Abstract
Bovine respiratory disease complex (BRDC) is a common, serious problem in feedlot cattle worldwide. Early diagnosis and outcome prediction are critical for making decisions to prevent economic loss and to limit antimicrobial use. Diagnosing BRDC is commonly based on visual signs and behavioral changes; both assessments are considered to have low diagnostic accuracy. Biomarkers are important for supporting the diagnosis of BRDC, determining the necessity and potential outcomes of treatment, and assisting in research in which differentiating diseased animals is required. There are few reviews summarizing the biomarkers available and utilized. We systematically evaluated the detection and prognostic potential of biomarkers from the literature published between January 1990 and December 2020. We performed a descriptive analysis of 5 biomarker categories: acute-phase proteins, stress-related hormones, other blood biomarkers, omics biomarkers, and non-blood biomarkers. The retrieved articles consisted of studies or trials that assessed the detection value and treatment and/or outcome prediction efficacy of biomarkers for BRDC in feedlot cattle; 23 manuscripts for review and analysis satisfied the selection criteria. Based on our review, we cannot recommend a specific biomarker as the sole method for the early detection or outcome prediction for BRDC, given that the application and efficacy of biomarkers varies in different situations. Our systematic review may serve as a reference for clinical and research investigations of early detection and outcome prediction of BRDC.
Bovine respiratory disease complex (BRDC) is the leading cause of economic loss to the beef feedlot industry in the United States, estimated at >$500 million per year. 36 BRDC is a multifactorial disease process initiated by compounding stressful events or immunosuppression that predispose cattle to infection by viral and bacterial agents. The most common viral pathogens include bovine herpesvirus 1 (BoHV1; infectious bovine rhinotracheitis virus, IBRV; Bovine alphaherpesvirus 1), bovine viral diarrhea virus (BVDV), bovine parainfluenza virus 3 (Bovine respirovirus 3), and bovine respiratory syncytial virus (Bovine orthopneumovirus). 38 Bovine coronavirus may also play a role, based on some epidemiologic studies. 21 These viral agents can cause local respiratory and systemic disease, compromise the immunity of the bovine host, damage the epithelial lining of the respiratory tract, and interfere with the normal defense mechanisms of the respiratory tract. Secondary infection of the lower respiratory tract by commensal bacteria of the upper respiratory tract commonly results in severe bronchopneumonia. 15 The most cited bacterial pathogens involved in BRDC are Mannheimia haemolytica (MH), Pasteurella multocida, Histophilus somni (HS), and Mycoplasma bovis. 24
Early diagnosis of BRDC in feedlot cattle is an important step in planning appropriate interventions to prevent the progression of disease and economic loss. 15 A common practice for diagnosing and monitoring BRDC is the observation of clinical signs and behavior changes including depression, anorexia, respiratory abnormalities, and increased rectal temperature (DART). 50 Although DART has been used extensively for BRDC diagnosis, DART has limited sensitivity (Se) and specificity (Sp). 24 Identifying methods with increased Se and Sp for detection of BRDC in feedlot cattle can facilitate appropriate treatment of affected cattle, avoid treating cattle that do not require intervention thus decreasing the use of antimicrobials in unaffected cattle, reduce the economic impact of the disease, and provide improved information for research endeavors.
Combinations of different detection methods have been shown to significantly improve the early detection of BRDC.27,41,52 Various biomarkers have been investigated to determine their ability to predict BRDC occurrence, to classify calves as sick or healthy upon arrival, or to detect disease early in the feeding period.2,37 Additionally, biomarkers have been assessed regarding their reliability to determine the requirement for treatment and to predict BRDC outcomes in calves.2,9
Numerous reports have explored the response of acute-phase proteins (APPs), including fibrinogen (Fb), haptoglobin (Hp), and serum amyloid A (SAA), throughout the course of BRDC.5,10,57 In addition to APPs, a wide range of other biomarkers associated with inflammation and stress have been studied, such as L-lactate, 9 proinflammatory cytokines, and blood metabolites.6,11 Transcriptomic and metabolomic profiles, which are emerging techniques in biological research, have been investigated and provide more reliable and accurate detection methodology, as well as insights regarding the pathogenesis of BRDC.6,51
We systematically evaluated the detection and prognostic value of various biomarkers as published in the BRDC literature. Our assessment provides a descriptive analysis and reference for diagnosticians and researchers on the value of biomarkers in BRDC early detection and outcome prediction (multiple treatments or death) in feedlot cattle.
