Abstract
Although bovine respiratory syncytial virus (BRSV) infection has been reported in cattle in Argentina, it has not been associated with pneumonia in Argentina. We report here 5 cases of bovine pneumonia associated with BRSV. Autopsies were performed on 35 beef cattle with gross and/or microscopic lesions of pneumonia from 3 commercial feedlots. Lung samples in 5 of 35 animals were BRSV-positive by reverse-transcription nested PCR. The lungs of 2 of these 5 animals were coinfected with Mannheimia haemolytica, and 1 with bovine viral diarrhea virus 1. Microscopically, the lungs of 3 of the 5 BRSV PCR–positive animals had fibrinosuppurative bronchopneumonia, with or without pleuritis; 2 of the 5 had interstitial pneumonia. We conclude that BRSV is part of the bovine respiratory disease complex in Argentina.
Bovine respiratory disease complex (BRDC) is one of the most prevalent diseases in feedlot cattle. BRDC is responsible for significant economic loss in the livestock industry, including costs associated with prevention, treatment, poor growth, and death of animals.9,15 BRDC is caused by a variety of pathogens, including viruses and bacteria. The viruses include bovine herpesvirus 1 (BoHV-1; Varicellovirus bovinealpha1), bovine respiratory syncytial virus (BRSV; Orthopneumovirus bovis), bovine parainfluenza virus 3 (BPIV-3; Respirovirus bovis), and bovine viral diarrhea virus 1 (BVDV-1; Pestivirus bovis). 15 The bacteria include Histophilus somni, Mannheimia haemolytica, Mycoplasma bovis, Pasteurella multocida, and Trueperella pyogenes.
Among the viruses, BRSV may play a major role in the pathogenesis of BRDC. 2 BRSV infects mainly epithelial cells of the respiratory tract, causing cell damage and immune cell chemotaxis, leading to inflammation and alteration of alveolar macrophage function. These changes are frequently followed by secondary bacterial infections. 17
The predisposing factors for BRSV infection have not been fully determined, but stress associated with temperature fluctuations, weaning, shipping, mixing of animals, dusty environments, and overcrowding are thought to be critical. 19 The prevalence of BRSV infection is correlated positively with bovine population density. 20
In Argentina, information about BRSV infection is limited.1,7 One report described high seroprevalence in bovine breeding herds from different regions of the country, 16 and another reported high BRSV seroconversion in feedlot cattle and described the associated risk factors for this infection. 6 BRSV isolation has been reported only once in Argentina, from the lungs of a calf with respiratory disease, but associated gross and microscopic lesions have not been reported to date. 2
BRSV is an enveloped, single negative-stranded RNA virus 19 that plays a prominent role in BRDC, and has a direct impact on animal welfare and the cattle industry.19,21,23 The morbidity caused by BRSV infection can be as high as 80% and the mortality can reach 20%, particularly in young calves. 22 The incubation period is 2–5 d. BRSV infection may be subclinical, limited to the upper airways, or involve both the upper and lower respiratory tract. Clinical signs include coughing and nasal and ocular discharge, and, in more severe infections, anorexia, decreased milk yield in lactating cows, and hyperthermia. 23 Factors favoring infection are particularly frequent in feedlots, where agent transmission is enhanced by transportation, lack of vaccination, mixing animals from different sources, continuous entrance of animals to the pen, including those purchased from sale barns, dietary changes, and crowding. 22
BRSV infection is endemic in most countries. In North America, BRSV has been identified as the leading cause of respiratory disease in calves, causing high seroconversion rates in cattle in feedlots during the first month after arrival. 19 Several European countries have also reported high seroprevalence. 23 In Argentina, there is little information about BRSV circulation. We have previously defined risk factors associated with BRSV infection, including source of animals, presence of respiratory signs, age, and herd size. 6
To determine the role of BRSV in the pathogenesis of BRDC in feedlot cattle in Argentina, we investigated the presence of BRSV in postmortem lung samples from feedlot cattle. During 2015, 35 feedlot cattle were submitted for autopsy and respiratory disease diagnostic workup to the Veterinary Pathology Laboratory of the School of Veterinary Sciences, National University of La Plata, Argentina. The animals came from 3 commercial feedlots, located in the districts of Magdalena (15), San Miguel del Monte (1), and La Plata (19; all within Buenos Aires Province). The cattle population in the 3 feedlots was 5,000–10,000 animals, with 135–200 head per pen. The animals were 5–18-mo-old and included 24 males and 11 females weighing 170 ± 45 kg. Positive BRSV serology had been found previously in cattle in 2 of the 3 feedlots included in our study (100% in Magdalena and 47% in La Plata; authors’ unpublished observations). Vaccination against BRDC was not performed in any of the animals in the 3 feedlots.
