Abstract
Clostridium perfringens and Clostridioides difficile cause significant morbidity and mortality in foals. Antemortem diagnosis of C. perfringens infection has been complicated by a paucity of tests available for toxin detection. Fecal PCR panels have assays for a variety of C. perfringens toxin gene sequences as well as for several other foal gastrointestinal pathogens. We evaluated results of a comprehensive fecal diarrhea PCR panel in 28 foals that had been presented to a referral hospital because of diarrhea. Sixteen (57%) foals were positive for C. perfringens and/or C. difficile toxin gene sequences on fecal PCR, including 3 foals positive for NetF toxin. These foals were younger (p = 0.0029) and had higher hematocrits (p = 0.0087), hemoglobin (p = 0.0067), and red blood cell concentrations (p = 0.028) than foals with diarrhea that tested negative for clostridial toxins. The foals had lower total protein concentrations (p = 0.045) and were more likely to have band neutrophils on a CBC (p = 0.013; OR: 16.2). All 3 foals with NetF toxin gene sequences detected in feces survived to discharge, indicating that diarrhea caused by NetF toxigenic C. perfringens isolates is not uniformly fatal.
Clostridium perfringens is a cause of necrotizing enterocolitis in foals and horses. Neonatal foals are the most commonly affected age group, frequently being <10 d old; foals within this age range often have the most severe clinical manifestations. The main types of C. perfringens isolates that affect horses are types A and C. Type C infection is associated with beta toxin and causes necrotizing enteritis and hemorrhagic diarrhea; it has been associated with high morbidity and mortality in foals. 1 Type A infections have been less well understood, with uncertainty about the pathogenicity of type A isolates in that they can be found in normal foals as well, whereas type C isolates are rarely found in healthy animals. Alpha toxin is not significantly toxigenic alone, and there is a question about the clinical significance of type A isolates that produce only alpha toxin. Such alpha toxin–only producing isolates can be found in normal foals and horses.7,12 Reports of additional virulence factors in some type A isolates, especially β2 and NetF toxin, have explained the increased virulence of certain type A C. perfringens isolates.3–5
The β2 toxin has been associated with diarrhea and colitis in foals as well as older horses.7,11 Enterotoxin (CPE), a toxin protein commonly associated with disease in humans, is not considered a major toxin in horses. Whereas the C. perfringens beta2 (cpb2) and enterotoxin (cpe) genes and associated toxins have been found in the feces of healthy foals, complicating the interpretation in sick foals, the netF gene has not been identified in healthy newborn foals, to our knowledge. 2 The identification of NetF-producing isolates has explained the virulence of some type A isolates in producing disease in foals. The netF gene is often present in conjunction with the cpb2 and cpe genes, suggesting a possible additive or synergistic pathogenicity role of these other toxins.3–6,15
NetF toxin is the most recently identified virulence factor, and it appears to render type A isolates of C. perfringens quite virulent.3–5 The netF gene was first described in 2015 associated with fatal necrotizing enterocolitis and hemorrhagic gastroenteritis in foals and dogs, respectively.3–5 Prior to that, the role of type A C. perfringens in enteric disease of horses and foals was poorly understood. 14 The publications describing type A, NetF-associated enterocolitis were based on retrospective analyses of banked isolates from foals that had succumbed to the disease. We found no reports of antemortem diagnosis or of survival of foals with enterocolitis associated with NetF-producing isolates in the peer-reviewed literature.
Clostridioides difficile can cause a similar spectrum of diarrhea severity in young foals. Similar to C. perfringens, it can cause hemorrhagic diarrhea and necrotizing enterocolitis. The virulence factors include 2 exotoxins, toxins A (TcdA) and B (TcdB). Coinfection with C. perfringens has been reported. 16 Other causes of foal diarrhea include rotavirus, Salmonella spp., Cryptosporidium sp., as well as Strongyloides westeri parasites. Coronavirus can also cause diarrhea in foals, but it may be more important in coinfections with other pathogens. Agents causing diarrhea in older foals include ascarids, Rhodococcus equi, and Lawsonia intracellularis, and rarely Neorickettsia risticii, the cause of Potomac horse fever.
