Abstract
Viral agents such as bovine respiratory syncytial virus (BRSV) and bovine parainfluenza virus 3 (BPIV-3) are considered primary infectious agents in bovine respiratory disease complex (BRDC). Information regarding the pathogenesis of BRDC is scarce, especially at an advanced chronicity stage, in addition to ongoing coinfection with other primary agents such as Mycoplasma bovis. Based on a retrospective review of histology slides from 104 autopsy cases, we classified cases according to type of pneumonia and chronicity. We performed immunohistochemistry (IHC) for BRSV, BPIV-3, and M. bovis as well as real-time PCR (rtPCR) for M. bovis on lung tissue of all 104 cases and correlated results with the morphologic type of pneumonia. Histomorphologically, 79 cases were classified as bronchopneumonia, 16 as bronchointerstitial pneumonia, and 9 as interstitial pneumonia. In 89 cases, at least 1 of the investigated agents was detected by IHC; 44 of these cases had a coinfection. BPIV-3 was the predominant agent present, as a single infection in 39 cases, and in coinfection with M. bovis in 39 cases. Comparing the detection methods for M. bovis, rtPCR was more specific and sensitive than IHC. The combination of both methods provided a good visual tool for assessing severity and distribution of M. bovis antigen within the tissue. Unlike BRSV, BPIV-3 and M. bovis persisted in chronic BRDC, suggesting ongoing impairment of defense mechanisms in the lung.
Introduction
Bovine respiratory disease complex (BRDC) is a major health problem of cattle worldwide. 10 In dairy calves in Switzerland, pneumonia is the most commonly observed disease. 12 BRDC is a multifactorial disease involving infectious agents, compromised host immune system, and environmental factors, which ultimately result in bronchopneumonia. 8 Viral pathogens are capable of causing primary infections. Generally, viruses are associated with mild clinical signs, 14 but can damage defense mechanisms and the immune system, which thereby increases susceptibility to secondary bacterial infections. 3 At the time of autopsy, particularly in severe and/or chronic stages of the disease, secondary bacterial infections often dominate the pathomorphologic appearance 1 and hinder the investigation of BRDC. This presents a challenge for the diagnostic pathologist as well as the clinician in practice interpreting the laboratory results.
The main viral pathogens associated with BRDC include bovine herpesvirus 1 (BoHV-1; family Herpesviridae, subfamily Alphaherpesvirinae, genus Varicellovirus, species Bovine alphaherpesvirus 1), bovine viral diarrhea virus (BVDV; family Flaviviridae, genus Pestivirus), parainfluenza virus 3 (BPIV-3; family Paramyxoviridae, subfamily Orthoparamyxovirinae, genus Respirovirus, species Bovine respirovirus 3), and bovine respiratory syncytial virus (BRSV; family Pneumoviridae, genus Orthopneumovirus, species Bovine orthopneumovirus). Bacteria frequently associated with BRDC are Histophilus somni, Mannheimia haemolytica, Mycoplasma bovis, and Pasteurella multocida.1,8 In a broad survey of microbiologic and histopathologic findings in BRDC in Canada, M. haemolytica was the predominant agent in peracute, acute, and subacute BRDC cases, whereas M. bovis predominated in chronic cases. 1 BVDV was the predominant viral agent identified, whereas BRSV and BPIV-3 appeared to play only a minor role in the disease. 1 In Switzerland, the infectious disease situation for RDC is unique and makes comparing current results to studies in other countries difficult: BoHV-1 has been eradicated; a successful BVDV eradication program is ongoing, with >99% of cattle farms being free of this virus (https://www.blv.admin.ch/blv/de/home/tiere/tierseuchen/bekaempfung/ausrottung-bvd.html), thus leaving BRSV and BPIV-3 as the most likely primary viral agents in BRDC. In a 1985 serologic survey from Switzerland, 77.4% of the tested cattle were positive for BRSV. 6 In Sweden, 76.8–85.3% of cattle herds were positive for BRSV, 18 indicating widespread distribution of this virus. Information regarding the prevalence and spread of BPIV-3 and M. bovis is scarce.
