Abstract
Feces are a noninvasive and easily collectible matrix that may help determine cumulative hormone metabolite concentrations over medium-to-long times. To date, 11-oxoetiocholanolone, an important metabolite of cortisol, has been measured in equine and bovine feces solely by an in-house enzyme immunoassay (EIA). Therefore, we validated the use of a commercial ELISA kit (11-oxoetiocholanolone ELISA kit; Cayman Chemical), which had been validated on sheep feces and human urine, to measure 11-oxoetiocholanolone in feces from 42 horses and 32 bulls. The ELISA kit had good precision (intra- and inter-assay CVs: 5.8% and 11.2% for equine feces; 9.9% and 11.2% for bovine feces, respectively), analytical sensitivity (0.186 ng/mL for both equine and bovine feces), and accuracy (parallelism and recovery tests) in determining 11-oxoetiocholanolone concentrations in feces from both species. We found ranges of 11-oxoetiocholanolone concentrations of 1–109 ng/g in equine feces and 40–302 ng/g in feces of bulls. The Cayman Chemical ELISA kit offers a simple and accessible means of analysis of 11-oxoetiocholanolone in equine and bovine fecal samples.
11-oxoetiocholanolone is an important metabolite of cortisol that has been reported in equine and bovine feces solely by an in-house enzyme immunoassay (EIA).5,6,11 The importance of measuring this hormone in animal studies is that cortisol, its precursor, is considered to be the main biomarker of hypothalamic–pituitary–adrenal (HPA) axis activity. In a study of adrenocortical function in cattle, the administration of ACTH resulted in the excretion of androstanes in feces, specifically 11,17-dioxoandrostanes. Steroids are metabolized in the liver, excreted via the bile into the gut, and then in feces; the lag time between the administration of ACTH and the appearance of cortisol metabolites in feces is influenced by the gut passage time and is species-specific.5,9,11 This group of cortisol metabolites reflects blood cortisol concentrations with a lag-time of 8–16 h in cattle 11 and a lag-time of 24 h for horses. 9
In a search of Google, PubMed, Web of Science, and Scopus, using search terms “cortisol or glucocorticoid” AND “feces or faeces or fecal or faecal” AND “horse or equine” AND “cattle or bovine,” we retrieved no reports of an 11-oxoetiocholanolone EIA measuring 11,17-dioxoandrostanes before 1997. The in-house assay published in 1997 10 for 11-oxoetiocholanolone in feces has since been evaluated in studies of animal transport, 13 animal handling, 3 social stress, 8 pain, 4 housing conditions, 2 and milk production. 1 Feces are a noninvasive and easily collectible matrix that may be helpful in determining cumulative hormone metabolite concentrations over medium-to-long times (with a species-specific lag-time). 7 In contrast to blood and saliva, feces are less prone to sudden changes or the pulsing release of certain hormones, such as glucocorticoids.9,14 This is a huge advantage because samples can be obtained in which glucocorticoid metabolites are not affected by the animal’s handling and can be collected with minimal or no disturbance to the study animal. 14 Adopting fecal assessments may help in understanding phenomena that occurred at a time before the sample was taken and which elicited HPA axis activation.
Our aim was to validate the use of a commercial competitive ELISA kit, which had been tested only on sheep feces and human urine, to assess 11-oxoetiocholanolone in equine and bovine feces as a noninvasively sampled matrix. Cattle and horses are often subjected to long-distance transportation (> 8 h) and their welfare conditions may need to be assessed.
Materials and methods
The care and use of experimental animals complied with local animal welfare laws, guidelines, and policies, and was carried out per EU Directive 2010/63/EU and Italian legislation on animal care (DL 26; 04/03/2014).
Sample type and sampling
Fecal samples were collected from 42 Trotter horses (age: 6.3 ± 6.3 y [x̄ ± SD]; range: 1.0–20.5 y) in northeastern Italy following the animals’ death at slaughter. Samples were obtained manually (nitrile glove; Chemil) from the rectum following the evisceration phase of the animal at the slaughterhouse. The horses had been considered fit for slaughter by the slaughter veterinarian immediately after ~15 min of transportation to the slaughterhouse.