The clinical history indicated that within 72 h of arrival at the feedlots, the animals had been given a single dose of ivermectin (Bagomectina; Biogénesis Bagó) or ricobendazole (Paraxane; Biogénesis Bagó) and a single dose of a polyvalent clostridial vaccine containing antigens of Clostridium chauvoei, Clostridium septicum, and Clostridium perfringens types C and D (Policlostrigen; Biogénesis Bagó). The diet was composed of dry corn, corn silage, sunflower pellets and/or malt, and vitamin and mineral supplements. The diet during the first 20–25 d contained 15 ± 2% protein and 25 ± 3% fiber. After 25 d, the animals received a diet with 12 ± 3% protein and 8 ± 3% fiber content.
The 35 animals included in our study were found dead (n = 25) or were euthanized in extremis (n = 10). All deaths occurred within 40 d of arrival at the feedlot. Euthanasia was performed with an overdose of concentrated procaine (Eutanasico; Equi Systems) after sedation with xylazine (Rompun; Bayer) and ketamine (Ketonal100; Richmond Vet Pharma). The euthanasia protocol was approved by the Committee for the Care and Use of Laboratory Animals, School of Veterinary Sciences, National University of La Plata, Argentina (protocol 56-5-16P).
In 26 of 35 animals, gross lesions were noted in the lungs but not in other organs; 25 had bilateral cranioventral pulmonary consolidation; 19 also had fibrinous pleuritis (Table 1). One animal had rubbery lungs that did not collapse when the thoracic cavity was opened. In cases in which gross pulmonary consolidation was observed, the percentage of pulmonary parenchyma consolidation was calculated as described previously, 5 and was 3–85% of the total area of the lung. Of the 9 animals without gross pulmonary lesions, we found rumenitis (1), traumatic pericarditis (1), endocarditis (1), and funiculitis (1; Table 1). No gross abnormalities were observed in the remaining 5 animals.
Morphologic patterns of lung disease, ancillary testing, and cause of death of 35 feedlot calves with bovine respiratory disease complex.
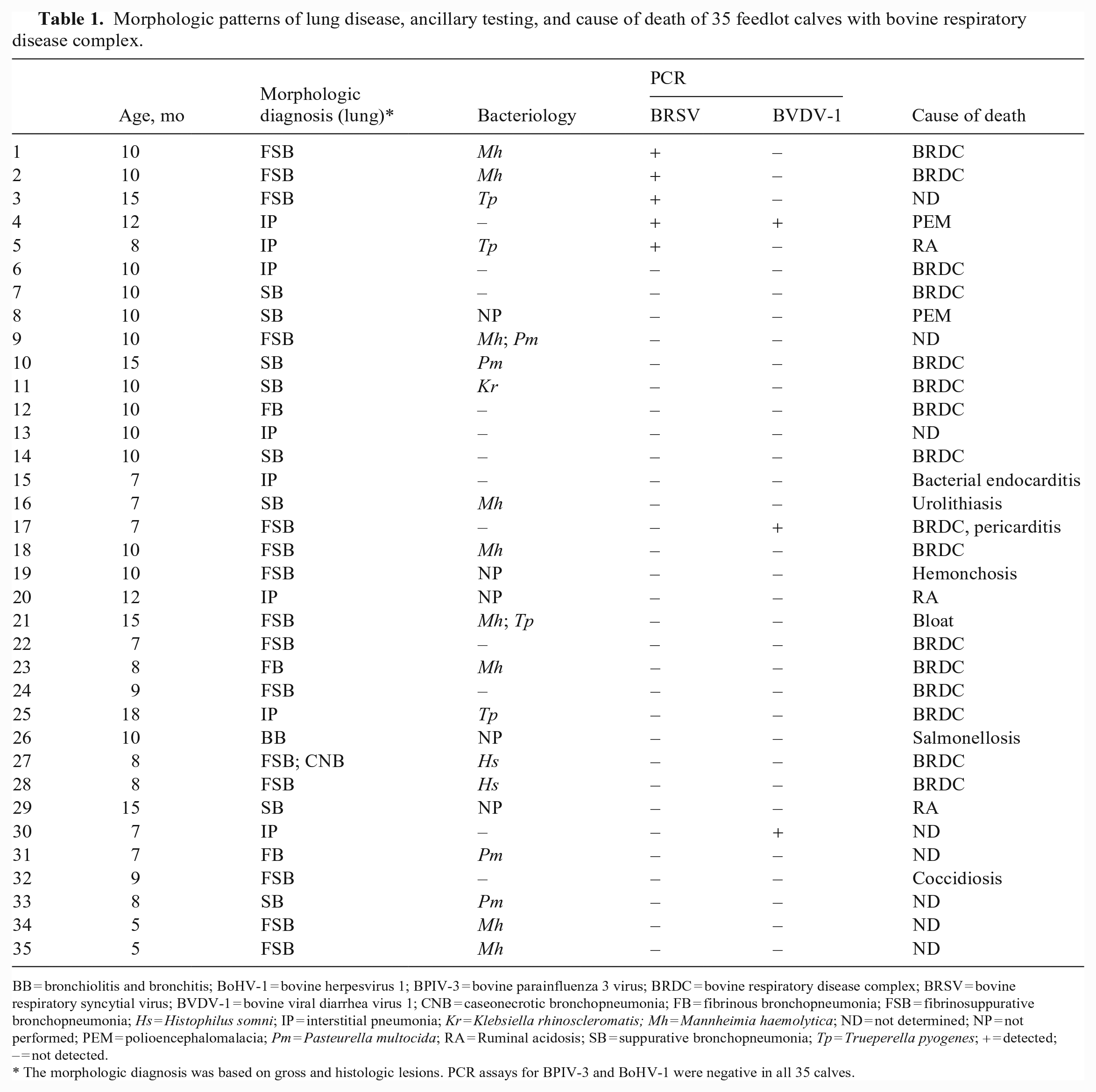
BB = bronchiolitis and bronchitis; BoHV-1 = bovine herpesvirus 1; BPIV-3 = bovine parainfluenza 3 virus; BRDC = bovine respiratory disease complex; BRSV = bovine respiratory syncytial virus; BVDV-1 = bovine viral diarrhea virus 1; CNB = caseonecrotic bronchopneumonia; FB = fibrinous bronchopneumonia; FSB = fibrinosuppurative bronchopneumonia; Hs = Histophilus somni; IP = interstitial pneumonia; Kr = Klebsiella rhinoscleromatis; Mh = Mannheimia haemolytica; ND = not determined; NP = not performed; PEM = polioencephalomalacia; Pm = Pasteurella multocida; RA = Ruminal acidosis; SB = suppurative bronchopneumonia; Tp = Trueperella pyogenes; + = detected; – = not detected.
The morphologic diagnosis was based on gross and histologic lesions. PCR assays for BPIV-3 and BoHV-1 were negative in all 35 calves.