M. bovis is consistently found in BRDC cases, 8 especially associated with chronic disease. 1 Calves with lung lesions caused by M. bovis often receive prolonged antimicrobial therapy and undergo nonresponsive or relapsing respiratory disease termed “chronic pneumonia.”2,8 This bacterium is often overlooked given that its culture requires specific growth media. In Europe, M. bovis is reportedly responsible for one-quarter to one-third of all calf pneumonias, although this might be an underestimate because very few laboratories routinely test for mycoplasmas. 11 In a Canadian study, in which immunohistochemistry (IHC) was used to detect M. bovis in chronic, antibiotic-resistant pneumonia in feedlot cattle, M. bovis antigen was found in 71% of investigated lung samples. 9 M. bovis can be a primary pathogen or occur as a coinfection. 8 More specifically, M. bovis seems to interplay with secondary bacterial pathogens, which was shown in experimentally induced BRDC in which M. bovis acted as a predisposing factor. 2 Therefore, we consider M. bovis as a possible primary pathogen in BRDC and focus on tools to address the detection of M. bovis in chronic BRDC cases.
A variety of tools, including bacterial and viral cultures, are used to identify the etiologic agents involved in BRDC. Molecular methods such as IHC and PCR are useful methods, especially for cases of pneumonia with severe necrosis, secondary bacterial infections, antibiotic pretreatment, and in which only formalin-fixed, paraffin-embedded (FFPE) tissues are available. Moreover, unlike serology or PCR, IHC allows a morphologic link between the infectious agent and the histologic lesion.
Considering the aforementioned facts, the focus of our study was on coinfections of primary infectious agents present in lethal BRDC of Swiss cattle herds: BRSV, BPIV-3, and M. bovis. Our aim was to determine the prevalence of these primary agents using IHC on formalin-fixed tissues and to link these results to the histomorphologic subtype of pneumonia. Additionally, we compared IHC and rtPCR as methods for the detection of M. bovis. We consider H. somni, M. haemolytica, and P. multocida to be secondary bacterial infections, and thus did not focus on these agents in our investigation.
Material and methods
Selection of cases and sampling
We searched the autopsy archive of the Institute of Animal Pathology (University of Bern, Switzerland) for cases of pneumonia with lethal outcome over a 5-y period (2012–2016). The criterion for inclusion in our study was the diagnosis of pneumonia as cause of death or reason for euthanasia.
Histology and IHC
Four-μm sections of FFPE lung samples were stained with hematoxylin and eosin (H&E) according to routine laboratory procedures. The H&E sections were evaluated and classified as bronchopneumonia, bronchointerstitial pneumonia, or interstitial pneumonia, as well as acute or chronic cases based on a classification system (Table 1) adapted from standard pathology literature. 4 For IHC, 4-μm sections of FFPE lung samples were mounted on positive loaded glass slides (Superfrost plus; Thermo Scientific, Braunschweig, Germany). The slides were deparaffinized at 55°C for 30 min, followed by 5 min of immersion in xylol. Subsequently, tissue samples were rehydrated at room temperature (RT) in descending concentrations of ethanol (100%, 95%, 80%, and 70%). Endogenous peroxidase blocking was performed with 3.25% H2O2 for 10 min at RT. Heat-induced epitope retrieval was carried out by incubating the slides for 10 min at 100°C in citrate buffer (pH 6.0). Nonspecific binding of the antibodies was blocked with 1% bovine serum albumin for 10 min. Primary antibodies were incubated overnight at 4°C in a humid chamber: BRSV, 1:200 (RSV monoclonal antibody, MA5-16512; Thermo Fisher Scientific, Zug, Switzerland), BPIV-3, 1:20 (Moab a-BPIV-3, BIO 290; Bio-X Diagnostics, Rochefort, Belgium), and M. bovis, 1:1,000 (MAB970, Merck Millipore, Molsheim, France). Antibody detection and visualization were performed (Dako LSAB kit, AEC-Chromogen; Dako, Glostrup, Denmark), as indicated by the manufacturer. Slides were counterstained with Ehrlich hematoxylin and covered with coverslips (Aquatex; Merck, Darmstadt, Germany). A light microscope (Nikon E400; Nikon Instruments, Tokyo, Japan) was used for histologic examination.
