Abstract
A 10-year-old male, neutered domestic shorthair cat was presented with fever, anorexia, vomiting, and diarrhea. Serologic testing for Feline immunodeficiency virus and Feline leukemia virus were negative. Fine-needle aspirates of mesenteric lymph nodes revealed the presence of banana-shaped apicomplexan parasites. The cat died after 4 days of hospitalization. Postmortem polymerase chain reaction (PCR) analysis confirmed the presence of Toxoplasma gondii in all examined organs. Parasites were ex vivo isolated in outbred mice and subsequently transferred into cell culture. Genotyping, using genetic markers for SAG2, SAG3, BTUB, GRA6, c22-8, c29-2, L358, PK1, and Apico for PCR–restriction fragment length polymorphism, revealed infection with type II T. gondii displaying type II alleles at all loci except Apico, which exhibited a type I allele. This is the most frequently identified genotype among cats acting as definitive hosts in central Europe, but to the authors' knowledge, it has never been associated with systemic toxoplasmosis in an adult, immunocompetent cat.
Toxoplasma gondii is an obligate intracellular protozoan parasite that can infect virtually any warm-blooded animal, including human beings. Felidae are the only definitive hosts, but they can also act as intermediate hosts. Infection of intermediate and definitive hosts occurs by oral ingestion of sporulated oocysts from the environment, by ingestion of meat harboring tissue cysts, or by transplacental infection. Cats seem to be more susceptible to infection by ingestion of tissue cysts than by oocysts, while the opposite holds true for mice. 9 Compared with other hosts, T. gondii oocysts are less infective and less virulent for cats. 8
In North America and Europe, 3 T. gondii clonal lineages, designated as clonal types I, II, and III, have predominantly been found; discrimination was based on polymerase chain reaction–restriction fragment length polymorphism (PCR-RFLP) and microsatellite typing. 1,16 These clonal types also showed different levels of virulence in outbred mice; type I always caused lethal infection, while the other 2 types were far less virulent. 