Samples of 7 areas from both lungs of each of the 35 animals were collected for histologic evaluation, including: left and right cranial lobes, cranial and caudal aspects of the left and right caudal lobes, and the right middle lobe. Samples of the left tracheobronchial lymph node, trachea, brain, pre-scapular lymph node, kidney, liver, rumen, small intestine, and large intestine were also collected. Samples were fixed by immersion in 10% neutral-buffered (pH 7.2) formalin for 48 h and processed routinely to produce 4-μm H&E-stained sections.10,15
BRSV immunohistochemistry (IHC) was performed on lung sections of the 5 BRSV PCR–positive animals, as described previously, 12 with the following variations: instead of heat-based antigen retrieval, 0.1% trypsin at 37°C for 10 min was used. Nonspecific binding sites were blocked using 1% BSA, 0.05% nonfat dry milk, and 2.5% normal horse serum (Vector) for 15 min each. Primary polyclonal goat anti-RSV (all antigens) antibody (EMD Millipore), diluted at 1:1,000 (Da Vinci green diluent; Biocare Medical), was incubated overnight at 4°C. A horse anti-goat secondary antibody (ImmPRESS reagent; Vector) was applied for 30 min at room temperature. A lung section from a calf that had tested positive for BRSV by PCR and IHC (UC Davis) was used as a positive control. A section from the same block processed with normal rabbit serum instead of hyperimmune serum was used as a negative reagent control. Furthermore, a section of a block of BRSV PCR–negative lung tissue was incubated with primary antibody (negative tissue control). BRSV IHC was negative in all 5 BRSV PCR–positive cases.
A sample from the left cranial lung lobe of all 5 BRSV PCR–positive animals was submitted for aerobic and microaerophilic culture. These specimens were inoculated onto blood and MacConkey agar and incubated aerobically or in 5–10% CO2 at 37°C for 48 h. Colonies were identified by morphology, detection of hemolysis, Gram staining, and conventional biochemical tests. Mycoplasma sp. culture was not available at the time of our study. M. haemolytica and T. pyogenes were isolated in 2 each of the 5 BRSV-positive animals (Table 2).
Bacteria and other viruses identified in 5 bovine respiratory syncytial virus PCR–positive feedlot calves.

BoHV-1-/5 = bovine herpesvirus 1 and 5; BPIV-3 = bovine parainfluenza 3 virus; BVDV-1 = bovine viral diarrhea virus 1; + = detected; – = not detected.
We attempted virus isolation on lung samples from the 5 BRSV PCR–positive animals. We used 5-cm3 lung specimens collected from the left cranial lobe and stored in liquid nitrogen. These samples were homogenized under sterile conditions and to each was added 1 mL of a Dulbecco modified eagle minimum essential medium (1:1) mixture, supplemented with 5% fetal bovine serum. After centrifugation at 6,000 × g for 20 min at 4°C, 0.1 mL of the supernatant was inoculated onto a Madin–Darby bovine kidney (MDBK) cell layer in a 24-well microplate. BRSV (A51908 strain)-infected and non-infected MDBK cells were included as positive and negative controls, respectively. Five blind passages were carried out, and the cells were observed for cytopathic effect (CPE). After cell passage, no viral CPE was detected. BVDV-1 was detected by PCR in the lung of only one BRSV PCR–positive animal (animal 4; Table 2).
