Abstract
A 3-y-old, intact female, American Pit Bull Terrier was presented because of acute onset of anorexia and a large subcutaneous submandibular mass that had been present for 3 wk. The submandibular mass, 2 engorged black-legged ticks on the dorsum of the neck, pyrexia, and icterus were seen on physical examination. Abnormal laboratory test results included a positive
Lymphoma is a common canine tumor, comprising 7–24% of all tumors and 83–90% of hematologic cancers. 13 Middle-aged to older dogs are most affected, with higher incidences seen in Boxers, Bullmastiffs, and Bulldogs. 12 Clinically, lymphoma with primary nodal involvement is more common than extranodal disease in dogs, but any organ can be affected. 12 B-cell–derived tumors are twice as common as those derived from T cells. 13 T-cell lymphoma-associated hemophagocytic syndrome (LAHS) has been identified very rarely in dogs; most cases have been reported in Asia. 14 LAHS has also been described in humans. 14
Hemophagocytic syndromes or hemophagocytic lymphohistiocytosis (HLH) represents an uncontrolled, overactive immune response as a result of immune dysfunction, resulting possibly from an immune deficiency or acquired failure of immune regulation.3,14 Major characteristics of HLH include histiocytic proliferation and pronounced hemophagocytosis by the mononuclear phagocyte system. 14 HLH may occur independent of, or as a part of, a neoplastic process. 7
A 3-y-old, intact female, American Pit Bull Terrier was presented to the Tuskegee University Veterinary Medical Teaching Hospital (TU-VMTH; Tuskegee, AL, USA) with a 2-d history of inappetence and a large subcutaneous mass ventral to the left mandibular ramus, noticed 3 wk prior to presentation. The patient was not current on preventive medications or vaccines. Physical examination revealed pyrexia and mildly