Abstract
Gas gangrene is a necrotizing infection of subcutaneous tissue and muscle that affects mainly ruminants and horses, but also other domestic and wild mammals. Clostridium chauvoei, C. septicum, C. novyi type A, C. perfringens type A, and C. sordellii are the etiologic agents of this disease, acting singly or in combination. Although a presumptive diagnosis of gas gangrene can be established based on clinical history, clinical signs, and gross and microscopic changes, identification of the clostridia involved is required for confirmatory diagnosis. Gross and microscopic lesions are, however, highly suggestive of the disease. Although the disease has a worldwide distribution and can cause significant economic losses, the literature is limited mostly to case reports. Thus, we have reviewed the current knowledge of gas gangrene in mammals.
Introduction
Several bacteria of the genus Clostridium are known in human and veterinary medicine because of their ability to cause diseases through the production of powerful exotoxins. 30 Among these diseases, gas gangrene (previously referred to as malignant edema) is a highly lethal histotoxic disease that affects several animal species with worldwide distribution.65,70,78
Although gas gangrene is most frequently observed in sheep, cattle (Figs. 1, 2), and horses (Fig. 3),20,53,65,70 it has also been described in other domestic mammals, including goats,27,30 pigs (Fig. 4), 54 dogs, and cats.53,56,62 Individual cases of gas gangrene have also been described in several wild animal species including an Indian elephant (Elephas maximus), 59 a guanaco (Lama pacos), 78 a lynx (Lynx canadensis), 33 and a brown bear (Ursus arctos). 10 Cases have also been described in a marmoset (Callithrix jacchus) 84 and a rhesus macaque (Macaca mulatta). 42 No age predisposition has been described in any animal species, except for cases of omphalophlebitis, which occur in neonatal animals.44,51
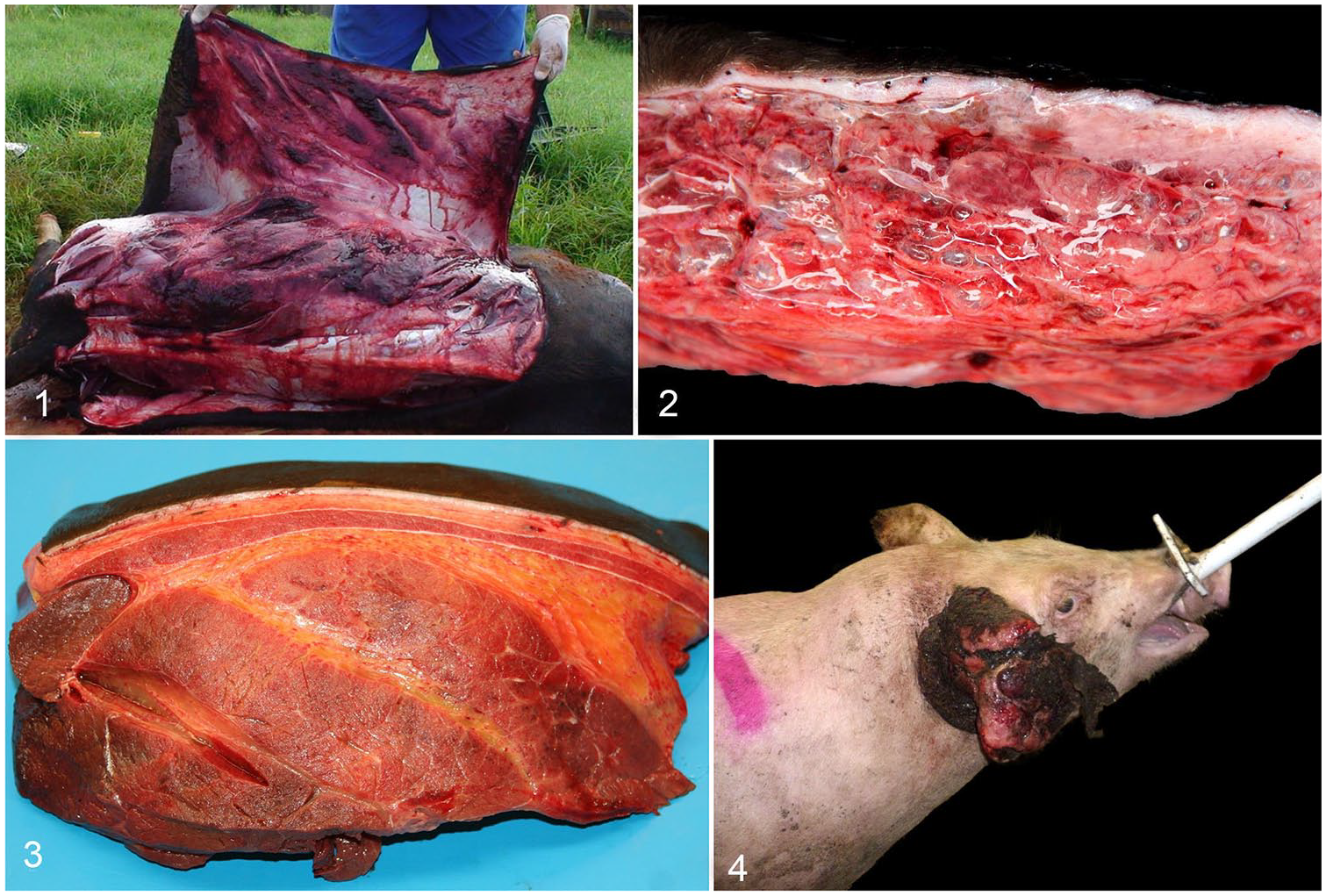
Gas gangrene in cattle, horse, and pig. Figures 1, 2. Gas gangrene caused by Clostridium septicum in a heifer.
