated from the testes in these chronically affected boars. This is supported by the testes having the lowest Ct values of all tissues tested when compared to other reproductive and systemic tissues. The epididymis and vesicular glands were positive in both boars that had testing performed on these tissues; however, the Ct values were much higher than those obtained from the testes and could have been the result of the continuous presence (epididymis) or potential reflux (vesicular glands) of testicular-origin materials. The ductus deferens, prostate gland, bulbourethral gland, glans penis, and prepuce were also positive for at least one of the boars for which these tissues were tested, but the high Ct values and inconsistent detection suggest that these tissues are not significant contributors to the viral RNA detected in the semen. The origin or relative contributions of the viral RNA detected prior to euthanasia may have been different earlier in the course of infection and should not be directly interpreted from the tissue results obtained during the chronically infected state. For example, viral RNA or intact virions may have passed from serum into the semen through the accessory sex glands during the viremic phase of infection, or there may have been active replication within and shedding from the penile or preputial epithelium.
Numerous viruses have been isolated from or detected in boar semen, including porcine reproductive and respiratory syndrome virus (PRRSV; Betaarterivirus suid), African swine fever virus, classical swine fever virus (Pestivirus C), bovine viral diarrhea virus (Pestivirus), pseudorabies virus (Suid alphaherpesvirus 1), porcine cytomegalovirus, porcine parvovirus (PPV; Ungulate protoparvovirus 1), porcine circovirus, Japanese encephalitis virus (JEV), porcine rubulavirus (Porcine orthorubulavirus), and several picornaviruses such as FMDV, swine vesicular disease virus, and porcine enterovirus.4,6,9 Of the viruses detected in boar semen, fewer are known to be isolated from or infect the testis and include PRRSV, JEV, porcine rubulavirus, and PPV.14,17,18,20 SVA now needs to be considered along with these viruses as a differential for testicular infection based on our RT-rtPCR results from the homogenized testicular tissue supernatant and our SVA ISH results on the FFPE testicular tissue sections.
The fine cellular detail of the testicular tissue was not well preserved after tissue processing was performed as part of the ISH protocol, hence the specific cell types with positive ISH signal cannot be determined definitively. However, the ISH signal was present primarily within the seminiferous tubule, and most was within cells in the basal portion of the tubule, which suggests that either Sertoli cells or early-stage germinal epithelial cells were the cells supporting persistent infection in these boars. Although the anthrax toxin receptor 1 (ANTXR1) has been identified as a necessary cellular receptor for SVA infection of human neuroendocrine tumor cell lines and may be a candidate host receptor in other species, the expression of this receptor in pig tissues for productive SVA infection is uncertain and the levels of ANTXR1 expression on testicular cells is unknown. 10 Additionally, SVA has been shown to infect cells through the release of virions within exosomes from infected cells and subsequent infection of the target cell, which was not blocked by the presence of SVA-neutralizing antibodies. 19 Sertoli cells and the Sertoli-Sertoli cell junctions are vital to the maintenance of the blood-testis barrier and help to protect germ cells from immune attack as well as prevent testicular infections. 12 However, Sertoli cells internalize extracellular materials through endocytosis and hence they may be susceptible to infection through internalization of exosomes containing SVA virions, or by direct infection. Viruses that are able to replicate within the seminiferous tubules, as we have demonstrated, may occur during SVA infections in boars, would have the potential to infect susceptible cells anywhere along the affected seminiferous tubule. Additionally, the blood-testis barrier could contribute to the inability of the host immune system to clear the virus quickly and efficiently from the testis, which may be a contributing factor for persistent testicular infections and prolonged shedding in semen observed in these boars.
Numerous questions remain to be answered about SVA transmission and pathogenesis in pigs. Our findings indicate that boars and boar semen may play important roles in the maintenance and spread of SVA in the U.S. and global swine populations. Further areas of research should include experimental infection trials, which will facilitate more rigorous investigations into the potential for SVA transmission to gilts or sows through breeding activities, as well as allow more comprehensive evaluation of the pathogenesis of infection in mature boars under controlled conditions.
Footnotes
Acknowledgements
We thank the staff in the UMN-VDL Molecular Diagnostics section for their expertise and assistance with PCR testing. We thank Ms. Wendy Wiese and the staff in the UMN-VDL Virology section for their expertise and assistance with virus isolation and serology. We thank the New Fashion Pork animal caretakers and staff for providing exceptional animal care and collection of samples.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to research, authorship, and/or publication of this article.
Funding
Laboratory testing was client-funded, with partial support by the University of Minnesota Swine Disease Eradication Center.