23 Likewise, in experimental infection of immunocompetent cats, parenteral infection with the RH strain of T. gondii (type I) resulted in severe to fatal disease, 20 but parenteral infection with the ME49 strain (type II) usually led to mild transient signs, of which chorioretinitis was the most specific. Severe to fatal disease has only been described 5 in immunocompromised cats. Little is known about the genotypes that may cause systemic toxoplasmosis in naturally infected immunocompetent cats.
The main pathologic features of systemic toxoplasmosis in cats after both experimental and natural infection have been well characterized 10,11,20 and include interstitial pneumonia, hepatitis, splenitis, lymphadenomegally, and ophthalmitis. In addition, myocarditis and central nervous system disease may occur. 10 Recently, generalized toxoplasmosis in immunocompetent adult human beings caused by strains of T. gondii exhibiting atypical genotypes that were highly virulent in mice has been described. 3,4 Similar atypical genotypes have been isolated from both domestic and wild mammals. 12,13
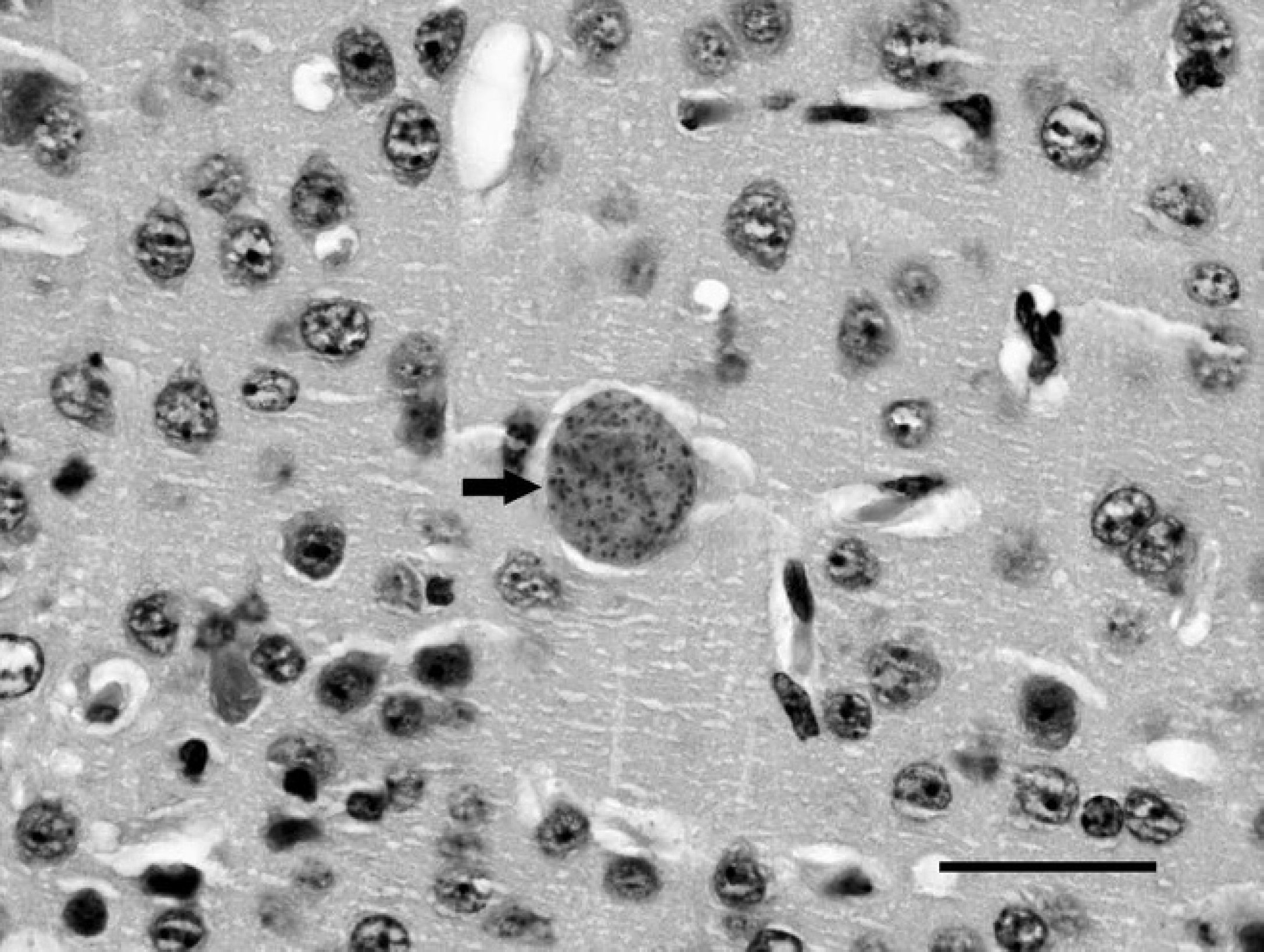
A 10-year-old male, neutered domestic shorthair cat was referred to the Small Animal Clinic at the University of Bern (Bern, Switzerland) with a 1-week history of anorexia, vomiting, diarrhea, and fever; the symptoms had been unresponsive to treatment with antibiotics (amoxicillin–clavulanic acid), nonsteroidal anti-inflammatory drugs (tolfenamic acid), and antiemetics (metoclopramide). The cat had outdoor access and was regularly vaccinated and dewormed. At presentation, the cat was lethargic. Physical examination revealed pale mucous membranes, mild hypothermia (37.7°C), tachypnea (40 breaths/min), and a faint systolic heart murmur (grade II/VI) with a normal heart rate (160 beats/min). Significant findings related to a complete blood count, serum biochemistry profile, and coagulation profile included a degenerative left shift, hypoalbuminemia, hypocholesterolemia, decreased urea nitrogen, elevated liver enzymes, and prolonged clotting times (Table 1). Serologic testing a for antibodies against Feline immunodeficiency virus (FIV) and antigen of Feline leukemia virus (FeLV) were negative. Subsequent PCR for FeLV in blood was also negative.
