Abstract
Objective:
Cannabidiol (CBD) administration (5 mg/kg) in healthy rats has been shown to significantly decrease lymphocyte subset numbers in peripheral blood without involvement of natural killer cells. The aim was to evaluate whether lymphocyte numbers also decrease in the spleen.
Introduction:
CBD, the major non-psychotropic compound of Cannabis sp., is an effective treatment for inflammatory and autoimmune diseases with various anti-tumor effects, but the mechanisms of its long-term actions in vivo remain unclear.
Methods:
To examine the effects of CBD on lymphocyte subsets in the spleen and NK cellular cytotoxicity (NKCC), adult male Wistar rats (n = 63) were administered intraperitoneal injections of CBD (2.5 or 5 mg/kg/day) for 14 consecutive days, and lymphocyte counts were obtained using flow cytometry. NKCC in the peripheral blood and spleen was quantified using a Chromium-51 release assay. Furthermore, the effect was similar to a decrease in lymphocytes caused by treatment with the selective receptor antagonist AM630 (1 mg/kg).
Results:
The results indicate that repeated administration of CBD at a dose of 5 mg/kg/day decreased splenic lymphocyte numbers, involving T and non-T/NK CD45RA+ lymphocytes but not NK cells. The effects of CB2 receptor antagonist were not significant, but it had a significant interaction with CBD. No changes in NKCC were observed following CBD administration.
Conclusion:
These results reveal that in healthy rats, CBD produces similar lymphopenic effects in the spleen as it does in peripheral blood without affecting NK cell counts or cytotoxicity.
Introduction
Cannabidiol (CBD) is one of the major non-psychotropic phytocannabinoids found in Cannabis sp. The plant has been used in medicine throughout history, but psychotropic side effects have largely limited its use in therapy. Unlike the major psychotropic compound of cannabis, THC (Δ9-tetrahydrocannabinol), CBD displays a low affinity to cannabinoid CB1 and CB2 receptors.1,2 Although it is commonly thought that CBD does not bind to CB1 and CB2 at physiologically relevant concentrations, it can inhibit the effects of CB1 and CB2 cannabinoid receptor agonists and was shown to behave as a CB2 receptor inverse agonist in vitro2,3 as well as a negative allosteric modulator of CB1. 4 It has also been suggested that CBD imposes allosteric-like effects on CB2 receptors. 5 Moreover, CBD is known to affect cannabinoid receptors indirectly by modulation of alternative pathways such as endocannabinoid signaling, for example, by inhibiting FAAH, but these discussed effects have not been demonstrated in vivo.6,7 Much of the therapeutic use of cannabinoids is focused on those devoid of psychotropic properties, namely CBD.8–10 Like many other cannabinoids, CBD produces analgesic, antiemetic, neuroprotective, and immunomodulatory effects,8,9 and because of its low toxicity in humans,11–13 it has been proven to have wide therapeutic applications.10,13,14
CBD demonstrated efficacy in animal models of autoimmune diseases like rheumatoid arthritis (RA), type I diabetes, and multiple sclerosis (MS) via its anti-inflammatory and immunomodulatory effects.15–17 It has also been shown to alleviate neuropathic pain both in animal models and humans.14,18–21 Moreover, CBD is known to produce antitumor effects, decreasing the proliferation and migration of various tumor cell lines in vitro, as well as tumor growth and metastasis in vivo.22,23 While the antitumor effects of CBD have been demonstrated extensively, its biological effects on different human cancers depend on cell type and origin. 24 Other shown benefits of CBD specifically are improved sleep and mood quality,25–28 as well as anxiolytic and antipsychotic effects.13,29,30 Most recently, CBD (Epidiolex), has been FDA-approved for the treatment of drug-resistant or refractory epilepsy. 31
CB2 receptors, which are the focus of this work, are predominantly expressed on immune cells and in lymphoid organs. 32 The mechanisms of CBD’s influence on the immune system have been investigated in vitro, where it was found to induce apoptosis, decrease cell proliferation, cytokine production, and induce regulatory cells.14,33,34 Moreover, in vivo effects of CBD are mostly immunosuppressive and anti-inflammatory.14,35 However, the exact mechanisms through which CBD affects lymphocyte number in vivo and whether in vitro observations are relevant to physiological conditions are unclear.33,36 Although data concerning CB2 receptor involvement in the effects of CBD on the immune system is limited, some studies indicate that CBD may exert effects via CB2 receptors.37–39
Despite its therapeutic potential in inflammatory and autoimmune diseases,8,9 the effects of prolonged, systemically administered CBD on the immune system and the underlying mechanisms remain unclear. Our previous studies revealed a significant decrease in lymphocytes of the peripheral blood of rats following systemic CBD administration for 14 consecutive days. 40 The present study examined the effects of repeated administration of CBD on the lymphocyte numbers (B, T, NK, and T CD4+ and T CD8+ subsets) in the spleen of rats. Furthermore, it assessed the mediation of these effects by CB2 receptors, as they play an important role in the regulation of immune response by cannabinoids. Moreover, because our previous studies indicated that CBD administration may increase the number of NK cells that are responsible for primary antitumor and antiviral innate immune responses, we investigated whether repeated treatment with CBD affects the cytotoxic activity of NK cells against tumor cells.