BRSV reverse-transcription nested PCR (RT-nPCR) was performed on lung samples of all 35 animals, as described previously. 25 Viral RNA was extracted from each sample (TRIzol; Invitrogen), according to the manufacturer’s instructions. Briefly, after homogenizing the sample with TRIzol, chloroform was added to separate a clear upper aqueous layer containing RNA, which was subsequently precipitated with isopropanol. Our PCR assay amplifies a 481-bp fragment of the fusion protein (F) gene. The cDNA was synthesized using a commercial Moloney murine leukemia virus reverse transcriptase (Promega) for 60 min at 42°C. The enzyme was inactivated by 5-min incubation at 95°C. The first stage was carried out in a 25-μL reaction mixture containing 1.5 µL of 25 mM MgCl2, 0.5 µL of 10 mM dNTPs (Promega), 0.5 µL (1.5 pmol) of each external oligonucleotide (5′-AATCAACATGCAGTGCAGTTAG-3′ and 5′-TTTGGTCATTCGTTATAGGCAT-3′), 0.25 µL (1.25 U) of GoTaq polymerase (Promega), 5 μL of 5× GoTaq buffer (Promega), 5 μL of cDNA, and 11.75 µL of diethyl pyrocarbonate–treated water (Thermo Fisher). After an initial denaturation at 94°C for 5 min, 25 cycles consisting of denaturation at 94°C for 45 s, annealing at 50°C for 45 s, and elongation at 72°C for 1 min and 30 s were performed. The second stage of PCR was done using 1 μL of the first PCR product, with 0.5 µL (1.5 pmol) of each F gene inner primer (5′-GTGCAGTTAGTAGAGGTTATCGTTGT-3′ and 5′-TAGTTCTTTAGATCAAGTACTTTGCT-3′) for 35 cycles, including denaturation at 94°C for 45 s and annealing at 50°C. The products were separated by electrophoresis in 2% agarose gel, stained (GelRed; Invitrogen), and visualized by UV light.
Lungs of animals 1–5 were BRSV-positive by RT-nPCR (Fig. 1), which indicated viral exposure. BRSV PCR–positive lung samples were also tested for BVDV-1, BPIV-3, and BoHV-1/-5 by PCR, as described previously.14,18,24 BVDV-1 was detected in only one BRSV-positive animal (animal 4; Table 2).
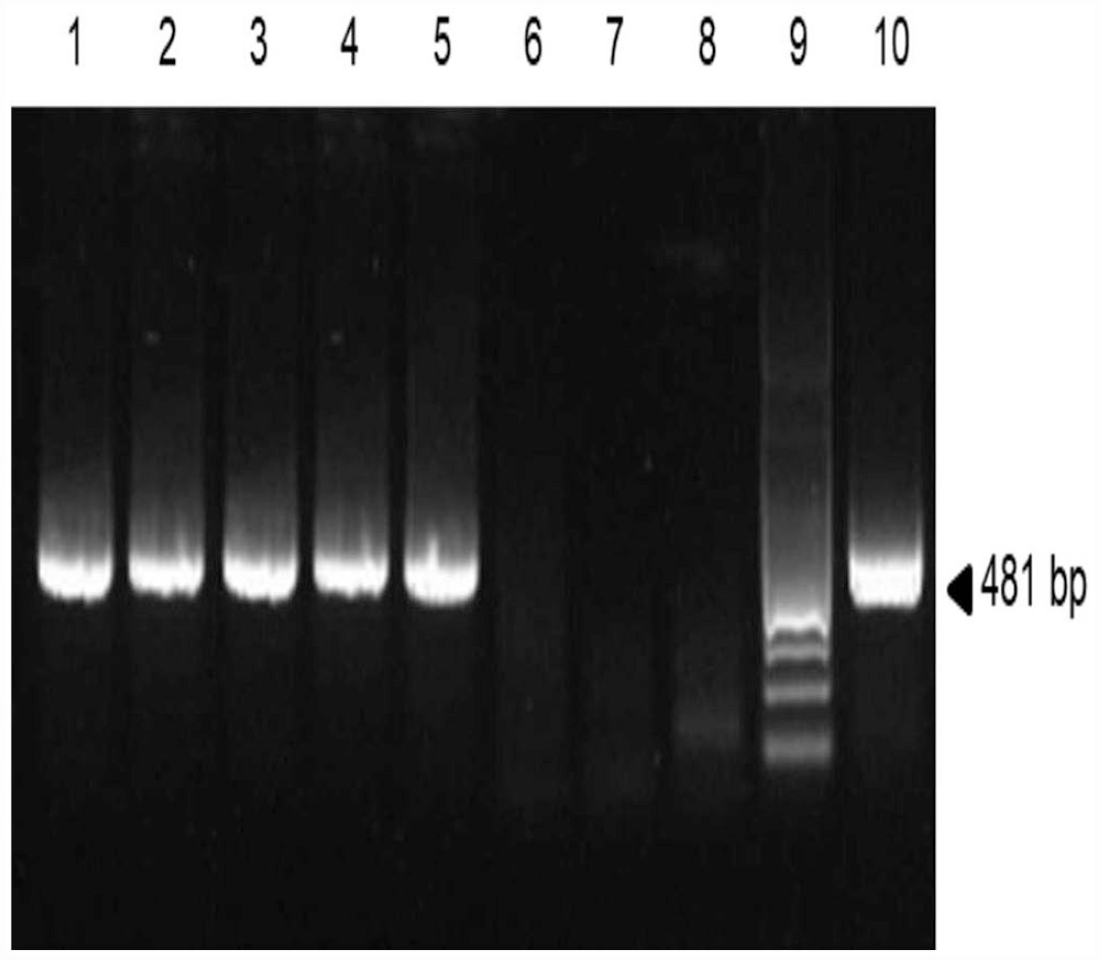
Reverse-transcription nested PCR (RT-nPCR) assays on lung of 5 feedlot cattle. Amplified products for bovine respiratory syncytial virus (BRSV) were analyzed by electrophoresis using 2% agarose gel and stained with GelRed (Invitrogen). Lanes: 1–5 = cDNA lungs from BRSV PCR–positive cases; 6–8 = non-template controls for each step of the RT-nPCR; 9 = 100-bp ladder; 10 = positive control (RNA extracted from BRSV-infected cells).
