Abstract
The spread of both infectious and noninfectious diseases through wildlife populations is of increasing concern. Neoplastic diseases are rarely associated with population-level impacts in wildlife; however, impacts on individual health can be severe and might reflect deteriorating environmental conditions. An adult male free-ranging Blanding’s turtle (Emydoidea blandingii) originally captured in 2005 and deemed healthy, was recaptured in 2018 with a 1 × 1.5 cm intra-oral broad-based right mandibular mass. An excisional biopsy was performed, and histopathology revealed squamous cell carcinoma (SCC). Consensus herpesvirus PCR identified a novel herpesvirus (proposed name Emydoidea herpesvirus 2 [EBHV-2]) within the tumor. EBHV-2 shares 85% sequence homology with Terrapene herpesvirus 2 (TerHV-2), a herpesvirus linked to fibropapillomas in eastern box turtles (Terrapene carolina carolina). Virus-associated fibropapillomas have been identified in multiple marine turtle species and have had debilitating effects on their populations, but to date, virus-associated SCCs are rarely reported.
The emergence of infectious and noninfectious diseases in wildlife threatens species conservation efforts and global biodiversity. 4 Neoplastic processes are rarely reported in wildlife, but they can cause significant morbidity and mortality at the individual level. Although rare, neoplastic diseases can also drive population declines. 13 For example, since its initial description in 1996, Tasmanian devil facial tumor disease (DFTD) has reduced population sizes of the world’s largest marsupial carnivore by >60%, resulting in classification of this species as endangered by the Australian state of Tasmania in 2008. 14
Several reptilian neoplasms have been reported in association with viral infections, including arenavirus-associated lymphoid neoplasms in red-tailed boa constrictors (Boa constrictor constrictor) and herpesvirus-associated fibropapillomas in sea turtles and eastern box turtles (Terrapene carolina carolina).2,9,19 Transmissible cancers have the potential to negatively impact reptile populations around the globe, though they remain understudied in many wild chelonians. 2 Understanding the epidemiology of virally associated neoplasia is important for designing conservation management strategies that support individual and population health.
The Blanding’s turtle (Emydoidea blandingii) is a semi-aquatic chelonian native to the northern United States and southern Canada. Populations are declining across much of their range as a result of urban development, road mortality, climate change, illegal poaching and collection, and nest depredation (https://www.iucnredlist.org/species/7709/155088836). The Blanding’s turtle population in Lake Plain (northeast Illinois) was in decline in 2010 and had a 95% probability of going extinct in the next 50 y without intervention. 7 In response, the Lake County Forest Preserve District initiated the Blanding’s Turtle Recovery Program (BTRP) to assess and mitigate threats. 7 A comprehensive health assessment program was added to the BTRP in 2016 to characterize individual and population health, as well as identify disease threats to free-living Blanding’s turtles. We provide here gross, histologic, and molecular descriptions of a squamous cell carcinoma (SCC) associated with a novel herpesvirus in a wild Blanding’s turtle identified during the BTRP health surveillance project.
All animal sampling was permitted by the following organizations: Illinois Department of Natural Resources (IDNR; Scientific Collectors Permits [SCP] NH17.5065, NH18.5065; IDNR Endangered and Threatened permits SBT-16-062, 1199, 14-046, and 1042), the Wisconsin Department of Natural Resources (WIDNR; SCP: SCP-SOD-004-2013; WIDNR Scientific Research License SRLN-18-026), and the University of Illinois Institutional Animal Care and Use Committee (Protocols 18000, 18165). The subject of our report was first found in 2005 and was tracked using radiotelemetry or encountered opportunistically at least once per year in 6 of the next 10 y. Upon each encounter, mass and location were recorded. Beginning in 2016, physical examinations were performed at each capture including an oral examination; however, this individual was not recaptured until 2018. In late May 2018, physical examination revealed asymmetrical nares and a 1 × 1.5 cm fleshy, broad-based, pink mass with a cobblestone appearance arising from the right mandibular gingiva (Fig. 1A, B). The mass had pale nodules throughout and was crusted with loosely adhered caseous material. The right oral commissure was also expanded by an encapsulated mass of firm, yellow material (suspect granuloma), which tracked beneath the gingiva and extended caudodorsally, potentially representing an extension of neoplastic disease (Fig. 1A). A combined oral/cloacal swab and a blood sample were collected and frozen at –20°C.