Methodologic review
Literature search, appraisal, and review process
Our systematic review was designed using the Preferred Reporting Items for a Systematic Review and Meta-analysis of Diagnostic Test Accuracy (PRISMADTA) studies methodology (Fig. 1). 40 To retrieve studies evaluating the utility of biomarkers in early detection or outcome prediction for BRDC, electronic searches were conducted using PubMed (https://www.ncbi.nlm.nih.gov/pubmed/), CAB (https://www.cabdirect.org/), and Agricola (https://agricola.nal.usda.gov/) databases. Relevant articles were also included by manually searching the bibliographies of retrieved articles from the electronic searches. Search terms were developed based on the 3 main concepts of “biomarkers,” “diagnosis,” and “bovine respiratory disease complex (BRDC)” (Suppl. Table 1). Three authors appraised and reviewed the literature based on 5 selection criteria (Suppl. Data). We retrieved and selected 23 English journal articles published between January 1990 and December 2020 for their relevance and accuracy in the early diagnosis and outcome prediction for BRDC in feedlot cattle. The relevant information of each article was extracted and summarized (Table 1; Suppl. Table 2). The studies were grouped into the following categories for description and analysis: APP biomarkers, stress-related hormones, other blood biomarkers, omics biomarkers, and non-blood biomarkers. The high variability of study designs, laboratory methods used to measure biomarkers, reported outcomes, and criteria used to define BRDC-positive animals limited our study to a descriptive review.
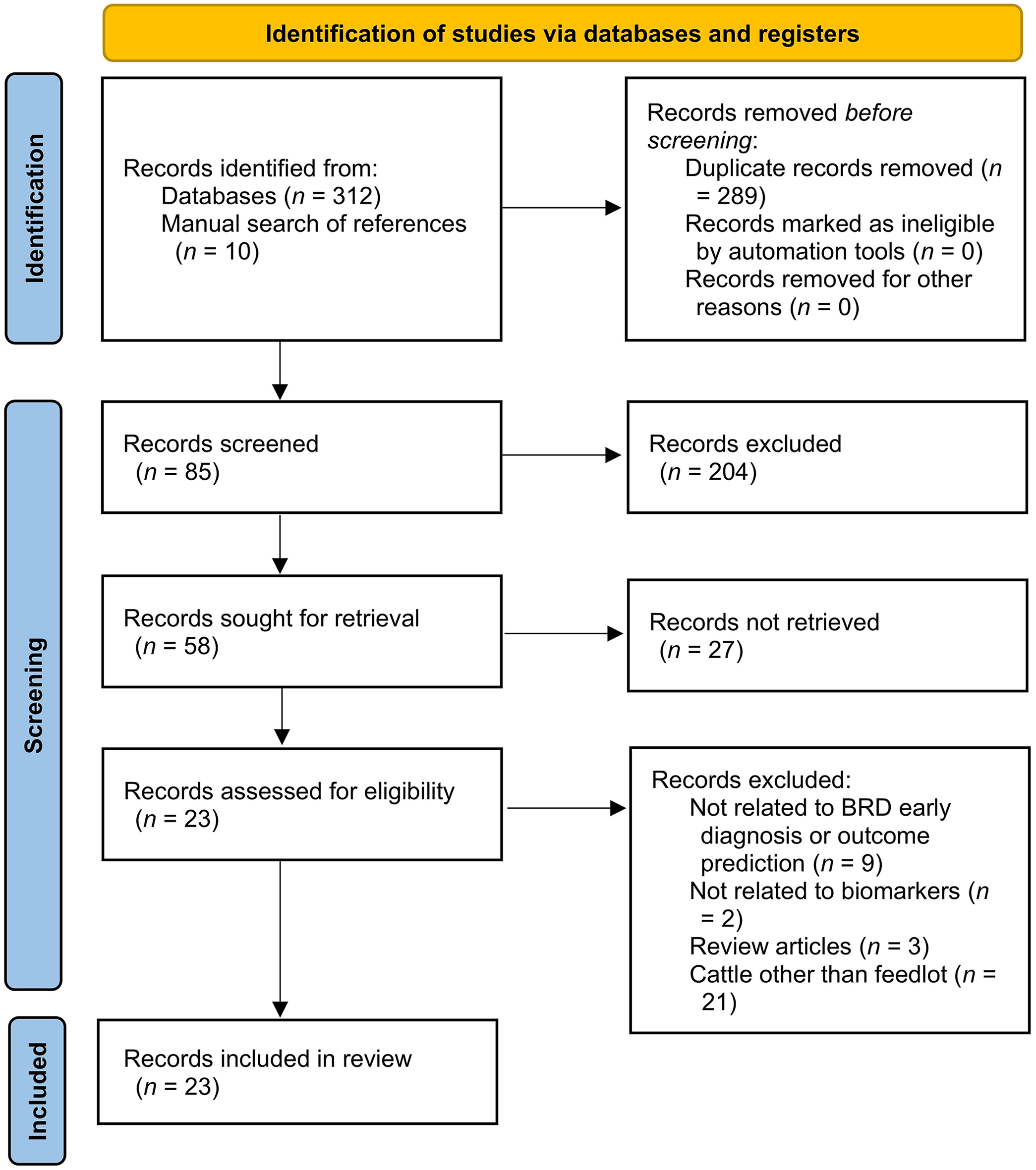
Stepwise literature exclusion of journal articles published in English between 1990 and 2020 that evaluated different biomarkers in bovine respiratory disease (BRD) complex early diagnosis or outcome prediction.
