Abstract
Anaplastic large-cell lymphoma or null-cell lymphoma is a clinical entity reported in people, classified according to the unique appearance of large pleomorphic cells that express CD30. Null-cell lymphoma has also been described in dogs when neither CD3 nor CD79α is expressed by the tumor. We describe a case of lymphoma in the dog in which neoplastic cells did not express routine B- or T-lymphocyte markers on flow cytometry or immunohistochemistry; however, cells immunohistochemically labeled for CD30. The dog in our case died 5 mo after initial presentation, confirming a poor prognosis. Identification of further similar cases in dogs would provide additional prognostic information for this subset of lymphomas. CD30 may also serve as a potential therapeutic target in anaplastic large-cell lymphomas.
A 10-y-old male neutered Beagle dog was presented to North Downs Specialist Referrals (Bletchingley, Surrey, UK) with unilateral right-sided mandibular lymphadenopathy. Prior medical history included dorsal laminectomy and disc fenestration of an intervertebral disc extrusion (T13/L1) the preceding year. He had also been diagnosed with sciatic neuritis by histologic examination of a sciatic nerve biopsy specimen 9 mo prior to presentation. The dog had received intermittent treatment with prednisolone and cyclosporine. Three months before presentation, the dog developed severe anemia as a result of gastrointestinal hemorrhage, and prednisolone therapy was stopped.
Hematologic examination at the time of presentation indicated no evidence of cytopenias, and cytologic evaluation of a fine-needle aspirate (FNA) of the enlarged mandibular lymph node was carried out by a board-certified veterinary clinical pathologist (B Szladovits). The nucleated cell population was dominated by medium-to-large lymphocytes (Fig. 1). Cells had mostly round, occasionally indented, and rarely lobed, central-to-paracentral nuclei that were typically 1.5–2× and occasionally ≥3× the size of red blood cells. There was moderate anisokaryosis and a finely granular to mildly clumped chromatin pattern. Nuclei had moderately distinct, small-to-medium–sized nucleoli, and occasional cells had a single large nucleolus. Cells had unusually large amounts of mid-blue cytoplasm with low-to-moderate numbers of small vacuoles, and prominent perinuclear clearing (Golgi zone), commonly around the entire nucleus. The mitotic rate was high (≥3 per five 40× fields). Small lymphocytes were rare; occasional neutrophils, rare eosinophils, and rare plasma cells were present. A diagnosis of lymphoma was considered most likely; however, the cellular morphology was considered unusual given the large amount of cytoplasm within the cells. Thoracic radiography and abdominal ultrasonography were unremarkable. Bone marrow evaluation was not performed, and the patient was diagnosed as having stage 1 lymphoma according to the World Health Organization’s clinical staging system for lymphoma in domestic animals. 4
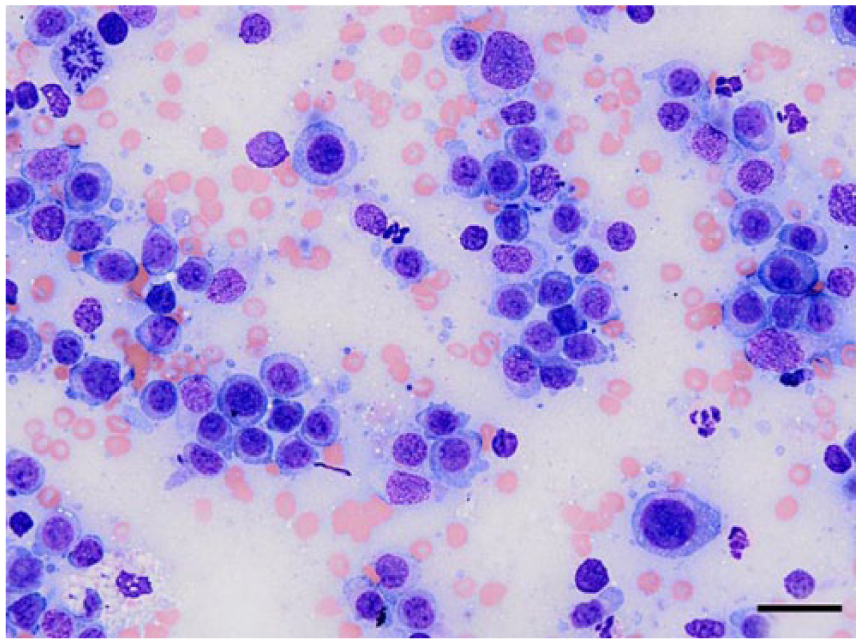
A population of medium-to-large lymphocytes on cytologic evaluation of mandibular lymph node aspirates from a dog. Modified Wright stain. Bar = 20 μm.