We detected BRSV in lung tissue from 5 of 35 (14%) dead animals by our RT-nPCR assay. In previous studies, the prevalence of BRSV infection was 4.6 8 –9%. 10 The higher percentage observed in our report could be explained by the fact that we considered not only fatal pneumonia cases, but also samples from animals without gross lung lesions, which could have enabled the detection of subclinical cases.
Of the 5 animals that were positive for BRSV, 2 had a clinical history of respiratory disease: 1 had been treated unsuccessfully with tilmicosin (Micotil; Elanco); the others had not received any treatment. A third animal had a history of neurologic disease. No other clinical abnormalities had been observed in any of these 5 BRSV-positive animals. The BRSV-positive cases occurred in autumn and winter.
All BRSV-positive calves in our study died within 40 d of arrival to the feedlot, which is when BRDC cases are more likely to occur. 19 The peak incidence of BRDC caused by BRSV in the United States occurs in autumn and early winter. 20 We have previously reported high rates of BRSV seroconversion in Argentina during the same seasons, particularly during the first month after arrival in the feedlot. 6 Grossly, 2 of the 5 animals that were BRSV PCR–positive had bronchopneumonia with pleuritis: 1 had bronchopneumonia, 1 had rumenitis (Table 3). One BRSV PCR–positive animal did not have any gross lesions. Histologic lung lesions included fibrinosuppurative bronchopneumonia characterized by neutrophil infiltration, oat cells, coagulative necrosis, thrombosis, bronchiolar necrosis, and fibrinous pleuritis in 3 BRSV PCR–positive animals (Tables 1, 3; Figs. 2–5). In 2 of the BRSV-positive animals without gross lung lesions, we observed marked congestion and thickening of the alveolar septa by lymphocytes, plasma cells, and macrophages. The left tracheobronchial lymph node had lymphoid hyperplasia in 3 of 5 animals. No significant microscopic lesions were observed in any of the other organs examined. We considered BRSV to have at least contributed to the death of 2 of 35 animals (Table 2).
Main lesions and cause of death of 5 bovine respiratory syncytial virus PCR–positive feedlot calves.