Definition of histologic classification criteria for different subtypes and chronicity stages of pneumonia.

DNA isolation, rtPCR, conventional PCR, and Sanger sequencing
DNA was isolated from two 20-µm thick scrolls from each paraffin block (n = 104; QIAamp DNA FFPE tissue kit; Qiagen, Hilden, Germany), according to the manufacturer’s instructions. To standardize PCR analyses, the concentrations of DNA extractions were adjusted to 50 ng/µL. Real-time PCR targeting the uvrC gene of M. bovis was performed as described previously. 13 TaqMan exogenous internal positive control reagents (Applied Biosystems, Foster City, CA) were used to assess PCR inhibition, according to the manufacturer’s instructions. Additionally, a standard curve was generated using a plasmid containing the uvrC gene fragment. The concentration of 10-fold dilutions of the plasmid DNA was quantified by spectrophotometry, and the plasmid copy number was calculated using online software (http://cels.uri.edu/gsc/cndna.html). The standard curve was determined (7500 System software v.2.0.5; Applied Biosystems).
A conventional PCR to detect Mycoplasma sp. was also performed in all cases, as described previously. 16 This PCR targets a 270-bp fragment of the gene encoding for the 16S rRNA. Amplicons were subsequently sequenced (ABI Prism 3100 genetic analyzer, BigDye Terminator cycle sequencing kit; Applied Biosystems). To confirm the presence of amplifiable DNA in the FFPE samples, rtPCR targeting the gene encoding for the 18S rRNA of eukaryotic cells was performed using the eukaryotic 18S rRNA endogenous control according to the manufacturer’s instructions (Thermo Fisher, Zug, Switzerland).
Statistical analysis
Using software (NCSS v.10; NCSS, Kaysville, UT), receiver operating characteristic (ROC) analysis was performed to investigate the value of IHC compared to rtPCR for M. bovis, which was taken as the real condition or gold standard. This was followed by calculation of the area under the curve (AUC) as well as the odds ratio, sensitivity, and specificity of the IHC. A chi-square test comparing the prevalence of infectious agents within acute and chronic cases of the different pathomorphologic subtypes was performed; the p-value for identification of statistically significant differences was set at ≤0.05.
Results
Case selection
Our search yielded 104 cases in which the animal died or was euthanized because of pneumonia, all of which we included in our study. The age of the animals ranged from 2 d to 8 y, with a mean of 391 d. Of 104 cases, 47 were dairy breeds, 32 were mixed breeds (Simmental), 11 were beef breeds, 12 were of unknown breeds, and 2 were of other breeds (Eringer [syn. Herens], used for cow fighting; Supplementary Table 2).
Histopathology
Of 104 BRDC cases, 79 were classified as bronchopneumonia, 16 as bronchointerstitial pneumonia, and 9 as interstitial pneumonia. Of all cases, 56 were characterized as chronic, whereas 48 were considered acute (Fig. 1; Supplementary Table 2).
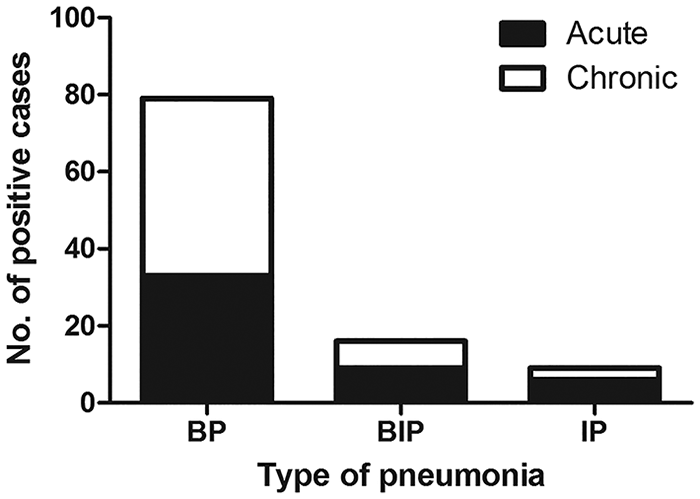
Histologic classification of pneumonia type and chronicity for 104 cases of bovine respiratory disease complex in Switzerland. BIP = bronchointerstitial pneumonia; BP = broncho-pneumonia; IP = interstitial pneumonia.
