Abstract
This study charted 237 fatal cases of bovine respiratory disease (BRD) observed from May 2002 to May 2003 in a single Oklahoma feed yard. Postmortem lung samples were used for agent identification and histopathology. Late in the study, 94 skin samples (ear notches) were tested for Bovine viral diarrhea virus (BVDV) by immunohistochemistry (IHC). Bovine respiratory disease morbidity was 14.7%, and the mortality rate of all causes was 1.3%, with more than half (53.8%) attributed to BRD (0.7% total of all causes). The agents isolated were the following: Mannheimia haemolytica (25.0%), Pasteurella multocida (24.5%), Histophilus somni (10.0%), Arcanobacterium pyogenes (35.0%), Salmonella spp. (0.5%), and Mycoplasma spp. (71.4%). Viruses recovered by cell culture were BVDV-1a noncytopathic (NCP; 2.7%), BVDV-1a cytopathic (CP) vaccine strain (1.8%), BVDV-1b NCP (2.7%), BVDV-2a NCP (3.2%), BVDV-2b CP (0.5%), and Bovine herpesvirus 1 (2.3%). Gel-based polymerase chain reaction (PCR) assays were 4.6% positive for Bovine respiratory syncytial virus and 10.8% positive for Bovine coronavirus. Bovine viral diarrhea virus IHC testing was positive in 5.3% of the animals. The mean values were determined for the treatment data: fatal disease onset (32.65 days), treatment interval (29.15 days), number of antibiotic treatments (2.65), number of different antibiotics (1.89), and day of death (61.81 days). Lesions included the following: 1) duration: acute (21%), subacute (15%), chronic (40.2%), healing (2.8%), normal (18.1%), and autolyzed (2.8%); 2) type of pneumonia: lobar bronchopneumonia (LBP; 27.1 %), LBP with pleuritis (49.1 %), interstitial pneumonia (5.1 %), bronchointerstitial pneumonia (1.4%), septic (0.9%), embolic foci (0.5%), other (2.8%), normal (10.3%), and autolyzed (2.8%); and 3) bronchiolar lesions: bronchiolitis obliterans (39.7%), bronchiolar necrosis (26.6%), bronchiolitis obliterans/bronchiolar necrosis (1.4%), other bronchiolar lesions (6.5%), and bronchiolar lesion negative (25.7%). Statistically significant relationships were present among the agents, lesions, and the animal treatment, disease onset, and mortality data. Clinical illnesses observed in this study were lengthier than those reported 16–20 years ago, based on fatal disease onset, treatment interval, and day of death.
Introduction
Bovine respiratory diseases (BRD) have a major impact on the feedlot industry in North America. 14,19 The economic losses from BRD are represented by mortality, cost of therapy, prophylaxis, reduced performance in the feedlot, and decreased carcass value. The BRD pneumonias constitute a significant proportion of feedlot diseases. The etiologic agents associated with feedlot pneumonias include viruses (Bovine herpesvirus 1 [BHV-1], Bovine parainfluenza virus 3 [BPIV-3], Bovine viral diarrhea virus 1 and 2 [BVDV-1–2], Bovine respiratory syncytial virus [BRSV], Bovine adenovirus A–D [BAdV-A–D], and Bovine coronavirus [BCoV]), bacteria (Mannheimia haemolytica, Pasteurella multocida, and Histophilus somni), and Mycoplasma spp. 1–6,8–18,20,21,24,25,27,28
Several previous studies examined the presence of specific BRD lesions in feedlot mortalities, along with the detection of infectious agents. Multiple publications came from an in-depth year-long study of feedlot diseases in Colorado in the 1970s. 17,18 Since then, other studies of feedlot BRD have also been published. 2,13 Selected studies reported on the lesions and infectious agents from multiple feedlots and/or the occurrence of cases relatively early in the feeding period. 2,13 In another study, fatal BRD cases were related to the time of initial illness, fatal disease onset (FDO), treatment interval (TI), time between the day of initial treatment and death, and interval from the day the animal entered the feedlot to day of death. 28 The present study of BRD in an Oklahoma feedlot had the following objectives: 1) description of lung pathology and detection of infectious agents from lungs during a year-long study, 2) detection of BVDV persistently infected (PI) cattle dying in the feedlot, and 3) the relationship of the fatal cases of pneumonia with mortality and treatment information.
Materials and methods
Feedlot and clinical information
A year-long study, from May 2002 to May 2003, included cattle with BRD clinical signs that died in the sick pen or that died suddenly in the regular feeding pen without treatment for BRD. The BRD morbidity rate over this interval was 14.7%, with a mortality rate of 0.7% for BRD of the 1.3% total for all causes. Samples of lung (fresh and formalin fixed) were collected at necropsy by feedlot personnel and were submitted to Oklahoma State University (Stillwater, OK). In the later stages of the study, when BVDV immunohistochemistry (IHC) became available, skin samples (formalin-fixed ear notches) were collected from necropsy cases. Samples collected during the study included 237 lung samples and 94 ear notches for BVDV IHC testing. The feed yard provided information on the 237 individual animals, including FDO, day first treated, TI, interval from first treatment day until day of death, treatment numbers (number of antibiotic treatments), number of treatments with different antibiotics, and day of death in the feedlot. The day of death was defined as the day after arrival in the feedlot when the animal died. The feedlot processing at entry included a viral vaccine that contained modified live virus strains of BHV-1, BRSV, BPIV-3, and BVDV-1a (NADL) cytopathic strain. a
Pathology
Two hundred and fourteen lung samples were submitted in 10% neutral buffered formalin. A single glass slide that contained 2–3 specimens of lung was prepared, stained with hematoxylin and eosin, coverslipped, and examined by microscopy. 6,9,11 The microscopic lesions were characterized as follows: 1) classifications of pneumonias, 2) duration (acute to chronic), and 3) bronchiolar lesions.
Agent identification
Two hundred and twenty fresh lung samples were submitted to the Oklahoma Animal Disease Diagnostic Laboratory (OADDL; Stillwater, OK) for cell culture isolation of viruses, and culture for bacteria and Mycoplasma spp. 6,9,11 For virus isolation, lung homogenates were inoculated onto bovine turbinate monolayers in 24-well plates. 6,9,11 After 5–7 days, a second passage was made by subculturing trypsinized cells and these cultures were observed for viral cytopathology for another 5–7 days. At the end of the second passage, the trypsinized cells were stained by an indirect fluorescent antibody test (IFAT) procedure with monoclonal antibodies (mAb) 15c5 and 20.10.6. Based on the presence or absence for cytopathology in cell cultures, the BVDV isolates were classified as either cytopathic (CP) or noncytopathic (NCP) biotypes of BVDV. Cytopathic agents other than BVDV, including BRSV, BPIV-3, and BHV-1, were identified by direct FAT by the OADDL. The BVDV isolates were typed as BVDV-1a, BVDV-1b, BVDV-2a, or BVDV-2b, based on the phylogenetic comparisons of sequences of the 5′-untranslated region (UTR) of the viral genome. 7,10,11 Reference BVDV strains were included in these comparisons, including those BVDV strains currently used in viral vaccines. The BVDV IHC testing was performed on formalin-fixed ear notches from 94 cattle in the study. 7 The IHC procedure was performed at the OADDL as described by using the primary anti-BVDV mAb 3.12 F1. 7 The BRSV and BCoV were assayed in 194 lung homogenates by using gel-based reverse transcription polymerase chain reaction (RT-PCR) assays. The RT-PCR product for the BRSV RT-PCR was a 481-bp region of the F gene. 29 The product for the BCoV assay was a 251-bp region of the N gene. 26
Statistical analyses
For all statistical analyses, SAS version 9 b was used. Means, standard error of the mean (SE), and ranges were calculated for each of the response variables (FDO, treatment interval, number of treatments, number of antibiotics, and death day). For each categorical independent variable (positive/negative), a 2-population t test was performed to assess whether the means associated with the response variables were significantly different for the 2 categories in question. The Satterthwaite degree of freedom adjustment was used when the equal variance assumption was violated. The relationships among the categorical variables were assessed with the use of contingency tables. Because the sample sizes were small and the percentages associated with the cells within the tables were often near zero, the Fisher exact test was used to compare the percentages in question. A significance level of 0.05 was used for all tests.