Summary of 23 selected studies of the use of biomarkers in the early diagnosis and/or outcome prediction of bovine respiratory disease complex (BRDC).
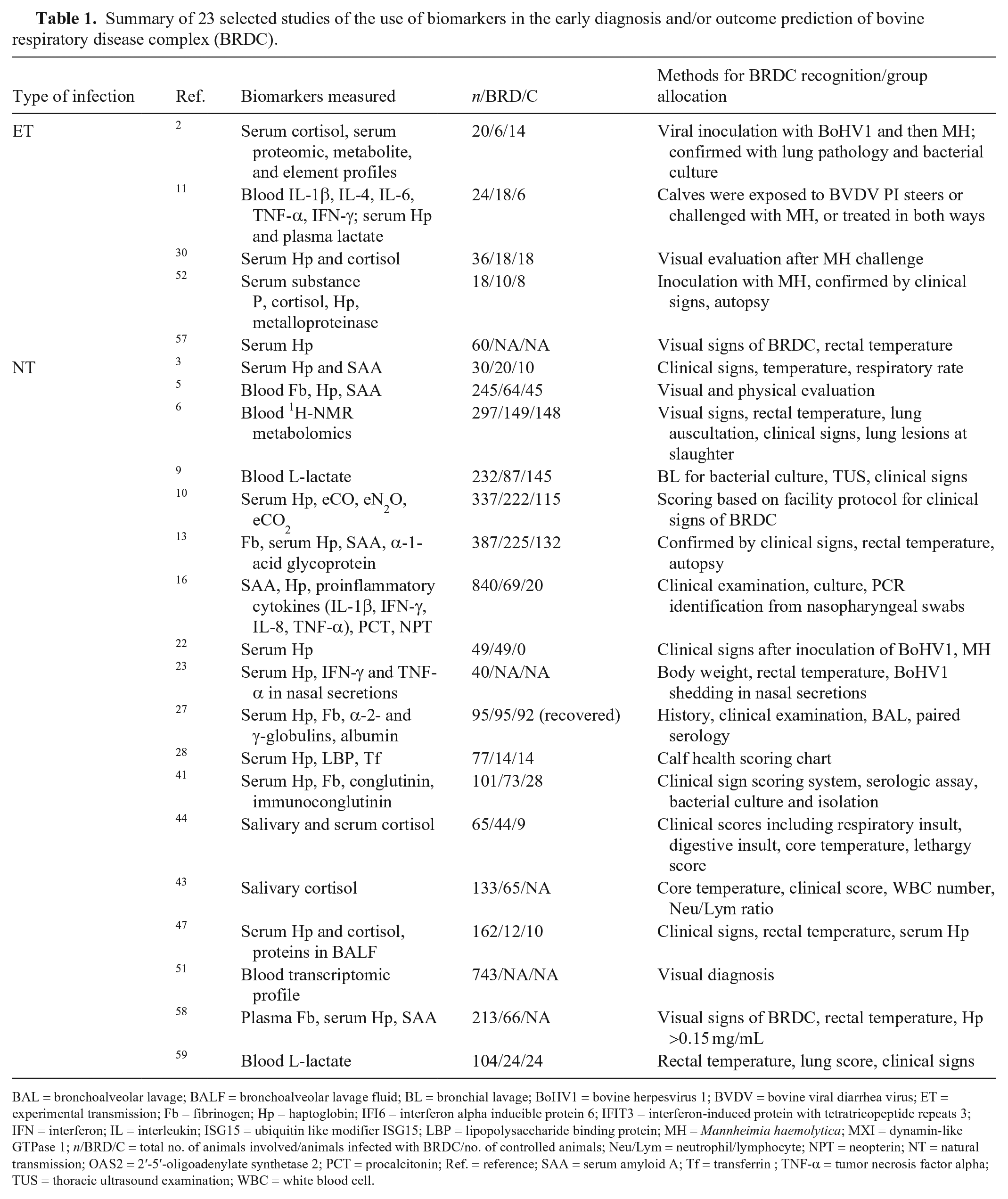
BAL = bronchoalveolar lavage; BALF = bronchoalveolar lavage fluid; BL = bronchial lavage; BoHV1 = bovine herpesvirus 1; BVDV = bovine viral diarrhea virus; ET = experimental transmission; Fb = fibrinogen; Hp = haptoglobin; IFI6 = interferon alpha inducible protein 6; IFIT3 = interferon-induced protein with tetratricopeptide repeats 3; IFN = interferon; IL = interleukin; ISG15 = ubiquitin like modifier ISG15; LBP = lipopolysaccharide binding protein; MH = Mannheimia haemolytica; MXI = dynamin-like GTPase 1; n/BRD/C = total no. of animals involved/animals infected with BRDC/no. of controlled animals; Neu/Lym = neutrophil/lymphocyte; NPT = neopterin; NT = natural transmission; OAS2 = 2′-5′-oligoadenylate synthetase 2; PCT = procalcitonin; Ref. = reference; SAA = serum amyloid A; Tf = transferrin ; TNF-α = tumor necrosis factor alpha; TUS = thoracic ultrasound examination; WBC = white blood cell.