Immunohistochemistry
At least 1 of the infectious agents was detected in 90 samples; the remaining 14 samples had no positivity for BRSV, BPIV-3, or M. bovis. Of the 90 positive cases, 44 had coinfection with 2 or 3 infectious agents, whereas in 46 cases, a single infectious agent was present. The highest prevalence of single agents was detected for BPIV-3 in 39 cases, followed by M. bovis with 6, and BRSV with 1. The most commonly detected coinfection was the combination of BPIV-3 and M. bovis, in 39 cases. In 3 of the 44 overall positive cases, all 3 investigated agents were present, whereas in 1 case each, a combination of BRSV and BPIV-3 or BRSV and M. bovis were present. There was no significant difference in the prevalence of infectious agents in acute versus chronic cases of bronchopneumonia (Figs. 2–4).
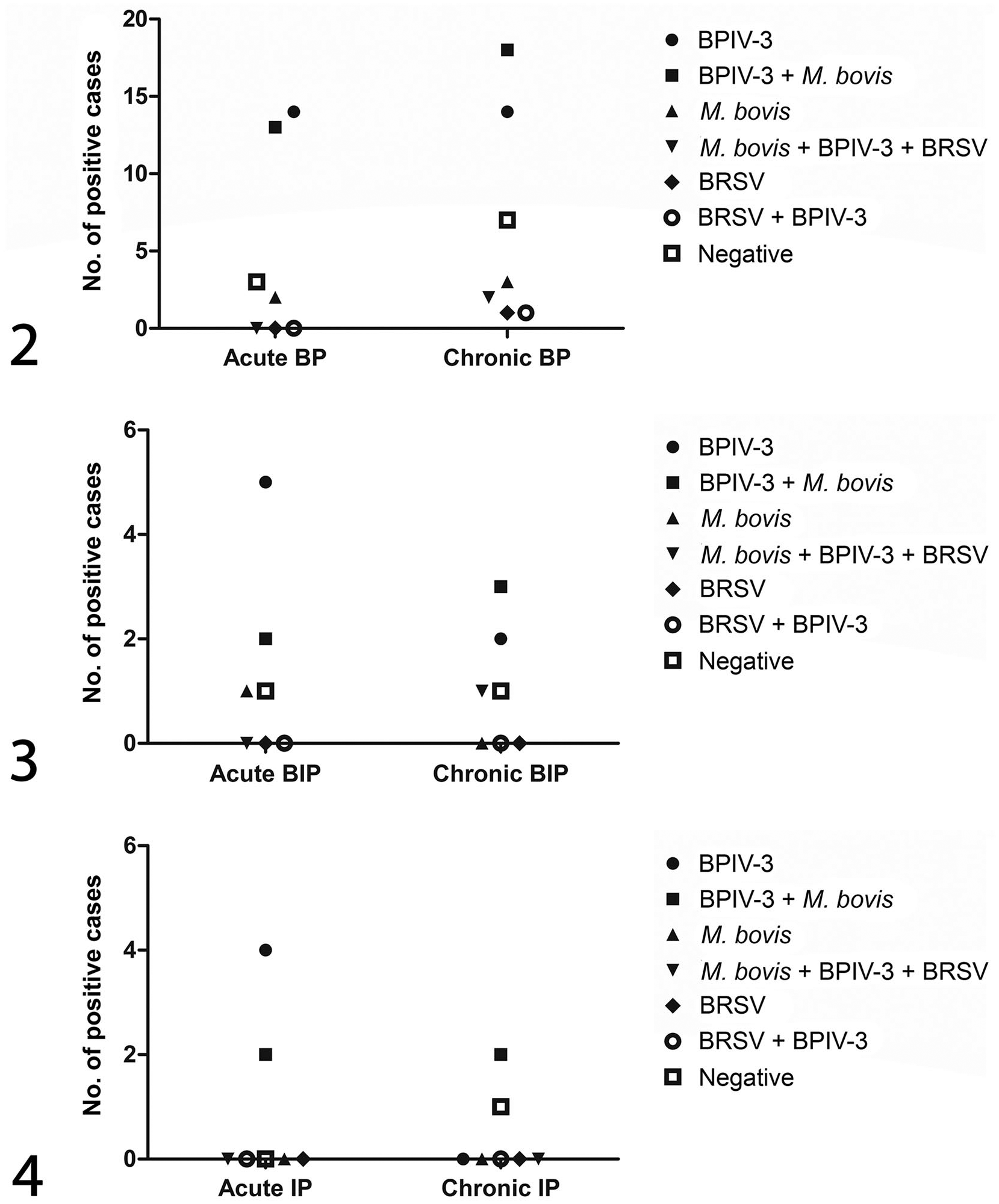
Prevalence of various respiratory pathogens in different patterns of bovine pneumonia.
BPIV-3 antigen was visible as a band-like area of positivity, apically on ciliated epithelial cells. BRSV antigen was mainly seen in alveolar and to a lesser degree in bronchial and bronchiolar epithelium, whereas M. bovis antigen was present in epithelial cells of bronchi and bronchioles as well as in a few alveolar macrophages (Figs. 5–8). The investigated infectious agents were distributed uniformly across the different pathomorphologic types of pneumonia.
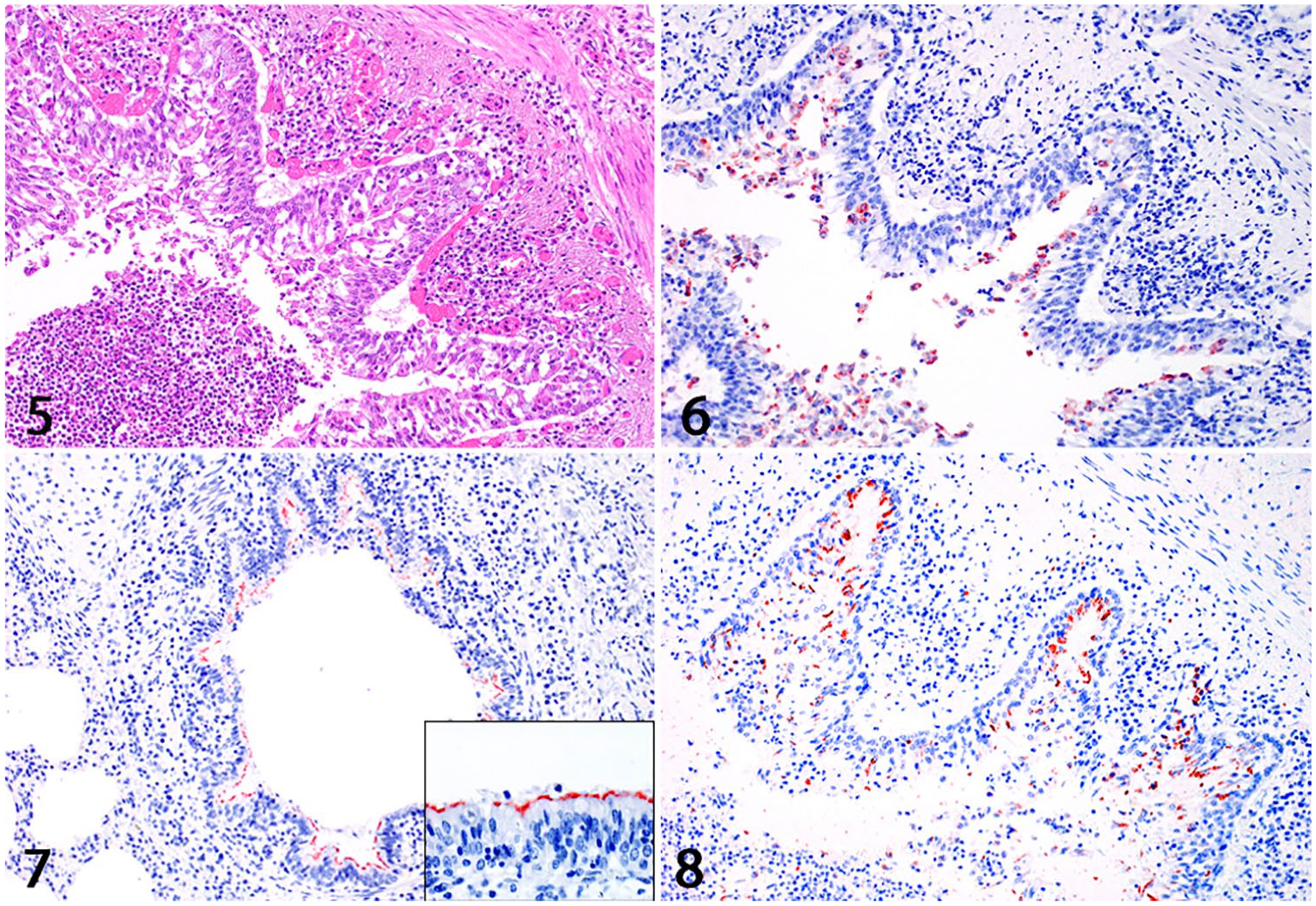
Sections of bovine lung with bronchopneumonia.
Real-time PCR for M. bovis; PCR and Sanger sequencing for Mycoplasma sp