Results
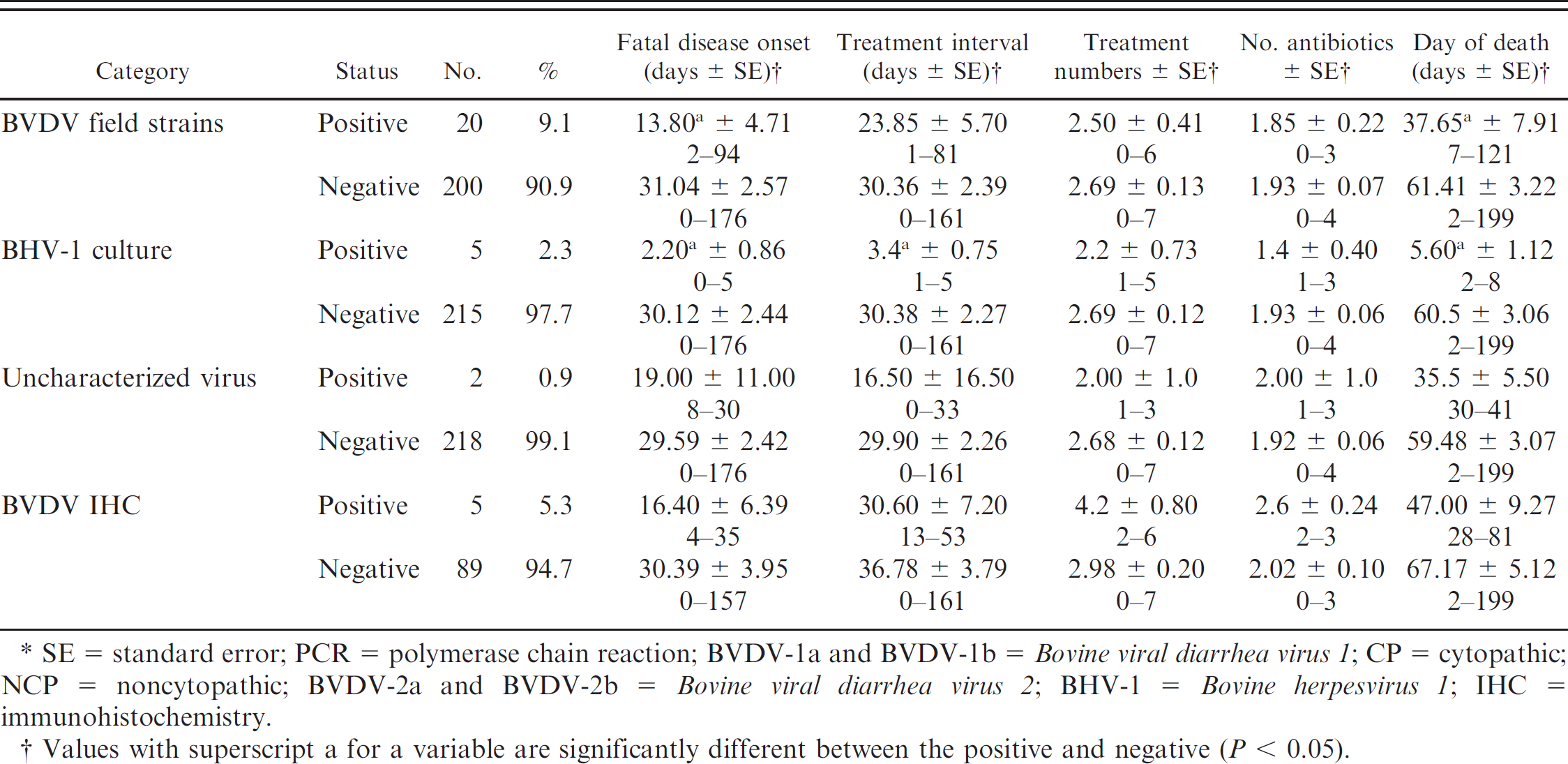
The animal mortality and treatment information from the feedlot for the 237 cattle dying of fatal pneumonia are listed in Tables 1–4. The BRD morbidity rate more than the 1-year interval was 14.7%. The mortality rate for all causes was 1.3%, of which over half (53.8%) was attributed to BRD (0.7% of all cases). The FDO day mean was 32.65 days ± SE 2.72, with a range of 0–241 days. The TI mean was 29.15 days ± SE 2.12, with a range of 0–161 days. The mean for the treatment numbers for antibiotics used was 2.65 ± SE 0.12, with a range of 0–7. The mean for the number of antibiotic treatments was 1.89 ± SE 0.06, with a range of 0–4. The mean for the day of death was 61.81 days ± SE 3.18, with a range of 2–255 days. The FDO and day of death for feedlot mortalities in the current study indicated early feeding period illness, with day of death within the first 60–62 days, mean FDO of 32.65 days, and mean death day of 61.81 days. In addition, several animals became ill and died much later in the feeding period (241–255 days).
Relationship of infectious agents and animal mortality and treatments.*
Continued.
SE = standard error; PCR = polymerase chain reaction; BVDV-1a and BVDV-1b = Bovine viral diarrhea virus 1: CP = cytopathic; NCP = noncytopathic; BVDV-2a and BVDV-2b = Bovine viral diarrhea virus 2: BHV-1 = Bovine herpesvirus 1: IHC = immunohistochemistry.
Values with superscript a for a variable are significantly different between the positive and negative (P < 0.05).
The agents identified in the lung homogenates are listed in Table 1. The following agents were isolated by bacterial culture and Mycoplasma culture in the available 220 fresh lung samples: 55 M. haemolytica (25.0%), 54 P. multocida (24.5%), 22 Histophilus somni (10.0%), 77 Arcanobacterium pyogenes (35.0%), 1 Salmonella sp. (0.5%), and 157 Mycoplasma spp. (71.4%). The following viruses were recovered by cell culture: 6 BVDV-1a NCP (2.7%), 4 BVDV-1a CP vaccine strains (1.8%), 6 BVDV-1b NCP (2.7%), 7 BVDV-2a NCP strains (3.2%), 1 BVDV-2b CP strain (0.5%), and 5 BHV-1 (2.3%). There were 2 agents that were CP in cell culture yet negative by all the FAT conjugates; these were listed as uncharacterized viruses. The BHV-1 strains isolated in the current study were not differentiated as field strains or vaccine strains. The BVDV-1a CP strains were identical to the 5′-UTR region of the BVDV-1a NADL strain, which is the virus in the modified live virus (MLV) vaccine used at processing. There were 20 BVDV strains categorized as BVDV field strains (total of 24 minus the 4 BVDV-1a CP vaccine strains). By using gel-based RT-PCR assays on the 194 lung homogenates for BRSV and BCoV, there were 21 (10.8%) BCoV positive and 9 (4.6%) BRSV positive by using lung homogenates. Approximately 5% (5.3% [5/94]) of the ear notches from the fatal cases were BVDV IHC positive.