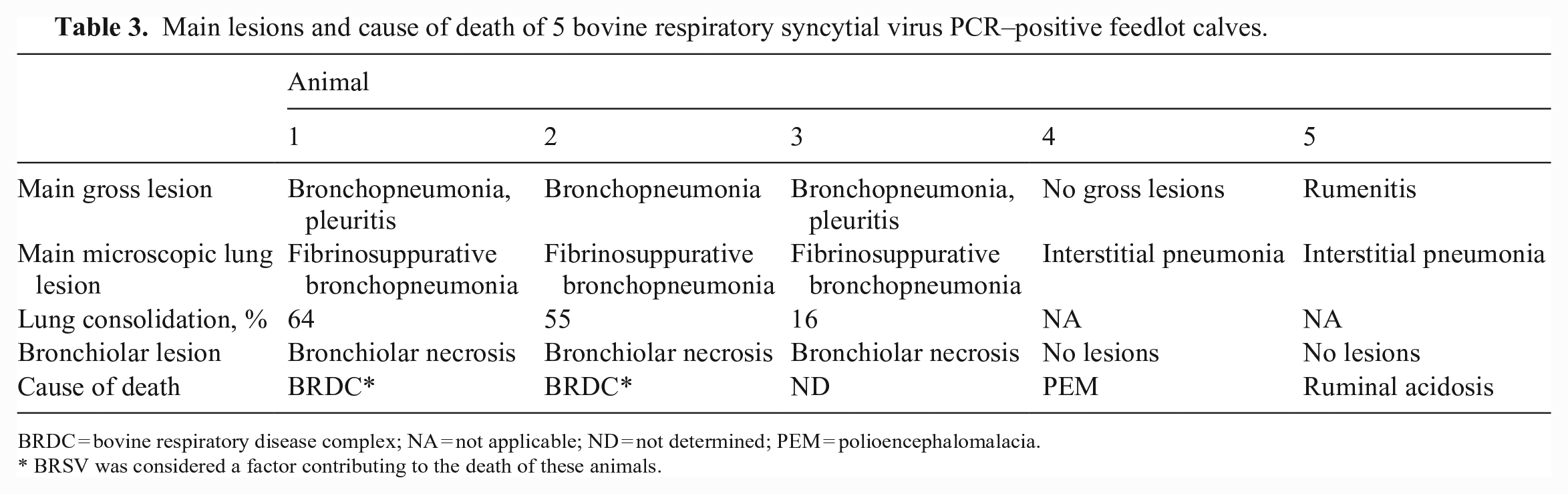
BRDC = bovine respiratory disease complex; NA = not applicable; ND = not determined; PEM = polioencephalomalacia.
BRSV was considered a factor contributing to the death of these animals.