Methods
Animals
The subjects (n = 63 (36 + 27)) were adult male Wistar rats (Experiment 1: R. Grabowski Laboratory Animals Breeding, Gdansk; Experiment 2: Trojmiejska Akademicka Zwierzetarnia Doswiadczalna, Gdansk, Poland), 10 weeks old, 250 ± 20 g at the beginning of experiments. Animals were housed in cages of 4, with free access to food and water. The animal room was maintained at a temperature of 21 ± 1°C with humidity 55 ± 10%, under a 12-h light cycle (lights on from 6:00 to 18:00). For 14 days prior to the start of experiments rats were handled daily and adapted to the presence of the experimenter to minimize stress evoked by experimental procedures.
The principles for the care and use of laboratory animals in research, as outlined by the Local Ethical Committee (permission number: 33/2008), were strictly followed and all the protocols were reviewed and approved by the Committee. All efforts were made to minimize animal discomfort and the number of animals used.
For experiment 1, animals were divided randomly into 3 groups, and for 14 consecutive days received intraperitoneal injections of CBD at a dose of 2.5 mg/kg/day (Group I, n = 9), 5 mg/kg/day (Group II, n = 9), or the vehicle (Group III, n = 9). 5 mg/kg is a commonly used dose of CBD in in vivo studies40–43 and was shown to be most effective in the previous study investigating lymphocyte subset numbers in the blood. 40 For experiment 2, animals were divided randomly into 4 groups and received 2 intraperitoneal injections for 14 consecutive days. The experimental group was pre-treated with CB2 receptor antagonist AM630 at a dose of 1 mg/kg/day, followed by injections of CBD at a dose of 5 mg/kg/day (Group I, n = 9). The three control groups were pre-treated with the vehicle, followed by 5 mg/kg/day of CBD (Group II, n = 8), pre-treated with 1 mg/kg/day of AM630 followed by vehicle (Group III, n = 9), or received vehicle for both treatments (Group IV, n = 11).
Drug administration and experimental design
CBD (Cannabidiol, Lipomed, Arlesheim, Switzerland) and AM630 ([6-iodo-2-metyl-1-[2-(4-morpholinyl)etyl]-1H-indol-3-yl](4methoxyphenyl)methanone; Tocris Bioscience, Bristol, United Kingdom) were dissolved in a vehicle solution containing Cremophor (Sigma Aldrich, Germany), 99.9% ethanol, and saline (0.9% NaCl) in a 1:1:18 ratio, which was also used in control groups. For 14 consecutive days, animals received intraperitoneal injections of vehicle or CBD at doses of 2.5 or 5 mg/kg/day. Doses were based on recent literature, the effects of CBD and AM630 in animal models, and our preliminary experiments. AM630, a selective antagonist of CB2 cannabinoid receptors, was administered intraperitoneally at a dose of 1 mg/kg, 15 min prior to injection of CBD or vehicle.1,6,44–46 All solutions were administered at fixed time points for each animal in a volume of 1 mL/kg and were prepared immediately before use.