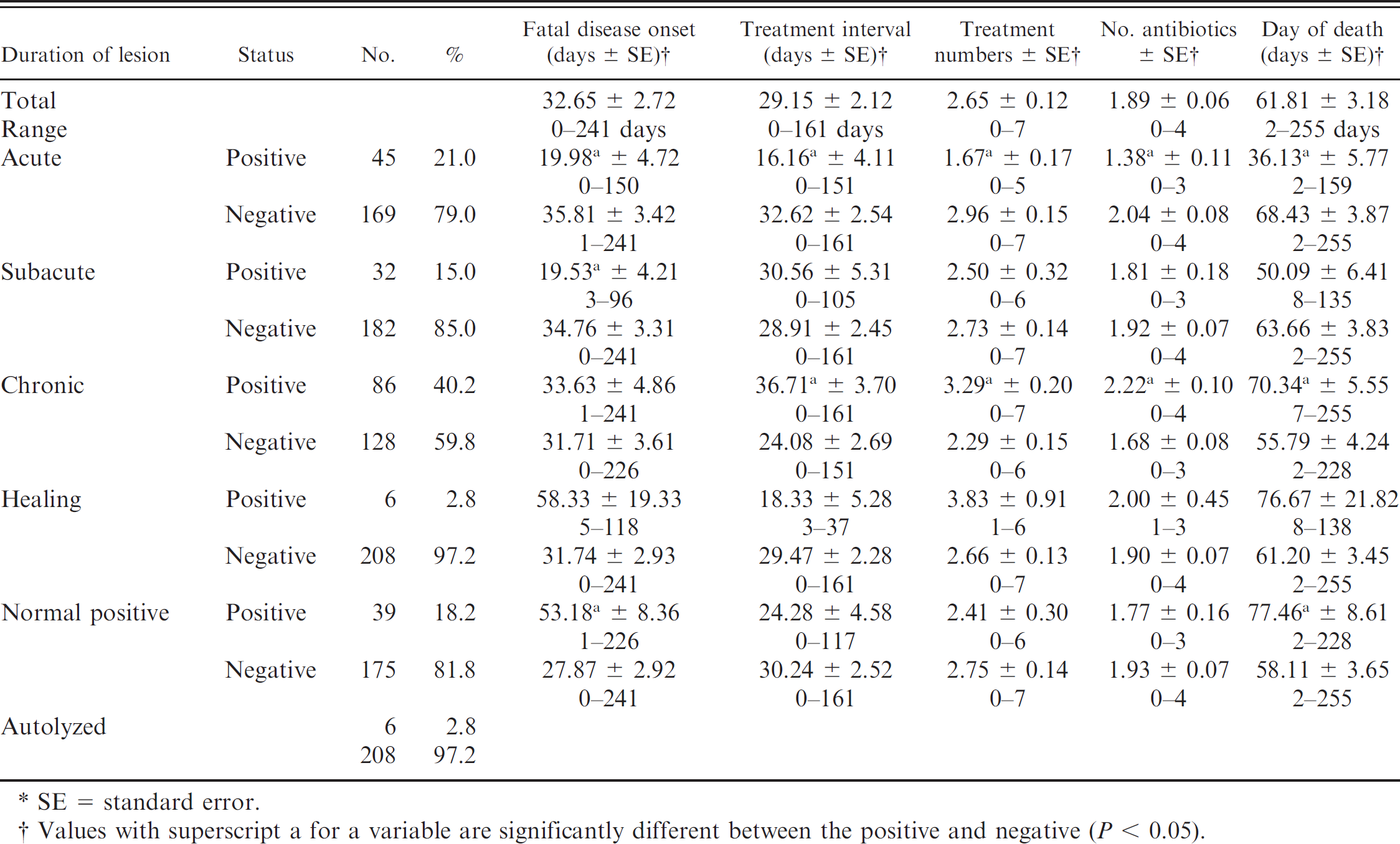
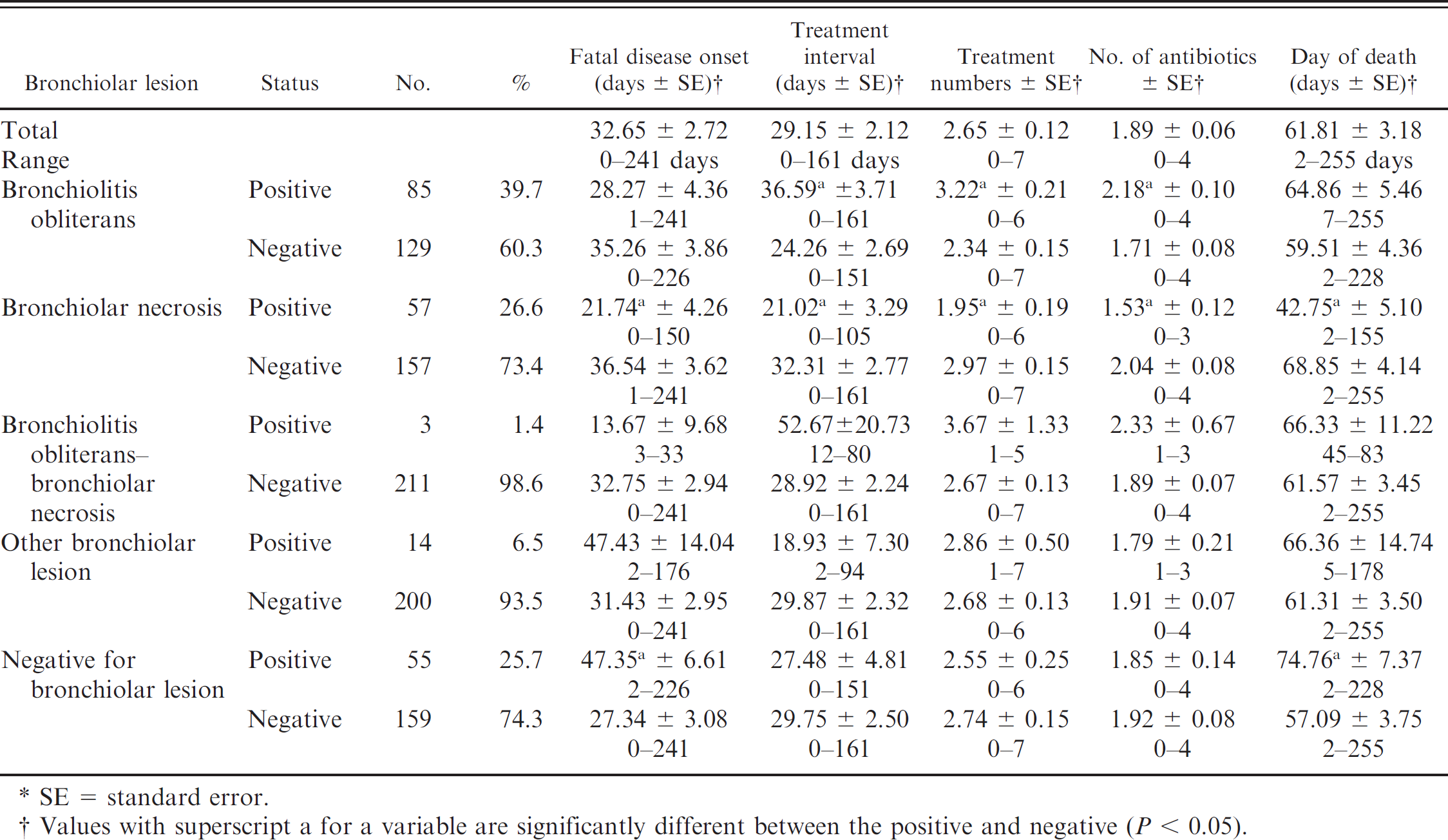
The types of pneumonia, duration of lesions (acute to chronic), and bronchiolar lesions are listed in Tables 2–4. The major types of pneumonia were as follows: 58 cases of lobar bronchopneumonia (LBP; 27.1%), 105 cases of LBP with pleuritis (LBPP; 49.1%), 11 cases of interstitial pneumonia (5.1%), 3 cases of bronchointerstitial pneumonia (1.4%), 2 cases of septic pneumonia (0.9%), 1 case of pneumonia with embolic foci (0.5%), 6 cases of other types of pneumonia (2.8%). The remaining specimens included 22 normal sections of lung (10.3%), and 6 autolyzed sections of lung (2.8%). Descriptions of the types of duration of pneumonias were 45 acute (21.0%), 32 subacute (15.0%), 86 chronic (40.2%), and 6 healing (2.8%). The remaining specimens included 39 normal lungs (18.2%) and 6 autolyzed lungs (2.8%). When the duration and type of pneumonia were classified together, the 7 most-common diagnoses were chronic LBPP (23.4%), chronic LBP (15.4%), acute LBPP (15.0%), subacute LBPP (9.8%), subacute LBP (5.1%), interstitial pneumonia (5.1%), and acute LBP (3.7%). Bronchiolar lesions included 85 cases of bronchiolitis obliterans (39.7%), 57 cases of bronchiolar necrosis, (26.6%), 3 cases of bronchiolitis obliterans–bronchiolar necrosis (1.4%), 14 cases of other bronchiolar lesions (6.5%). The remaining 55 specimens were negative for bronchiolar lesions (25.7%).