Acute-phase protein biomarkers
Positive APPs are expected to increase during inflammatory conditions.29,48 Fb, Hp, and SAA are all positive APPs that are studied commonly in cattle with BRDC.
Haptoglobin
Hp, an α-2 globulin that has a bacteriostatic effect by binding hemoglobin, 53 has received the most study. In an experimental transmission trial, Hp increased significantly on day 4 (p < 0.05) after MH inoculation; an increase was not observed with BoHV1 inoculation on day 0. 2 In another experimental trial, a significant increase of Hp concentration (p < 0.001) was detected on the first day of MH challenge, which was 4 d after BoHV1 inoculation. 22 Other experimental transmission trials conducted with MH challenge also demonstrated significant increases (p < 0.01) of Hp concentration during the early stage of infection,11,30,52 which indicates the utility of Hp as an early biomarker of BRDC, especially for bacterial infection.
In clinical trials of naturally occurring BRDC, sick calves that required treatment had significantly higher concentrations of Hp than healthy calves.3,16,57 The Hp concentration was also positively associated with the severity of naturally occurring BRDC. Calves that required multiple treatments for BRDC had higher Hp concentrations than calves that needed only one treatment or did not require treatment.5,13 Additionally, BRDC calves had higher (p < 0.05) serum Hp concentrations during the course of disease than did calves that had recovered from BRDC,10,41 indicating the positive correlation of the Hp concentration with the severity of BRDC and its capability to predict disease outcome.
In a study with naturally occurring BRDC, a Hp concentration (0.15 mg/mL) was utilized as a component of the case definition of disease. BRDC-positive steers (n = 124) were tested using commercial ELISA kits within 35 d of arrival at the feedlot; 94% had a Hp concentration >0.15 mg/mL. 58 A cutoff concentration of Hp has yet to be determined; various values are reported in the primary literature. The reported concentration of 0.15 mg/mL lacks validation against control, BRDC-negative cattle, and thus cannot be supported as a definitive detection concentration. In another study, Hp concentration was quantified in cattle with clinical signs of BRDC.27,49 A threshold of 25 mg/L was chosen to help determine naturally occurring BRDC cases; Se was low (~60%), Sp was relatively high (>80%), and higher efficacy could be achieved when analyzed in conjunction with other biomarkers (Fb, α-2- and γ-globulins in the same study). 27 In contrast, Se and Sp were 92.9% and 85.7%, respectively, of Hp with a cutoff 0.81 mg/mL measured with ELISA kits in a study to evaluate different APPs (Hp, lipopolysaccharide-binding protein [LBP], and transferrin [Tf]) in feedlot calves with naturally occurring BRDC. 28 The different Hp concentrations examined might be the result of different kits or methods applied to measure Hp, different case definitions for inclusion, and different pathogens in the BRDC cases studied. Further refinement of the Hp cutoff concentrations between healthy and BRDC-positive cattle needs to occur to further elucidate the utility of Hp as a reliable detection biomarker.
Fibrinogen
Fb, another positive APP, has also been studied extensively in the detection of BRDC.5,13,41 Conflicting reports of Fb utility exist in the literature. In one study, Fb could be used to identify feedlot calves requiring multiple antimicrobial treatments for naturally occurring BRDC, 5 whereas Fb concentration was not correlated with the number of required treatments in another study. 13 Both studies applied a heat precipitation method to measure the Fb concentration. 8 Fb may not be a very sensitive biomarker to indicate the need for BRDC treatment when used alone, thus it is often used in combination with other biomarkers and clinical signs in BRDC diagnosis.3,5,13
Serum amyloid A
SAA is another positive APP that has been studied during inflammatory processes in cattle. 45 SAA has been evaluated predominantly in BRDC feedlot calves with natural transmission and is considered capable of modulating the immune system during tissue injury or infection.16,39 In one study, 24.4 μg/mL and 44.4 μg/mL were determined as thresholds of SAA for detection and need for treatment of BRDC, respectively, with Se and Sp >80%. 16 However, SAA concentration was not correlated with the number of treatments required for naturally occurring BRDC.5,13
Other acute-phase proteins
In a natural transmission study, LBP and Tf were measured along with Hp. 28 LBP is a class-1 APP 46 that presents lipopolysaccharide to monocytes, macrophages, and granulocytes. 17 The LBP concentration increased along with Hp in BRDC-positive cases, indicating the potential of LBP to aid in the detection of BRDC diagnosis under field conditions. An LBP cutoff of ≥0.33 μg/mL was used to detect BRDC-positive cases with 93% Se and Sp.
Tf is an iron-binding negative APP in plasma and is consumed in the acute phase of inflammation to inhibit bacterial iron utilization.18,29 Tf was not able to differentiate accurately between BRDC-positive and healthy calves.