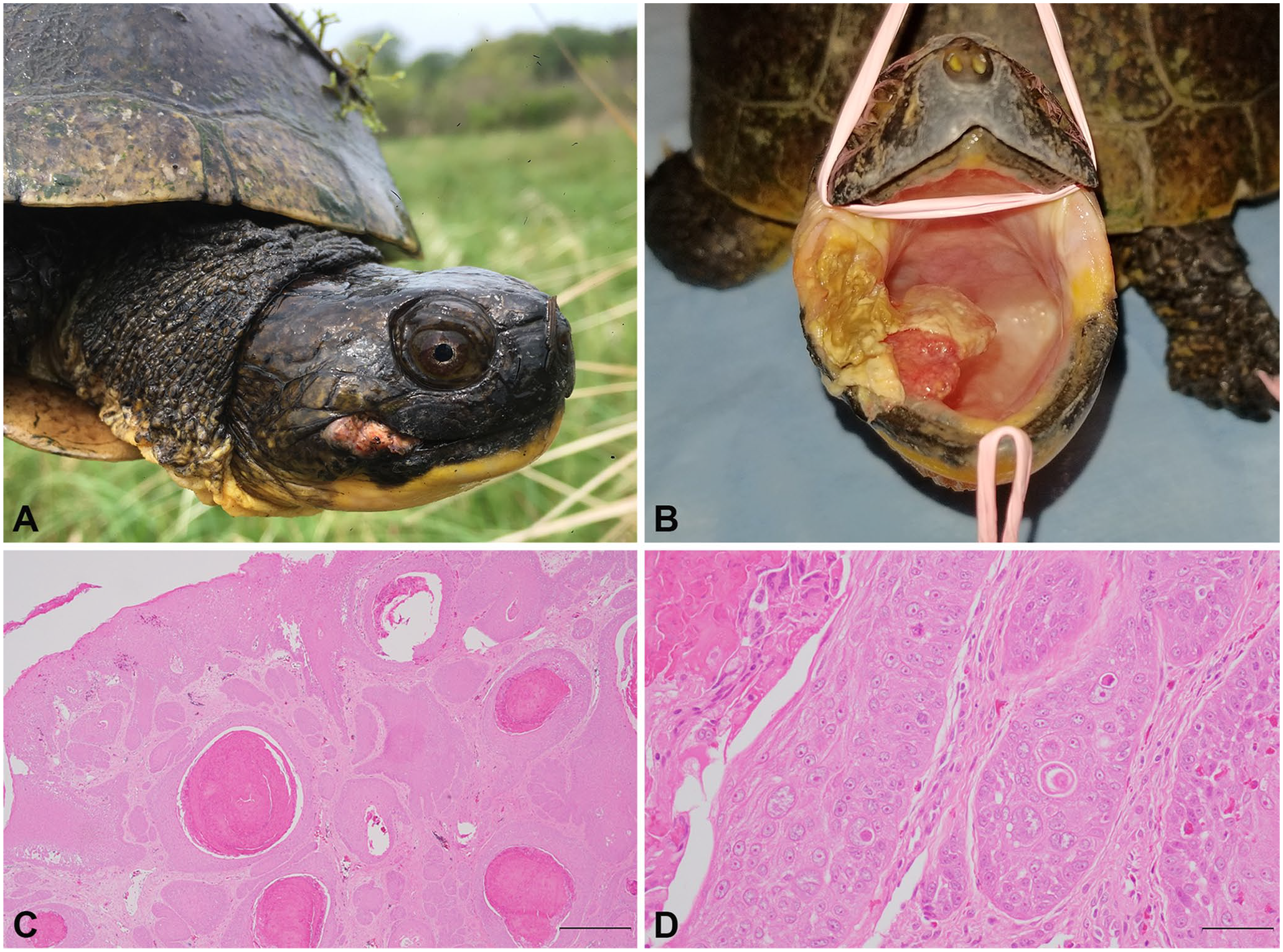
Adult male Blanding’s turtle with oral squamous cell carcinoma (SCC)-associated herpesvirus (Emydoidea herpesvirus 2) in Lake County, IL in 2018.
Based on the mass’s location and the concern that the turtle’s weight was the lowest on record to date (decreased 111 g or 9% since March 2016), the animal was transferred to the University of Illinois (Urbana, IL) for surgical debridement. Surgical sampling of the oral lesion was performed under general anesthesia, and tissue samples were stored fresh-frozen at –20°C and in 10% neutral-buffered formalin. The turtle was released following computed tomography imaging. Two months later, mild tumor recurrence was noted, but the turtle’s weight had increased by over 200 g (16% of body weight), and additional treatment was not pursued. A second combined oral/cloacal swab and blood sample were collected during the exam and frozen at –20°C. The formalin-fixed sample was processed routinely with hematoxylin and eosin stain and assessed via histopathology.