Flow cytometric analysis was carried out on the FNA samples of the enlarged lymph node in order to immunophenotype the lymphoma. Samples were stained with canine-specific or cross-reactive fluorochrome-conjugated monoclonal antibodies (mAbs) against both extra- and intracellular antigens (Table 1). Seven-color staining was performed. Cells were stained with mAbs for cell surface antigens for 20 min. Following incubation in buffer (Intracellular fixation and permeabilization buffer, Thermo Fisher Scientific, Paisley, UK) overnight, the cells were stained with mAbs against intracellular antigens for 30 min. All incubations were performed at 4°C in the dark, and cells were washed twice following each of the incubation periods. Flow cytometric data were acquired (FACS Canto II flow cytometer, BD Biosciences, Oxford, UK) and analyzed (Flowjo software, Tree Star, Ashland, OR). All gates were defined by using isotype and “fluorescence minus one” controls. Flow cytometric analysis of viable cells of the FNAs yielded the impression of a predominantly medium-sized population, consistent with the cytologic appearance. The population was negative for all lymphoid antigens, with a high proportional expression of the proliferation marker Ki-67.
Flow cytometry: staining antibodies, isotype controls, and fluorochromes.
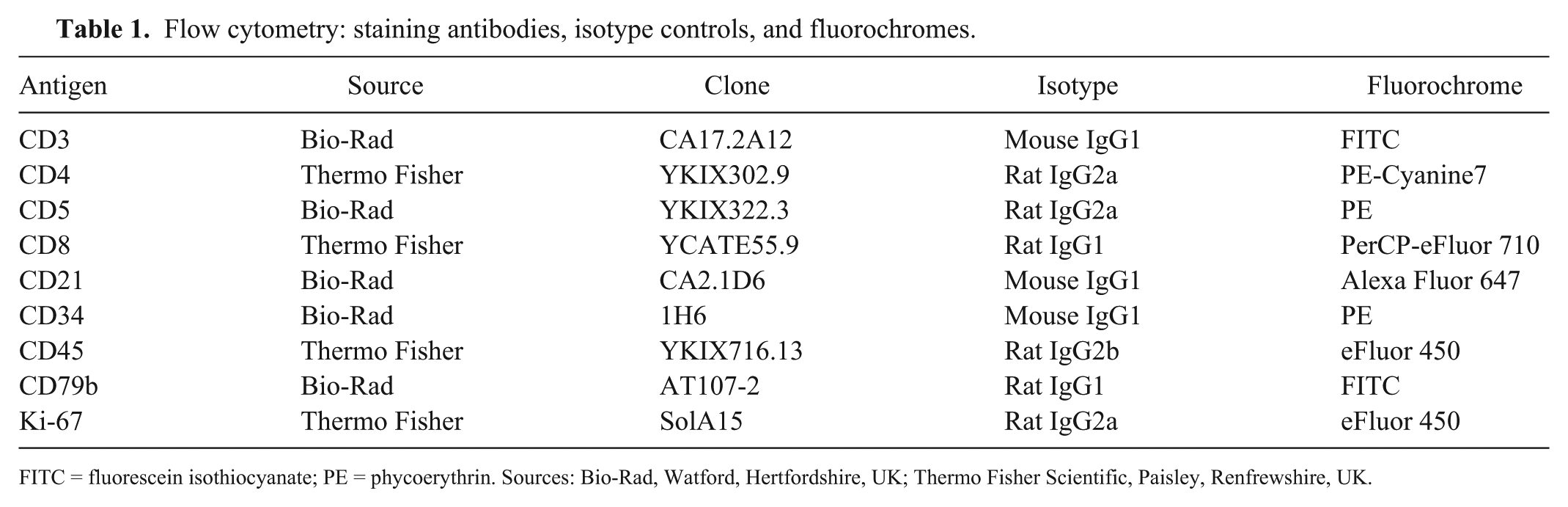
FITC = fluorescein isothiocyanate; PE = phycoerythrin. Sources: Bio-Rad, Watford, Hertfordshire, UK; Thermo Fisher Scientific, Paisley, Renfrewshire, UK.