Procalcitonin (PCT), another APP, has been used as a biomarker in bacterial and parasitic infections given that it increases rapidly with the induction of specific cytokines. 42 PCT was highly sensitive and specific in the diagnosis (Se = 100%; Sp = 96%) and prognosis (Se = 100%; Sp = 96%) of BRDC, especially when bacterial pathogens were present. 16
Stress-related hormones
Cortisol
Cortisol is a hormone regulated by the hypothalamic–pituitary–adrenal axis in response to stressful events. 54 Cortisol has been used as a common stress indicator for cattle upon arrival at a feedlot.44,52 In an experimental transmission study, the serum cortisol concentration increased on days 0.5 and 1 (p < 0.01) after MH inoculation, supporting a role as an early indicator of BRDC infection. 52 In another study, serum cortisol concentration increased significantly in calves with fatal BRDC experimentally challenged with BoHV1 and MH. 2 In a natural transmission study, the Se and Sp of serum cortisol were 100% and 54%, respectively. 44 Salivary cortisol concentration increased significantly (p < 0.01) 4–6 d before observation of clinical signs in steers with natural infection; however, there was not a significant difference between calves with BRDC signs and healthy calves.43,44 The Se and Sp of salivary cortisol to detect true-positive BRDC calves were only 70% and 53%, respectively. 43 Hence, cortisol was not found to be a useful biomarker in the diagnosis of BRDC.
Substance P
Substance P is a proinflammatory tachykinin peptide hormone that is secreted during painful or inflammatory conditions.12,14,34,52 Substance P concentration increased on day 0.5 after MH inoculation in an experimental trial. 52 However, substance P is not considered a specific biomarker of infection or inflammation given that it can change with numerous conditions or factors.
Other blood biomarkers
Proinflammatory cytokines
In addition to APPs, proinflammatory cytokines have been measured in some studies to facilitate BRDC diagnosis and outcome prediction.11,16 In an experimental transmission trial, interleukin-1 beta (IL-1β), IL-6, and tumor necrosis factor–alpha (TNF-α) concentrations increased significantly (p < 0.04) in calves exposed to BVDV persistently infected calves compared to BVDV-negative calves. 11 IL-1β, IL-6, TNF-α, and interferon-gamma (IFN-γ) concentrations increased significantly (p < 0.05) in MH-infected calves 72 h after inoculation compared to MH-negative calves. 11
A natural transmission study evaluated cytokines, including IL-1β, IL-8, TNF-α, and IFN-γ, in 69 feedlot calves. 16 IFN-γ, IL-1β, and TNF-α increased significantly (p < 0.05) at 72 h post bacterial infection. Likewise, IL-1β, IL-6, and TNF-α concentrations increased significantly (p < 0.05) with naturally transmitted BVDV. IL-1β, IFN-γ, and IL-8 had high diagnostic Se (>90%) and Sp (>90%); TNF-α had lower Se (85%) and Sp (85%) in calves with MH and/or HS infection. Among these proinflammatory cytokines, IL-8 was most accurate (high Se and Sp) in BRDC prognosis. Proinflammatory cytokines might be utilized with APPs to help detect BRDC.
Immune-related proteins
Neopterin (NPT) is produced by macrophages. 56 Increased concentrations of NPT potentially indicate inflammatory and cell-mediated immune responses that occur in BRDC. NPT increased significantly (p < 0.001) in MH- and/or HS-infected calves compared with healthy calves and calves that had recovered from treatment. 16 NPT also had high Se (100%) and Sp (90%) at a threshold of 4.2 ng/mL in the same study, which revealed its potential to monitor treatment response and prognosis of BRDC.
Other immune-related proteins investigated for the purpose of BRDC diagnosis in feedlot calves include conglutinin and immunoconglutinin. 41 Conglutinin is a β-globulin that mediates bactericidal activity against gram-negative bacteria. 32 Immunoconglutinin is an antibody that can agglutinate complement and enhance phagocytosis of bacteria.20,25 Blood samples were collected at 1, 8, 15, and 28 d after arrival at the feedlot. 41 The conglutinin concentration was not significantly different between healthy and sick calves as confirmed by clinical signs and pathogen identification. Significantly higher titers (p < 0.05) of immunoconglutinin were detected in calves treated for 6 d compared to calves treated for 3 d, with the titer peaking on day 15 during recovery, which indicates the potential of immunoconglutinin as a biomarker in BRDC prognosis.
Metalloproteinase, which is expressed by alveolar macrophages and responds rapidly to inflammation, is induced by endotoxin. 31 The metalloproteinase concentration was significantly greater (p < 0.01) in calves inoculated with MH on days 1, 2, and 3 compared to healthy calves 52 ; hence, metalloproteinase may be an early indicator of BRDC.