icteric, moist mucous membranes, with a normal capillary refill time. Mandibular and popliteal lymphadenopathies were palpable; the subcutaneous left submandibular mass was ~6 × 6 cm and was firmly fixed. Two blacklegged ticks were found and removed from the dorsum of the neck. A complete blood count (CBC), serum biochemical profile, SNAP 4Dx Plus test (
Selected hematology and serum chemistry parameters in a canine lymphoma-associated hemophagocytic syndrome patient.
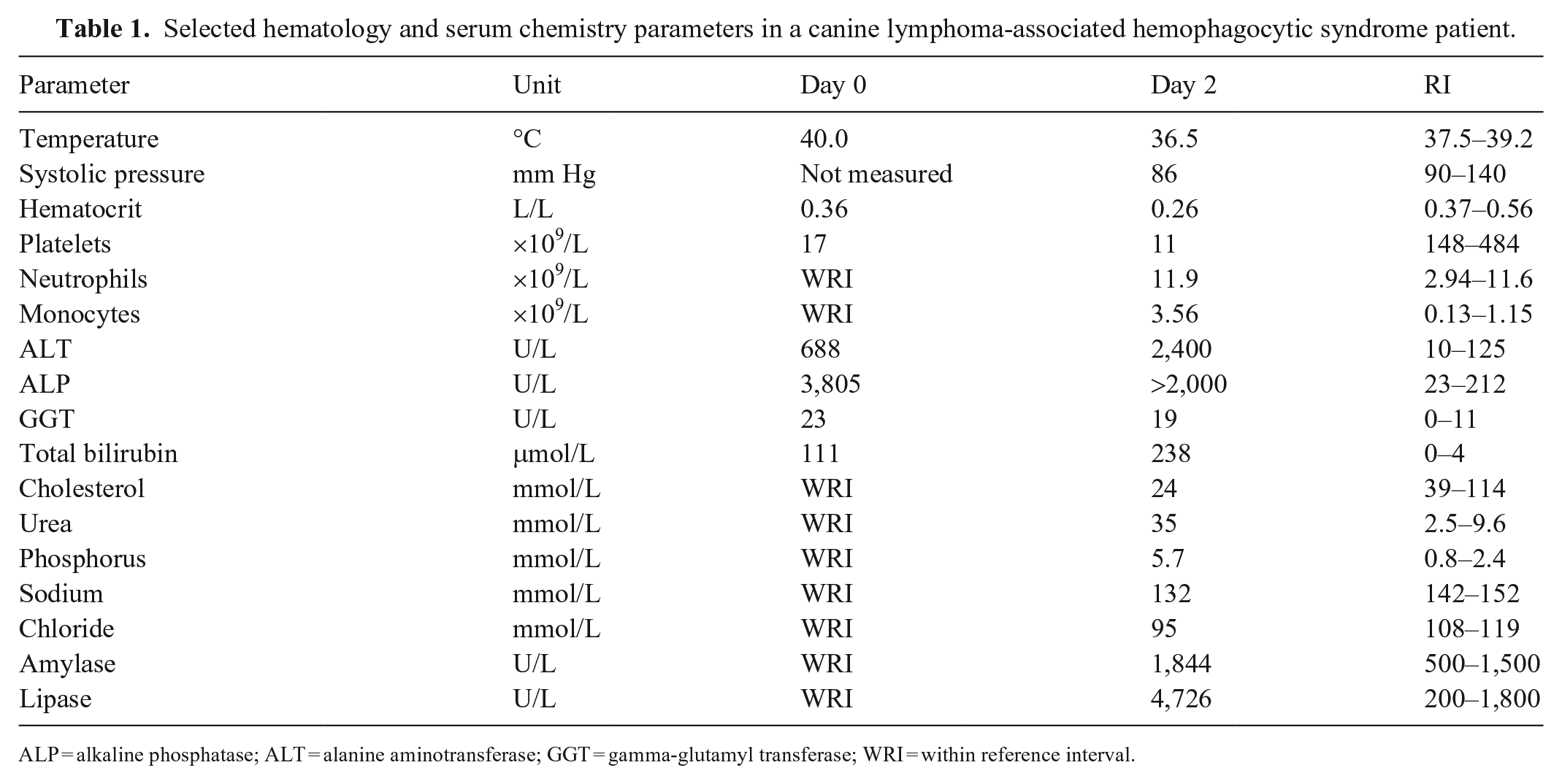
ALP = alkaline phosphatase; ALT = alanine aminotransferase; GGT = gamma-glutamyl transferase; WRI = within reference interval.
Cytologically, the FNA of the submandibular mass had marked numbers of karyolytic neutrophils, often with intracellular cocci, clusters of extracellular cocci, and low numbers of macrophage-type cells. There were low numbers of lymphoid cells and rare, small aggregates and individualized spindle-shaped mesenchymal-like cells scattered throughout. The mesenchymal-like cells had a moderate nucleus:cytoplasm (N:C) ratio with moderately basophilic cytoplasm and oval nuclei with coarse chromatin and 1 or 2 prominent nucleoli (Fig. 1). The interpretation was marked septic purulent and mild-to-moderate mixed inflammation with evidence of acute hemorrhage and mild mesenchymal proliferation.
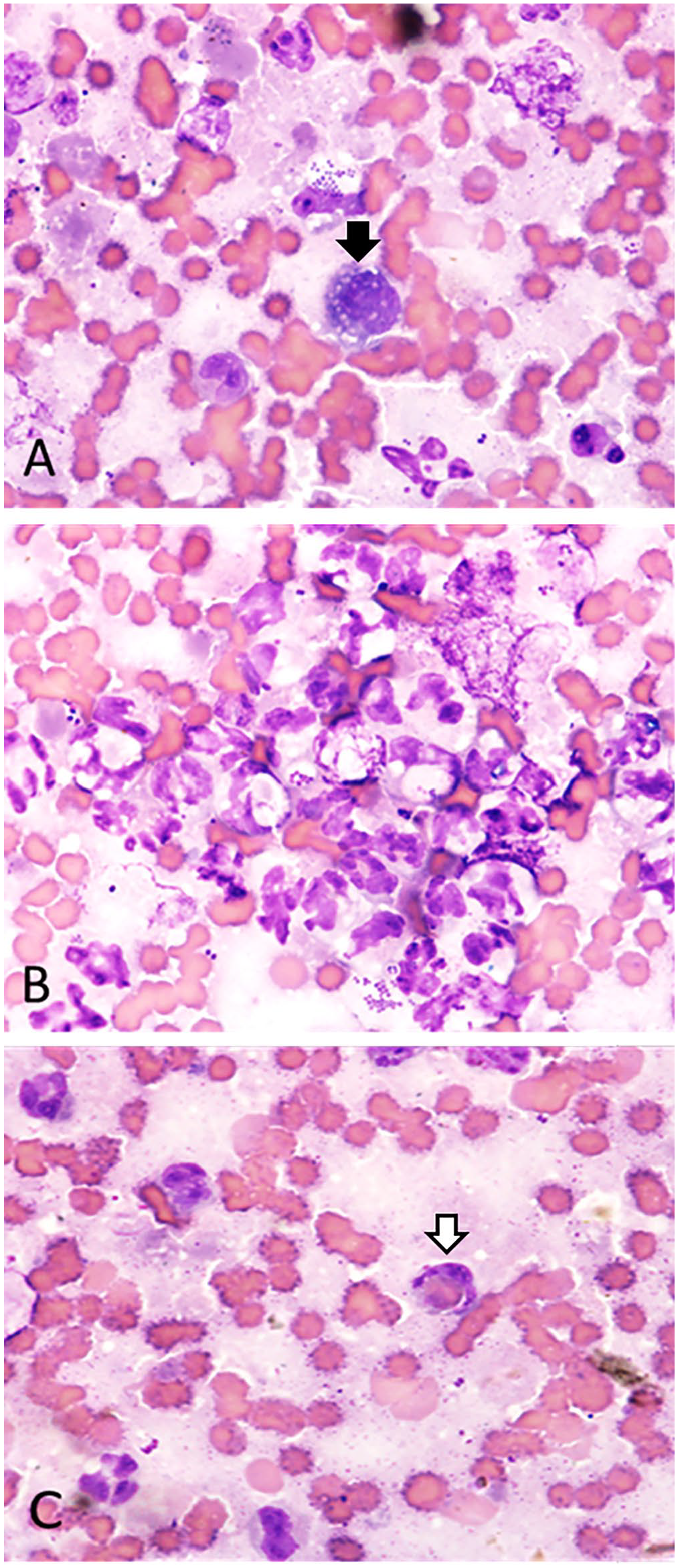
Cytology of the left submandibular mass in T-cell lymphoma-associated hemophagocytic syndrome in an American Pit Bull Terrier.
In light of the profound thrombocytopenia, performing an incisional or excisional biopsy for histopathology of the jaw mass was postponed. The patient was presumptively diagnosed with anaplasmosis and discharged with medications: doxycycline (q12h, PO; Harris Pharmaceutical), ivermectin–pyrantel (q30d, PO; Boehringer Ingelheim), and imidacloprid–permethrin–pyriproxyfen (q30d, topically; Bayer). A one-week reassessment was scheduled with the TU-VMTH Internal Medicine Service to evaluate the patient’s progress and treatment efficacy.