Blood and spleen sampling
Blood samples (3 mL) were drawn to measure the cytotoxic activity of NK cells on experimental day 14, 60 min after the last CBD or vehicle injection. Samples were collected by cardiac puncture under halothane anesthesia (Narkotane Zentiva, Prague, Czech Republic) according to the standard procedure used in our laboratory. 47 Blood sampling was limited to 2 min after taking the animal from the home cage, to avoid the effects of stress on immune parameters. Immediately after blood sampling, the rats were infused with a lethal dose of intracardial pentobarbital (450 mg/kg, ~160 mg per animal, Morbital, Biowet, Pulawy, Poland) and their spleens were harvested. Spleen weight and volume were recorded immediately, followed by homogenization, filtration by pushing through nylon mesh bags, and suspension in Hanks’ Balanced Salt Solution (HBSS, Sigma-Aldrich) with sodium bicarbonate (Merck, USA) for further analysis. Spleen volume was measured using the water displacement method with a calibrated glass cylinder, allowing for subsequent calculation of spleen density. 48
Analysis of leukocyte and lymphocyte subset number
In spleen samples, the lymphocytes were gated, and the distribution of lymphocyte subsets was determined by flow cytometry (Supplemental Figure 1–3). A three-color immunofluorescent antibody staining procedure was used for determination of T (CD3+), B (CD3-CD45RA+), NK (CD3-CD161a+), as well as T helper (CD3+CD4+) and T cytotoxic (CD3+CD8+) lymphocyte subsets (CD3-FITC/CD45RA-PC7/CD161A-APC and CD3-FITC/CD4-PC7/CD8-APC antibodies, IO Test kit, Beckman Coulter, Immunotech, Marseille, France) as described previously.40,48 All antibodies were added and incubated with spleen samples according to the manufacturer’s instructions. Erythrocytes were lysed (1 mL Versalyse, Beckman Coulter), and the lymphocytes were fixed (25 μL Fixative Solution, Beckman Coulter). Samples were analyzed within 2 h, using an FC500 flow cytometer (Beckman Coulter). Total leukocyte number was assessed by a haematocytometer (Neubauer chamber). The percentage numbers of leukocyte populations in blood and spleen: lymphocytes, neutrophils, eosinophils, and monocytes were determined by morphological methods (May-Grünwald and Giemsa staining). The total number of each leukocyte subset was calculated by multiplying the total leukocyte number and the percentage of the individual subset.
NKCC assay
Cytotoxic activity of NK cells in peripheral blood and spleen was quantified using a chromium-51 release assay. As target cells YAC-1 murine lymphoma cell line was used. Washed in complete medium, target cells (5 × 106) were labeled with 100 Ci of Na251CrO4 (Radio Chemical Center, Otwock—Swierk, Poland) at 37°C for 1 h. Labeled target cells were washed with RPMI containing 2% FCS and adjusted to 1 × 105/mL in complete medium. Target cells were cultured in round-bottomed micro-well plates (Nunc, Roskilde, Denmark) with concentration of effector cells E: T = 50:1 (in a total volume of 200 µL) (Exp) in triplicate under standard culture conditions for 4 h. Spontaneous chromium-51 release wells (Sp) had target cells plus 100 µL of complete medium and the maximum release wells (Max) contained target cells plus 100 µL of complete medium with 5% Triton X-100 (Serva Feinbiochemia, Heidelberg/New York). The assay was terminated at 4 h by centrifuging the plates. Subsequently, 100 µL of supernatant was removed from each well, and triplicate samples were counted in a gamma-counter (Wizard 1470) for 1 min. The percentage of cytotoxicity was calculated as [(Exp–Sp)/(Max–Sp)] × 100. Results of NKCC assay are presented as lytic units, LU20 which were calculated as Estd/E:T20 × Tstd, where Estd stands for the standard number of effector cells (107), Tstd stands for the standard number of target cells (2 × 104), and E:T20 effector-to-target cell ratio, where lysis of 20% of target cells occurs.
Corticosterone concentration measurements
Blood samples (0.4 mL, with EDTA) were centrifuged for plasma sample collection and storage at −70°C until use. Quantitative determination of corticosterone concentration in rat plasma was performed using a commercial radioimmunoassay kit (ICN Biochemicals, Inc., Costa Mesa, CA, USA) and Wizard 1470 gamma counter (Pharmacia LKB, Turku, Finland). The assay was performed according to the manufacturer’s instructions.
Statistical analysis
Data were analyzed with one-way or two-way analysis of variance (ANOVA) followed by Tukey’s or Dunnett’s post hoc tests as appropriate. Results are presented as Mean ± SEM, and the statistical significance threshold was p < 0.05. The sample size was selected based on previous experiments and existing literature to have sufficient samples and statistical power. Furthermore, a power analysis was performed using the blood leukocyte count changes due to 2.5 and 5 mg/kg CBD treatment. 40 From the raw data in this study, an expected effect size was calculated. Although the blood leukocyte count data represent a different population from the splenic lymphocyte population assessed in the current study, it was used as preliminary data to estimate the required group sizes. This was due to its relevance in systemic immune responses and the absence of prior splenic data. G*Power software was used to calculate the total sample size with an effect size of ~0.7, an alpha value of 0.05, power of 0.8, and three groups (vehicle, CBD 2.5 mg/kg, CBD 5 mg/kg) for the splenic lymphocyte study.49,50 The calculated sample size was n = 8 per group. A minimum of n = 9 per group was used to account for potential animal loss. No data or animals were excluded from the study. Any missing data was not due to exclusion but due to data loss.