Lactate
Lactate is another analyte that has been used to monitor BRDC. Lactatemia was evaluated during the early feeding period of 232 steers. 9 When lactate concentrations were measured in serum over time; the hazard of death increased as the serum lactate concentration increased, based on the Cox proportional hazard survival analysis. 4 In one report, a lactate threshold of >5 mmol/L was selected as an indication for the use of tilmicosin. However, animal temperament influences lactate concentration given that both stoic and excitable animals had variations of lactate concentration. The reported threshold was most accurate in cattle with a normal temperament. Nonetheless, this threshold was implied to be useful as a guide to identify calves that were more likely to benefit from therapy. In another study, the blood lactate concentration, as determined by a hand-held lactate analyzer, was evaluated for its value in BRDC detection 59 ; the blood lactate concentration was significantly higher (p = 0.02) in BRDC calves than in healthy calves. The authors recommended that blood lactate could be utilized with other variables to improve the accuracy of BRDC identification and treatment evaluation. 59
Omics biomarkers
Proteomic, metabolomic, and transcriptomic profiles have also been analyzed in various studies to screen for substances associated with BRDC infection.2,6,51 These profiles assessed biologic and physiologic pathways during the course of disease. These omic assessment profiles might result in early detection and prediction of BRDC onset and outcome.
Metabolomics biomarkers
Proton nuclear magnetic resonance ( 1 H-NMR) spectroscopy for metabolomics analysis was conducted in an experimental BRDC model in which calves were inoculated with BoHV1 on day 0 and MH on day 4. 2 Concentrations of metabolites, including lactate and glucose, were significantly different between dead and survived calves on both days 0 and 4, which indicates the potential of metabolic biomarkers to predict BRDC outcomes.
In a 2020 study, 1H-NMR metabolomics was applied to identify the blood biomarkers that could be used to detect BRDC. 6 The study included 149 feedlot cattle with naturally occurring BRDC and 148 clinically healthy cattle. Biomarkers that were selected to classify BRDC-positive and BRDC-negative cases in this study included phenylalanine, lactate, hydroxybutyrate, tyrosine, citrate, and leucine. Blood metabolomics seemed to be more accurate, Se (>80%) and Sp (>85%), when used in conjunction with visual assessment and diagnosis of BRDC. Hence, blood metabolomics might be a useful method to detect BRDC.
Transcriptomics biomarkers
RNA-Seq based transcriptomics enables identification of expressed gene biomarkers associated with disease susceptibility. 7 In a 2020 study, transcriptomics was assessed as a potential tool in the early detection of BRDC. 51 Blood samples were collected at entry, treatment, and close-out stages, which represented subclinical, clinical, and healthy status. Key genes identified were OAS2, IFI6, IFIT3, ISG15, and MXI as excellent potential individual biomarkers to predict sick cattle upon entry (area under the receiver operator characteristic curve > 0.9).
Proteomics biomarkers
In one study, proteomics was performed by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS). 2 Only 2 APPs (Hp and apolipoprotein AI) had significantly increased concentrations (p < 0.05) on day 4 post viral (BoHV1) inoculation compared to the concentrations on day 0. 2 Further investigation of proteomic biomarkers is necessary to determine the value of proteomics in BRDC early detection.
Non-blood biomarkers
Given that BRDC is a disease complex with multiple etiologies, biomarkers from multiple organ systems have been explored. Breath analysis and serum biomarker analysis have been performed together in certain studies. In one study, breath analysis was conducted to determine the accuracy of exhaled gases for BRDC detection and outcome prediction among newly received feedlot cattle. 10 Exhaled CO2 (eCO2), eCO, and eN2O were measured to assess the detection potential of these gases, which are often related to proinflammatory effects 26 and may aid in the detection of BRDC. Cattle that remained healthy had a relatively constant ratio of eN2O:eCO2, likely reflecting a normal respiratory pattern. The eN2O:eCO2 ratio and eCO:eCO2 ratio increased in cattle that required more treatments, which may reflect the extent or severity of BRDC. These ratios may not be useful in the early detection of BRDC given that, in this study, samples were only collected at times of antimicrobial treatment. 10
Bronchoalveolar lavage and analysis of bronchoalveolar lavage fluid (BALF) is a common diagnostic procedure that provides information about the etiologic agent and animal response to respiratory infection. Annexin A1 and A2, detected in BALF, are expressed in epithelial cells of the bovine bronchus 55 in response to stress and glucocorticoids, 47 and are responsible for local immune response and fibrinolysis.19,33 Annexin A1 and A2 displayed a strong negative association with BRDC outcomes. 47 Concentrations were higher in calves that remained healthy throughout the study, which suggested a role in the pathogenesis, possibly by control of neutrophil infiltration and collateral lung injury. 47 Based on these results, annexin A1 and A2 are potential biomarkers in BRDC outcome prediction.