In total, 29 cases were positive for M. bovis by rtPCR; 18 of those cases were also positive for M. bovis by IHC (Supplementary Table 1). Of the 75 rtPCR-negative cases, IHC positivity for M. bovis was observed in 31 cases. Conventional PCR for Mycoplasma sp. of those 31 rtPCR-negative, but IHC-positive cases, yielded 8 additional Mycoplasma-positive cases. Of these 8 cases, subsequent Sanger sequencing revealed 1 case of M. bovis, 1 case of M. dispar, and in 6 cases, no identifiable sequences could be obtained. The conventional PCR for Mycoplasma sp. performed on the remaining rtPCR-negative cases yielded 4 additional positive cases. However, the quality of the amplicons was insufficient for sequencing. Further 18S rRNA PCR was negative in 3 cases, leaving those cases with no clear results. The standard curve represented a linear regression (slope −3.18), with corresponding efficiency of 106 and R2 of 0.98. The limit of detection was 10 copies per reaction.
Evaluation of impact of IHC versus rtPCR for M. bovis
In comparison to rtPCR, IHC for M. bovis had low sensitivity (60.7%) and specificity (57.9%). The AUC value was only 59.3%, with p = 0.04, meaning that IHC is not a good predictor of the presence of M. bovis.
Discussion
Across the different gross and pathomorphologic appearances of BRDC in our study, BPIV-3 was the infectious agent detected most commonly by IHC. This is in contrast to previous studies on BRDC in Canada, in which only 1% of the investigated calves with BRDC were positive for BPIV-3 by IHC. 1 As in most countries, BVDV is the predominant viral agent in BRDC; BPIV-3 