Results
To comprehensively assess the effects of CBD (5 mg/kg) administration on splenic lymphocyte counts, corresponding groups from Experiments 1 and 2 were merged. Namely, the vehicle and 5 mg/kg dose groups from Experiment 1, as well as the vehicle-vehicle (VV) and vehicle-5 mg/kg dose (VC) groups from Experiment 1 and 2, respectively, were combined as presented in Figure 1.

Decreased lymphocyte numbers in the spleen following administration of CBD (5 mg/kg) for 14 consecutive days. (a) Total number of splenocytes. (b) Total numbers of splenic lymphocyte subsets: B, T cells, T CD8+, and T CD4+. Unpaired t-test with Welch’s correction for unequal variances.
Repeated administration at a dose of 5 mg/kg resulted in a significant 31.93% decrease from 1.28 ± 0.07 × 106 cells/μL in the vehicle-treated to 0.86 ± 0.03 × 106 cells/μL in the group receiving CBD (Unpaired t-test with Welch’s correction, p < 0.0001, t(27.05) = 5.13, Figure 1(a)). A significant decrease was also observed in B, T, CD8+, and CD4+ lymphocytes counts (Figure 1(b)). The biggest change was in the B lymphocyte count with a decrease of 36.58% (47.98 ± 5.61 × 104 in vehicle, 30.43 ± 2.11 × 104 in CBD, p = 0.0074, t(24.21) = 2.93). T and CD8+ lymphocyte counts decreased 32.81% (59.79 ± 3.16 × 104 in vehicle, 40.14 ± 2.91 × 104 in CBD, p < 0.0001, t(35.98) = 4.57) and 32.26% respectively (22.72 ± 2.17 × 104 in vehicle, 15.39 ± 1.61 × 104 in CBD, p = 0.0104, t(34.17) = 2.71). The smallest decrease of 27.31% was observed in the CD4+ lymphocyte counts (33.07 ± 1.63 × 104 in vehicle, 24.04 ± 1.51 × 104 in CBD, p = 0.0003, t(35.99) = 4.05).
Experiment 1
In Experiment 1, comparing CBD at a dose of 2.5 mg/kg versus 5 mg/kg, total splenic leukocyte (splenocyte) number (including 97%–98% lymphocytes, 1.3%–2.3% neutrophils, <1% monocytes, and eosinophils) in rats treated with CBD at a dose of 5 mg/kg decreased by 23.8% as compared to vehicle (1.189 ± 0.087 × 106 in vehicle, 1.188 ± 0.131 × 106 in 2.5 mg/kg, 0.906 ± 0.062 × 106 in 5 mg/kg groups), but this effect was not significant (One-way ANOVA, Dunnett’s post hoc test, p = 0.058, F2.22 = 3.25, Figure 2(a)). Similarly, there was no significant effect on total splenic lymphocyte number (Supplemental Figure 5).

Lymphocyte numbers in the spleen following administration of CBD for 14 consecutive days with doses of 2.5 and 5 mg/kg. (a) Total number of splenocytes. (b) Total numbers of splenic lymphocyte subsets: B, T cells, T CD8+, and T CD4+.
Within the splenic lymphocyte subpopulations, repeated administration of CBD at a dose of 5 mg/kg resulted in a significant 28.5% decrease in total B lymphocyte number in the spleen as compared to the vehicle-treated group (p = 0.025, F2.22 = 4.36). This was a decrease from 32 ± 0.2 × 104 cells/μL in the vehicle-treated group to 22.9 ± 1.2 × 104 cells/μL in the group receiving 5 mg/kg of CBD (Figure 2(b)). A dose of 2.5 mg/kg did not change the number of B cells (32.4 ± 2 × 104 cells/μL) or any other of the assessed splenic lymphocyte subset numbers (Figure 2(b)). The change in total T cell number was not significant (One-way ANOVA, Dunnett’s post hoc test, p = 0.078, F2.22 = 2.88, Figure 2(b)). This slight decrease in T cell number was mainly due to a decrease in T CD8+ lymphocyte number by 31.8% compared to the vehicle (One-way ANOVA, Dunnett’s post hoc test, p = 0.094, F2.22 = 2.65) but also T CD4+ lymphocyte number decreased by 15.8% compared to the vehicle (One-way ANOVA, Dunnett’s post hoc test, p = 0.37, F2.22 = 1.03, Figure 2(b)). However, these effects were not significant. Overall, no significant change in the percentage distribution of B, T, T CD8+, and T CD4+ cells in the lymphocyte population was observed (not shown), suggesting a proportional decrease in the numbers of B and T cell subsets. Furthermore, treatment at either dose of CBD caused no significant changes in counts or percentages of total splenic lymphocytes, as well as granulocytes or monocytes (Supplemental Figure 5).