Histopathology revealed an irregular, infiltrative, multinodular neoplasm arising from a markedly hyperplastic and mildly dysplastic oral mucosa. The neoplasm was comprised of polygonal cells arranged in islands, trabeculae, and small nests within a scirrhous stroma (Fig. 1C, 1D). Neoplastic cells had conspicuous intercellular desmosomal junctions, abundant eosinophilic cytoplasm with tonofilaments, and large euchromatic nuclei with prominent nucleoli. There was moderate-to-marked anisocytosis and anisokaryosis, and 25 mitotic figures in 10 high-power (400×) fields (2.37 mm2). Throughout neoplastic islands, there were dyskeratotic cells and multiple cores of keratin plugs and pearls, consistent with a SCC.
Computed tomography images were acquired in the cranial-to-caudal plane using a 16-detector computed tomography system (Aquilion LB; Toshiba) using the following technical factors: 120 kVp, fluctuating current with maximum setting of 300 mA, and 16 detectors × 0.5 mm collimation. Intravenous iohexol (700 mg I/kg; Omnipaque, 300 mgI/mL; GE Healthcare) was administered as a contrast medium, and contrast-enhanced studies were obtained immediately. All studies were reconstructed into a soft algorithm with display settings of window width of 2,500 and window level of 250, and window width of 400 and window level of 40, with a 512 × 512 matrix. There was mild faint, amorphous mineral attenuation associated with soft tissue thickening within the right side of the oral cavity (Suppl. Fig. 1). The mineral contoured to the right commissure, abutting subtle loss of bone along the internal margin of the maxilla and the right angular bone and prearticular bone of the lower jaw. No additional significant findings were identified.
DNA extraction from frozen tissue, oral/cloacal swabs, and blood samples was performed using a commercial kit (DNeasy blood & tissue kit; Qiagen). DNA concentration and purity were evaluated using a spectrophotometer (NanoDrop 1,000; Thermo Fisher). PCR was performed for herpesviruses and papillomaviruses using existing consensus assays, inclusive of positive and negative controls, as described previously.16,18 PCR products were electrophoresed on 1% agarose gel. Samples producing bands of the appropriate size (~400 bp for herpesviruses, ~600 bp for papillomaviruses) were treated with ExoSAP-IT (USB Corporation) and sequenced commercially in both directions. Forward and reverse sequences were aligned, trimmed of primers, and compared to existing sequences in GenBank using BLASTN (http://www.ncbi.nlm.nih.gov/blast/Blast.cgi).
A 481-bp product was obtained from the oral mass using herpesvirus consensus PCR targeting the DNA polymerase gene, but no products were amplified from the oral/cloacal swabs or blood samples. Similarly, no products were amplified using papillomavirus primers. The herpesviral sequence shared 85% homology to TerHV-2 (KM507472.1). We deposited the sequence for this virus, tentatively named Emydoidea herpesvirus 2 (EBHV-2), in GenBank under accession MN456821.
The translated amino acid sequence of the EBHV-2 partial DNA polymerase gene was aligned with additional chelonian herpesvirus sequences for this gene, as reported in GenBank, using Geneious Prime 2020.1.2. 11 Bayesian phylogenetic analysis of the amino acid alignment was performed using the MrBayes plugin for Geneious, 10 and maximum-likelihood phylogenetic analysis of the alignment was performed using the PHYML plugin for Geneious. 8 Parameters for both methods were set as described previously. 15 Similar dendrogram topology was recovered by both Bayesian and maximum-likelihood methods (Suppl. Figs. 2, 3). Sequences for EBHV-2 and TerHV-2 were recovered in a monophyletic group with high posterior probability (1) and bootstrap support (98.6).
The turtle overwintered successfully, but in May 2019 physical examination revealed a 200 g weight loss (15% of body weight), and the oral cavity mass had returned to its original size. More aggressive surgical debridement and medical care was provided. The excised tissue was subjected to herpesvirus PCR as described above, and sequencing confirmed the same herpesvirus that had been detected 1 y prior (GenBank MN456821). Oral/cloacal swabs and whole blood samples collected from the turtle in July, August, and September of 2019, following the removal of the tumor, were all negative for herpesvirus. The turtle improved and was therefore released at the site of initial capture 5 mo after surgery with no noticeable lesions. As of June 2020, the oral mass had not recurred.
