Abstract
Cryptosporidium parvum is a zoonotic, protozoan parasite that causes potentially life-threatening diarrhea in the host and can be transmitted via the fecal-oral route. C. parvum can infect cattle and may be detected in their feces using a variety of tests. We compared the level of agreement, ease of procedure, and cost among PCR, lateral flow immunoassay, fluorescent antibody, and Kinyoun acid-fast stain direct smear tests. Over the course of 9 mo, 74 calf fecal samples were submitted and tested for C. parvum using all 4 tests. A Fleiss kappa value of 0.813 was obtained, indicating an excellent level of agreement among tests. Overall, the best test based on cost and ease of procedure was the Kinyoun acid-fast stain direct smear.
Cryptosporidium parvum is a zoonotic, protozoan parasite and a common cause of diarrhea (called cryptosporidiosis) in calves, 1–4-wk-old.2,3,10 Calves shed the infective oocysts in their feces and can transmit C. parvum through the fecal-oral route, often in water and food, to other cattle and to humans. 1 The ingested parasite colonizes the small intestine, attaching to villi and causing villus atrophy. 2 C. parvum is a resilient parasite, capable of tolerating many disinfectants. 2 C. parvum can occur on its own or in association with other pathogenic bacteria and viruses (e.g., Escherichia coli, rotavirus, coronavirus); however, the availability of vaccines for other enteropathogens has resulted in C. parvum becoming one of the most important causes of calf diarrhea. 4 In fact, a study in Ontario, Canada found that 40.6% of calves on surveyed farms were infected with C. parvum and within-farm prevalence was 0–70%. 11 Given the ubiquitous nature and environmental persistence of this hardy parasite, it is vital to have an accurate and cost-effective method of detection that is trusted by veterinarians and producers.
Common methods used to detect C. parvum oocysts in the feces of live calves include direct smears, PCR, and antigen detection methods such as immunochromatographic and immunofluorescence assays. PCR appears to have the highest sensitivity and specificity, followed by antigen detection methods and direct smears.1,2,6,10,12 However, others have pointed out that inhibitory substances found in feces may impact the performance of PCR, and that PCR may overestimate infection by identifying very low concentrations of C. parvum or non-infectious genetic material. 2
The Animal Health Centre (AHC), British Columbia Ministry of Agriculture, Food, and Fisheries (Abbotsford, BC, Canada) uses the Kinyoun acid-fast stain direct smear for the routine detection of Cryptosporidium, and uses a PCR assay upon request. The objective of our study was to evaluate this workflow by using 2 other modalities (Crypt-a-Glo fluorescent antibody test, Waterborne; Xpect lateral flow immunoassay, Remel), and to determine the degree of agreement among all 4 assays for the detection of Cryptosporidium in calves with diarrhea. The tests used are not specific for the detection of C. parvum, rather the tests can detect Cryptosporidium spp. oocysts up to the genus level. However, given the young ages of the calves submitted, it is likely that a positive result indicates C. parvum. Previous studies have found that C. parvum comprised 97% of all Cryptosporidium spp. found in pre-weaned calves. 10
Seventy-four fecal samples from 1–6-wk-old calves with diarrhea were submitted to the AHC in April–December 2020. Each sample was tested using the 4 assays, for the detection (but not quantification) of Cryptosporidium spp. in bovine feces. To ensure that the sample size was appropriate, a minimum required sample number of 39 was calculated using the Power4Cats function in v.1.2.5033 of R as part of the kappaSize (v.1.2; https://cran.r-project.org/web/packages/kappaSize/kappaSize.pdf) package.
For the Kinyoun acid-fast–stained direct smear, feces were smeared on a glass slide, air-dried, heat-fixed, and then, along with a control slide, flooded with carbol-fuchsin. The slide was allowed to stand for 5 min before being rinsed with water and drained.7,8 The slides were then decolorized with acid-alcohol until no more stain appeared in the wash water. The slides were counterstained with methylene blue for 1 min, rinsed with water, drained, and air-dried. Slides were examined for the presence of oocysts with a 100× oil immersion objective (Fig. 1). All samples tested were run in conjunction with a known positive control (i.e., feces from a calf diagnosed with cryptosporidiosis by PCR).