Despite the undeniable importance of gas gangrene in mammals, information regarding this disease is dispersed and mostly in the form of case reports. We review and discuss herein the most relevant aspects of gas gangrene in mammals.
Etiology
Gas gangrene can be caused by one or more of the following clostridial species: Clostridium septicum, C. chauvoei, C. novyi type A, C. perfringens type A, and C. sordellii.19,53,70 These agents are widespread in the environment, and some of them are frequently found as part of the intestinal microbiota of many animal species.29,68,74,80,81 All of these clostridial species can also be frequent postmortem invaders, 74 and their presence should be interpreted with caution in carcasses because invasion of tissues from the intestine begins shortly after death or during the last minutes of life. 52 These 5 clostridial species exert their effect via the production of powerful toxins (Table 1).57,58
Gene location, molecular weight, and mode of action of the main clostridial toxins responsible for gas gangrene in animals.

GTPase = guanosine triphosphatase.
Clostridium septicum
The involvement of C. septicum in gas gangrene was confirmed in 1881 by Robert Koch.31,67 C. septicum produces 4 major toxins, (i.e., alpha, beta, delta, and gamma), but alpha-toxin, a lethal and necrotizing pore-forming cytolysin, is recognized as the factor that is essential for the virulence of this microorganism. 35 This toxin uses glycosyl-phosphatidylinositol–anchored proteins on the membrane of target cells as the receptor.34,57 After proteolytic activation, many activated monomers form a transmembrane pore that has lytic and vacuolating properties.28,34 Gamma-toxin, a hyaluronidase, might play a role in the spreading of bacteria or their toxins through tissues. 34 If this is the case, this toxin would be at least partially responsible for the hallmark lesions of gas gangrene, which are edema and hemorrhage. 57 The roles of the other toxins of C. septicum in the virulence of this microorganism are largely unknown.
C. septicum is widespread in the environment, and it is considered by some authors as the main cause of gas gangrene in ruminants,52,70 probably because it is frequently isolated in pure culture from tissues of animals with this disease. This, however, may just represent the capacity of this organism to overgrow other histotoxic clostridia when they act together, given that C. septicum grows very quickly.11,12,29,44,68 Nevertheless, several cases of gas gangrene in which only C. septicum was detected by fluorescent antibody (FA) testing or immunohistochemistry (IHC) seem to contradict the above statement.61,62,78,81 FA testing and IHC provide a snapshot of the relative proportion of different clostridial species present in animal tissues and are not affected by the presence of other microorganisms. 8
Clostridium chauvoei
C. chauvoei is commonly recognized as the cause of blackleg, a so-called “endogenous” myonecrosis that affects mainly cattle 6- to 24-mo-old.2,12,76 However, this microorganism is also commonly associated with gas gangrene of ruminants, usually acting in combination with other histotoxic clostridia.6,38,70
C. chauvoei produces several potent exotoxins, but toxin A (CctA), a pore-forming toxin of the leucocidin toxin superfamily, is the main virulence factor. 25 Other toxins, such as the neuraminidase of C. chauvoei, are thought to play an important role in spreading C. chauvoei in the tissues of infected animals. 77 C. chauvoei is able to survive in bovine macrophages, a fact that is considered critical for the pathogenesis of blackleg, which is thought to involve activation of spores dormant in macrophages when conditions of reduced redox potential occur. 55 However, in most cases of gas gangrene associated with C. chauvoei, the pathogenesis of the disease is believed to be exogenous; that is, the microorganism or its spores gain entry into the tissues via cutaneous or mucosal wounds.6,46 Although it is possible that an endogenous mechanism also exists for other clostridia, particularly in cases of gas gangrene in non-human primates, 84 this has not been confirmed.