Cattle samples were obtained from 32 Salers bulls (age: 10.5 ± 1.9 mo [x̄ ± SD]; range: 8.2–15.3 mo) in northeastern Italy and were collected immediately after defecation. The bulls had been housed in pens for ≥ 1 mo. The cattle were considered to be clinically healthy at the farm by the farm veterinarian. Fecal samples were stored in clear polyethylene sampling bags (Pool Pack Nord Est) and immediately frozen (−20°C) until processed.
Feces extraction
Fecal samples must be homogenized before extraction because fecal glucocorticoid metabolites may not be distributed evenly. 12 Equine feces (~0.5 g) were placed in glass vials, covered with 5 mL of 80% methanol (1 mL of water + 4 mL of methanol), vortexed for 2 min, and centrifuged at 2,500 × g for 15 min. Then a 1-mL aliquot of the supernatant was extracted with 5 mL of diethyl ether and 0.5 mL of 5% NaHCO3; 4 mL of water was added and the tube was inverted 4 times. The aqueous phase was frozen at −20°C, and the ether phase was decanted and dried down at 37°C under an air-stream suction hood. The dried ether phase was dissolved in 500 µL of Tris buffer (1×) and stored at −20°C until analysis.4,5
Cattle feces (~0.5 g) were placed in glass vials, covered with 5 mL of 80% methanol (1 mL of water + 4 mL of methanol), vortexed for 2 min, and centrifuged at 2,500 × g for 15 min. An aliquot of the supernatant was diluted 1:4 in Tris buffer (1×) and stored at −20°C until analysis.11,13
Both for equine and bovine samples, weight was considered repeatable after obtaining at least 3 successive equal weights with the same third significant figure after the decimal point (the one corresponding to mg). This procedure was carried out given the sensitivity (readability 1 × 10−4 g; reproducibility(s) 1 × 10−4 g) of the balance (ABJ 80-4NM; Kern) used to weigh the samples. 9
To ensure the quality of the results, pipettes are calibrated by a specialized service provider every 6 mo; a calibration certificate includes an assessment of the measurement uncertainty of the pipette. Between calibrations, weekly routine checks of the pipettes ensure that the pipettes continue to perform within specifications. Analytical balances are calibrated once per year by a professional scale calibration company, and we check scale calibration daily to 2 × 10−4 g accuracy with calibrated weights.
11-oxoetiocholanolone EIA characteristics
The 11-oxoetiocholanolone concentrations in equine and bovine feces were measured with a commercial ELISA (501420, 11-oxoetiocholanolone ELISA kit; Cayman Chemical). The kit included a clear plastic 96-well microplate coated with mouse monoclonal anti-rabbit IgG.
The reagents in the kit included: 1) 20 μg/mL of 11-oxoetiocholanolone standard in methanol, which was used to generate the standard curve [standard was diluted (as indicated in the kit instructions) to obtain 8 standards: 1 (200 ng/mL), 2 (57.1 ng/mL), 3 (16.3 ng/mL), 4 (4.67 ng/mL), 5 (1.33 ng/mL), 6 (0.38 ng/mL), 7 (0.11 ng/mL), and 8 (0.03 ng/mL)]; 2) a rabbit polyclonal antibody specific for 11-oxoetiocholanolone, to be added for competitive binding of antigen (11-oxoetiocholanolone) in the standards, in the samples, and in the maximum binding wells; 3) 11-oxoetiocholanolone alkaline phosphatase (AP) conjugate (11-oxoetiocholanolone tracer), to be added to the standards, the samples, the nonspecific binding, and the maximum binding wells, to compete with binding of the 11-oxoetiocholanolone antibody; 4) Tris buffer concentrate (10×), diluted 1×, which was used to create the dilutions of the 11-oxoetiocholanolone standards and to dissolve the dry residues of the extracted fecal samples; 5) AP wash buffer concentrate (150×), diluted 1×, which was used to physically separate the free fraction in each well (after being diluted following the manufacturer's instructions); and 6) pNPP substrate solution added to each well to generate a detectable signal.