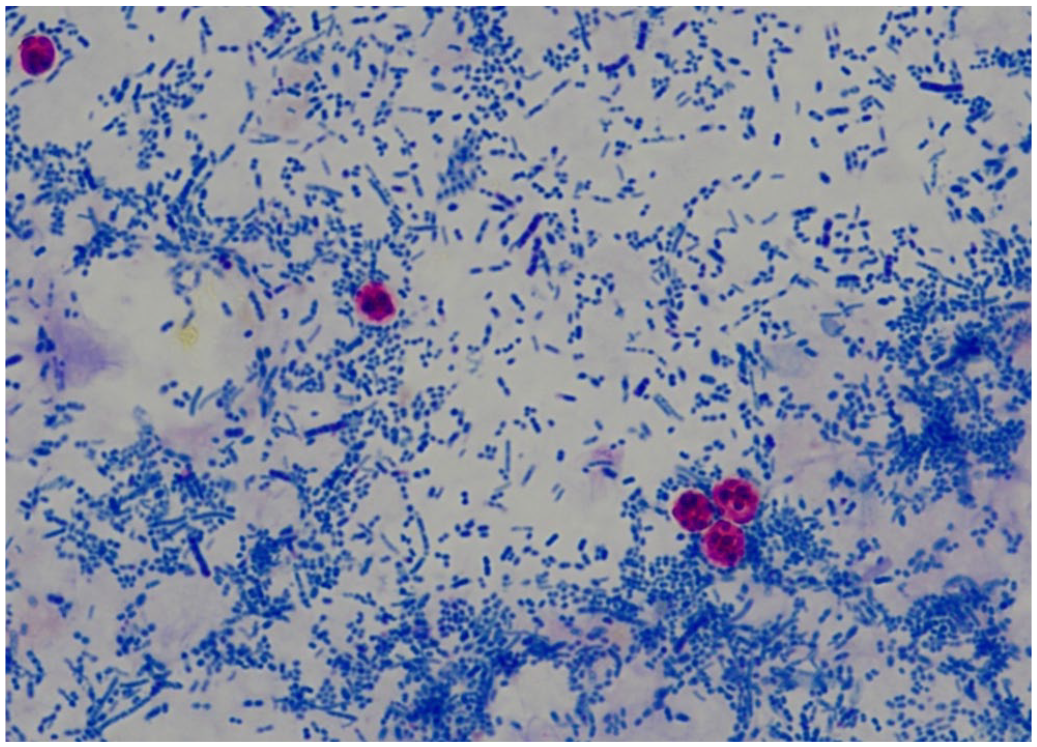
Cryptosporidium spp. oocysts (round, red-staining structures) in calf feces with the Kinyoun acid-fast stain, against a background of bacteria. Original objective 100×.
The Crypt-a-Glo fluorescent antibody test was performed according to the manufacturer’s instructions. Briefly, the specimen was prepared by placing ~5 g of feces into a specimen cup and adding ~10 mL of PBS. Feces was emulsified and then passed through gauze to remove debris. The remaining contents were poured into a test tube and centrifuged, the supernatant was decanted, and the fecal pellet was resuspended in 1 mL of PBS; 15 µL of the prepared specimen was applied to the well of a the SuperStick slide included in the kit, alongside a positive control, and the slides were air-dried. The slide was fixed with 45 µL of absolute methanol and allowed to dry. The sample well was stained with 50 µL of 1× diluted DAPI (4′,6-diamidino-2-phenylindole, dihydrochloride). After 1 min, the slide was rinsed with 50–100 µL of wash buffer. After 1 min, 45 µL of the antibody reagent was applied. The slides were then incubated at room temperature for a minimum of 25 min. The slides were rinsed free of the antibody reagent using 50–100 µL of wash buffer. After 1 min, 1 drop of counterstain was applied per well and incubated at room temperature for 1 min. Following this step, 50–100 µL of wash buffer was used to rinse the slides free of counterstain. Slides were then air-dried. After applying 45 µL of mounting medium, the slides were viewed using 20× and 40× objectives under fluorescence microscopy. Note that the reagents, slide, and positive control were all supplied with the kit; no negative control was used.
The Xpect lateral flow immunoassay was also performed according to the manufacturer’s instructions. Briefly, unpreserved fecal samples were diluted 1:4 using demineralized water. Four drops of the provided dilution buffer were then added to labeled tubes. In each tube, 0.1 mL of the diluted fecal sample was added; 4 drops of conjugate were added to each tube and mixed; 0.2 mL of the mixture of the diluted fecal sample and conjugate was transferred into the well of the lateral flow device. Results were available after 15 min, and a positive result was indicated by a complete line present at the CTRL position and CRYP position. No specific positive or negative control was required given that the kit uses an internal test control as indicated by a complete line present at the CTRL position.
PCR was performed using the following procedure. Feces were diluted 1:10 in minimum essential medium and homogenized in a stomacher for 2 min at medium speed. Homogenized tissues were centrifuged briefly, then DNA was extracted (QIAamp DNA mini kit; Qiagen) with the tissue protocol. DNA extracts were evaluated by a conventional PCR assay targeting the 18S rRNA gene of Cryptosporidium. 5 PCR was performed using the manufacturer-recommended protocol (illustra PuReTaq Ready-to-Go PCR bead; GE Healthcare). Briefly, each PCR reaction contained 1 Ready-to-Go PCR bead, forward and reverse primer (1 µL of each at a starting concentration of 20 µM), 21 µL of nuclease-free water, and 2 µL of DNA template for a total run volume of 25 µL. The assay was cycled at 95°C for 5 min, followed by 50 cycles of 95°C for 1 min, 55°C for 1 min, and 72°C for 1 min, with a final extension of 7 min at 72°C. The assay produced a 435-bp product that was evaluated on 2% agarose gel with ethidium bromide, and then analyzed using a UV photo documentation system. All primers were made by Integrated DNA Technologies. Each assay was run with 3 controls: a negative extraction control, a negative template control, and a positive amplification control.