Relationship of lesions of pneumonia and animal mortality and treatments.*

SE = standard error.
Values with superscript a for a variable are significantly different between the positive and negative (P 0.05).
Relationship of duration of lesions and animal mortality and treatments.*
SE = standard error.
Values with superscript a for a variable are significantly different between the positive and negative (P < 0.05).
The various infectious agents and the lung lesions were analyzed for a positive or negative relationship for each respective variable compared with each of the 5 clinical categories, including FDO, TI, number of antibiotic treatments, number of antibiotics, and death day (Tables 1–4). Significant differences between positive and the negative values (P < 0.05) were found as follows. Cattle that were virus positive in the lung had earlier FDO and death days compared with cattle with negative test results. Cattle from which bacteria were isolated had earlier FDO, a longer TI, greater number of antibiotic treatments, and more antibiotics administered compared with cattle with negative test results. Cattle with Mycoplasma spp. were treated more often and with more antibiotics than the Mycoplasma spp.–negative cattle. The cattle with M. haemolytica–positive lungs were treated earlier, treated less frequently, and died earlier compared to cattle that were negative for M. haemolytica. Cattle with P. multocida had a longer TI and were treated more frequently compared with cattle that were negative for P. multocida. Cattle that were positive for A. pyogenes had a longer TI, received more treatments, and required more antibiotics compared with cattle that were negative for A. pyogenes. The BCoV-positive cattle became ill earlier than the BCoV-negative cattle. Cattle that were positive for the BVDV-1a CP vaccine strain were treated earlier, had a shorter TI, and died earlier compared with cattle that were test negative. The BVDV-1a NCP–positive cattle were treated earlier than the BVDV-1a NCP–negative cattle. The BVDV-1b NCP–positive cattle were treated considerably earlier, had a shorter TI, required fewer treatments, and died earlier than cattle that were negative for BVDV-1b NCP. The group of BVDV field strain–positive cattle was treated earlier and died earlier compared with BVDV field strain–negative cattle. Cattle positive for BHV-1 were treated earlier than BHV-1–negative cattle, had a shorter TI, and died earlier.
The types of pneumonia were analyzed with respect to their relationships to mortality and treatment information (Table 2). Significant (P < 0.05) findings included cattle with LBPP, which were treated earlier and died earlier than the LBPP-negative cattle; and cattle with bronchointerstitial pneumonia (BIP), which had a shorter TI, received fewer treatments, and received fewer antibiotics than BIP-negative cattle. In addition, the cases of septic pneumonia appeared earlier in the feeding period, had a shorter TI, and died earlier than cattle without septic pneumonia.
Duration of acute, subacute, and chronic lung lesions, were analyzed with respect to mortality and treatment information (Table 3). Several significant differences (P < 0.05) were found between the positive and negatives for each variable. Cattle with acute lung lesions compared with negative cattle were treated earlier, had a shorter TI, had fewer treatments, received fewer antibiotics, and died earlier in the feeding. Cattle with subacute lung lesions were treated earlier than negative cattle. Cattle with chronic lesions had a longer TI, were treated more frequently, received more antibiotics, and died later in the feeding period compared with cattle negative for chronic lesions.
Relationship of bronchiolar lesions and animal mortality and treatments.*
SE = standard error.
Values with superscript a for a variable are significantly different between the positive and negative (P < 0.05).
The types of bronchiolar lesions were compared with mortality and treatment information (Table 4) with several significant relationships (P < 0.05). Cattle with bronchiolar necrosis had earlier onset of disease, had a shorter TI, were treated fewer times, received fewer antibiotics, and died earlier in the feeding period compared with cattle negative for bronchiolar obliterans. Cattle positive for bronchiolitis obliterans lesions had a longer TI, were treated more frequently, and received more antibiotics compared with cattle negative for bronchiolar obliterans. Cattle without bronchiolar lesions had a later FDO and died later in the feeding period compared with cattle with bronchiolar lesions.