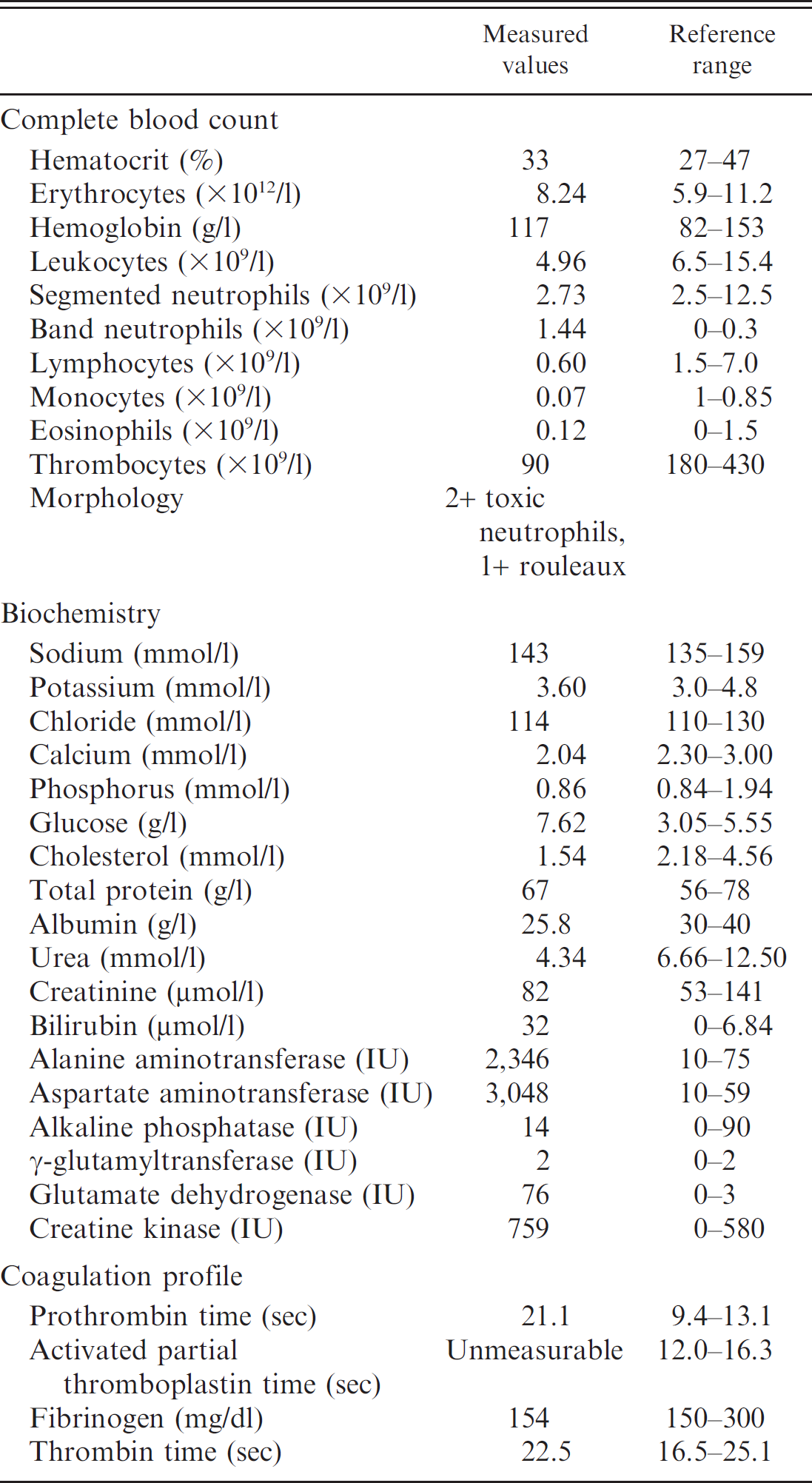
The cat was treated with intravenous crystalloid infusions, a plasma transfusion, intravenous antibiotics (amoxicillin–clavulanic acid, metronidazole, and enrofloxacin), and vitamin K1. During the first 3 days of hospitalization, the cat's general condition deteriorated, with the animal displaying increasing weakness and respiratory distress. On the third day, thoracic radiographs revealed generalized osteosclerosis and moderate pleural effusion. Cytologic examination of the free thoracic fluid revealed hemorrhagic effusion. Ultrasonographic examination of the abdomen showed a hypoechogenic liver parenchyma, a thickened gallbladder wall, and splenomegaly, as well as generalized lymphadenomegally. Cytologic examination of fine-needle aspirates of the ileocecal and jejunal lymph nodes taken under ultrasonographic guidance revealed lymphoid hyperplasia and banana-shaped intracytoplasmic protozoan organisms in macrophages (Fig. 1). Clindamycin was administered based on suspected toxoplasmosis, but the cat died on the evening of the fourth day.