is considered to play a minor role 1 and is often not investigated at all. 7 To our knowledge, similar results have not yet been reported. With the eradication of BVDV and BoHV-1 from Switzerland, the expectation of reduced BRDC has not been fulfilled, and pneumonia is still the most commonly observed disease in dairy calves. 12 Therefore, it is possible that, in the absence of BVDV and BoHV-1, and in addition to BRSV, BPIV-3 takes on a prominent role as an important primary agent in the pathogenesis of BRDC, persisting until later stages of the disease. As described for human parainfluenza viruses, 15 we observed BPIV-3 antigen apically in ciliated epithelial cells. This selective tropism may limit exposure of viral antigen to antigen-presenting cells, possibly leading to reduced immune response that may allow persistence of the virus and re-infection. 15 This may partially explain the high IHC prevalence of BPIV-3 in our study, especially compared to BRSV. Despite high serologic prevalence worldwide, 18 and in Switzerland, 6 we detected BRSV antigen in only a few of our cases. Experimental studies show that BRSV antigen is cleared within 15 d post-infection, which leaves a very short time for demonstrating BRSV by virus isolation or antigen detection. 17 Therefore, our study supports the hypothesis that BRSV is an initiating agent of BRDC, with rapid clearance and reduced detectability in later stages of disease. We conclude that BRSV does not persist in lethal and chronic BRDC in cattle, and does not lead to further progression of the disease at a chronic stage.