Our aim was to describe the antemortem diagnosis of NetF infections in foals and the results of comprehensive fecal PCR testing in foals with diarrhea, as well as to document survival of foals with NetF-producing clostridial infections. Our hypothesis was that C. perfringens is a common cause of diarrhea and enteritis in young foals, but that NetF toxin is not necessarily fatal in this age group.
Materials and methods
We performed a retrospective analysis of records from foals ≤30 d old with diarrhea that had a comprehensive fecal PCR panel performed at the William R. Pritchard Veterinary Medical Teaching Hospital (Davis, CA) in 2017–2020. The diarrhea PCR panel consists of analysis for gene sequences for Salmonella spp., C. difficile toxins A and B (reported as C. difficile toxin A and/or B gene sequence present because primers and probes are mixed), C. perfringens and its toxins (alpha [CPA], beta [CPB], β-2 [CPB2], NetF, and enterotoxin [CPE]), rotavirus, coronavirus, Lawsonia intracellularis, and Rhodococcus equi (Table 1).
PCR assays for detection of Clostridium perfringens and toxin gene sequences in foal feces.

Foals were categorized into 2 groups, Clostridial and Other. Foals that tested positive for clostridial toxin gene sequences, either for C. perfringens or C. difficile toxins, comprised the Clostridial group. Foals that were negative for all tested pathogens, positive for other non-clostridial pathogens, or were positive only for C. perfringens antigen (positive for DNA gyraseB gene, but negative for all toxin gene sequences) were placed in the Other group. Data were tested for normality using the Kolmogorov–Smirnov test. Age, vital parameters, and hematologic parameters were compared using an unpaired t-test (for parametric data) or Mann–Whitney U test (for nonparametric data). The presence or absence of band neutrophils on the CBC, as well as age at development of diarrhea, were compared using a Fisher exact test, with calculation of odds ratios (ORs) and 95% CIs. A p ≤ 0.05 was considered statistically significant. The CBCs were obtained using an automated analyzer (Advia 120 and 2120i; Siemens), with manual differential counts.
Results
Twenty-eight foals with diarrhea had the comprehensive diarrhea PCR panel submitted during the timeframe of our study. Results were available within one business day. Sixteen (57%) of the 28 foals were positive for clostridial toxin gene sequences (comprising the Clostridial group); 7 foals had both C. perfringens and C. difficile toxin gene sequences, 6 had only C. perfringens toxin gene sequences, and 3 only C. difficile toxin gene sequences (Table 2). In the Clostridial group, there were 9 colts and 7 fillies. Breeds included Thoroughbreds (7), Quarter Horses (3), and 1 each of Arabian, Andalusian (Pura Raza Española), Haflinger, Gypsy, Appaloosa, and Quarter Horse–Morgan cross.
Results of PCR testing of feces from 16 foals for clostridial and toxin gene sequences.