IFN-γ and TNF-α have been measured in nasal secretions to help monitor BRDC infection. 23 The concentration of TNF-α increased significantly (p < 0.05) on day 3 after BoHV1 inoculation in all calves; IFN-γ increased on day 4 and peaked on day 5 post-inoculation. BoHV1-challenged calves had higher proinflammatory cytokine concentrations when processed with different stressful events, such as transportation and weaning or combination of stressful events, than when processed with less or no stress. Hence, proinflammatory cytokine concentrations might help predict BRDC infection in calves upon arrival at the feedlot. 23
Biomarkers in BRDC early diagnosis or outcome prediction
Biomarkers that could be used in early diagnosis (Table 2) or outcome prediction (Table 3) were summarized. Hp,11,52 some proinflammatory cytokines (IFN-γ, IL-1β, and TNF-α), 11 serum cortisol, substance P, and metalloproteinase 52 demonstrated possible utility to detect early infection by bacterial BRDC pathogens. In other experimental trials, higher concentrations of IFN-γ and TNF-α in blood or nasal secretions were associated with early viral infection.11,23 Although experimental trials allow for early monitoring and tracking of the course of infection, natural infections may lead to different results because of unpredictable development of disease and complex mixtures of pathogens. 1
Summary of biomarkers in early diagnosis of bovine respiratory disease complex (BRDC).

BoHV1 = bovine herpesvirus 1; BVD = bovine viral diarrhea; ET = experimental transmission; Hp = haptoglobin; IL-1β = interleukin-1 beta; IL-4 = interleukin-4; IL-6 = interleukin-6; LPS = lipopolysaccharide; MH = Mannheimia haemolytica; NT = natural transmission; TNF-α = tumor necrosis factor–alpha.
Biomarkers with potential for bovine respiratory disease complex (BRDC) outcome prediction.
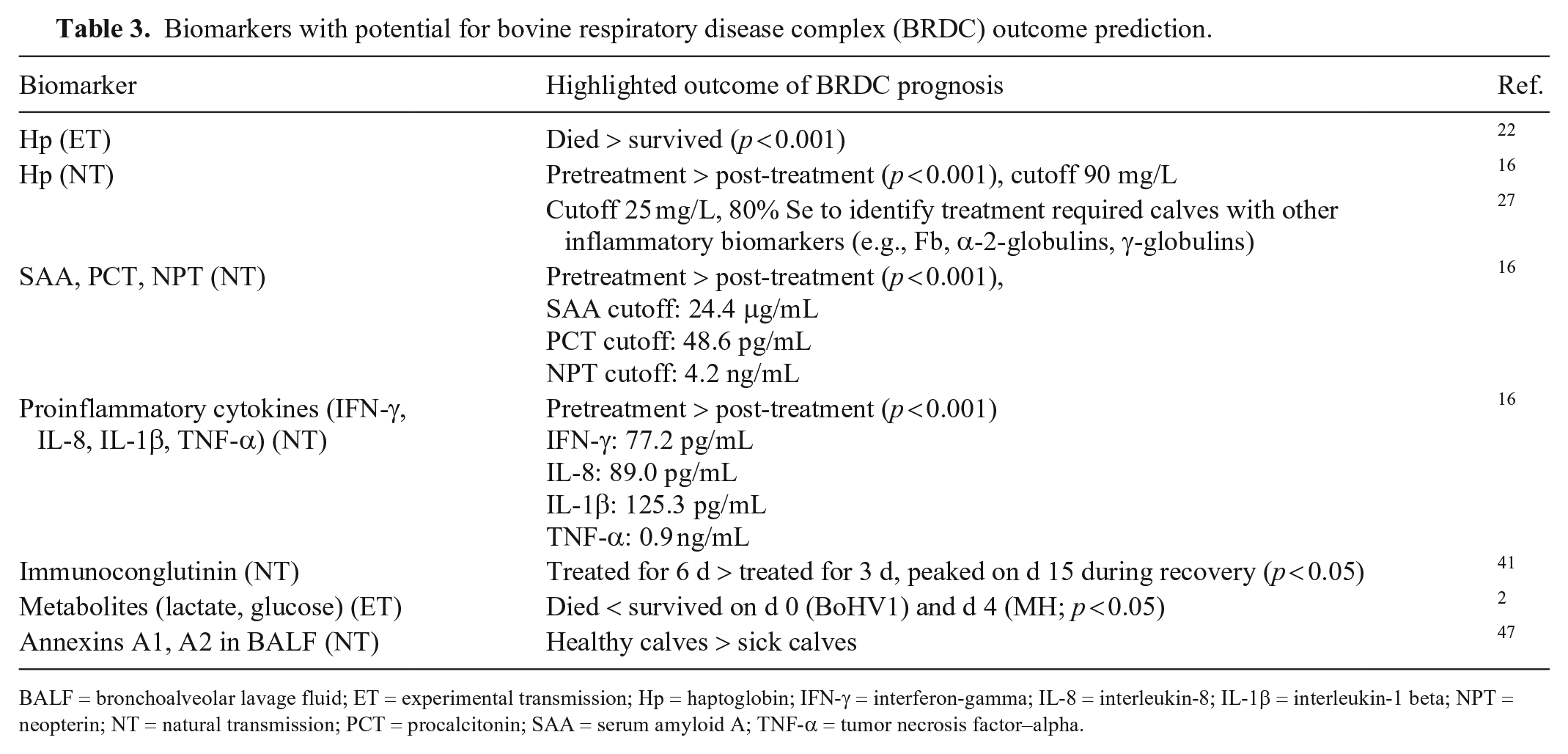
BALF = bronchoalveolar lavage fluid; ET = experimental transmission; Hp = haptoglobin; IFN-γ = interferon-gamma; IL-8 = interleukin-8; IL-1β = interleukin-1 beta; NPT = neopterin; NT = natural transmission; PCT = procalcitonin; SAA = serum amyloid A; TNF-α = tumor necrosis factor–alpha.