CBD at either dose did not affect total or percentage splenic NK cell numbers (p = 0.324, F2.22 = 1.19) or fraction of total lymphocytes (p = 0.790, F2.22 = 0.24; Figure 3(a) and (b)). Furthermore, no significant changes in NK cytotoxic activity in the spleen or peripheral blood were observed (p = 0.464, F2.24 = 0.79, p = 0.154 and F2.39 = 1.962, in the spleen and blood, respectively, Figure 3(c) and (d)).

NK cell counts of rats treated with CBD for 14 consecutive days. (a) Total NK cell counts. (b) Percentage of total lymphocytes. n = 7–9 per group for (a) and (b). (c) Cytotoxic activity of NK cells toward YAC-1 murine lymphoma tumor target cells in the spleen, n = 9 per group. (d) Cytotoxic activity of NK cells in the blood. n = 9 per group for (c) and n = 14 per group for (d).
CBD administration induced a significant decrease in total spleen weight of 1580 ± 111 mg in the vehicle group, to 1253 ± 77 mg and 1243 ± 77 mg in groups treated with 2.5 and 5 mg/kg/day of CBD, respectively (p = 0.021; F2,24 = 4.56, Supplemental Figure 4(a)). Similarly, spleen weight relative to total bodyweight also decreased from 0.5 ± 0.031% in the vehicle group to 0.41 ± 0.018% and 0.41 ± 0.023% both in animals treated with 2.5 or 5 mg/kg/day of CBD, respectively (p = 0.023; F2,24 = 4.46, Supplemental Figure 4(b)). There were no changes in spleen volume among the groups (p = 0.68, F2,22 = 0.39), so significantly reduced spleen density was observed in animals treated with CBD at a dose of 5 mg/kg. Spleen density decreased from 1.079 ± 0.038 μL in the vehicle-treated group to 0.853 ± 0.031 μL in the 5 mg/kg dose-treated group (p = 0.0046, F2,22 = 6.925). Additionally, there were no significant differences between blood corticosterone concentrations between the vehicle, 2.5 mg/mL, and 5 mg/mL groups (Supplemental Figure 6).
Experiment 2
Experiment 2 assessed the effect of CBD at a dose of 5 mg/kg with pretreatment with CB2 receptor antagonist AM630. As a result of CBD administration (VC), a significant decrease in the number of splenic leukocytes was observed (Figure 4(a), Two-way ANOVA, p = 0.018, F1,33 = 7.119 for effect of CBD). The decrease in splenic leukocyte number caused by CBD reached 38.1% as compared to the group receiving double injections of vehicle (VV; Two-way ANOVA, Tukey’s post hoc test, VV vs VC p = 0.004). The same effects were observed in the total lymphocyte counts (Supplemental Figure 7, p = 0.004), as well as in the B (p = 0.043), T (p = 0.002), CD8+ T (p = 0.003), and CD4+ T (p = 0.01) lymphocyte subsets. Furthermore, a two-way ANOVA revealed a significant interaction between CBD and AM630 treatment in the case of total numbers of splenic leukocyte (p = 0.021, F1,33 = 5.87), lymphocyte (p = 0.020, F1,33 = 6.02), B lymphocyte (p = 0.041, F1,33 = 4.54), T lymphocyte (p = 0.029, F1,33 = 5.22), and CD8+ T lymphocyte (p = 0.011, F1,33 = 7.18), but not CD4+ T lymphocyte subset (p = 0.076, F1,33 = 3.35). However, in the CD4+ T lymphocyte subset, a significant main effect of CBD was observed (p = 0.0097, F1,33 = 7.52; Figure 4(b)).