The Fleiss kappa value was calculated using the kappam.fleiss function in v.1.2.5033 of R 9 using the readxl (v.1.3.1; https://cran.r-project.org/web/packages/readxl/readxl.pdf) and irr (v.0.84.1; https://cran.r-project.org/web/packages/irr/irr.pdf) packages to assess statistical agreement among all 4 tests. The cost per test, based on materials and hands-on technical time (i.e., not counting time waiting between steps), was also calculated.
We tallied the various combination of test results that were observed as well as the number of samples associated with each combination of test results (Table 1). Results agreed for 62 of the 74 samples across all 4 tests (Table 1); there were 12 samples for which the results of 1 or more tests were discordant, resulting in a Fleiss kappa value of 0.813 (values >0.75 indicate excellent agreement). Samples with discordant results may be attributable to either differences in test sensitivity, or to uneven distribution of C. parvum within the feces.
Comparison of the results of 4 assays for the detection of Cryptosporidium parvum in the feces of scouring calves.

– = negative result;+ = positive result.
Number of samples associated with each combination of test results.
Advantages of the Xpect lateral flow immunoassay include simplicity in conducting the assay and interpreting the results, as well as short turnaround time. The main disadvantage is the expense of the test kit (Table 2) especially if many animals must be tested to assess the prevalence of C. parvum on the farm. In contrast, the Crypt-a-Glo immunofluorescent assay and PCR are relatively less expensive but also more time consuming with a number of preparation steps (Table 2). Interpretation of slides (via visual inspection) is also more subjective for Crypt-a-Glo compared to Xpect and PCR. Interpretation of the Kinyoun acid-fast stain direct smear is also subjective; however, the test is inexpensive, fast, and simple to perform (Table 2). Performing the Kinyoun acid-fast stain direct smear is not contingent upon expensive and specific equipment, hence the test can be set up and completed on the farm.
Comparison of the cost and time required to perform 4 assays for the detection of Cryptosporidium parvum in the feces of scouring calves.
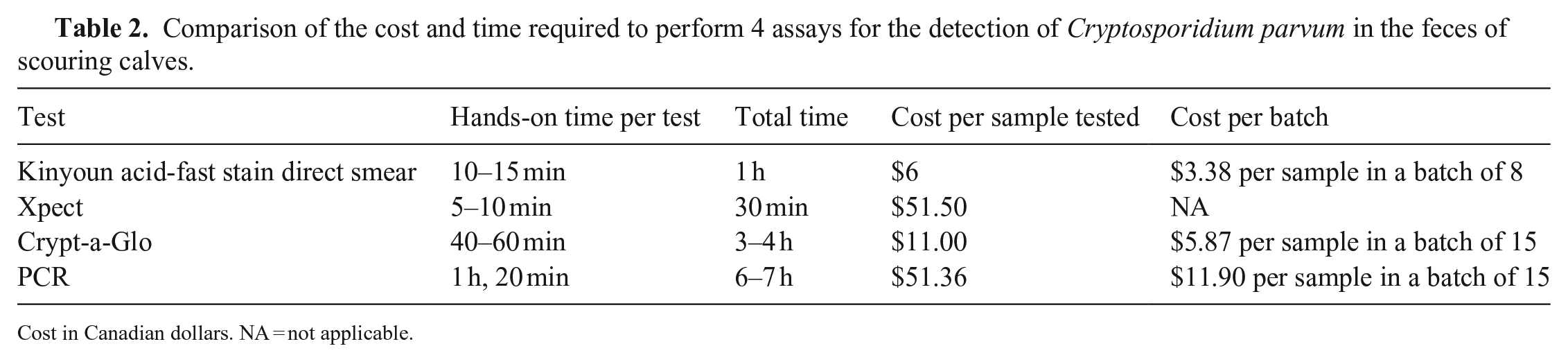
Cost in Canadian dollars.
NA = not applicable.
Given that there is no gold standard for the detection of C. parvum in fecal samples from cattle, relative sensitivity and specificity could not be determined. 4 However, given high agreement among tests, low cost, repeatability on the same sample, and simplicity of the Kinyoun acid-fast stain direct smear, the AHC has elected to continue using this assay for routine C. parvum testing. Recommendations to improve test performance include using fecal concentration methods and including a second evaluator.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