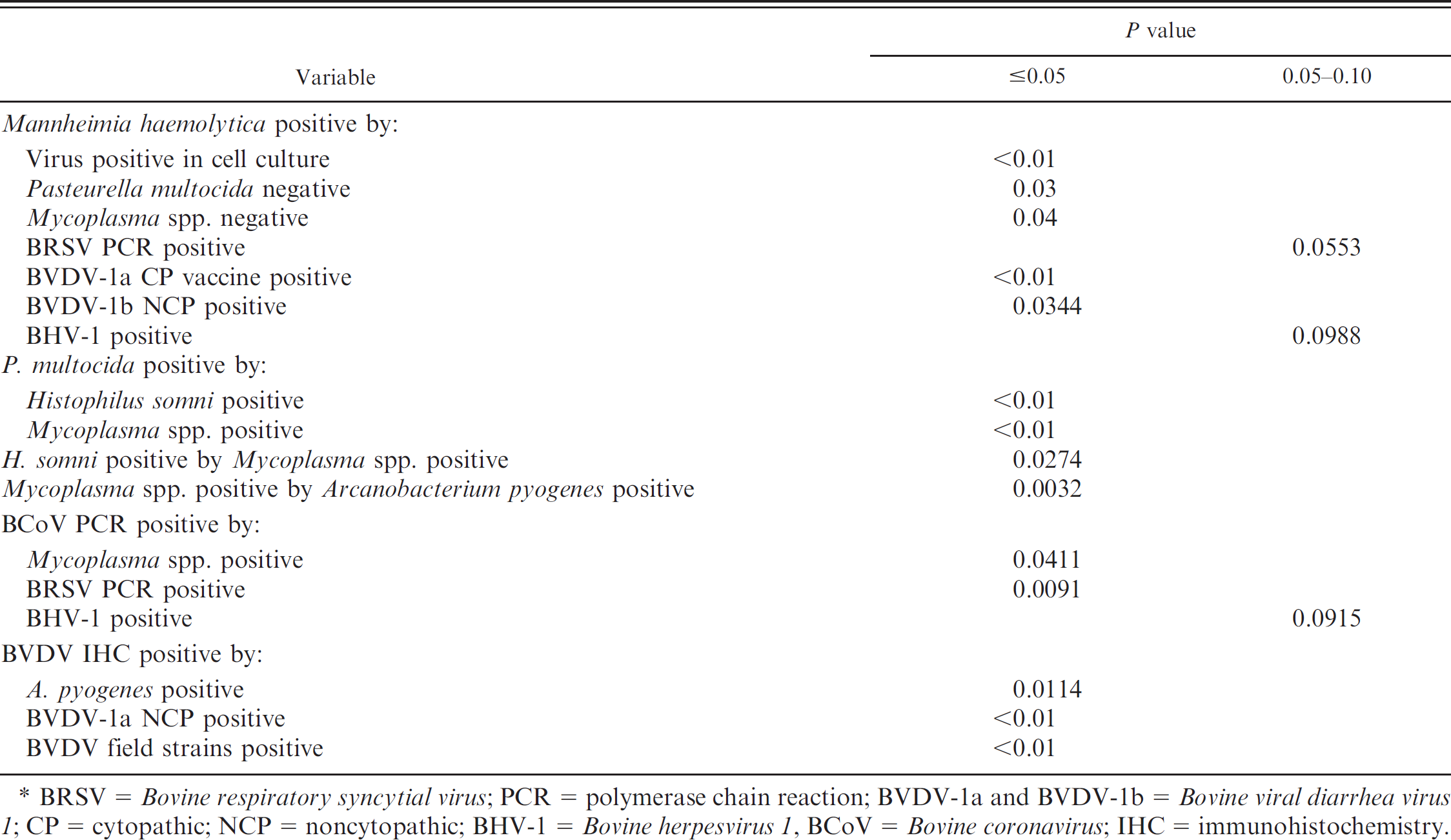
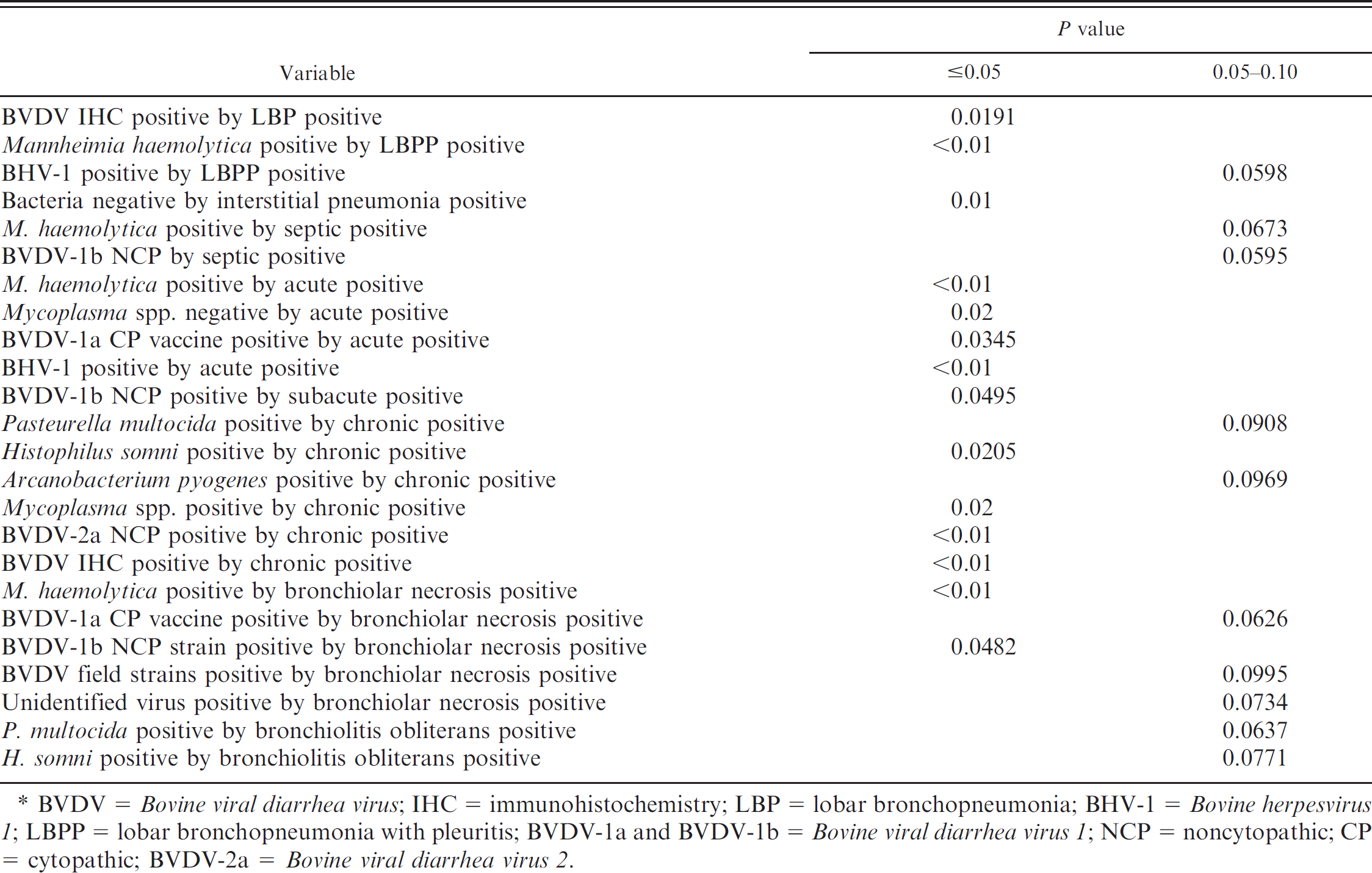
The linear correlations of pairs of variables are listed in Tables 5–7, and these are reported by P values of <0.05 or between 0.05 and 0.10. The relationship of concurrent isolation of a specific infectious agent with another infectious agent is indicated in Table 5. Mannheimia haemolytica isolation was positively correlated with several agents (P < 0.05): virus-positive cases detected by isolation in cell culture, BVDV-1a CP vaccine, and BVDV-1b NCP. The PCR-positive BRSV and BHV-1 cases were positively correlated with M. haemolytica infection at P > 0.05–0.10. There was statistical significance (P = 0.03) that M. haemolytica isolation occurred in the absence of P. multocida or Mycoplasma spp. infections. Pasteurella multocida isolation was positively correlated with H. somni and Mycoplasma spp. isolation (P < 0.01). Histophilus somni isolation was positively correlated with isolation of Mycoplasma spp. (P < 0.05). The isolation of Mycoplasma spp. was positively correlated with the isolation of A. pyogenes (P < 0.05).
Bovine coronavirus detection by PCR was positively correlated to Mycoplasma spp. isolation (P < 0.05) and BRSV PCR-positive status (P < 0.05), as well as with BHV-1 recovery (P = 0.0915). The positive BVDV IHC results were positively correlated to the isolation of A. pyogenes, BVDV-1a NCP, and BVDV field strains (P < 0.05).
The etiologic agent association with the type of pneumonia is presented in Table 6. The detection of BVDV by IHC was positively correlated with positive LBP (P < 0.05). The cases of LBPP were positively correlated with recovery of M. haemolytica (P < 0.01) and BHV-1 (P = 0.0598). The absence of bacterial recovery correlated with the presence of interstitial pneumonia (P < 0.05). There was no correlation between positive BRSV PCR and interstitial pneumonia (P < 0.05; data not shown in Table 6). Cases with septic lesions were related to M. haemolytica–positive (P = 0.0673) cases and BVDV-1b NCP (P = 0.0595).