The presence of the oral SCC appeared to minimally impact behavior and overall wellness of this turtle. There was, however, a significant weight gain of over 200 g following the first surgery and again after the second, suggesting physical disruption of eating or pain associated with the mass. Cutaneous SCC has been identified in multiple species of squamates and chelonians, 6 and its presence in a Blanding’s turtle may signify further conservation challenges for this imperiled species.
There has been an increase in the number of reported cases of reptile neoplasia since the 1970s. 17 The increase can most likely be attributed to improved veterinary care and husbandry, resulting in longer life spans as well as more detailed postmortem examinations. 17 A retrospective study identified SCC in several species of reptiles including snakes, monitor lizards, skinks, and turtles. 6 Although there was no definitive histologic evidence of viral involvement in any of these cases, molecular investigation was not performed in most. 6 A novel alphaherpesvirus associated with oropharyngeal SCC was observed in smooth green snakes (Opheodrys vernalis); 12 2 cases of epithelial tumors, a spiny-tailed lizard (syn. North African mastigure; Uromastyx acanthinura) with multiple papillomas and a Dumeril’s boa (Acrantophis dumerili) with SCC, were both associated with papillomaviruses. 5 Thus, viral-associated SCC might be underdiagnosed.
The DNA polymerase gene of EBHV-2 is most closely related to TerHV-2, which has been linked to the development of fibropapillomas.3,19 Interestingly, our analysis indicates that EBHV-2 and TerHV-2 are more closely related to each other than to Emydoidea herpesvirus 1 and Terrapene herpesvirus 1. The same pattern is present for sea turtle herpesviruses, with the fibropapillomatosis-associated chelonid herpesvirus 5 (ChHV-5) arising from a lineage separate from all other sea turtle herpesviruses (Suppl. Figs. 2, 3). Previous work on ChHV-5 indicates that this genetic variation from benign host-adapted herpesviruses facilitates tumor pathogenesis and immune system evasion, potentially promoting the development of neoplasia. 1 Further research may reveal the presence of oncogenes in EBHV-2 and TerHV-2 similar to those involved in tumorigenesis in ChHV-5, providing context for the relative importance of these viruses for host health.
Although viruses have been detected in reptilian tumors, challenge studies have not been performed to evaluate the causal association between viral infection and development of neoplasia. It is possible that EBHV-2 was coincidentally detected in neoplastic tissue but was not involved in tumorigenesis alone or in conjunction with other viruses in this Blanding’s turtle. Indeed, the detection of alphaherpesviruses in oral/cloacal swabs is common in emydid turtles. 19 However, EBHV-2 DNA was only detected in tumor tissue, and not in oral/cloacal swabs or whole blood samples, supporting the association between viral infection and neoplasia in this individual.
The prognosis of both SCCs and herpesviral infections in reptilian species is largely unknown. Except for a fluctuation in weight, the behavior of the Blanding’s turtle in our case appeared largely unaffected by the presence of the tumor, and the turtle was consistently bright, alert, and responsive on physical examination. However, it is unclear how long this turtle would have survived without medical care. Although continued monitoring and additional intervention is planned for this genetically valuable individual, this management strategy may not be appropriate if larger numbers of animals were affected, underscoring the need for additional research on EBHV-2 epidemiology.
Conservation management strategies that account for all important threats to individuals and populations are likely to be the most successful. Pathogens within a landscape may be common, but the impact on wellness for many disease agents is unknown. Subsequent studies should investigate the prevalence of EBHV-2, determine its associated clinical signs, and investigate its relationship with tumorigenesis.
Supplemental Material
sj-pdf-1-jvd-10.1177_1040638721989302 – Supplemental material for Detection of a novel herpesvirus associated with squamous cell carcinoma in a free-ranging Blanding’s turtle
Supplemental material, sj-pdf-1-jvd-10.1177_1040638721989302 for Detection of a novel herpesvirus associated with squamous cell carcinoma in a free-ranging Blanding’s turtle by Kirsten E. Andersson, Laura Adamovicz, Lauren E. Mumm, John M. Winter, Gary Glowacki, Rachel Teixeira-Neto, Michael J. Adkesson, Eric T. Hostnik, Ellen Haynes and Matthew C. Allender in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