Approximately half of the BPIV-3–positive lungs were coinfected with M. bovis or other Mycoplasma sp. This indicates a high prevalence of Mycoplasma sp., especially M. bovis, within severe, and more importantly, chronic cases of BRDC, and also supports the hypothesis that M. bovis is under-detected in routine testing. A high prevalence of M. bovis has also been reported in studies from Canada 1 and the United States. 7 Given that no specific pattern for coinfection with M. bovis can be observed histologically, molecular methods such as IHC or PCR are necessary to gain further insight. Comparing IHC and rtPCR for detection of M. bovis, IHC showed low sensitivity and specificity with respect to rtPCR on FFPE lung tissue. The rtPCR showed a detection limit of 10 uvrC gene copies per reaction. This is satisfactory analytical sensitivity, and therefore we considered rtPCR as the gold standard for analysis of FFPE tissue material. Although rtPCR is fairly sensitive, it must be noted that cross-linking and degradation of DNA can increase the amount of un-amplifiable DNA, as can be seen with our results obtained with the 18S rRNA PCR. Interestingly, the results of samples negative for M. bovis by rtPCR, but positive by IHC for the presence of other Mycoplasma spp., revealed an overlap with M. dispar in one case. This indicates probable cross-reactivity of the antibody utilized in IHC for M. bovis with M. dispar and possibly other Mycoplasma spp. Cross-reactivity may partially explain the discrepancy between IHC and rtPCR. Our results leave the possible involvement of Mycoplasma spp. other than M. bovis in cases of BRDC open to interpretation, presenting potential research avenues.
Overall, rtPCR is a valuable method for detecting M. bovis in tissue samples with high sensitivity, 5 whereas IHC has the advantage of assessing the morphologic distribution and intensity within different areas of the affected lung. Ultimately, IHC is a good tool to detect BRSV and BPIV-3 in FFPE lung sections. To detect M. bovis, we recommend the combination of IHC and rtPCR, especially in cases in which culture is inconclusive or cannot be performed (i.e., when only formalin-fixed tissue is available). However, our results also indicate that in the case of clinical or pathology suspicion of the presence of M. bovis, the analysis of fresh material might be a better option. We conclude that BPIV-3 and M. bovis are widely distributed, whereas BRSV is rarely detected in chronic and lethal BRDC cases in Switzerland. Our results should be considered while developing vaccination and/or immune therapy strategies for the various infectious agents of BRDC.
Supplemental Material
DS1_JVDI_10.1177_1040638719861686 – Supplemental material for Coinfection of Swiss cattle with bovine parainfluenza virus 3 and Mycoplasma bovis at acute and chronic stages of bovine respiratory disease complex
Supplemental material, DS1_JVDI_10.1177_1040638719861686 for Coinfection of Swiss cattle with bovine parainfluenza virus 3 and Mycoplasma bovis at acute and chronic stages of bovine respiratory disease complex by Kemal Mehinagic, Paola Pilo, Beatriz Vidondo and Nadine Stokar-Regenscheit in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Manuela Bozzo, Erika Bürgi, Bettina de Breuyn, and Evelyne Rohrer for technical support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
We thank the Specialization Committee of the Vetsuisse Faculty, University of Bern, for financial support.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