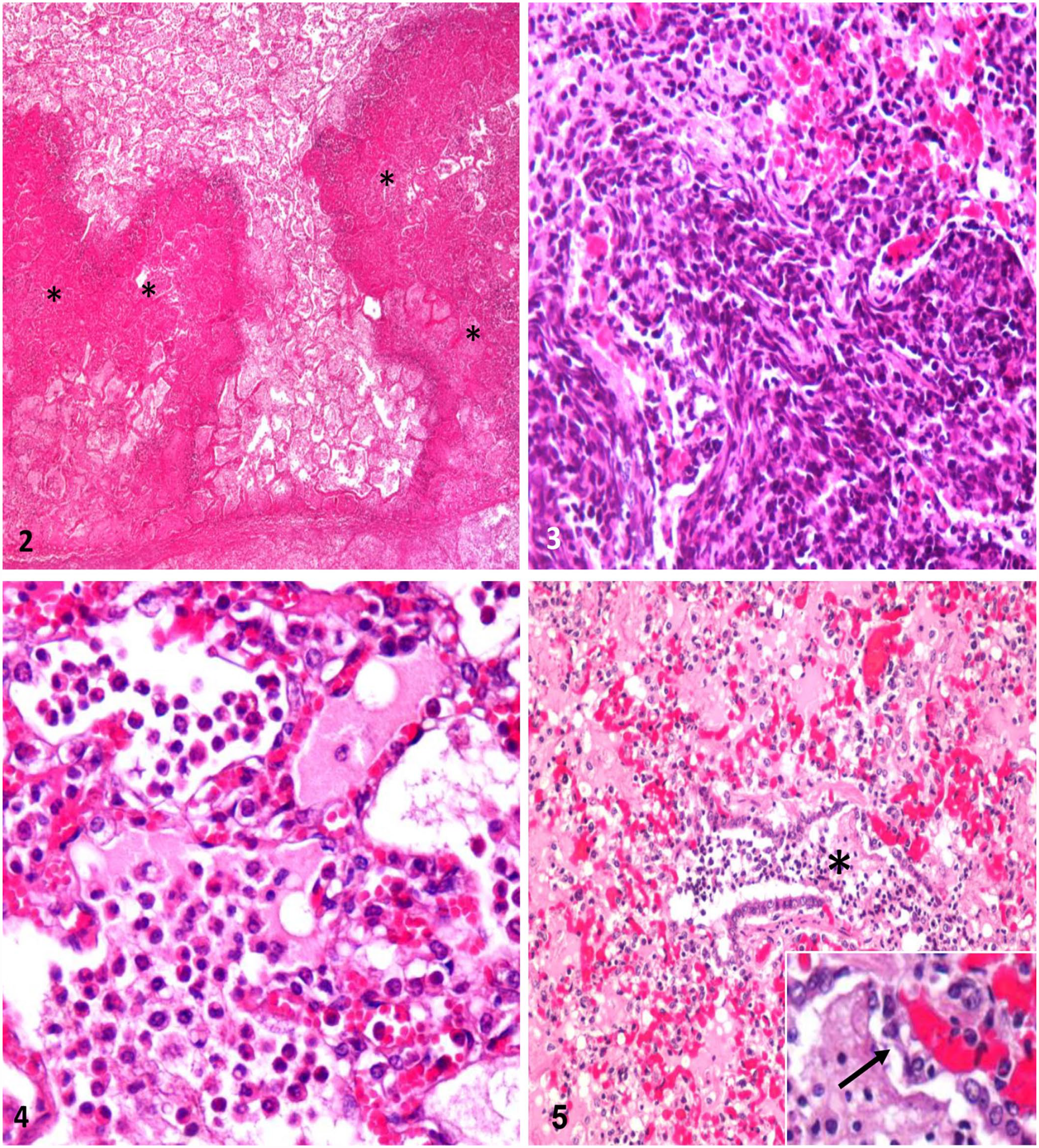
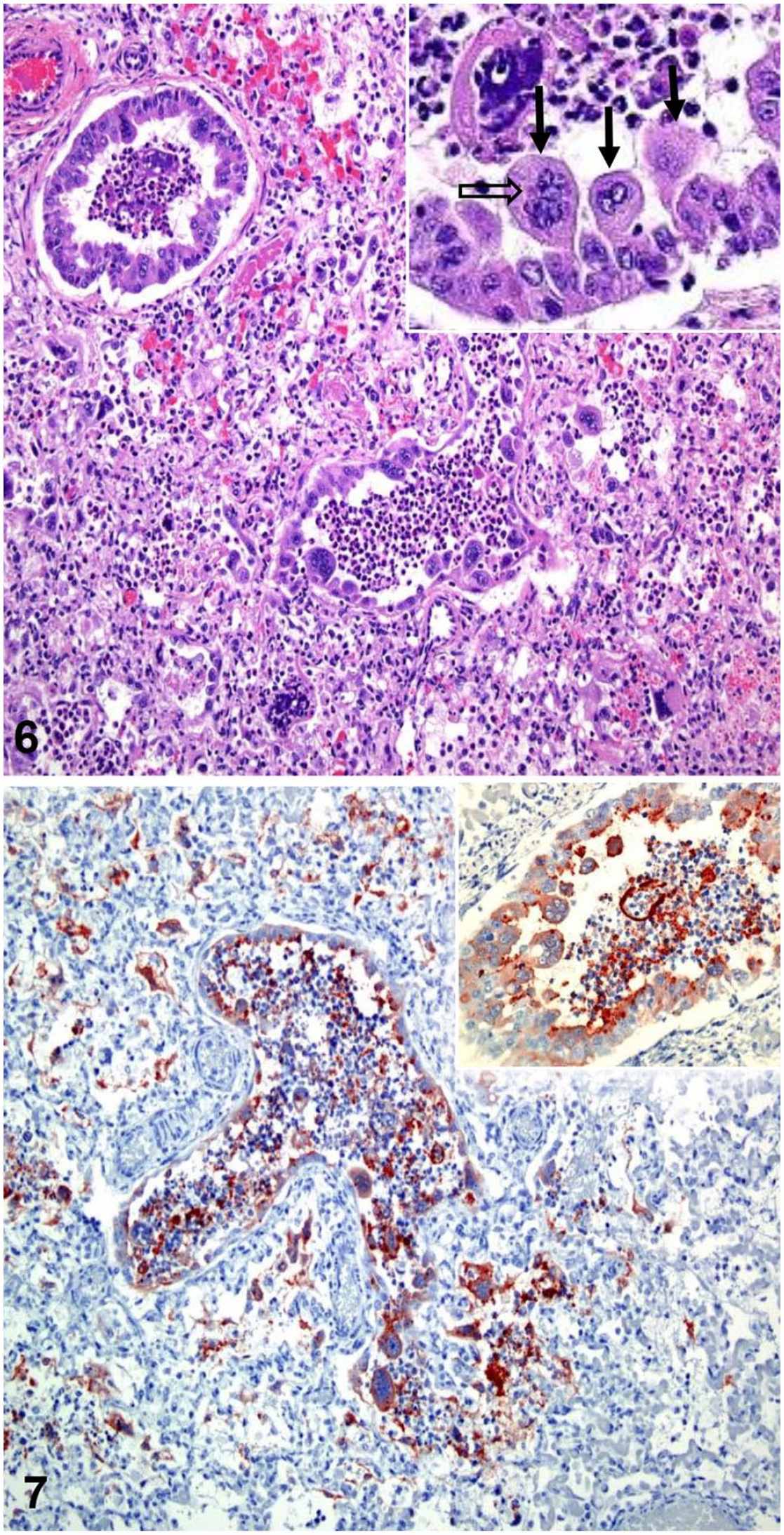
Microscopic lesions in bovine respiratory syncytial virus PCR–positive feedlot calves with fibrinosuppurative bronchopneumonia. H&E.