Relationships among infectious agents.*
BRSV = Bovine respiratory syncytial virus; PCR = polymerase chain reaction; BVDV-1a and BVDV-1b = Bovine viral diarrhea virus 1: CP = cytopathic; NCP = noncytopathic; BHV-1 = Bovine herpesvirus 1, BCoV = Bovine coronavirus; IHC = immunohistochemistry.
The relationship of the etiologic agent status and duration of lesion is presented in Table 6. Cases of acute duration were positively correlated with M. haemolytica, BVDV-1a CP vaccine, and BHV-1 (P < 0.05). Acute cases were not associated with recovery of Mycoplasma spp. (P = 0.02). For cases of subacute duration, a positive correlation existed with BVDV-1b NCP (P < 0.05). Cases of chronic duration were positively correlated (P < 0.05) with recovery of H. somni, Mycoplasma spp., and BVDV-2a NCP, as well as a positive test result for BVDV IHC and isolation of P. multocida or A. pyogenes (P > 0.05–0.10).
The relationship of the etiologic agent–positive status and the presence of bronchiolar lesions is presented in Table 6. Recovery of M. haemolytica and BVDV-1b NCP strain was positively correlated (P < 0.05) with bronchiolar necrosis. Bronchiolar necrosis also was positively correlated with BVDV-1a CP vaccine–positive cases, BVDV field strains, or unidentified virus (P > 0.05 0.10). The lesions of bronchiolitis obliterans were positively correlated with the recovery of P. multocida or H. somni (P > 0.05–0.10).
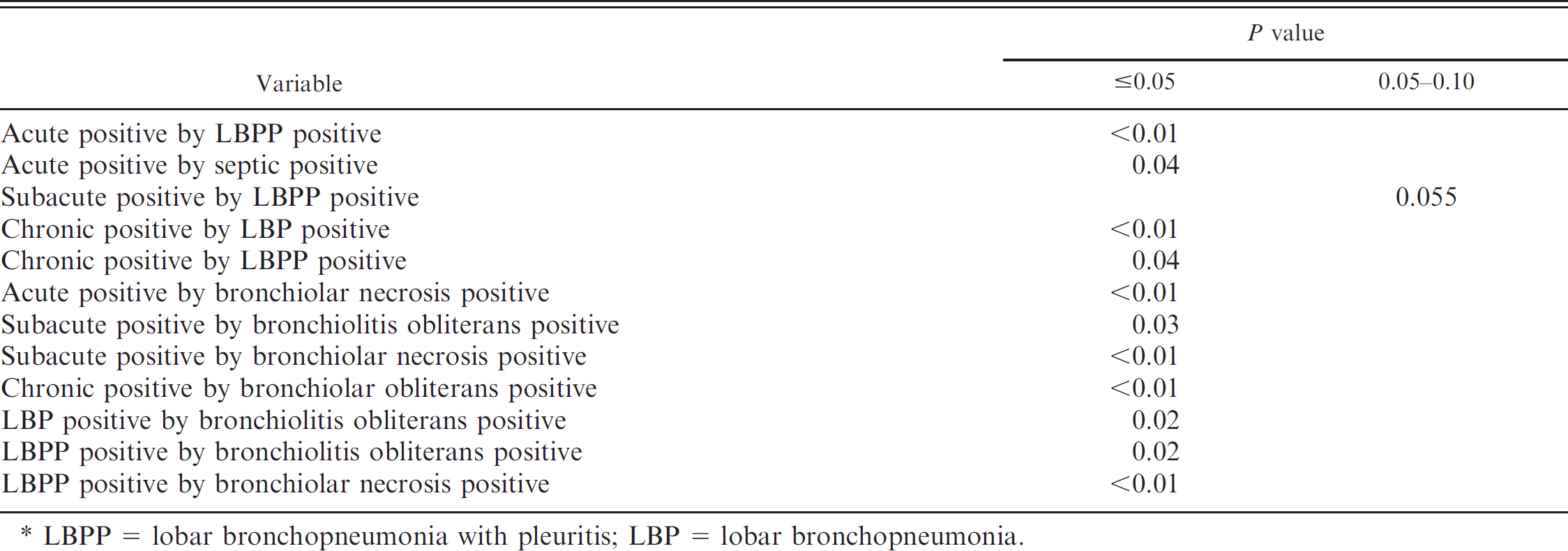
The relationship of the duration of lesions and the type of pneumonia is presented in Table 7. Cases of pneumonia that were acute in duration were positively correlated with LBPP or septic disease (P < 0.05). Subacute cases of pneumonia were positively correlated with LBPP at P > 0.5–0.10. Chronic cases of pneumonia were positively correlated with LBP or LBPP (P < 0.05).
The relationship of the bronchiolar lesions, duration of lesions, and pneumonia type is presented in Table 7. Cases with bronchiolar necrosis were positively correlated with acute duration, subacute duration, or LBPP (P < 0.05). Bronchiolitis obliterans positive lesions were positively correlated with subacute or chronic duration, LBP, or LBPP (P < 0.05).
Discussion
The role of BRD in U.S feedlots remains an important problem in the production beef cattle. The role of various infectious agents in BRD is broad, and numerous viruses, bacteria, and Mycoplasma spp. have been reported from diverse cattle populations. Many reports of BRD investigations exist and are too numerous to entirely document or reference. These various studies should be considered collectively to determine the causation of BRD by one or multiple agents that interact in the pathogenesis of BRD. Publications include case reports of single animals and larger surveys of live-animal populations as single or multiple groups. These diverse publications also include serologic evidence of infection by or isolation of pathogens from the upper respiratory tract (nasal swabs) as well as summaries of necropsy reports with gross lesion descriptions and recovery of infectious agents from dead animals. Studies of the end stage of BRD (fatal cases of pneumonia) have the shortcoming of not being able to determine if the agent(s) recovered was the primary pathogen, etiologic agent of the disease, or present concurrently only in the end stage of disease. Similarly, studies in which the diagnosis of infectious agents were based on serocon-version or agent detection in nasal swabs might not have been predictive of the infectious agents that cause pathogenic processes in the lung or result in concurrent infection in previously diseased individuals.
Relationship of infectious agents and lesions.
BVDV = Bovine viral diarrhea virus; IHC = immunohistochemistry; LBP = lobar bronchopneumonia; BHV-1 = Bovine herpesvirus 1; LBPP = lobar bronchopneumonia with pleuritis; BVDV-1a and BVDV-1b = Bovine viral diarrhea virus 1; NCP = noncytopathic; CP = cytopathic; BVDV-2a = Bovine viral diarrhea virus 2.
Relationship between lesion and duration.*
LBPP = lobar bronchopneumonia with pleuritis; LBP = lobar bronchopneumonia.
The current study was designed to detect several infectious agents and various pneumonic lesions in lungs collected from cases of fatal pneumonia in a commercial feedlot and to relate the agents and lesions to animal mortality and treatment information. Another purpose of the present study was to correlate any agents with 3 aspects of lung lesions, including the type of pneumonia, duration of the lung lesions, and type of bronchiolar lesions. The present study represents the first comprehensive study of feedlot pneumonias in the United States since the 1970s. 17,18 Recently, 2 published studies from Canada reported the infectious agents and lesions from cattle in feedlots of Ontario and Western Canada (Alberta). 2,13 Both of the Canadian studies evaluated information derived from the cattle that died within 60 days of entering the feedlot and that originated from several different feedlots. Therefore, these 2 study populations differed from the current study, which focused on fatal BRD cases in only 1 feedlot over a 1-year duration. In addition, the BRD cases in the present study were collected over the entire feeding period (200+ days) rather than the first 60 days.
