Abstract
Parasites resembling Neospora caninum or Toxoplasma gondii were detected by cytologic examination of cerebrospinal fluid (CSF) from a dog with neurologic disease. The dog became severely ill and was euthanized. Canine tissue homogenates were used for direct parasite isolation in cell culture, bioassay in 2 mouse lineages, and PCR. T. gondii was isolated in monkey kidney cells, and species identity was confirmed by PCR. Inoculated parasites were highly virulent for mice, which developed clinical signs and were euthanized immediately. PCR-RFLP for T. gondii using the cultured isolate (TgDgBA22) was conducted with 12 genetic markers, and a unique recombinant strain was identified. Detection of T. gondii by CSF cytology, although described in humans, had not been reported previously in dogs, to our knowledge, and was crucial for the diagnosis of toxoplasmosis in the examined dog.
Toxoplasma gondii, which is one of the most successful protozoan parasites, uses a great variety of animals as hosts, is vertically and horizontally transmitted, possesses opportunistic behavior, and is distributed globally. 2 Since ~2000, outbreaks of human toxoplasmosis in Brazil have affected hundreds of people.12,14
Companion animals, such as dogs, usually share with their owners some means of parasite transmission and may also be affected clinically by toxoplasmosis. Scarce data are available on the isolation of viable T. gondii from clinical cases of canine toxoplasmosis. 3 Serology is the major tool used to assess clinical toxoplasmosis; however, analysis of cerebrospinal fluid (CSF) is an alternative method for patients with central nervous system infection. 5 Despite the importance of CSF analysis in veterinary medicine, we found no published report on T. gondii detection by CSF cytology in dogs. We describe here T. gondii identification based on CSF cytology, bioassay in 2 mouse lineages, in vitro isolation of the parasite, and analysis by PCR–restriction-fragment length polymorphism (PCR-RFLP). The use of animals in our study was approved by the Ethical Committee of the Federal University of Bahia, under license 32/2014.
In 2015, a stray 1-y-old female mixed-breed dog (body score: 4 of 9) was presented to the Animal Teaching Hospital at the Federal University of Bahia, Brazil, because of a fracture of the right femur. The animal had been rescued from the street, in Salvador, Bahia, after being hit by a car. Three days after surgical femur fracture repair, the dog had hyporexia and rigidity in the neck and mandible. Prescribed treatment for the dog included dipyrone (25 mg/kg, PO, q8h, for 5 d), tramadol (4mg/kg, PO, q8h, for 5 d), diazepam (0.5 mg/kg, PO, q12h, for 4 d), and metronidazole (15 mg/kg, PO, q12h, for 8 d). On day 4 after surgery, the clinical condition of the dog had worsened; the dog was in lateral recumbency with hyperextension of the fore- and hindlimbs. On day 5, physical examination findings included rectal temperature of 40.1°C, heart rate of 140/min, respiratory rate of 36/min, opisthotonus, bilateral miosis, dehydration, pale mucous membranes, prostration, semicomatose state, and intense edema, hematoma, and erythema in the right hindlimb.
A blood sample from the dog was characterized by hypochromic normocytic anemia, leukopenia with left shift, and thrombocytopenia. Blood cytology revealed moderate anisocytosis and polychromasia, activated monocytes, Lentz inclusions in leukocytes and erythrocytes, and toxic granules in the cytoplasm of segmented neutrophils. A CSF sample collected from the cisterna magna under general anesthesia showed pleocytosis with mixed populations of neutrophils and macrophages; numerous structures resembling protozoal zoites were observed in the cytoplasm of macrophages and outside the cells (Fig. 1). As the clinical condition of the dog deteriorated and it was comatose, euthanasia was elected.