At home, the owner attempted feeding the doxycycline with food, but the patient regurgitated on each attempt. Two days after the initial presentation, the patient was returned to the TU-VMTH Emergency Service, non-ambulatory, with a distended abdomen, anorexia, worsened icterus, hypotension, hypothermia, and ~10% dehydration. The patient’s heart rate and sounds were normal, but heart sounds were muffled by harsh lung sounds in all fields. A rapid leptospirosis IgM-detection immunochromatographic test and a slide agglutination test were both negative, ruling out leptospirosis and Evans syndrome, respectively. On abdominal radiographs, hepatomegaly, splenomegaly, and a mid-abdominal mass were observed. Repeat bloodwork (CBC and serum biochemistry [Table 1] with the addition of a coagulation panel) revealed anemia, thrombocytopenia, and leukocytosis, with mild mature neutrophilia and moderate monocytosis. Biochemistry revealed moderately elevated serum urea nitrogen, marked hyperphosphatemia, moderate hypocholesterolemia, ALP activity too high to read, moderately elevated GGT activity, more significantly increased ALT activity, profound hyperbilirubinemia, moderate hyperamylasemia, marked hyperlipasemia, moderate hyponatremia, and mild hypochloremia. Prothrombin time (>100 s; RI: 11–17 s) and partial thromboplastin time (>300 s; RI: 72–102 s) were both prolonged beyond the machine’s ability to read, suggesting disseminated intravascular coagulation (DIC). The patient became tachypneic and went into cardiac arrest shortly after instituting supportive care. Cardiopulmonary resuscitation (CPR) was administered but was unsuccessful; the owner consented to an autopsy.