In studies focusing on naturally occurring infections, Hp, 41 SAA, PCT, proinflammatory cytokines, 16 metabolomics biomarkers, 6 and transcriptomics biomarkers 51 have been considered biomarker candidates for early detection of BRDC. It is generally recommended that SAA, proinflammatory cytokines, and PCT be used in combination with other methodologies to improve the accuracy of these test methods. 16 Hp might be used as a single biomarker to predict BRDC infection 28 ; however, different studies provided different cutoffs of Hp concentration,27,28,58 which confounds its utility as a BRDC diagnostic aid. Possible causes of the various Hp cutoffs may include the lack of an antemortem gold standard of defining and diagnosing BRDC, different study objectives, and different test methods. In general, combinations of biomarkers, such as APPs, proinflammatory cytokines, stress-related hormones, or immune-associated proteins, could increase the Se and Sp of early detection of BRDC.11,27,52
Additionally, biomarkers might also be used in outcome prediction of BRDC infection. APPs, 5 exhaled gases (eN2O:eCO and eCO2), 10 metabolites (lactate and glucose), 2 immune-related proteins, 41 proinflammatory cytokines (IL-8), 16 and annexin A1 and A2 in BALF 47 could be used to help predict the risk and outcome of BRDC. Higher concentrations of Hp have been associated with repeated treatments in various studies of naturally occurring BRDC.5,10,13 Decreased concentrations of annexin A1 and A2 have a strong association with increase susceptibility to pneumonia. 47 Further investigation of naturally occurring BRDC infection will be required to determine the prognostic value of exhaled gases, metabolite biomarkers, and immune-related proteins.
Proteomic, metabolomics, and transcriptomics have promising potential to provide more insight in early detection and outcome prediction of BRDC. Compared to metabolomic and proteomic profiles, transcriptomic biomarkers may serve as better indicators of early infection of BRDC by providing a transcriptional profile of BRDC infection stages. 51 Nonetheless, the 1H NMR metabolomics technique, as a new research modality, provides better accuracy to detect multiple blood biomarkers simultaneously. 35 Despite the great potential of the omics biomarkers in BRDC early detection, risk assessment, and outcome prediction, the technologies used for omics analysis are currently not clinically applicable given the complex and time-consuming methods employed. Currently, the omics biomarkers are mainly research tools used in the investigation of the biological responses to BRDC infection; they are not clinically applicable in BRDC diagnosis or outcome prediction.
Implications and limitations
Our review may serve as a resource regarding the potential methods available to detect or predict the outcome of BRDC. It is critical to realize the important limitation of using biological markers. For example, the general inflammatory analytes are not specific to BRDC, and therefore, may incorrectly classify the animals if other inflammatory processes are occurring. Further studies are necessary to assess the efficacy of biomarker combinations on early detection and outcome prediction of BRDC.
The most significant limitation of our review is the lack of a gold standard method for antemortem diagnosis of animals as affected or not by BRDC. One study provided APP reference intervals to detect BRDC, which many studies have since utilized as a parameter for BRDC detection. 27 Most prospective clinical trials rely on clinical signs, a diagnostic scoring system, and/or rectal temperature for BRDC diagnosis, which may affect the accuracy of animal selection and subsequently the results of those studies. Attrition bias, which occurred in several studies, may be a problem when small numbers of diseased animals die before the planned sampling dates. Our review design included clear inclusion criteria for literature selection to avoid publication bias. We only included studies that were published in peer-reviewed journals. Language bias could be a problem given that we reviewed only papers that were published in English. All biases undoubtedly had an impact on the findings of our review, and these challenges highlight the need for more standardized study design and reporting to maximize the quality of future research data.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387221081232 – Supplemental material for A systematic review of the utility of biomarkers as aids in the early diagnosis and outcome prediction of bovine respiratory disease complex in feedlot cattle
Supplemental material, sj-pdf-1-vdi-10.1177_10406387221081232 for A systematic review of the utility of biomarkers as aids in the early diagnosis and outcome prediction of bovine respiratory disease complex in feedlot cattle by Jing Li, Yiping Zhu, Brian Shoemake, Bo Liu, Pamela Adkins and Laurie Wallace in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