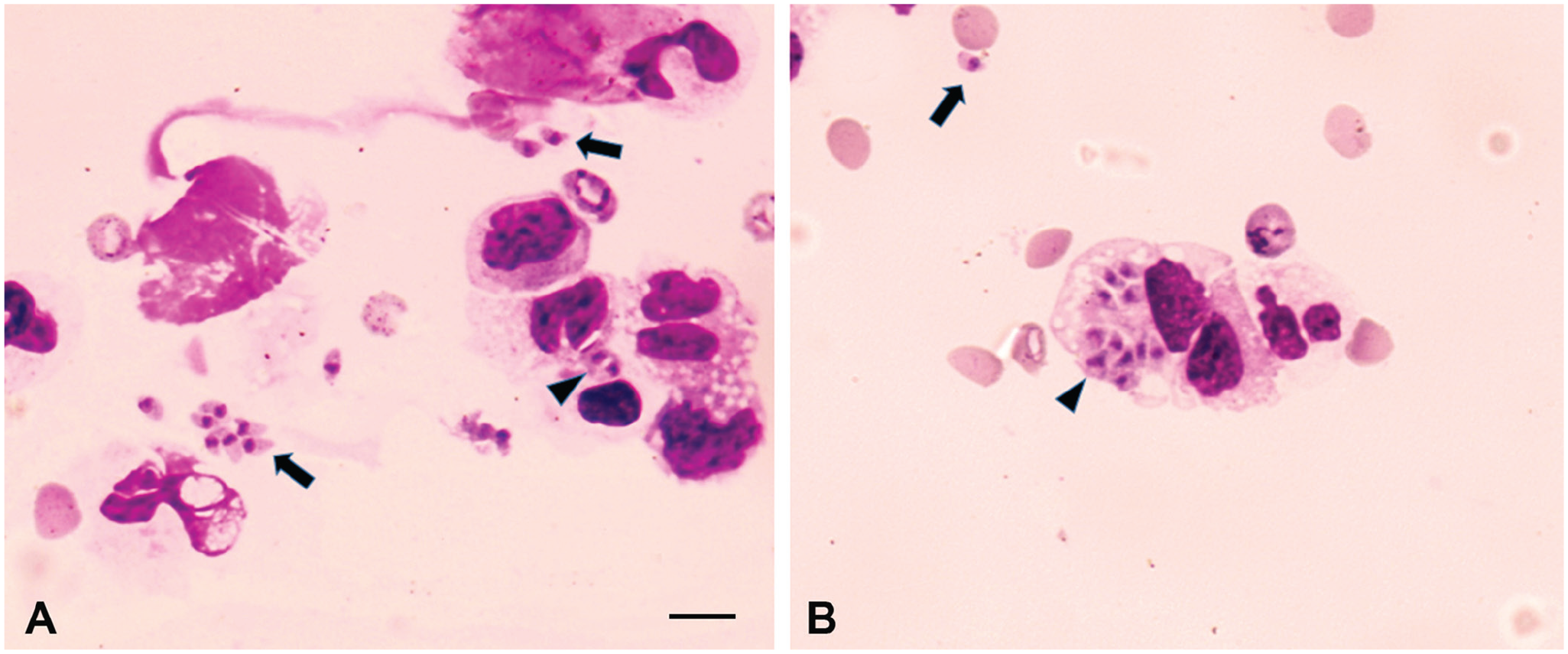
Tachyzoites resembling Neospora caninum or Toxoplasma gondii in the cerebrospinal fluid from a dog with neurologic disease.
Samples of brain, heart, lungs, and spinal cord were collected 12 h after euthanasia and placed in separate bottles containing a 2% solution of antibiotic–antimycotic (200 units/mL of penicillin, 200 μg/mL of streptomycin, 0.50 μg/mL of amphotericin B; Gibco by Thermo Fisher). During collection of tissue samples, tissue imprints were prepared on glass slides and stained using a Romanovsky-based commercial hematologic staining kit (Instant Prov; Newprov); tachyzoites were found in all examined organs of the dog (brain, spinal cord, heart, lungs), indicating systemic infection by the parasite.
The zoites observed in tissue imprints were morphologically compatible with T. gondii or Neospora caninum, given that some parasitophorous vacuoles contained organisms in pairs, suggestive of multiplication by endodyogeny. Tissue samples were processed for cell culture and bioassay. In brief, samples from brain, spinal cord, lungs, and heart were ground with PBS containing 2% antibiotic–antimycotic (Thermo Fisher), filtered through sterile gauze, and then centrifuged; 1 mL of sediment from each tissue was added into six 25-cm2 flasks containing monolayers of Vero cells (Table 1). For bioassays, 4 Swiss mice and 2 C57 gamma interferon knockout mice (Y-IFN-KO) were employed to isolate T. gondii or N. caninum. The Y-IFN-KO mice were included in the bioassay because this mouse model is more appropriate than Swiss mice for N. caninum isolation. 6 Tissue suspensions were administered to mice intraperitoneally and/or subcutaneously (Table 1).
Isolation of parasite in cell culture and by mouse bioassay using tissues from a Brazilian dog with neurologic disease.