We added 2 controls obtained from extracted sheep feces to the plate, one with a low 11-oxoetiocholanolone concentration (18.9 ng/g) and the other with a high 11-oxoetiocholanolone concentration (258 ng/g). The cross-reactivities of the 11-oxoetiocholanolone antibody with other steroids (as established by the manufacturer) were as follows: 5β-androstan-3α-ol-11,17-dione (11-oxoetiocholanolone) 100%, 5β-androstan-3,11,17-trione 54.7%, 5α-androstan-3α-ol-11,17-dione (11-ketoandrosterone) 2.56%, 5α-androstan-3,11,17-trione 2.49%, 5α-androstan-3β-ol-11,17-dione (11-ketoepiandrosterone) 1.4%, 5β-androstan-3α,11β-diol-17-one (11β-hydroxy etiocholanolone) 0.12%, 5α-androstan-3α-17β-diol (17β-dihydro androsterone) < 0.01%, 5α-androstan-3β-17β-diol (17β-dihydroepiandrosterone) < 0.01%, 5β-pregnan-3α,11β,17,21-tetrol-20-one (tetrahydrocortisol) < 0.01%, 5β-pregnan-3α,17,20α,21-tetrol-11-one (α-cortolone) < 0.01%, 5β-pregnan-3α,17,20β,21-tetrol-11-one (β-cortolone) < 0.01%, 5β-pregnan-3β,11β,17,21-tetrol-20-one (3β-tetrahydrocortisol) < 0.01%, 5β-3α,17,21-trihydroxy-pregnane-20-one (tetrahydro-11-deoxy cortisol) < 0.01%.
Fecal 11-oxoetiocholanolone analysis by ELISA
The 11-oxoetiocholanolone standard (20 μg/mL) was dilut-ed as indicated in the kit instructions to obtain 8 standards, 200–0.03 ng/mL. Then, 50 μL of standards, controls, or sa-mples were pipetted in duplicate into different wells of a clear plastic 96-well microplate coated with mouse monoclonal anti-rabbit IgG. Subsequently, 50 μL of 11-oxoetiocholanolone tracer and 50 μL of 11-oxoetiocholanolone antibody were pipetted into each of those wells on top of standards, controls, samples, and maximum binding wells. The plate was then incubated for 20 h at 4°C to allow the competitive binding between the antigen (11-oxoetiocholanolone) and the 11-oxoetiocholanolone antibody. At the end of the incubation time, the microplate was washed 5 times with 300 μL wash buffer 1×, 200 μL of the pNPP was added to each well, and 5 μL of 11-oxoetiocholanolone tracer was added in the total activity well. This step was followed by another incubation of the plate on an orbital shaker at room temperature for 120 min to allow the reaction between the pNPP and 11-oxoetiocholanolone tracer in the wells; the microplate was then read at 405 nm (Ensight multimode plate reader; Perkin-Elmer).
Fecal 11-oxoetiocholanolone ELISA validation
All of the validation tests for the assay that we used for the fecal 11-oxoetiocholanolone measurements were conducted in the same way for the equine and bovine specimens and all of the tests used different pools of samples, each comprising 5 fecal extracts.
We performed a parallelism test to identify any deviations from the standard curve among the series of fecal extracts, which contained known amounts of 11-oxoetiocholanolone. The test was prepared using serial dilutions of the extracts from equine feces and extracts from bovine feces in Tris buffer (1×), which had high concentrations of 11-oxoetiocholanolone (98.5 ng/g and 257 ng/g for equine and bovine feces, respectively). Each dilution was pipetted in quintuplicate. Linear regression was used to determine if the fecal extracts and the standard 11-oxoetiocholanolone curve deviated from parallelism.
We conducted a recovery test to evaluate the analytical system response to an increasing amount of 11-oxoetiocholanolone standard, which was added to fecal extracts with low 11-oxoetiocholanolone concentrations. Each spiked or non-spiked sample was pipetted in quintuplicate. The percentage of recovery was determined as follows: ([measured 11-oxoetiocholanolone in spiked sample]/[measured 11-oxoetiocholanolone in non-spiked sample + 11-oxoetiocholanolone added] × 100%).
We determined the analytical sensitivity of the assay as the hormone concentration that resulted in the displacement of the labeled hormone by at least 2 SD from maximal binding (as calculated in RiaSmart; Canberra-Packard).