The enlarged lymph node was surgically excised and, following fixation in 10% neutral-buffered formalin, was processed routinely and embedded in paraffin wax. Sections (4 µm) were stained with hematoxylin and eosin. Histologically, ~90% of the node was effaced by a dense, highly infiltrative, nonencapsulated, poorly differentiated round-cell neoplasm. The neoplastic cells were round-to-oval with distinct cell borders and variable amounts of eosinophilic cytoplasm that usually contained a single, hyperchromatic nucleus that was 2–3× the size of a red blood cell. There was marked anisocytosis and anisokaryosis, including frequent macrokaryosis (Fig. 2). Frequently, some cells exhibited large vesicular nuclei with distinct indentations or binucleation with multiple distinct, magenta, ovoid nucleoli. Fifteen mitotic figures were observed within 10 high-power (400×, 2.37 mm2) fields, including occasional bizarre mitoses. Numerous individual neoplastic cells were either necrotic or apoptotic. There were many fewer small (mature) lymphocytes within the remaining nodal architecture and large central areas of coagulative necrosis (tumor necrosis and/or the result of prior FNA sampling). The capsule was thickened by fibrous connective tissue that was infiltrated by moderate numbers of small (reactive) lymphocytes and small foci of neutrophils. Subcapsular and medullary sinuses were congested or contained low numbers of hemosiderophages. The neoplastic tissue appeared contained by the thickened capsule within the sections examined. A diagnosis of high-grade large-cell lymphoma was given.
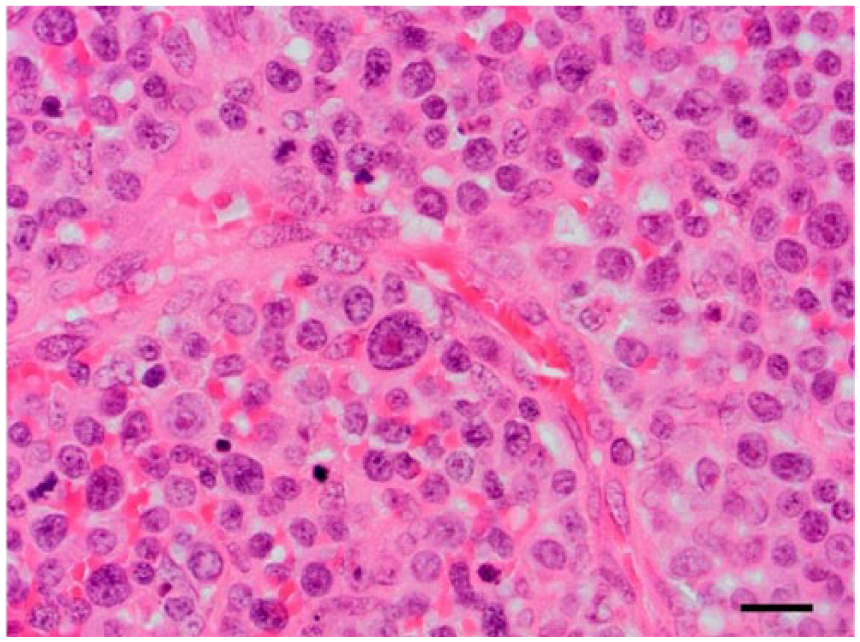
Neoplastic cells within the mandibular lymph node of a dog exhibit marked anisocytosis and anisokaryosis. Nucleoli are prominent, and mitotic figures are present. H&E. Bar = 20 μm.