Postmortem examination revealed a firm, dark-red, 7 × 7-cm mass affixed to the left mandible. There was ~40 mL of blood within the abdominal cavity and another 20 mL within the oral and nasal cavities. The gallbladder was distended, red-purple, with a diffusely roughened mucosal surface. The spleen was enlarged and contained pale 0.5 × 0.5-cm nodules. A firm, dark multinodular 12 × 5 × 4-cm mass between the liver and duodenum was attached to the proximal end of the pancreas. Frothy red fluid was seen within the trachea, bronchi, and bronchioles. Both lungs were pale pink with petechiae and ecchymoses, possibly the result of severe thrombocytopenia, abnormal coagulation, and/or chest compression trauma during CPR. A 2.5-cm dark-red mass was adjacent to the stomach within the mesentery. Petechiation was present throughout the small intestinal mucosa, and there was diffuse, dark-red to purple discoloration of the jejunal mucosa. Melena and hematochezia were observed within the large and small intestines. Organ samples were collected, formalin-fixed, and processed routinely for histologic review. Bone marrow was not collected.
Histologically, the submandibular and pancreatic masses were poorly demarcated, densely cellular, nodular, and composed of cells arranged in packets within a scant fibrovascular stroma. The cells were round-to-polygonal, 15–45 µm diameter, with moderate, amphophilic, homogeneous cytoplasm. The nuclei were single or binucleate, oval-to-reniform, and central, with finely stippled to coarse chromatin. Anisokaryosis and anisocytosis were prominent, and there were 13 mitotic figures in 2.37 mm2 (10 hpf; Fig. 2A). Neoplastic cell morphology was consistent with leukocytic origin. These neoplastic cells were intermixed with large amounts of blood, fibrin, and necrotic debris (Fig. 2B). There were abundant schistocytes, and sporadic, mild phagocytosis of erythrocytes by neoplastic cells, with moderate hemosiderosis in the liver, spleen, and mesenteric lymph nodes (LNs; Fig. 2C). Several blood vessels contained neoplastic cells and/or fibrin thrombi. Perivascular infiltration of neoplastic cells was seen within LNs, lungs, pancreas, jaw mass, myocardium, endocardium, spleen, kidneys, and liver. Intraluminal neoplastic cells were identified within blood vessels in the liver and lungs. Megakaryocytes were prominent in the spleen and lungs. The mucosa of the stomach had enlarged foci of presumed lymphoid aggregates. Based on these findings, histiocytic sarcoma was suspected.