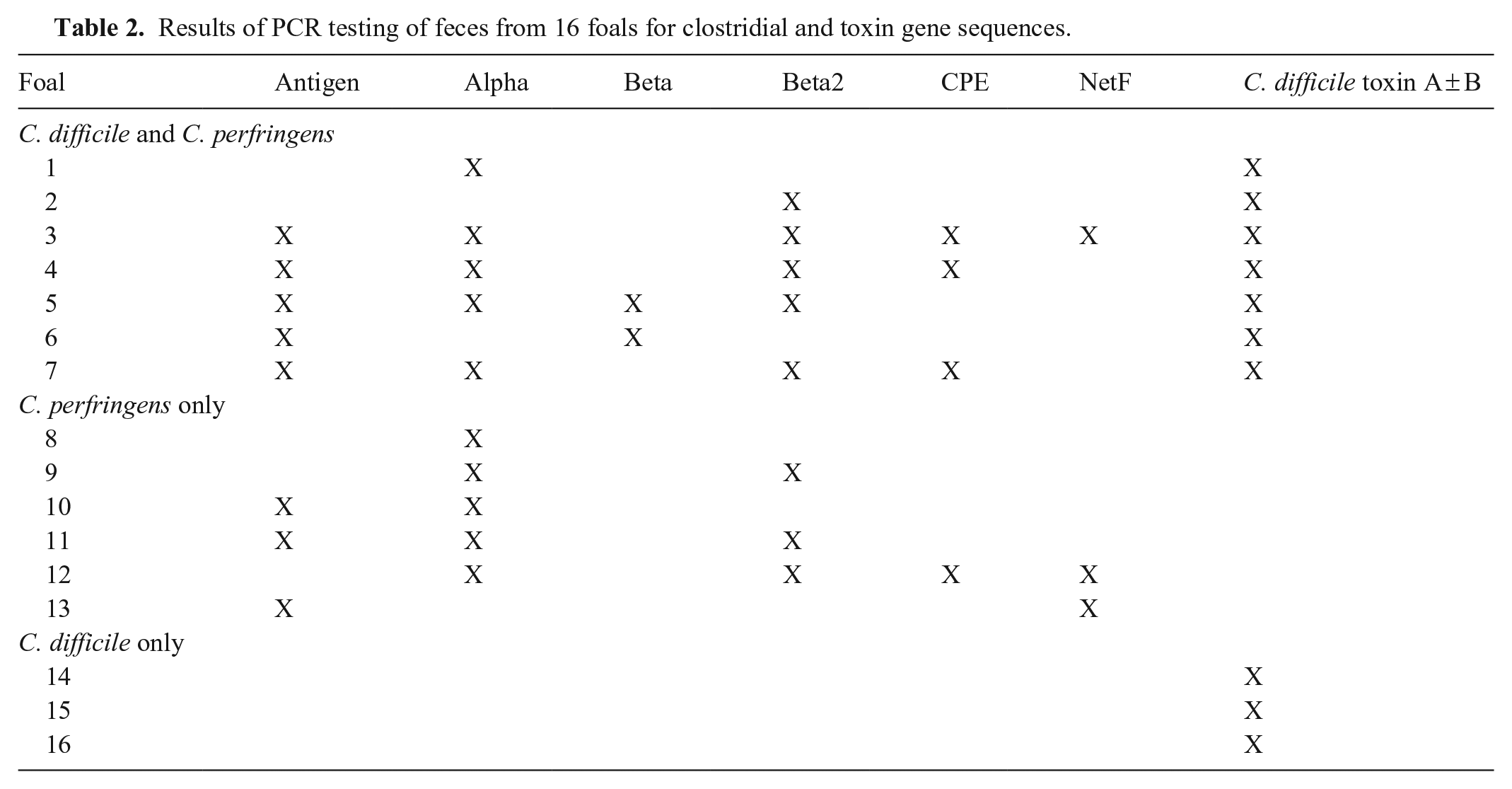
The Other group was comprised of 12 foals: 1 positive for Cryptosporidium sp., 1 for Salmonella spp. and C. perfringens antigen only, 1 for C. perfringens antigen only, and 9 negative for all tested pathogens. In the Other group, there were 3 colts and 9 fillies. Breeds consisted of Thoroughbreds (3), Quarter Horses (2), Arabians (3), Andalusians (2), and 1 each of Hanoverian and Knabstrupper.
Three foals in the Clostridial group were euthanized because of poor prognosis or clinical deterioration. One of these was positive for C. difficile, one was positive for C. difficile and type A C. perfringens (with cpb2 and cpe toxin genes), and one was positive for C. difficile and type C C. perfringens (cpb toxin gene). Two foals in the Other group died, and one was euthanized. The foal that was euthanized had a fatal genetic disease (glycogen branching enzyme deficiency, GBED); one of the foals that died was also found to have GBED. The third foal that died was found to have unspecified neutrophilic enterocolitis and frequent Alzheimer type II cells in the brain, consistent with hyperammonemia. These non-surviving foals were negative for all of the tested pathogen and toxin gene sequences.