Immunohistochemistry (IHC) was performed on serial sections (Bond-Max autostainer, Bond polymer refine detection system, Leica Biosystems, Newcastle-upon-Tyne, UK). Primary antisera were specific for CD3 (polyclonal rabbit anti-human, Dako, Ely, UK; 1 in 500 dilution; antigen retrieval in buffer pH 9.0 buffer [ER2, Dako] for 30 min); CD79α (monoclonal mouse anti-human, Dako; 1 in 100 dilution; antigen retrieval in pH 9.0 buffer [ER2] for 10 min); CD18 (mouse anti-canine, clone CA1.4E9, University of California Davis, CA; 1 in 20 dilution; with enzymatic antigen retrieval [Enzyme 1, Dako] for 10 min); and CD30 (monoclonal mouse anti-human, clone Ber-H2, Dako; 1 in 30 dilution; antigen retrieval in pH 9.0 buffer for 20 min). Immunohistochemically, the neoplastic cells diffusely exhibited specific membranous labeling for CD30 (Fig. 3) and did not express CD3, CD79α, or CD18. Negative (Fig. 4) and positive controls were processed with the evaluated slides and were labeled appropriately. A diagnosis of high-grade, poorly differentiated null-cell lymphoma or anaplastic large-cell lymphoma (ALCL) was given.
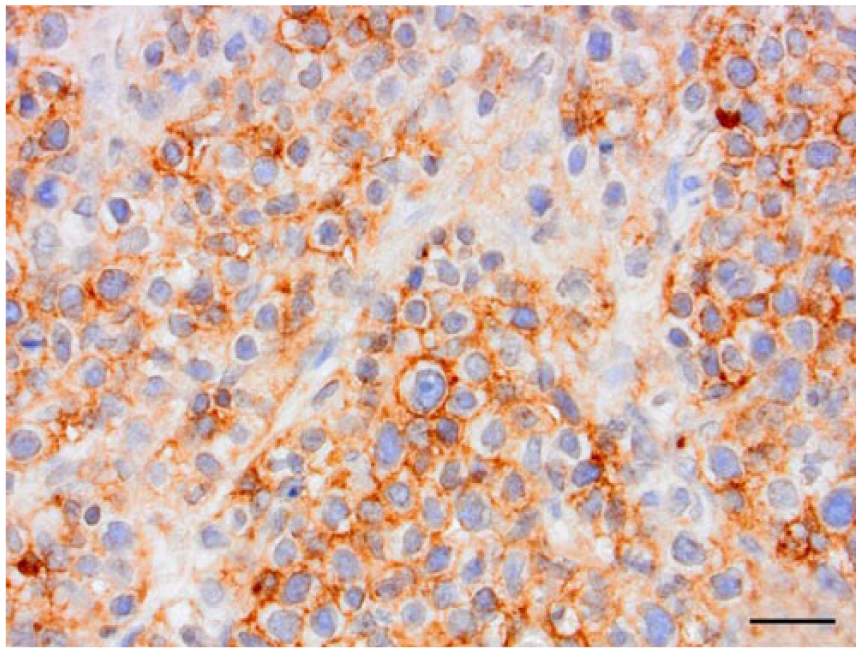
Neoplastic cells within the mandibular lymph node of a dog exhibit specific membranous labeling following CD30 immunohistochemistry. Bar = 20 μm.
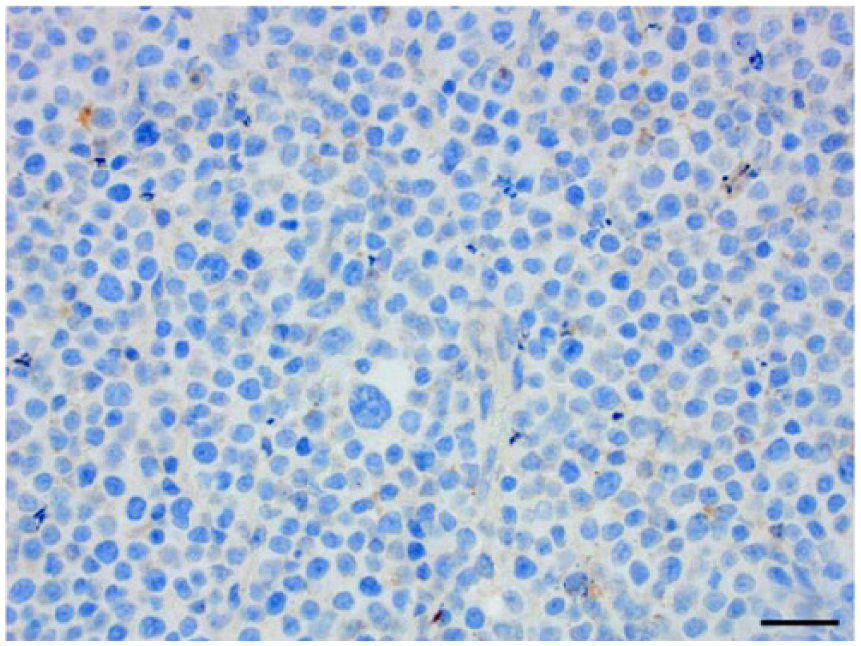
Neoplastic cells within the mandibular lymph node of a dog do not label for CD30 immunohistochemistry in the negative control. Bar = 20 μm.