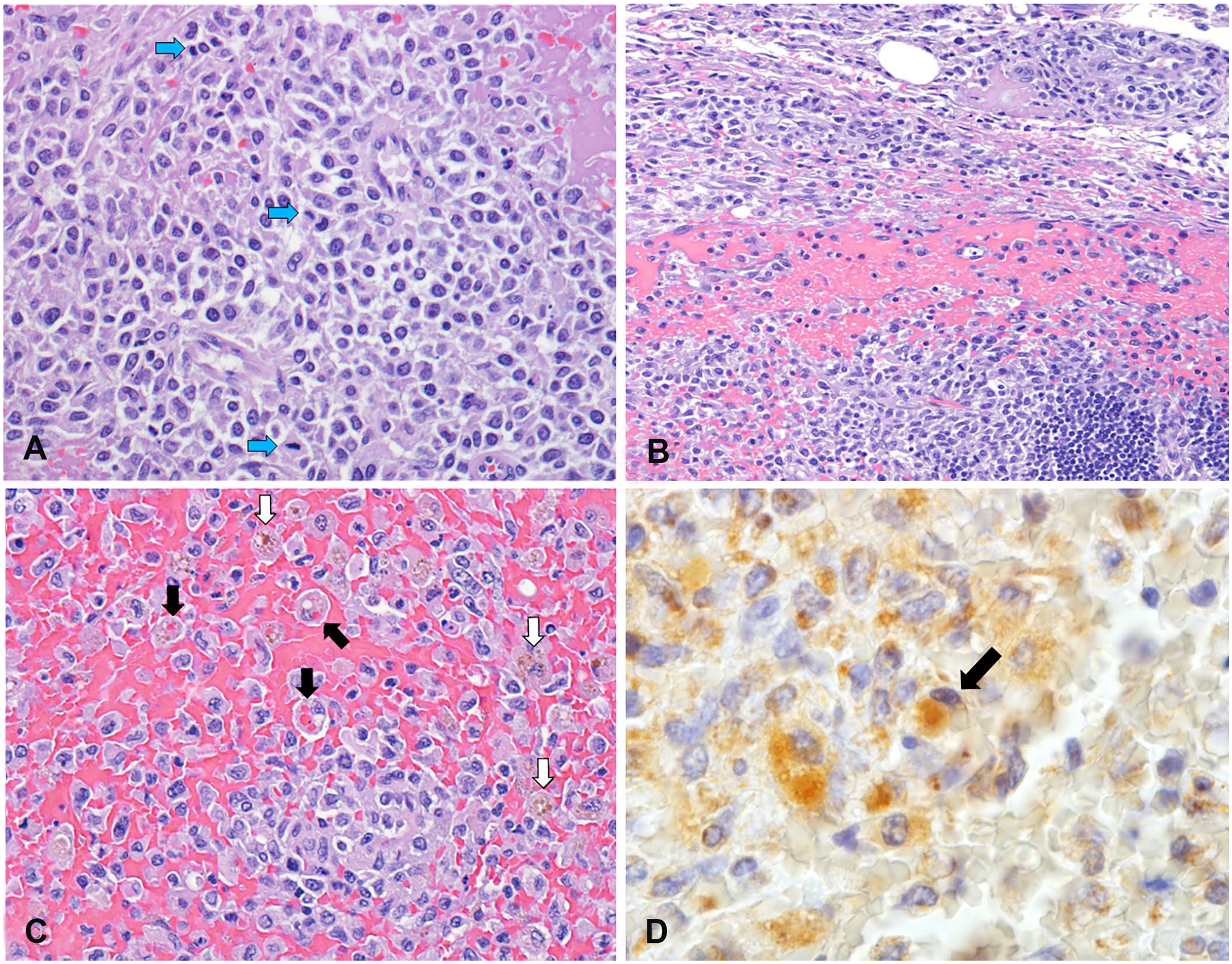
T-cell lymphoma-associated hemophagocytic syndrome in an American Pit Bull Terrier.
Formalin-fixed samples of the spleen, lungs, mesenteric LNs, and mandibular mass were submitted for immunohistochemistry (IHC) staining at the Michigan State University Veterinary Diagnostic Laboratory (MSU-VDL; Lansing, MI, USA). Based on stain availability and consultation with an MSU anatomic pathologist, CD18 (panleukocyte marker) and CD204 (histiocyte marker) staining were performed, followed by CD3 (T-lymphocyte marker) and CD20 (early B-lymphocyte marker) staining. Most of the proliferating, and often perivascular, neoplastic cells, had weak-to-strong perimembranous labeling for CD3 (Suppl. Fig. 1) and CD18; there were a few scattered CD20+ cells. The proliferating cells, including spindle-shaped cells, were CD204– in all tissues. These findings (CD3+, CD18+, CD204–) were deemed consistent with T-cell lymphoma. T-cell PCR for antigen receptor rearrangement (PARR) was additionally performed at the MSU Veterinary Molecular Pathology Laboratory on tissue samples submitted, but PARR did not confirm clonality for the T-cell receptor gene.
To verify the IHC findings, sections of lung, liver, and the submandibular mass were submitted to the North Carolina State University Anatomical Pathology Laboratory (Raleigh, NC, USA) for Iba1 (ionized calcium-binding adapter molecule 1) and CD3 staining. There were 17 mitotic figures per 2.37 mm2 (10 hpf) reported in the immunostained lung sections. Neoplastic cells were CD3+ and Iba1–. Hemophagocytes (>2%) were observed in the liver, spleen, and mesenteric LN of CD3– (Fig. 2D) and CD18-stained sections, similar to reports of the bone marrow of other canine LAHS patients. 13 Based on the laboratory findings, bicytopenia (anemia and thrombocytopenia), splenomegaly, and erythrophagia, T-cell LAHS was diagnosed.