We estimated the precision of the assay by assessing intra- and inter-assay variation, which was expressed as the CV. The intra-assay precision was evaluated by testing 20 replicates of a sample with a known medium 11-oxoetiocholanolone concentration (19.9 and 126 ng/g for equine and bovine feces, respectively) in the same assay. The inter-assay precision was evaluated by running the same equine and bovine extracts in 20 assays in duplicate over 10 d in 5 wk (the extracts were kept frozen).
Statistical analysis
The dose-response curve (4PL curve) was fitted using the statistical software for the RiaSmart immunoassay data. The software was used to interpolate the analyte concentrations in the unknown samples. The arithmetic x̄ and SDs, along with the CVs and linear regression, were calculated in a spreadsheet using routine descriptive and predictive statistical analysis procedures (Excel 2019; Microsoft).
Results
Parallelism was acceptable for equine feces and standards, which reacted identically to antibodies. A high correlation (r = 0.99) was also observed between the obtained and expected concentrations. The relationship between the fecal 11-oxoetiocholanolone concentrations and the expected concentrations was given by the equation y = 1.074x – 0.094 (Fig. 1). In the recovery test, the recovery rate was 122 ± 6% (x̄ ± SD). The assay analytical sensitivity was 0.186 ng/mL. The equine feces extracts (19.9 ± 1.2 ng/g; x̄ ± SD), in repeated determinations, had intra- and inter-assay CVs of 5.8% and 11.2%, respectively. The feces 11-oxoetiocholanolone concentrations in horses were 1.0–109 ng/g.
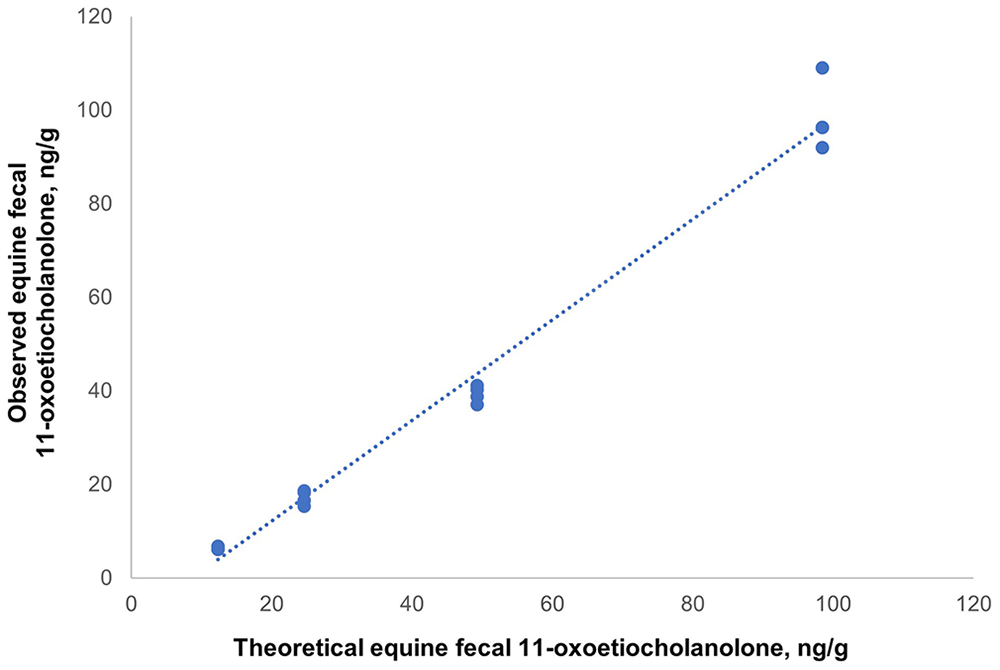
Graphical plot for the parallelism obtained with a commercial ELISA kit for 11-oxoetiocholanolone (Cayman Chemical) on equine feces. The relationship between equine fecal 11-oxoetiocholanolone concentrations and the standard 11-oxoetiocholanolone curves was given by the equation y = 1.074x – 0.094.