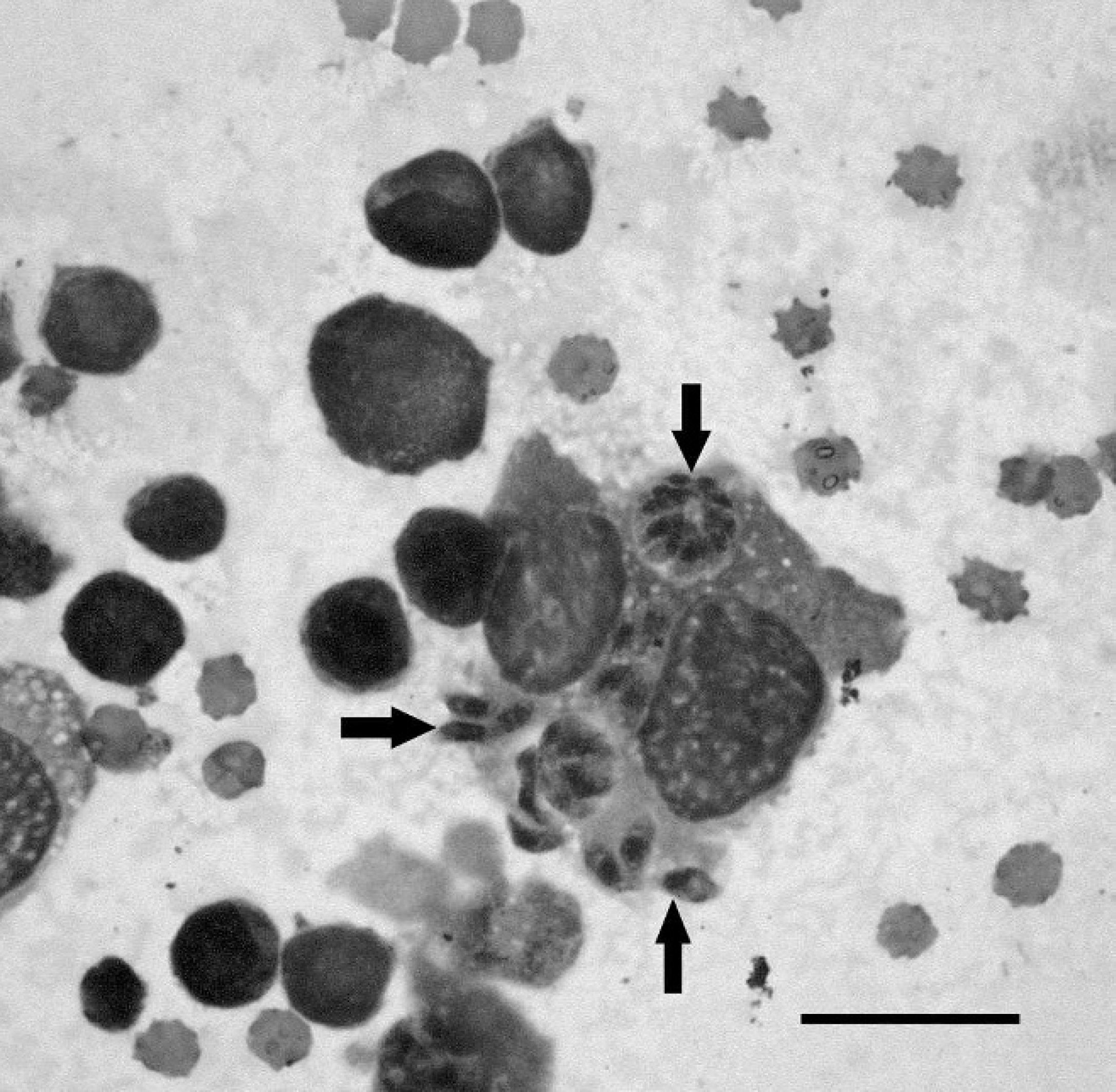
At necropsy, multiple small gray foci, measuring up to 2 mm in diameter, were present in all lung lobes (Fig. 2) and throughout the liver. Bronchial, intestinal, liver, and lumbar lymph nodes were moderately enlarged to approximately 1.5 cm in diameter. Routine histopathological evaluation of formalin-fixed tissues revealed multiple foci of acute coagulation necrosis (up to 100 μm in diameter), with infiltration of small amounts of neutrophilic granulocytes and macrophages in lymph nodes, liver, and lung. Throughout the brain, multiple small foci of gliosis and accumulation of a few macrophages and neutrophilic granulocytes were present. Protozoan cysts of up to 40 μm in diameter containing Toxoplasma organisms were occasionally detected within these foci. Systemic toxoplasmosis was confirmed by T. gondii–specific real-time PCR targeting the B1 gene 2,22 on fresh samples of intestine, lung, liver, spleen, bone marrow, and mesenteric and lumbar lymph nodes.
Blood parameters of the cat measured on the day of presentation at the Small Animal Clinic at the University of Bern.
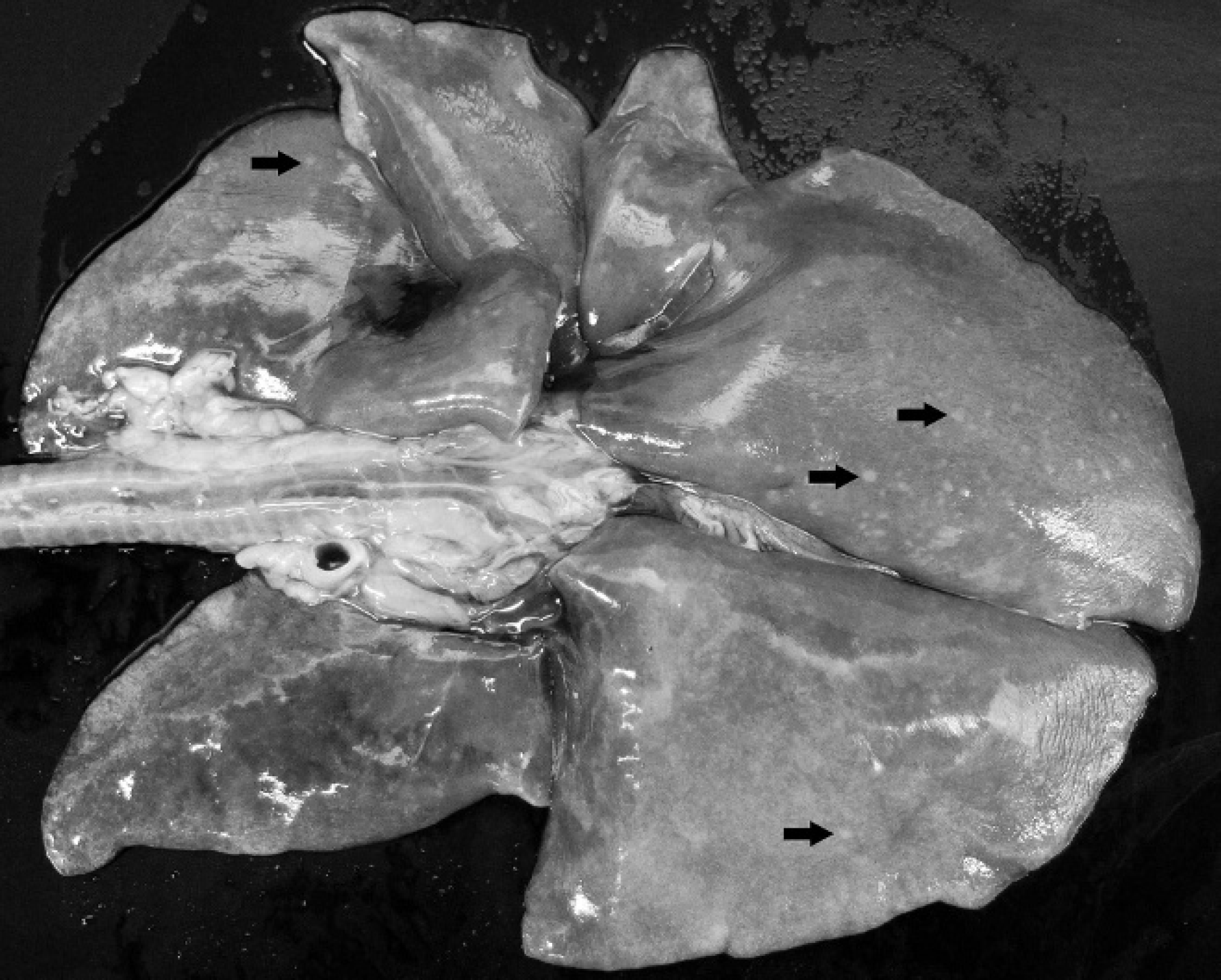