Clostridium perfringens type A
C. perfringens, previously known as Clostridium plagarum and Clostridium welchii,47,72 is commonly found in soil and microbiota of many healthy mammals; its role as an enteropathogen has not been clarified fully.71,81 C. perfringens is classified into 7 types according to the production of 6 so-called major typing toxins, namely alpha, beta, epsilon, iota, enterotoxin, and necrotic enteritis–like beta. 63 Of these, C. perfringens type A produces only alpha-toxin (CPA), which has been proven to be essential for the pathogenesis of gas gangrene in humans. 9 Although there is ample evidence of the significant role of CPA in human gas gangrene, the same does not hold true for gas gangrene of other mammals, and the role of CPA in animal gas gangrene remains speculative. CPA is a phospholipase C with sphingomyelinase activity that affects host signaling, contributing to tissue necrosis, thrombosis, and reduction or absence of leukocyte influx into the infection site.49,80 Because these changes are seen in cases of animal gas gangrene associated with C. perfringens type A, it is likely that, in those cases, CPA is an important virulence factor for animal gas gangrene. Perfringolysin O (PFO), a pore-forming toxin, is not essential for gas gangrene in humans, but in mice inoculated experimentally with C. perfringens type A, it acts synergistically with CPA, causing macrophage cytotoxicity at the beginning of the infection, coupled with thrombosis and a decrease in inflammatory cell influx at later stages of the infection. 82 Although there is no information available on the role of this toxin in spontaneous cases of animal gas gangrene, it is possible that PFO also plays a role in some of these cases.
C. perfringens type A seems to be more prevalent in human than in animal cases of gas gangrene. 37 Among domestic mammals, C. perfringens type A gas gangrene has been described in goats, sheep, cattle, pigs, and horses. 70 This microorganism was the most common cause of gas gangrene in 1 study of 37 horses. 53 The pathogenesis of C. perfringens type A in gas gangrene of ruminants has not been thoroughly studied, although in most cases in which this microorganism produces gas gangrene, other histotoxic clostridia, including C. septicum, C. chauvoei, and others, are involved. 70
Clostridium sordellii
C. sordellii was first isolated in 1922 by Alfredo Sordelli from an acute edematous wound infection of a human patient and was initially named Bacillus oedematis sporogenes.4,73 C. sordellii produces at least 5 toxins, 2 of which are essential for virulence, namely lethal toxin (TcsL) and hemorrhagic toxin (TcsH).21,83 These 2 toxins are closely related to Clostridium difficile toxins A (TcdA) and B (TcdB), respectively.7,30 TcsL and TcsH have glucosylation (UDP-glucose) activity, and their main targets are endothelial cells. 58 Both toxins bind to as-yet uncharacterized receptors on endothelial cells, where they are internalized and cause disruption of the cytoskeleton. The result is loss of cell adhesion, which leads to hemorrhage and edema with a drop in blood pressure. 7 This effect at the cellular level explains, at least in part, the severe edema and hemorrhage in most cases of C. sordellii–associated disease. 65 C. sordellii can cause gas gangrene in several domestic and wild mammals.10,20,24,39,43,51,65
Clostridium novyi type A
C. novyi type A, originally named Bacillus oedematis number 2, 72 was isolated in 1894 by Frederick Novy, from lesions of guinea pigs that had died after injections of casein. 30 This microorganism is commonly found in soil and rarely in the microbiome of healthy animals.11,29,41,48 C. novyi type A produces alpha-toxin, a large glucosylating toxin with necrotizing and lethal activity, which is considered the main virulence factor of this microorganism. 9 Similar to TcsL and TcsH of C. sordellii, the alpha-toxin of C. novyi type A acts on endothelial cells, causing loss of cell-to-cell contact, which leads to edema, followed by a drop in blood pressure, multi-organ failure, and death.7,58 C. novyi type A also produces novylysin, a cytolysin that damages cell membranes.7,58
Cases of gas gangrene caused by C. novyi type A in humans linked to the use of injectable drugs are common.41,64 In domestic mammals, reports of gas gangrene caused by C. novyi type A are scarce, with a few reports in horses, cattle, and sheep.23,43 An intra-abdominal abscess caused by this agent in a horse has been reported. 5 In this case, it was speculated but never proven, that the horse picked up C. novyi from a pasture in which cattle had been grazing. 5 However, the presence of C. novyi in those cattle was not investigated, and the suggestion that they could have been the origin of the equine infection is purely speculative.