Two months after lymph node removal, lymphadenopathy was noted in the same anatomic site. Cytologic evaluation confirmed a diagnosis of lymphoma. Blood evaluations, thoracic radiography, and abdominal ultrasonography were repeated. Bone marrow aspirate and biopsy of inflamed gingivae were also performed with no evidence of lymphoma at other sites.
Treatment was implemented with a modified L-CHOP protocol. No measurable improvement was noted in response to single L-asparaginase (400 IU/kg IM; Medac, Wedel, Germany), vincristine (0.7 mg/m2 IV; Hospira UK, Warwickshire, UK), or cyclophosphamide (250 mg/m2 PO; Star Pharmaceuticals, Harrow, UK) treatments, so the protocol was abandoned. Further treatments using doxorubicin (30 mg/m2 IV; Pharmachemie, Haarlem, The Netherlands), lomustine (70 mg/m2 PO; Medac), and chlorambucil (5 mg/m2 q48h PO; Aspen Pharma, Dublin, Ireland) also failed to achieve a beneficial effect. The patient underwent a course of palliative radiation therapy (5× once wk 7 Gy fractions) by which time lymphoma was also noted in the left mandibular and the right prescapular lymph nodes. All detectable lymphoid tissues responded favorably to radiotherapy; however, complete remission was not achieved.
While receiving treatment, a mass arose on the left upper eyelid and, 2 d after completion of the radiotherapy course, the patient showed markedly reduced alertness. The patient was anesthetized for MRI of the head and neck and cerebrospinal fluid analysis, which gave no indication of lymphoma in the central nervous system; however, there were multiple enlarged lymph nodes of the head and neck. The eyelid mass was removed and was composed of neoplastic cells similar to those described within the mandibular lymph node. A plasma cell origin to this neoplasm was also considered at this stage; however, cells did not exhibit immunohistochemical labeling for MUM-1 (performed at an external commercial laboratory; MUM-1/IRF-4 monoclonal mouse anti-human, Dako; 1 in 100 dilution; antigen retrieval in pH 9.0 buffer [Envision FLEX target retrieval solution, Dako] for 30 min). Neoplastic cells exhibited similar expression of CD30 and did not express CD3 or CD79a. The patient failed to make a satisfactory recovery from anesthesia and died 3 d later. Autopsy was not performed.
ALCL was first recognized as a clinical entity in people in 1985 and was classified according to the unique appearance of large pleomorphic cells that express CD30, originally termed Ki-1. 3 ALCL has previously been described in dogs, with 0.8% of 608 cases of canine lymphoma being classified as null-cell type given the lack of expression of either CD3 or CD79α. 6 Further reports of null-cell lymphomas in dogs are rare. The use of CD30 in dogs has been described in a study characterizing testicular neoplasms, in which CD30 was not expressed, 8 and in a case of canine pulmonary lymphomatoid granulomatosis in which a population of cells expressed CD30. 5 CD30 is a transmembrane protein receptor, of the tumor necrosis factor receptor superfamily, which is normally expressed by activated B- or T-lymphocytes. 2 , 7 Upon activation, CD30 influences cell growth and survival. 1
We suggest that in cases in which lymphoma is strongly suspected histologically, despite neoplastic cells failing to express routine T- or B-lymphocyte markers, CD30 may be used to provide a diagnosis of null-cell lymphoma or ALCL. Given positive CD30 expression and the unique appearance of large, pleomorphic neoplastic cells, a diagnosis of ALCL was made. Identification of further cases of ALCL in dogs would provide additional prognostic information for this subset of lymphomas. Therapeutics that target CD30 in people with Hodgkin lymphoma and ALCL have been developed. 1 The dog in our case had a survival time of 5 mo following initial presentation, despite both chemotherapy and radiotherapy, confirming a poor prognosis.