Immediately after the death of the cat, liver samples were aseptically transferred into cold Eagle minimal essential medium b supplemented with 10% heat-inactivated fetal calf serum, c 50 U/ml of penicillin, and 50 μg/ml of streptomycin d and transported to the Institute of Parasitology at the University of Bern. The sample was homogenized using individual sterile instruments and inoculated intraperitoneally into 10-week-old female BALB/c mice, e which were maintained under conventional day- and night-cycle housing conditions, according to the animal welfare legislation of the Swiss Veterinary Office. Throughout the experiment, no clinical signs, including local reactions, nodules, or other complications, were observed. However, seroconversion could be observed in a P-30 enzyme-linked immunosorbent assay that was performed as previously described, 21 with the following minor modifications: positive and negative mice sera f for T. gondii were used as controls, and reactive antibodies were detected with alkaline phosphatase–conjugated, goat-derived, anti-mouse immunoglobulin G. g Three months after inoculation, 1 of the 5 mice was euthanized. Routine histopathological evaluation of liver, spleen, lung, and brain revealed multiple randomly distributed small foci composed of clusters of neutrophils, macrophages, and lymphocytes. Few toxoplasmic cysts without associated inflammatory lesions were present in the brain (Fig. 3). Under aseptic conditions, the brain was homogenized in trypsin, washed twice in cold Roswell Park Memorial Institute (RPMI) 1640 medium, h and the resulting homogenate was resuspended in 5 ml of cold RPMI medium. The cell suspension was repeatedly passaged through a 25-gauge needle and finally pipetted into tissue culture flasks containing human foreskin fibroblasts (HFF) maintained in RPMI medium (5% immunoglobulin G–free horse serum, 2 mM glutamine, 50 U/ml of penicillin, 50 μg/ml of streptomycin d ) at 37°C and 5% CO2. The cultures were evaluated for parasite growth by light microscopy. After 2 weeks, HFF cells exhibited visible parasite pseudocysts. Parasites were maintained by serial passages in Vero cells cultured in RPMI medium and harvested as previously described. 6,14 A DNA extraction and quantitative real-time PCR targeting the T. gondii B1 gene 2,22 confirmed the presence of Toxoplasma DNA in the study sample.
Fine-needle aspirate of an ileocecal lymph node showing intracellular Toxoplasma gondii tachyzoites (arrows) in a macrophage. May–Grünwald/Giemsa, 1,000×. Bar = 20 μm.