Making comparisons to other studies, especially those that date back 30 years or more, is difficult. Likewise, making comparisons with the 2 recent Canadian studies is difficult because various analytical methods were used to identify any agents (molecular technologies and IHC vs. traditional culture assays) and data collection times differed (the initial 60 days in the feedlot vs. the entire feeding period of > 200 days). In addition, other factors such as climate, origin of the animals, and geographical region, differed for the Canadian studies and the present study in the south central United States. Also, the sample sizes are at variance with the Ontario and the Alberta studies (99 cases 2,13 vs. 237 cases in the current study).
The present study identified viruses either by cell-culture isolation or by RT-PCR assays and found 20 (9.3%) cattle with BVDV field strains, 4 (1.8%) with BVDV vaccine strains, 5 (2.3%) with BHV-1 strains, 21 (10.8%) with BCoV, and 9 (4.6%) with BRSV. Laboratory testing in the Ontario report indicated that 35 of 99 cases (35.4%) were positive for BVDV, 9 of 99 (9.1%) for BRSV, 6 of 99 (6.1%) for BHV-1, 3 of 99 (3.0%) for BPIV-3, and 2 of 99 (2.0%) for BCoV. 13 The Ontario study classified BVDV as either major subtype BVDV-1 or BVDV-2 based on PCR. The Ontario study reported 11 of 24 strains as NCP and 13 of 24 as CP strains. In the Ontario study, the 4 BVDV-2 strains were NCP, with 7 BVDV-1 NCP and 13 BVDV-1 CP. The BVDV-1 CP isolates could possibly have been of vaccine origin, because MLV vaccines usually are BVDV CP strains. The Ontario study classified the BVDV strains by major type (BVDV-2 and BVDV-1) as well as biotype (CP or NCP). Twenty of 24 BVDV-1 strains and 4 BVDV-2 strains were identified. However, the Ontario study did not differentiate the BVDV strains or vaccine strains as in the current study. In the present study, the incidence of BVDV field strains and vaccine strains were lower (10.2%) than in the Ontario study (35.4%). The Alberta study used IHC testing on tissues to identify BHV-1, BVDV, BRSV, BPIV-3, M. haemolytica, H somni, and Mycoplasma bovis, which was a different procedure to detect infectious agents 2 compared with the current study. IHC testing for P. multocida, A. pyogenes, or BCV was unavailable in the Alberta study, and the detection of these agents was not reported. In the Alberta study, M. haemolytica and M. bovis were the most commonly identified infectious agents. The association of concurrent agents was noted in the Alberta study and the present study, even though different analytical techniques were used. In the Alberta study, 25 of 26 (96%) BVDV IHC–positive cattle were also IHC positive for M. haemolytica. In the current study, there was a positive correlation (P < 0.05) with M. haemolytica–positive isolation, BVDV-1a CP vaccine strain, or BVDV-1b NCP strain. In the Alberta study, 12 of 15 (80%) H. somni IHC–positive cattle were also positive for M. bovis, whereas 0 of 15 H. somni–positive cattle were IHC positive for M. haemolytica or BVDV positive. There was no significant (P > 0.10) association between positive IHC results for M. haemolytica and M. bovis. The current study had similar results, with a positive correlation (P < 0.05) between H. somni–positive and Mycoplasma spp.–positive cattle; however, no correlation was found between positive status for H. somni or BVDV field strains (P < 0.05), or between M. haemolytica–positive and Mycoplasma spp.–positive cattle (P > 0.05). This is most likely because of the positive correlation of M. haemolytica with acute lesions, and the positive correlation of Mycoplasma spp. with chronic lesions.
In the Ontario feedlot study, 26 of 99 cattle (26.3%) tested positive for M. haemolytica, 19 of 99 (19.2%) for P. multocida, 14 of 99 (14.1%) for H. somni, and 19 of 99 (19.2%) for A. pyogenes. Thus, it appears that the percentage recovery of M. haemolytica, P. multocida, and H. somni was within 5–6% of the current study. However, in the Ontario study, several Mycoplasma spp. were listed: M. bovis (82/99), M. arginini (72/99), M. bovirhinis (1/99), and Ureaplasma diversum (15/99). The report of feedlot diseases in the 1970s indicated that 62% of the lungs contained Pasteurella spp. in some combination, Mycoplasma spp. in some combination in 50% of the lungs, and Pasteurella spp. and Mycoplasma spp. in some combination in 35% of the lungs. 18 The high percentage of Mycoplasma spp. recovery in the 1970s report is similar to the current study, wherein 71.4% of cattle were positive for Mycoplasma spp. 18 With further identification of Mycoplasma spp. isolates in the present study, 72% were identified as M. bovis (Fulton, unpublished data).
The current study is unique in that BVDV subtypes were identified and the sequences of isolated BVDV were compared with the vaccine strain and allowed identification of virus infections that resulted from MLV vaccination. Previously identified BVDV isolates (BVDV-1a, BVDV-1b, and BVDV-2a diagnostic laboratory isolates) were identified in cases of respiratory disease. 10 Likewise, in the current study, the BVDV NCP strains were isolated more frequently (19/24), and BVDV-1a NCP, BVDV-1b NCP, and BVDV-2a NCP were recovered. Results of the current study, which used the molecular analysis of BVDV, underscores the importance of separating BVDV vaccine strains from BVDV field strains. Four BVDV-1a CP strains were isolated, yet they were determined to be the BVDV-1a vaccine strain that is CP and identical to the NADL strain in the vaccine. Isolation of the BVDV vaccine strain may not indicate cause and effect but rather coincidental isolation that reflected vaccination. In addition, BVDV-2b CP was isolated, which is the first to have been identified in the United States. The source of this unique viral strain was not determined, based on available procurement records.
Bovine respiratory syncytial virus is not a highly recovered virus from affected tissues by using cell-culture isolation. The use of PCR assays to detect BRSV could be a useful diagnostic procedure, because 4.6% of the lung homogenates were PCR positive for this virus but cell culture failed to detect this pathogen. There was a lack of statistical significance for BRSV with interstitial pneumonia in the current study, with only 1 of 11 cases testing positive for BRSV by PCR (P > 0.10). In a study of deaths in Saskatchewan feedlots in 1994–1995, only 2 of 28 cases of interstitial pneumonia were positive for BRSV by IHC testing. 23 In that study and the current study, an association between BRSV and interstitial pneumonia could not be made. In addition, an association between recovery of bacteria or Mycoplasma spp. and the presence of interstitial pneumonia could not be made in the current study.
Several findings in the present and previous studies associate the isolation of selected bacteria with pulmonary lesions. The current study found a positive correlation of M. haemolytica isolation and LBPP, the most common type of pneumonia in this study (49.1% [105/214]). The Alberta study found a significant relationship between M. haemolytica IHC–positive cattle and fibrinonecrotizing pneumonia and fibrinous pleuritis. Cases of acute or chronic duration give an expectation for the recovery of selected pathogens. In the present study, M. haemolytica recovery was positively correlated with acute lung lesions (P < 0.05). In contrast, chronic lesions were positively correlated with the recovery of P. multocida and A. pyogenes (P > 0.051–0.10), as well as H. somni and Mycoplasma spp. (P < 0.05).