Grossly, animals 1–3 had fibrinosuppurative bronchopneumonia. This is the most frequently observed lesion in fatal cases of pneumonia in feedlot cattle in the United States. 10 However, animal 3 had < 30% of pulmonary compromise, which is the threshold proposed by some authors to consider this lesion as the cause of death. 10 Animals 4 and 5 did not have gross lung lesions. Because respiratory disease was not the cause of death of animals 3–5, we considered BRSV infection to be subclinical. This agrees with other authors who found that BRSV can be detected in lungs with or without gross lesions. 15 Gross lesions of BRSV infection usually have a cranioventral distribution and are characterized by sunken, well-delineated, deep-red areas of parenchymal consolidation.3,4 Occasionally, however, other parts of the lung are affected, and lesions may also be diffuse. 3 Because most BRSV infections are complicated by secondary bacterial infections, most cases have fibrinosuppurative exudate, restricted mostly to the cranioventral pulmonary lobes. 4
We did not find syncytial cells or viral inclusion bodies histologically in any of the BRSV-positive cases. BRSV infection is short-lived, and syncytial cells disappear within 8 d of infection as viral antigens are cleared. 4 This could explain the lack of syncytia and inclusion bodies, as well as the negative IHC results. Microscopically, typical, uncomplicated BRSV lesions are characterized by bronchointerstitial pneumonia with necrotizing bronchiolitis, bronchiolar and alveolar epithelial syncytia, and eosinophilic intracytoplasmic viral inclusion bodies in syncytial cells and the bronchiolar epithelium (Fig. 6).4,19 When secondary bacterial infections occur, as described here, fibrinosuppurative inflammation is usually observed. 4
Typical pulmonary lesions in the lung of a calf infected with bovine respiratory syncytial virus (BRSV). This is a case from the California Animal Health and Food Safety Laboratory System, UC Davis, and is not part of the 5 animals included in the study from Argentina.
Given its lability and poor growth in vitro, isolation of BRSV is difficult and laborious. 2 This was exemplified in our study when BRSV was detected by PCR in 5 animals, but could not be isolated. Failure to isolate the virus could indicate that the acute infection had already been cleared and the amount of viable virus present in the tissues was low. Alternative detection methods include immunofluorescence, IHC (Fig. 7), and molecular assays.20,21 Positive PCR results for BRSV indicate viral circulation among animals but not necessarily disease; therefore, PCR results should always be interpreted together with clinical and anatomopathologic findings.19,20
Microscopically, animals 1–3 had fibrinosuppurative bronchopneumonia suggestive of bacterial infection. We isolated M. haemolytica from animals 1 and 2; in animal 3, which had received antibiotic therapy, bacterial isolation was negative. Bronchiolar necrosis was observed in these 3 cases, suggesting viral infection. Nevertheless, neutrophil-mediated damage to the epithelium resulting from M. haemolytica infection could also cause this lesion. 4
Gross and microscopic examination demonstrated different types of lung injury, severity, and course in our BRSV-positive cases from Argentina. BRSV can generate a favorable environment for bacterial colonization and favors bacterial adhesion to infected cells and colonization in areas of eroded mucosa. Furthermore, the innate and adaptive immune systems are impaired. 17 We identified other pathogens in all of the BRSV-positive cases in our study.
Mixed infection with > 1 virus was observed only in animal 4, which was BRSV PCR– and BVDV PCR–positive (Table 3). Microscopic lesions consistent with transient BVDV infection, such as perivasculitis and vasculitis, were not observed. In another study, a negative effect on the production of superoxide ion (O2-) in alveolar macrophages coinfected with both viruses was demonstrated in vitro. 13 It is also possible that synergism between these 2 viruses occurred in our case. Bacterial coinfection occurred in 4 of 5 BRSV PCR–positive animals (Table 3); 2 cases were fatally infected with BRSV and M. haemolytica, strongly suggesting a synergistic effect between these pathogens. Even though T. pyogenes was detected in the other 2 animals, we considered T. pyogenes infection to be incidental in these cases, given that none of the T. pyogenes–infected animals had severe pneumonia or any other concomitant bacterial agent. 11 M. bovis is an important component of the BRDC, and 1 of 35 animals in our study had caseonecrotic bronchopneumonia (animal 27; Table 1), highly suggestive of M. bovis infection. Involvement of M. bovis in some of our cases cannot be ruled out. Unfortunately, ancillary testing for M. bovis was not available at the time of our study.
In our study, BRSV detection in lung tissue from clinically and subclinically infected animals strongly suggests that the local relevance of BRSV in BRDC pathogenesis has been underestimated in Argentina.
Footnotes
Acknowledgements
We thank Dr. Carlos Juan Perfumo for his academic contribution and Sebastián Streitenberger for language corrections.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our study was supported by the School of Veterinary Sciences, National University of La Plata through research grant 11/V257 (Argentina).