IP = intraperitoneally; ND = not done; SC = subcutaneously; Y-IFN-KO = C57 gamma interferon knockout mouse.
Three days after direct inoculation of cell monolayers with homogenates of canine tissue samples, tachyzoites were visualized in 1 of 2 culture flasks inoculated with canine brain homogenate. The remaining flasks inoculated with tissue macerates of spinal cord, heart, and lungs had bacterial contamination and were discarded.
Six days after bioassay in mice with brain homogenate, one Y-IFN-KO mouse had a head tilt and ascites and was euthanized immediately. Tachyzoites were detected in the peritoneal fluid of the mouse and were used to inoculate 4 culture flasks containing Vero cell monolayers. Tachyzoites were seen in one culture flask 24 h after inoculation. Of the 6 mice used in the experiment, 5 had clinical signs (ruffled hair coat, ascites, or head tilt) and were euthanized immediately. One of the mice was found dead. The clinical signs and death were observed 3–12 d post-inoculation of tissue homogenates.
PCR assays for T. gondii and N. caninum were performed using cultured parasites from the dog. Cells from each culture flask were scraped, and DNA was extracted (Invitrogen Easy-DNA; Thermo Fisher), according to the manufacturer’s instructions. PCR for N. caninum was conducted using the primer pair Np6/Np21, which targets the NC-5 gene of the parasite, with an expected product of 328 bp. 22 The reactions were performed in 25-µL volumes (PCR master mix, 50 units/mL Taq DNA polymerase in a proprietary reaction buffer, pH 8.5, 400 µM each of dATP, dGTP, dCTP, dTTP and 3 mM MgCl2; Promega). DNA from N. caninum tachyzoites (NC-Bahia strain) and sterile double-distilled H20 were used as positive and negative controls, respectively. Reaction conditions were 1 cycle at 94°C for 2 min; 40 cycles of 94°C for 30 s, 53°C for 30 s, 72°C for 30 s; and a final 72°C for 5 min. PCR for T. gondii was conducted in similar conditions, except by using the primer pair TOX4/TOX5, 10 an annealing temperature of 55°C, and T. gondii tachyzoite DNA (RH strain) as positive control. The expected size of the amplicon is 529 bp. PCR products were stained (Invitrogen SYBR Gold; Thermo Fisher) and analyzed on 1.5% agarose gels under UV light.
DNA extracted from parasites derived from the dog and grown in cell culture resulted in positive reactions in the PCR assay for T. gondii. No amplification was observed in the PCR assay for N. caninum. The T. gondii isolate, referred to here as TgDgBA22, was multiplied in cell culture and stored in liquid nitrogen.
The PCR and nested PCR were conducted as reported previously. 18 The nested PCR products were treated with restriction enzymes to reveal the PCR-RFLP patterns of the isolates, using 12 genetic markers (SAG1, 5’3’SAG2, Alt.SAG2, SAG3, BTUB, GRA6, c22-8, c29-2, L358, PK1, Apico, CS3).16,21 Negative controls (water and human foreskin fibroblast strain) and positive controls for T. gondii genotypes I (RH strain), II (PTG strain), III (CTG strain), and reference strains (TgCgCa 1, MAS, TgCatBr 5) were used in the reactions. No amplification was observed for the marker L358 (Table 2). The obtained pattern of DNA bands was compared with genotypes deposited in ToxoDB (http://toxodb.org/toxo/). The TgDgBA22 strain represents a unique genotype based on the employed genetic markers (ToxoDB, accessed on June 21, 2020).
Multilocus genotyping of Toxoplasma gondii from a dog with neurologic disease.
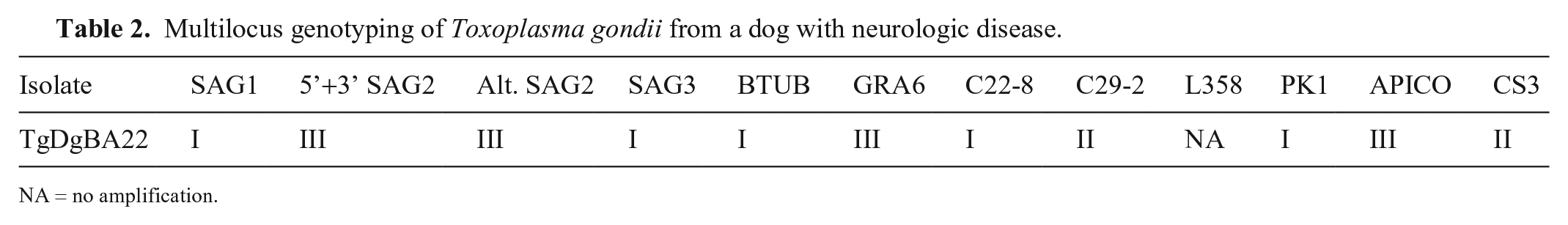
NA = no amplification.