The detection of BCoV has been performed by using special susceptible cell monolayers and trypsin treatment. 13 The current study did not have access to such cell culture techniques, yet 10.8% of the lungs were positive for BCoV by PCR assay, which was higher than the Ontario study (2.0% [2/99]), which used cell culture isolation. 13 It is possible that the BCoV assay was more sensitive than the cell-culture assay used in the Ontario study, or perhaps there are differences in the ecology of BCoV (regional differences) or study design.
The current study provides additional evidence for the role of BVDV in BRD as reported in other studies. 9–12,20 The distribution of BVDV subtypes in the study suggests possible variation in pathogenicity of different BVDV subtypes. BVDV-1a NCP and BVDV-1b NCP strains had an earlier FDO compared with cattle that tested negative for these viruses. However, BVDV-1b NCP had additional differences from other BVDV subtypes, with a shorter TI (5.50 vs. 30.16 days) and earlier day of death in the feed yard (9.83 vs. 60.64 days). The TI and day of death for cattle with BVDV-1a NCP and BVDV-2a NCP were not statistically significant from cattle that tested negative for these viruses. There was only 1 animal with BVDV-2b CP, and it died early in the feeding period (day 10) after onset of illness (day 6). In general, the BVDV field strains were related to early disease in the feedlot (13.8 vs. 31.04 days for FDO) and earlier death (37.65 vs. 61.41 days) for the BVDV field strain–negative cattle. Those cattle dying with BRD had a BVDV IHC–positive rate of 5.3% when using skin samples as the diagnostic specimen. This percentage is similar to the feedlot deaths previously reported, with a 2.5% BVDV IHC positive test result in fatal cases. 20
The relationship between recovery of infectious agents with clinical data, such as FDO, TI, number of treatments, number of antibiotics, and day of death, had numerous positive or negative differences for the variables that were evaluated (P < 0.05). Results from the current study differ somewhat in that cases of pneumonia had a mean FDO of 32.65 days, mean TI of 29.15 days, mean treatments of 2.65, mean number of antibiotics of1.89, and mean day of death of 61.81 days. These findings compare similarly with cases of fatal fibrinous pneumonia in an Alberta feedlot study (1985–1988) in which the day of death ranged from 19 to 22 days. 22 In a Saskatchewan feedlot study of BRD cases (1990–1992), the day of death was 25 for 1990, 14 for 1991, and 56 for 1992. 28 These 2 studies had markedly earlier days of death in the feedlot compared to the current study (61.81 days). Even using only the cases in the current study (LBP mean 70.52 and LBPP mean 51.25 of day of death), the death days were considerably longer than the 1985–1988 Alberta study 22 or for 1990 and 1991 in the Saskatchewan studies. 28
The Ontario study reported in 2006 included data of FDO for several necropsy diagnoses for pneumonias. 13 When using the criteria of acute fibrinosup-purative or caseonecrotic bronchopneumonia and caseonecrotic bronchopneumonia, mean FDOs were 14.6 days and 15.5 days, respectively, which were shorter than for all of the pneumonia cases in the current study. Use of different lesion classification criteria may complicate comparisons; however, it is obvious that pneumonia lesions in the present study were more chronic than in previous studies.
Several etiologic agents had decided differences in the animal and treatment information. Several infectious agents were associated with differences in the FDO for cattle with a positive versus negative test result. Cattle with infectious agents had a uniformly shorter FDO (P < 0.05) when they were positive for bacteria culture, cell culture virus, M. haemolytica, H. somni, BCoV PCR, BVDV-1a CP vaccine, BVDV-1a NCP, BVDV-1b NCP, BVDV field strains, or BHV-1. Thus, it appears that bacterial, viral, and mycoplasma infections were involved in the development of early illness in the feedlot. However, bacteria were impacted by TI, with longer treatments as a cumulative group and as individual bacterial strains. Cattle with a longer TI (P < 0.05) had a positive bacterial culture or isolation of P. multocida, H somni, and A. pyogenes. In contrast, cattle with viral infections (BVDV-1a CP vaccine, BVDV-1b NCP, and BHV-1) appeared to have a shorter TI. The current study of BRD cases with a TI of 29.15 days is quite different from the TI in the Saskatchewan study (1990–1992), which reported a TI of 6–20 days.
Treatment numbers is a relatively new parameter used to evaluate BRD cases. In recent years, additional antibiotics have become available. The impact of the number of antibiotic treatments and agent involvement was examined in the current study. In general, it appeared that the increased number of treatments was correlated with the recovery of bacteria and Mycoplasma spp. Cattle with an increased recovery of infectious agents had increased treatment numbers (P < 0.05). These included cattle that were positive for bacterial culture, Mycoplasma spp., P. multocida, and A. pyogenes. However, cattle with M. haemolytica infection were associated with fewer treatment numbers. This is likely because of a shorter TI and day of death, which resulted in fewer numbers of antibiotic treatments. Another example is fewer treatments associated with BVDV-1b NCP, which also had an extremely short day of death.
The number of antibiotics for BRD therapy was correlated with recovery of certain agents. The increased number of antibiotics was associated with increased numbers of cattle with a positive bacterial culture or isolation of Mycoplasma spp and A. pyogenes (P < 0.05). Although an association exists between treatment members and the number of antibiotics versus agent recovery, it should be noted that the appropriate antibiotic therapy used in the current study may likely reduce clinical illness and mortality from several agents.
Death in the feedlot for the fatal pneumonia in the current study was 61.81 days, which is in contrast to the Saskatchewan study (1990–1992), which had days of death of 25 (1990), 14 (1991), and 56 (1992). 22 Differences may be related to regional or management practices, distance of travel for cattle, extent of commingling, or antibiotic therapy. Selected animals may have survived longer, yet died from infections after antibiotic use. In addition, the virulence of the infectious agents may have differed from those of the current study.
The relationship of various etiologic agents with each other and/or with different lesions were numerous. It is possible that many positive correlations may have been more related to the time of death, which occurred very early or long after arrival in the feedlot. An early or late death would reflect opportunities for treatment. It is noted that the relationship with the number of treatments and number of antibiotics was often evident with agents such as P. multocida, Mycoplasma spp., and A. pyogenes. For example, an animal dying quite early in the feeding period and with an extremely short TI would not have been treated often and probably would not have received multiple antibiotics.
New findings from the current study included the distribution of the BVDV NCP strains BVDV-1a, BVDV-1b, and BVDV-2a in fatal feedlot pneumonia cases. The prevalence of BVDV-1b strains is important, because the most commonly used BVDV vaccines in the United States contain BVDV-1a and BVDV-2a and not BVDV-1b. Potential control of BRD in feedlots should use vaccines that protect against the BVDV-1b strains under field conditions. Also, monitoring for additional BVDV strains by use of viral subtyping is important to detect emerging BVDV in the United States. Furthermore, the current study demonstrated the benefits of viral subtyping, because BVDV-2b CP was identified for the first time in the United States.
The present study supports the development and use of molecular diagnostic tests such as RT-PCR and real-time PCR as molecular diagnostic assays. These tests for genomic material could be used in addition to traditional culture tests. Likewise, the use of IHC staining to detect specific antigens in tissues is increasing. The presence of BVDV in the feedlot also can be identified by using skin biopsy specimens and BVDV IHC staining. This analytical technique can identify PI BVDV in dead or persistently infected cattle in the feedlot.
Acknowledgements
This study was funded by a grant from The Noble Foundation (Ardmore, OK) and the Oklahoma Agriculture Experiment Station (Oklahoma State University, Still-water, OK). The technical assistance and expertise of Patricia Federico (USDA, ARS, NADC, Ames, IA) is greatly appreciated.
Footnotes
a.
BoviShield™ 4, Pfizer Animal Health, New York, NY.
b.
SAS Institute Inc., Cary, NC.