Epidemiology
All of the etiologic agents of gas gangrene described above are widespread; most of them can be found, albeit with different prevalences, in the intestinal content of animals and in the environment, including fresh water and soil.29,81 The spores are found more often in wet soils rich in organic matter, and regions where the soils are seasonally flooded are more prone to be contaminated by some of these microorganisms than are dry soils. 52 The use of treated wastewater for irrigation of pastures is a factor that can increase environmental contamination because most spores are not destroyed during the treatment of contaminated water. 32 Because several clostridial species responsible for gas gangrene are present in the intestines of wild and domestic animals,4,70 the presence of these animals on pastures is likely to also increase soil contamination. In addition, earth movement associated with construction or other human activities may also increase the risk of infection by some of the clostridial species responsible for gas gangrene.11,45
The occurrence of gas gangrene is dependent on close contact between the clostridial species involved and animals, typically contamination of wounds after vaccination, parturition, shearing, marking, neutering, docking, bleeding, and other traumatic interventions.20,43,50,68 A lack of, or a suboptimal, vaccination program, idiopathic individual lack of immune response, or high challenge doses can also contribute to the occurrence of gas gangrene.20,45 The disease generally occurs sporadically, but outbreaks have been reported in association with the injection of contaminated products or the use of contaminated needles.14,18,20
In cattle and sheep, cases of gas gangrene are more common in extensive production systems, and C. septicum is the most common etiologic agent.45,50 In both host species, outbreaks involving several animals are not unusual, and the main risk factor in these cases is vaccination without proper asepsis, although cases associated with other veterinary interventions43,52 and parturition 50 are also common.
In horses, C. perfringens type A and C. septicum are the most common causes of gas gangrene.14,53 C. sordellii, which was previously associated with multiple cases of omphalitis in foals, 51 has recently been identified as a significant cause of gas gangrene in horses (Fig. 2). 65 Although C. sordellii–associated omphalitis was described in male and female newborn foals, 51 no association between other forms of gas gangrene and age or sex of horses has been identified. However, in a study of 37 cases of equine gas gangrene, 43% of cases occurred in Quarter Horses, 53 a heavily muscled breed that is predisposed to several muscle diseases. 69 Based on this, it was suggested that Quarter Horse individuals may be more predisposed to gas gangrene than other breeds. 53 This, however, is speculative and has not been proved. Intramuscular injections seem to be the most common antecedent of gas gangrene in these horses.14,23,53,65
Pathogenesis
Contamination of wounds with spores or vegetative forms of histotoxic clostridia represents the starting point of most cases of gas gangrene.20,43,78 Low redox potential, metabolites of decomposing protein, and acid pH promote germination of spores and stimulate proliferation of vegetative forms of clostridia. 73 This is followed by the production of toxins (Table 1), which are ultimately responsible for the lesions and clinical signs characteristic of gas gangrene.
Cases have been described in which no evidence of wounds was found, which lend credence to the suggestion that some cases of gas gangrene, notably in non-human primates 84 and rarely in horses,16,53 may have a pathogenesis similar to that of blackleg. In blackleg cases, spores of C. chauvoei are ingested and, after one or more cycles of reproduction in the intestine, are absorbed into the systemic circulation through which they reach several tissues, including cardiac and skeletal muscle, where they stay dormant for variable periods of time, sometimes years. When blunt trauma or other injuries that do not produce skin or mucosal wounds occur and produce a reduction of the redox potential, the spores germinate, proliferate, and produce the toxins that are responsible for the disease.2,3 A similar pathogenesis, although suggested,1,16,53,84 has not been proven for other clostridial species.