Given the observation of Lentz inclusions in leukocytes and erythrocytes, which are indicative of morbillivirus (canine distemper virus [CDV]; Canine morbillivirus) infection, a commercial test for morbillivirus antigen (Rapid CDV Ag test kit; Bionote) was employed; serum from the dog tested positive. Morbillivirus induces immunosuppression in dogs, which predisposes these animals to opportunistic agents such as T. gondii.15,19 It is possible that morbillivirus infection contributed to immunosuppression of the dog in our case and reactivated a latent infection by T. gondii.
The newly isolated T. gondii strain TgDgBA22 was grown through direct inoculation of processed canine tissues in cell culture, as well as after bioassay in Y-IFN-KO and Swiss mice. Positive PCR results for T. gondii and negative DNA amplification using primers for N. caninum were conclusive for the diagnosis of toxoplasmosis. Coinfection with CDV was detected by blood cytology and confirmed by a commercial test for the virus antigen.
PCR-RFLP of TgDgBA22 using 12 genetic markers revealed a novel T. gondii genotype, which differed from previous isolates from dogs and other animal species in Brazil (ToxoDB). The genetic diversity of Brazilian strains of T. gondii has been reported in several studies and is characterized by high genetic recombination of the parasite.4,15,20 The virulence of the Brazilian strains of the parasite is of particular relevance, given that they have been associated with severe lesions in animals and humans.7,8
We observed intra- and extra-cellular tachyzoites during cytologic examination of CSF from the dog, which is a rare finding in canine toxoplasmosis. The bone injury and surgery might have immunosuppressed the dog and favored T. gondii and CDV growth. In a short time, from the bone fracture repair until the onset of clinical signs (3 d), rapid multiplication of T. gondii might have occurred, given that systemic toxoplasmosis was confirmed by detection of parasites in several organs of the dog. The strain TgDgBA22 was highly virulent for mice, which died or developed neurologic signs of toxoplasmosis 3–12 d post-inoculation.
Despite the detection of toxoplasmosis in dogs worldwide, isolation and genetic characterization of clinical cases in dogs are rare. 3 In studies performed outside South America, the most common T. gondii isolates, characterized by PCR-RFLP or by microsatellite analysis, were classified as type II1,9; however, a T. gondii strain characterized as type I has been detected in a dog from Italy. 13 In Brazil, genetic characterization of T. gondii in dogs has been conducted mostly after postmortem examination.9,11 In one case, toxoplasmosis was detected in a Brazilian dog with cutaneous toxoplasmosis. 17 A common finding in canine toxoplasmosis cases in Brazil is the high level of genetic recombination of the detected parasites. The only study on canine toxoplasmosis conducted in the state of Bahia, northeastern Brazil, resulted in the isolation of 4 T. gondii strains from 7 seropositive dogs 4 ; no information was provided about neurologic impairment of the tested animals. These 4 isolates were virulent for mice and had a high degree of genetic recombination.
To our knowledge, T. gondii has not been previously reported cytologically from the CSF from a dog. The isolated strain was highly virulent for mice and represents a unique genotype of the parasite, based on PCR-RFLP analysis. Research samples are available from the corresponding author on reasonable request. Given that the studied animal was a stray dog that had been rescued from the street, limitations of our study include the unknown vaccination status, nutrition, environment (rural or urban) of the dog, as well as contact with cats.
Footnotes
Acknowledgements
We thank the veterinary residents from the Veterinary Teaching Hospital at Federal University of Bahia, Brazil, for their excellent assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our work was financed by Bahia Research Foundation (FAPESB) under grant PNE0003/2011. Waléria B. Silva, Mariana M. Rezende-Gondim, and Gideão S. Galvão were recipients of fellowships from Coordination for the Improvement of Higher Education Personnel (CAPES). George R. Albuquerque and Luís F.P. Gondim are recipients of productivity fellowships from the National Council for Scientific and Technological Development (CNPq).