Parallelism was acceptable for bovine feces and standards, which reacted identically to antibodies. The correlation between the obtained and expected concentrations was high (r = 0.99). The relationship between the fecal 11-oxoetiocholanolone concentrations and the expected concentrations was given by the equation y = 1.091x – 0.076 (Fig. 2). In the recovery test, the recovery rate was 121 ± 11% (x̄ ± SD). The assay analytical sensitivity was 0.186 ng/mL. The bovine fecal extracts (126 ± 12 ng/g; x̄ ± SD), in repeated determinations, had intra- and inter-assay CVs of 9.9% and 11.2%, respectively. The fecal 11-oxoetiocholanolone concentrations in cattle were 40–302 ng/g.
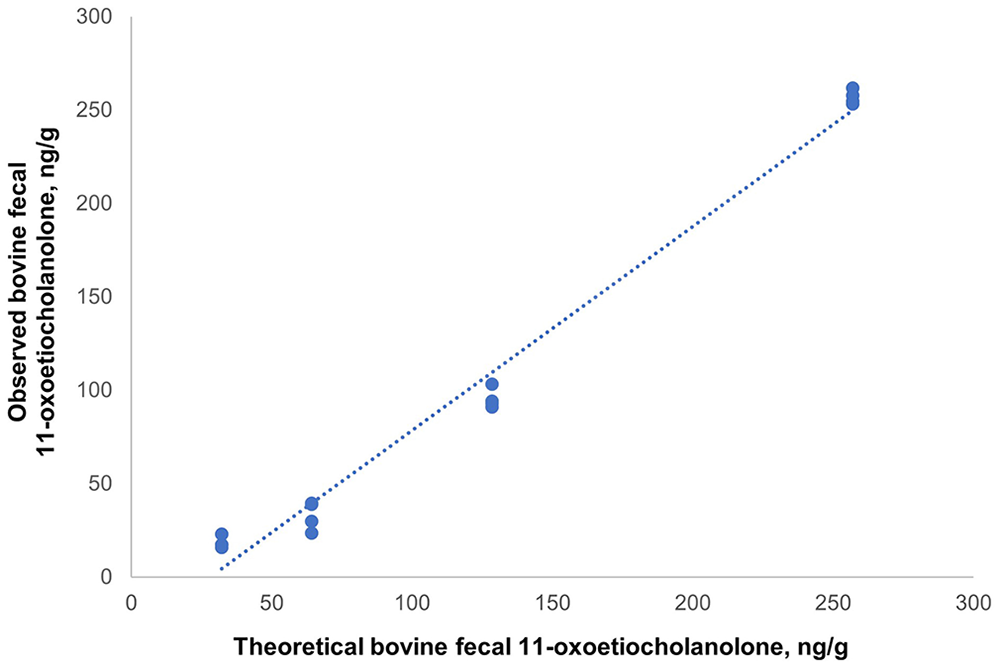
Graphical plot for the parallelism obtained with a commercial ELISA kit for 11-oxoetiocholanolone (Cayman Chemical) on bovine feces. The relationship between bovine fecal 11-oxoetiocholanolone concentrations and the standard 11-oxoetiocholanolone curves was given by the equation y = 1.091x – 0.076.
Discussion
We found that the Cayman Chemical ELISA kit, which has been validated for sheep feces by the manufacturer, had good precision, sensitivity, and accuracy (by parallelism and recovery tests) for determining 11-oxoetiocholanolone in both equine and bovine feces. In the samples that we tested, the range of 11-oxoetiocholanolone concentrations in equine feces was narrower than that observed in bovine feces. These results, however, need to be confirmed by a larger sample of data and the same selection and sampling criteria of the 2 groups (e.g., age range, physiologic status). Similar ranges and peaks of values of 11,17-dioxoandrostanes were reported in horses after castration or with colic, 4 and in cattle after transportation. 13 Note that the Cayman 11-oxoetiocholanolone ELISA kit and specifically, the 11-oxoetiocholanolone antibody, has a certain level of cross-reaction with 5β-androstan-3,11,17-trione, a cortisol metabolite of the 11,17-dioxoandrostanes group excreted in the feces.
The major challenge of the described protocol is the step of sample weighing before extraction. This step requires particular attention, and its accuracy can influence the final results; samples of <0.05 g may be inadequate for accurate results. 9
Limitations of our validation study include the lack of diversity of the subjects; also, the number of included individuals was too limited to provide ranges that would represent the RIs for 11-oxoetiocholanolone in horses and cattle.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