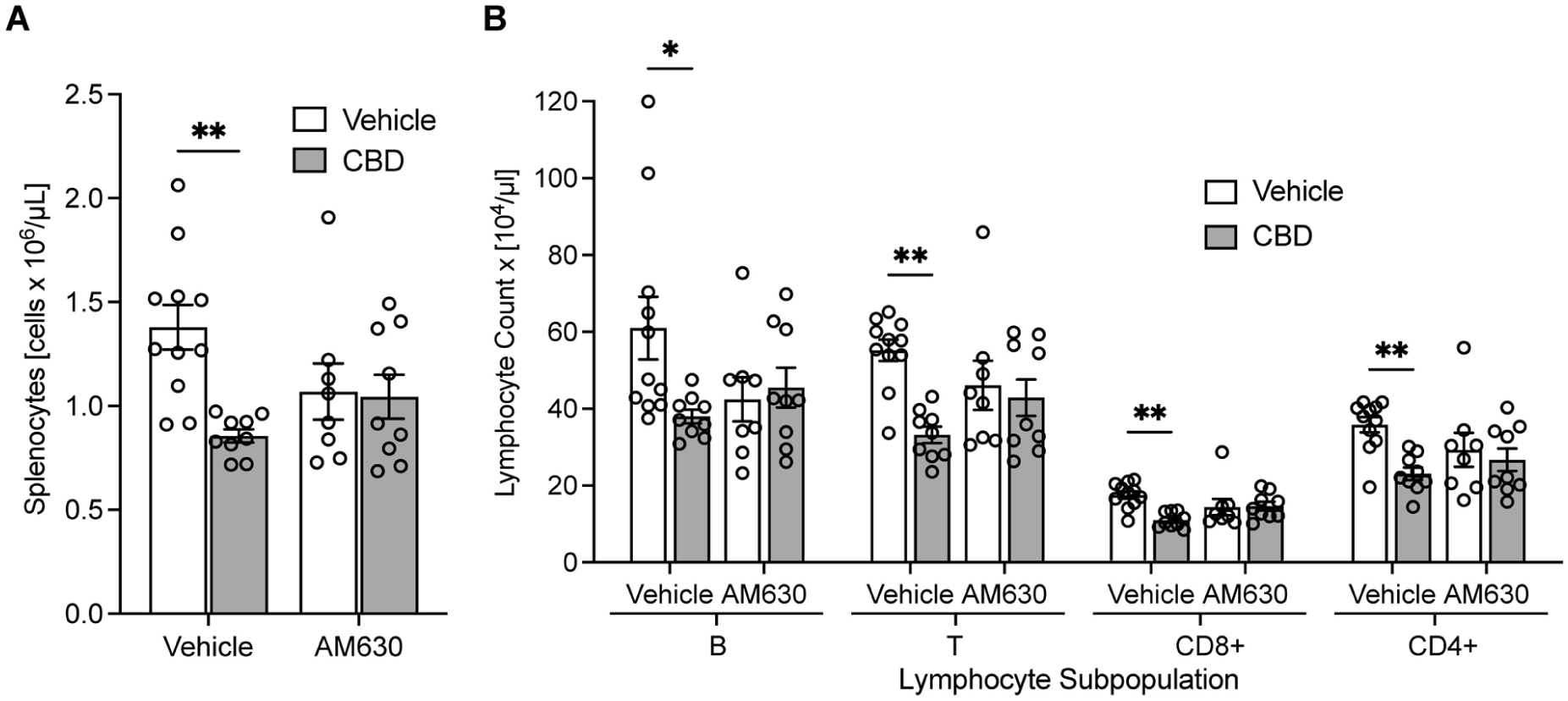
Leukocyte and lymphocyte numbers in the spleen after administration of CBD (5 mg/kg) and pretreatment with CB2 receptor selective antagonist AM630 (1 mg/kg). (a) Total splenic leukocyte number, (b) total number of splenocyte subsets: B, T, T CD8+, and T CD4. Two-way ANOVA followed by Tukey’s post hoc test.
Pretreatment with AM630 before CBD (5 mg/kg, AC) did not significantly affect the reduction in cell counts observed in the VC group. However, splenic leukocyte numbers decreased in the groups treated only with AM630 (1 mg/kg, AV) and both AM630 and CBD (AC) compared to the VV group by about 22.47% and 24.29%, respectively, but this decrease was not statistically significant (Tukey’s post hoc test, VV vs AV p = 0.159, VV vs AC p = 0.097). Similar trends were observed in lymphocytes (Supplemental Figure 7), as well as all splenic leukocyte subsets (Figure 4(b)).
No significant change in total or percentage NK cell numbers was observed (p = 0.086, F1,33 = 2.39, not shown) and did not affect total or percentage numbers of granulocyte or monocyte numbers in the spleen (data not shown).
CBD administration produced a decrease in total spleen weight (from 890 ± 43 mg in VV to 723 ± 34 mg in VC) (Two-way ANOVA, Tukey’s post hoc test, p = 0.002, F1,33 = 12.03 for the effect of CBD, Supplemental Figure 8). In terms of relative spleen weight (percentage of total body weight), the effect of CBD administration was significant, (Two-way ANOVA, p = 0.009, F1,33 = 7.78 for the effect of CBD), but in the post hoc analysis, the decrease from 0.28 ± 0.01 to 0.25 ± 0.01, for VV and VC, respectively, was not significant (Two-way ANOVA, Tukey’s post hoc test, p = 0.088, Supplemental Figure 8). CBD did not affect spleen volume (p = 0.59, F1,33 = 0.296, not shown), and spleen density was reduced by CBD administration, but this tendency was not significant (p = 0.078, F1,33 = 3.31, not shown). AM630 pretreatment did not affect spleen weight significantly (Two-way ANOVA, p = 0.479, F1,33 = 0.51 for the effect of AM630 pretreatment) but partially prevented the decrease in spleen weight in animals treated with CBD. Furthermore, no significant change was found between the VV and AC groups (Supplemental Figure 8).