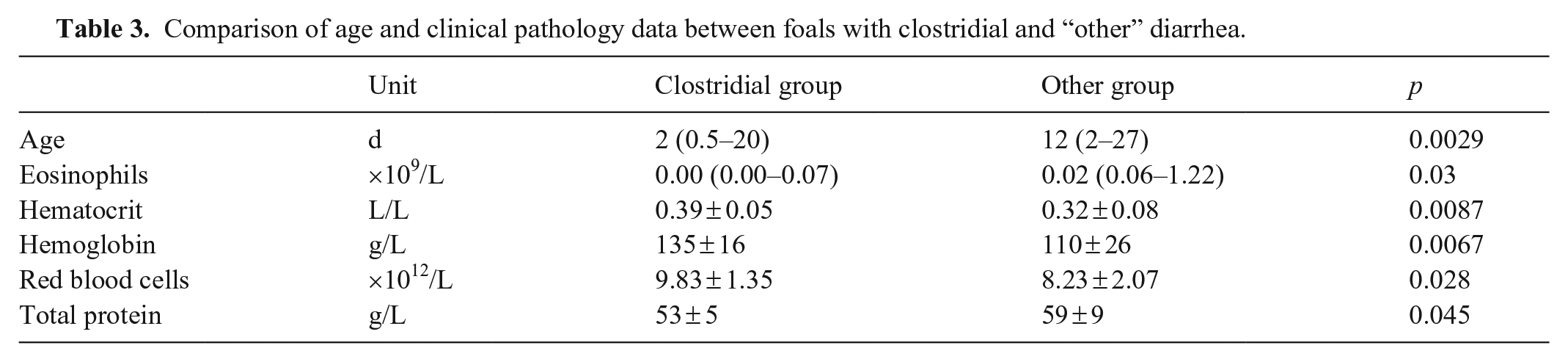
Regarding clinical pathology differences between groups (Table 3), foals with clostridial diarrhea had significantly higher hemoglobin concentration (p < 0.01), RBC count (p = 0.03), and hematocrit (p < 0.01) than those in the non-clostridial (Other) group. In addition, the total protein concentration (p = 0.045) on the CBC was lower, as was the eosinophil count (p = 0.03) in the Clostridial group foals. Foals with clostridial diarrhea were more likely to have a left shift with band neutrophils (p = 0.013; OR = 16.2; 95% CI = 1.56–168). Finally, foals with clostridial diarrhea were younger than those with non-clostridial diarrhea (p = 0.0029). Fourteen of the 16 foals in the Clostridial group were ≤5 d old, whereas only 3 of 12 foals in the Other group were ≤5 d old. There were no significant differences between groups in heart rate, respiratory rate, or any other CBC findings. Thirteen of 14 foals with recorded blood IgG concentrations had adequate passive transfer (>8 g/L) in the Clostridial group, and 6 of 7 with recorded values in the Other group had an IgG >8 g/L. Most of these IgG results had been determined through a stall-side immunoassay (SNAP; Idexx).
Comparison of age and clinical pathology data between foals with clostridial and “other” diarrhea.
Discussion
The comprehensive fecal PCR panel provided a means of identifying clostridial infections rapidly, as well as Cryptosporidium and Salmonella, in the feces of young foals, allowing early diagnosis and treatment. Foals with clostridial infections were younger, had greater evidence of hemoconcentration, lower plasma total protein concentrations, and had a greater OR of having band neutrophils on a CBC than did foals that tested negative for clostridial toxin gene sequences.