Cytologically and histologically, some T- and B-cell lymphomas resemble histiocytic sarcoma morphologically.11,12 Prior to IHC staining, histiocytic sarcoma was our top differential diagnosis. Immunocytochemistry and IHC are needed to support a diagnosis of LAHS and to rule out histiocytic tumors. CD18 and major histocompatibility complex II staining is helpful in diagnosing histiocytic tumors and indicating their cell of origin.11,12 Lymphocytes and histiocytes stained CD18+, which ruled in lymphoma. CD3 and CD20 staining is specific for T- and B-cell lymphoma, respectively, and so were necessary to differentiate between the lineages.4,10 Negative labeling of macrophage scavenger receptors (CD204) gave additional support to a non-histiocytic origin.11,12 The Iba1 stain is histiocytic malignancy specific, and unlike many other markers, has demonstrated consistency in staining and assessing formalin-fixed tissues.4,9 Staining Iba1– further supported non-histiocytic origin of the neoplastic cells.
In many T-cell lymphomas, the PARR assay amplifies the variable regions of the T-cell receptor, thus identifying a clonal population of lymphocytes. 5 Based on MSU quality control assays, their PARR test has ~90% sensitivity in detecting T-cell lymphoma. Although no PARR clonality was confirmed, results of histologic examination and immunophenotyping are consistent with T-cell lymphoma. Failure to demonstrate T-cell clonality was likely because the specific gene rearrangement in this cell population was undetectable. Approximately 9% of canine samples with confirmed lymphoid neoplasia will have a negative PARR result, which may reflect neoplasms that have V or J regions (to which the primers do not bind), lymphomas that have deleted antigen receptor genes, or NK-derived lymphomas that do not have rearranged antigen receptor genes. 2
Human HLH has been associated with malignancies, autoimmune diseases, and infections. 8 Diagnosis of human HLH is based on demonstration of any 5 of the following features: fever, splenomegaly, hypertriglyceridemia (≥3 mmol/L) and/or hypofibrinogenemia (≤1.5 g/L), low-to-nonexistent natural killer cell activity, hyperferritinemia (>500 μg/L), bicytopenia or pancytopenia, high soluble CD25, and erythrophagocytosis in lymphoid tissue.8,9,14 Clinical signs associated with canine hemophagocytic syndromes include fever, icterus, splenomegaly, hepatomegaly, and diarrhea. 16 In canine LAHS, the proposed diagnostic criteria includes a diagnosis of lymphoma, presence of bicytopenia or pancytopenia, and hemophagocytosis within the mononuclear phagocytic organs (seen in spleen and liver here). 14 Hemophagocytosis in LNs, therefore, may be an additional criterion to include in canine LAHS features. Although only suspected in our case, DIC, along with gastrointestinal and alveolar hemorrhage, was reported in 10 of 77 (13%) human LAHS cases in a retrospective study. 10
Without antigen testing and the absence of organisms on smear review, active anaplasmosis could not be confirmed for our case.
Chemotherapy and immunosuppression are the recommended treatment options in human LAHS, but poor outcomes have been reported.3,14,15,17 No treatments have improved the survival time in dogs with LAHS. A median survival time (MST) of 20 d was established in a single retrospective study of 6 cases of LAHS in dogs who received glucocorticoids or chemotherapy. 14 In one human retrospective study of LAHS, a MST of 43 d was noted. 3 However, a MST of 231 d after the initial diagnosis has been reported in 30 human LAHS patients, with improved survival time attributed to treatment using rituximab and stem cell transplantation. 17 These therapies, therefore, may be promising in extending canine LAHS MST and merit further research.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387211061264 – Supplemental material for T-cell lymphoma-associated hemophagocytic syndrome in an American Pit Bull Terrier
Supplemental material, sj-pdf-1-vdi-10.1177_10406387211061264 for T-cell lymphoma-associated hemophagocytic syndrome in an American Pit Bull Terrier by Dominique A. Hemmings, Athema L. Etzioni, Gbemisola Akingbade and Fredrick E. Tippett in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The TU RCMI Core Laboratory with grant U54MD007585 26 was used to fund microscopic imaging of Figures 1 and ![]() .
.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