Discussion
The results of this study revealed that repeated systemic administration of CBD at a dose of 5 mg/kg reduces the total number of lymphocytes and their subsets (B, T, NK, T CD4+, and T CD8+) in the spleen of healthy adult rats. A lower CBD dose of 2.5 mg/kg did not affect spleen leukocyte numbers. NK cells were not involved in this proportional decrease in T and B lymphocytes in the spleen, as their total number, percentage, or cytotoxic activity remained unchanged, paralleling previous findings in peripheral blood. 40 A decrease in not only spleen weight, but also density, due to no observed significant difference in volume, supports the depletion of lymphocytes in the spleen, potentially due to migration. In vitro studies using B lymphocytes have demonstrated the regulatory role of CB2 in cell migration, aligning with the decrease in B lymphocytes observed in Figures 1, 2, and 4.51,52 Furthermore, since CB2 has been shown to mediate the retention of immature B cells in the bone marrow in mice, CBD-induced CB2 activation could enhance this retention, potentially explaining the observed reduction in splenic B lymphocytes. 53 However, this mechanism remains to be tested.
CBD was found to reduce lymphocyte numbers without an effect on granulocytes and monocytes (Figure 1, Supplemental Figure 5). B and T lymphocytes play an essential role in adaptive immunity, targeting specific antigens and rendering them effective against pathogens. The overactivity of this process may also contribute to autoimmune diseases, where host cells are targeted instead of foreign antigens. Meanwhile, granulocytes and monocytes are associated with the mechanisms behind innate immunity.54–57 Administration of CBD at a lower dose of 2.5 mg/kg has been shown to significantly increase the percentage of NK cell numbers in peripheral blood. 40 This led to the hypothesis that CBD may affect NK cell function. In this study, no effects of CBD on NK cell numbers and NKCC were found, suggesting that antitumor effects of CBD may not be associated with increased activity of this cell population, supporting specific effects on adaptive immune response, without affecting some elements of the innate immune response.
Unexpectedly, pre-treatment with the CB2 antagonist AM630 caused a trend for decrease, albeit insignificant, in the cell counts, which makes the interpretation of its significant interaction with the effects of CBD non-trivial. An effect of AM630 alone was not anticipated, as previous literature has not demonstrated effects on immune responses in vitro of CB2 receptor antagonists such as AM630 or SR144528.17,34,58 In vivo, AM630 has been shown to block anti-rolling effects of CBD on leukocytes, but not their adherence to epithelia in blood vessels of the rat knee joint. 59 In this study, while the decrease in adherence after CBD administration was not blocked by AM630, the number of adherent leukocytes did increase with AM630+CBD, similarly to the trends observed in the present study (Figure 4).
A question remains about the fate of these lymphocytes that were no longer detected in the spleen. A similar decrease in lymphocyte numbers has been reported in the blood, 40 and pro-apoptotic effects of CBD on monocytes have been shown to be CB1 and CB2 receptor-independent, 60 but less is known about the role of CB2 receptors in human lymphocytes. 61 Induction of apoptosis of splenocytes has been supported by evidence of caspase-8 activation and depletion of intracellular glutathione and thiols in vitro, supporting our hypothesis of lymphocyte count decrease due to programmed cell death, but further experiments should be performed to address these possibilities, as for now, they are speculative.33,62
As an alternative cause of lymphopenia, endocannabinoid 2-arachidonylglycerol (2-AG) has been shown to cause immune cell chemotaxis in a manner that is CB2 receptor-dependent, so it is possible that CBD indirectly caused lymphocyte migration, but this could require further examination.63–66 There is evidence of the role of CB2 receptors in inhibiting the migration of lymphocytes in vivo and in vitro.67,68 However, it has been difficult to characterize the role of cannabinoids in their abilities to recruit immune cells as well as interfere with chemoattractants, rendering their role complex and context-dependent. 69 Therefore, the fate of the decreased lymphocytes remains an open question to be addressed in further studies.