Approximately 57% of foals with diarrhea that had been presented to this referral clinic had clostridial infections based on the fecal PCR results for toxin gene sequences. Of these 16 foals, 6 had C. perfringens only, 3 had C. difficile only, and 7 had coinfections. The foals with C. perfringens toxin gene sequences detected in feces included those with cpa (alpha toxin gene), cpb (beta toxin gene), cpb2 (β2 toxin gene), cpe (CPE toxin gene), and netF (NetF toxin gene), alone or in a variety of combinations. Coinfections of C. difficile and C. perfringens have been reported previously.9,16 In our study, 7 of 16 foals were positive for toxin gene sequences of both microbes.
The netF gene was identified in 11 of 15 isolates from foals with fatal necrotizing enterocolitis in the original publication describing its role in foals. 3 The NetF toxin is part of the leukocidin/hemolysis superfamily, to which Staphylococcus aureus α-toxin, as well as CPB and the NetB toxins, belong.3–5 These are pore-forming toxins, which are potent cytolytic agents. They protect the microbes against cell-mediated immunity, disrupt epithelial barriers in the gastrointestinal tract, and liberate nutrients for growth of the microbe.
The CPE toxin may have synergistic or contributory effects in disease caused by C. perfringens in foals. In a study evaluating type C C. perfringens enteritis in humans, purified CPB and CPE showed synergistic interactions in causing histologic damage and fluid accumulation in rabbit epithelial cells. 8 In the original study describing NetF as a toxin in foals, the cpe gene was present in all of the netF-positive strains, suggesting it may contribute to the disease in foals, as well.3–5 However, in one of the foals of our study, cpe was not detected, whereas netF was.
The foals in the Clostridial group of our study were younger than those in the Other group, and most were ≤5 d old. This is consistent with prior reports of clostridial enterocolitis in foals, because of the hypothesis that colostral trypsin inhibitors protect the toxins from degradation by trypsin in the stomach.10,13
The foals in the Clostridial group had a greater degree of hemoconcentration than those in the Other group, as indicated by a higher hemoglobin concentration, red cell mass, and hematocrit. They also had a lower total protein concentration and were more likely to have a left shift on the CBC. The lower eosinophil count may be the result of a stress response and endogenous corticosteroids. Taken together, all of these findings are suggestive of more severe disease in the Clostridial group as indicated by a greater degree of dehydration, protein loss, and systemic inflammation. Despite this indication of more severe disease, there was no difference in mortality rate between the 2 groups. The majority of foals in the Clostridial group (13 of 14 with recorded results) had an IgG concentration >8 g/L, consistent with adequate passive transfer. This has been a consistent finding among foals with C. perfringens infections, given that trypsin inhibitors in colostrum are believed to leave the clostridial toxins intact as they enter the small intestine, just as trypsin inhibitors in the diet (e.g., sweet potato) protect beta toxin in the pathogenesis of human enteritis necroticans.10,13
It is interesting to note that all 3 foals with netF found in their fecal samples survived to discharge. To date, all of the reports of foals with NetF-producing C. perfringens infections had necrotizing enterocolitis and were fatal, although these were postmortem studies, which explains the associated mortality.3–5 Our study indicates that diarrheic foals testing positive for netF can survive the disease if given intensive care. Further study of NetF-producing C. perfringens in foals is warranted, including age-associated susceptibility and factors associated with morbidity and mortality.
Limitations of our study include the relatively low number of foals within groups, reflecting the relatively recent use of PCR for detection of netF gene sequences. Also, we evaluated the results of fecal PCR testing only; it would have been optimal to have fecal ELISA performed; however, there are currently no commercial laboratories offering such assays for NetF or β2 toxins. Finally, a control population of healthy, non-diarrheic foals would have been optimal.
Footnotes
Acknowledgements
We acknowledge the veterinary and laboratory staff at the William R. Pritchard Veterinary Medical Teaching Hospital and the Lucy Whittier Molecular Core Facility, School of Veterinary Medicine, University of California, Davis.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our study was supported by the Center for Equine Health at UC Davis with contributions from the Oak Tree Racing Association and State of California pari-mutuel wagering fund, as well as the Henry Endowed Chair in Emergency Medicine and Critical Care at UC Davis.