Postmortem photograph of the lung of the cat. Multiple small foci of necrosis within all lung lobes (arrows).
Subsequently, DNA samples obtained from frozen samples of cat organs and from in vitro parasite culture were sent to the Friedrich-Loeffler-Institut (Wusterhausen, Germany), where genotyping was performed using genetic markers for SAG2, SAG3, BTUB, GRA6, c22-8, c29-2, L358, PK1, and Apico for PCR–RFLP analysis, as previously described. 15 Genotyping revealed type II alleles at all loci except Apico, where a type I allele was found. Toxoplasma gondii oocysts shed by cats from Germany and Switzerland frequently belong to the group of T. gondii type II isolates. 15 Some of the cats suffered from diarrhea, but no other clinical signs associated with generalized toxoplasmosis have been reported thus far, 15 indicating that natural infection with this strain is usually avirulent in cats. Furthermore, the BALB/c mice infected experimentally with T. gondii type II, Apico I, remained asymptomatic. Immunocompetent cats infected with T. gondii ME49 (clonal type II) usually remain asymptomatic as well, but disease can be provoked if the cats are immunocompromised. 5 However, natural infection with T. gondii type II, Apico I, led to fatal systemic toxoplasmosis in the cat reported in the current study, and all pathological findings were consistent with those of cats suffering from generalized toxoplasmosis. Despite this, concurrent FIV or FeLV infections, which may impair the immune system, were excluded, and no history of pharmacological immunosuppression was recorded. An underlying disease that may have affected the immune system could not be totally ruled out, but no evidence of this was found. Another possible reason for the susceptibility of the cat in the present study might be found in the genetic polymorphism of the host. Indeed, previous experiments have shown that the mouse-virulent strain RH (clonal type I) was not more pathogenic in pigs than was T. gondii type II, and this strain was devoid of cyst-forming capacity. 25 Furthermore, strains nonpathogenic in sheep were virulent in mice, but a strain causing abortions in sheep was shown 19 to be nonpathogenic in mice. This indicates that host factors must play a crucial role in the outcome of infection. In the mouse model, resistance or susceptibility between different BALB and B10 mouse strains toward infection with T. gondii type II and cyst formation appeared to be associated with the major histocompatibility complex (MHC) haplotype and with multigenic regulation outside the MHC. 7 During the chronic phase of infection, the L d -restricted cytolytic activity was elicited by type II but not by type I T. gondii. 18 Moreover, certain gene polymorphisms are associated with a higher susceptibility to clinical infection with T. gondii in human beings. Some of these are located within the MHC complex, 24 but others were found in unrelated loci. 17 To date, no such polymorphisms are known in the cat, but findings in the cat reported in the current case report indicate that this could be a possible reason for severe disease in this species. Toxoplasmosis should be included as a differential diagnosis in immunocompetent cats that present with signs consistent with severe multiorgan inflammation or sepsis.
Histophotograph of a bradyzoite cyst (arrow) within the cerebral cortex of an experimentally infected mouse. Hematoxylin and eosin, 1,000×. Bar = 30 μm.
Acknowledgements. Daland C. Herrmann was supported by the German Bundesministerium für Bildung und Forschung (grant 01 Kl 0765, Toxonet01 to G. Schares) and would like to thank Aline Beckert for excellent technical support.
Footnotes
a.
SNAP® FIV/FeLV Combo Test, IDEXX Laboratories Inc., Westbrook, MA.
b.
DMEM, Lubioscience GmbH (Invitrogen), Luzern, Switzerland.
c.
FCS Superior, Biochrom AG, Berlin, Germany.
d.
Penstrept, Lubioscience GmbH (Invitrogen), Luzern, Switzerland.
e.
BALB/c, Charles River, Sulzfeld, Germany.
f.
University of Bern, Bern, Switzerland.
g.
Goat-derived anti-mouse IgG (1 mg/ml), Promega Corp., Madison, WI.
h.
RPMI 1640, Lubioscience GmbH (Invitrogen), Luzern, Switzerland.