Both endo- and phytocannabinoids are known to directly inhibit immune cell function via CB2 receptors, which are located mainly in the periphery and are very abundant on the immune cells.70–73 CBD may affect the immune system through CB2 receptors either by direct interaction with receptors or indirectly by affecting endocannabinoid signaling. CBD has been shown to have negative allosteric-like effects on CB2 receptors, as well as act as an inverse agonist of both CB1 and CB2 receptors in vitro.3,5 Despite its low affinity to CB2 receptors, CBD may affect them by prolonging anandamide (AEA) signaling via inhibition of fatty acid amide hydrolase (FAAH), which is responsible for the enzymatic breakdown of AEA.6,9,74 AEA has been shown to have immunosuppressive effects in the spleen, such as a decrease in antibody production, which was successfully blocked by a CB2 receptor antagonist. 72 Although this work focused on CB2 receptors and subsequently the immune context, CBD could also be asserting its effects through other receptors such as GPR55.75,76 CBD has also been shown to activate peroxisome proliferator-activated receptor gamma (PPARγ), with effects on the immune system; however, much of this work has been in various models of inflammation, and not in healthy subjects. 77 Potential involvement of CB1 receptors cannot be ruled out. Furthermore, the study of CBD in the treatment of intestinal inflammation in vivo has suggested that the mechanism of FAAH inhibition by CBD is physiologically relevant and promising.78,79 Beyond the scope of this study, the mechanisms behind the role of CB2 affecting specific lymphocyte subpopulation numbers should be further studied in vivo to understand their physiological relevance. Additionally, while this study confirms the ability of CBD to induce lymphopenia in the spleen of healthy adult rats, its findings necessitate further validation for its conclusions to be considered for clinical examination or future therapeutic applications.
This study was conducted to test the effects of CBD and AM630 on immune cell numbers in the spleen in vivo, but more work is required to assess the molecular pathways that may be involved. It was unexpected that AM630 alone would cause a slight decrease in splenic lymphocyte counts in such a way that it did not differ significantly from CBD alone. Despite the sample size of this study being limited, we were able to demonstrate statistically significant decreases in some lymphocyte subset numbers. However, it would be valuable to replicate these experiments with a higher sample size to confirm our findings regarding the CB2 antagonist, as this may have affected robustness. Future experiments may benefit from technical improvements, including using more specific antibodies such as CD19 or CD20 for more specific labeling of B cells. Finally, the observed effects of AM630 alone complicated the interpretation of the effects of CBD on CB2 receptors. Although AM630 is a commonly used CB2 antagonist, it has also been described as a partial agonist as well as an inverse agonist of CB1, so potential off-target effects of this interaction cannot be ruled out.80,81
Conclusion
The present study revealed that repeated CBD treatment decreases lymphocyte numbers not only in the peripheral blood but also in the spleen. We found that CB2 antagonist had significant interaction with CBD, but it had no significant effect by itself. Alternative mechanisms besides CB2 are likely involved, and further studies are necessary to establish that. Since the lymphopenic effect is observed both in the peripheral blood and spleen, it is possible that it may not be an effect of lymphocyte redistribution but rather due to apoptosis, which should be further examined. Our studies also revealed that the effects of CBD on leukocyte numbers are limited to B and T cells, and the numbers of NK cells and their function were unaffected by CBD administration, suggesting a selective effect of CBD on cells related to adaptive immunity.
Supplemental Material
sj-doc-1-iji-10.1177_03946320251411441 – Supplemental material for Repeated administration of cannabidiol decreases splenic lymphocyte subset numbers in rats
Supplemental material, sj-doc-1-iji-10.1177_03946320251411441 for Repeated administration of cannabidiol decreases splenic lymphocyte subset numbers in rats by Tara H Turkki, Maciej M Jankowski, Wojciech Glac, Piotr Badtke, Viviane M Saito, Artur H Swiergiel and Bogna M Ignatowska-Jankowska in International Journal of Immunopathology and Pharmacology
Footnotes
Author contributions
To outline the contributions of the authors’ work THT: data analysis, visualization, manuscript writing; MMJ: conducting experiments; PB: conducting experiments; GW: conducting experiments; VMS: manuscript writing, AHS: experimental design, manuscript writing; BMIJ: experimental design, conducting experiments, data analysis, manuscript writing, visualization, funding. All authors revised the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Polish Ministry of Science and Higher Education Grants N N303 394036 and N N303 417137 to B. Ignatowska-Jankowska and M. Jankowski. The research was partly financed by the European Union within the European Social Fund in the framework of the project “InnoDoktorant – Scholarships for PhD students, I edition” and the Program of implementing the elements of modern education forms at the University of Gdansk, task: Research grants for PhD students—a chance for the economic development. The project was also supported by the Foundation for Polish Science Ventures Programme (Ventures/2009-4/3), co-financed by the EU European Regional Development Fund. Finally, the authors would like to thank the National Institute on Drug Abuse (NIDA), USA, for the drug supply.
Ethics approval
Ethical approval for this study was obtained from the University of Gdańsk Local Ethical Committee for Animal Experiments (permission number: 33/2008).
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