Most clostridial toxins involved in cases of gas gangrene act first on endothelial cells, producing circulatory alterations and, consequently, edema, hemorrhage, ischemia, and local necrosis.4,65 This environment provides the ideal conditions for survival and multiplication of these microorganisms and the production of more toxins. 7 In addition, several enzymes produced by histotoxic clostridia, including collagenases, DNases, hyaluronidases, and neuraminidases also contribute to initial evasion of host defenses, tissue damage, and nutrient acquisition, allowing the spread of the infection.7,30,58 When the toxins gain access to the blood circulation, toxemia ensues, followed by shock and death.7,58 Bacteremia is also common, with the agents of gas gangrene being found in blood and several organs. 52 In cases in which C. perfringens type A is involved, intravascular hemolysis may also occur as a result of the highly hemolytic nature of CPA and PFO. 80
Clinical signs
Depression, tachycardia, respiratory distress, muscle tremors, anorexia, and fever are clinical signs often seen in cases of gas gangrene.6,19,56 Within a few hours after infection, the inoculation site and adjacent tissues are swollen, erythematous, painful, and hot. As the infection progresses, there is increased swelling because of subcutaneous edema and emphysema, the latter becoming evident as crepitation during palpation. If the lesions are in the limbs, this usually leads to reluctance to move, lameness, and eventually recumbency.18,20,23 The skin is usually taut and diffusely red or black as dermal necrosis ensues. 19 In later stages of the disease, the affected areas become cold. 52 In most cases, death occurs as a consequence of toxemia and shock between a few hours to 3 d after the onset of clinical signs. Rare cases can have a clinical course of 30 d or more.18,23,43 Occasionally, animals die without showing any clinical signs. 52
In post-parturient gas gangrene, a form of necrotizing vulvovaginitis and metritis after parturition in heifers, vulvar swelling is the most common clinical sign. It starts 1–3 d after calving, accompanied by fever, depression, recumbency, discharge of red-brown fluid and, finally, death. 50 In these cases, it has been postulated that trauma associated with parturition produces wounds in the birth canal, which are the portal of entry for C. septicum. 50
Gross changes
The main gross changes associated with gas gangrene are diffuse, blood-stained, and gelatinous subcutaneous edema and emphysema (Figs. 1–3).19,23,56 Underlying muscles frequently have petechiae, ecchymoses, and/or multifocal-to-coalescing dark-red, gray, or blue discoloration indicating areas of necrosis with or without edema and gas bubbles (Figs. 2, 3).6,19,43 Serosal and subendocardial hemorrhages, and congested and edematous regional lymph nodes, spleen, lungs, and liver can be observed as a consequence of the severe toxemia.18,19,53,65 Gross findings are similar regardless of the clostridial species involved.52,70
In post-parturient gas gangrene of cattle, diffuse, gelatinous, and hemorrhagic perineal and perivaginal edema is common, which sometimes extends to adjacent musculature (Fig. 5). Multifocal necrosis and ulceration are observed in vulvar, vaginal, and uterine mucosae, which may be covered by a fibrinous pseudomembrane; systemic lesions are similar to those described above. 50
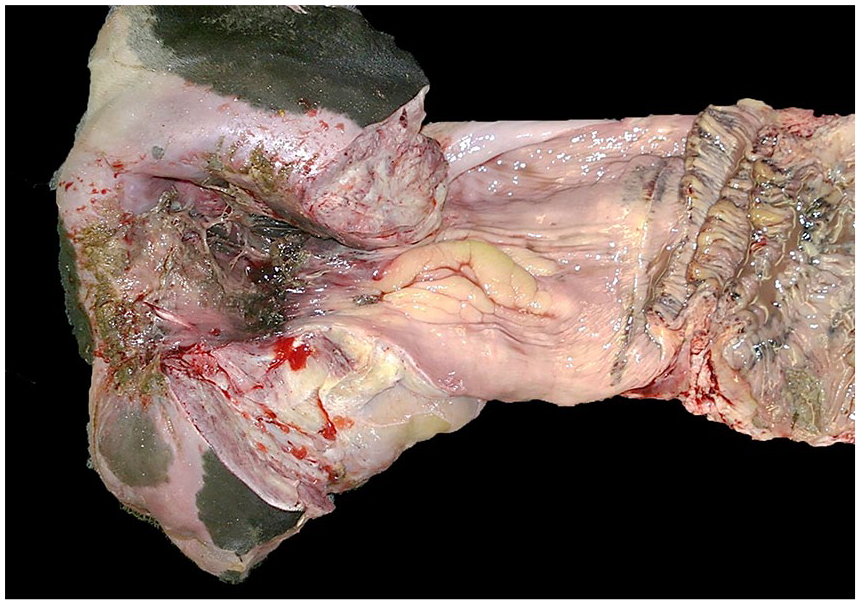
Postpartum gas gangrene in a heifer. Note mucosal tears covered by inflammatory exudate in vulva and vagina. Reproduced with permission. 50
In cases of clostridial omphalophlebitis in foals, yellow gelatinous edema is usually found in the parietal peritoneum, subcutaneous tissue, and abdominal muscles around the internal umbilical remnant, which is swollen and hemorrhagic. An excess of blood-stained and odoriferous peritoneal fluid is also common. Petechiae and ecchymoses are observed in abdominal serosae. 51
Microscopic changes
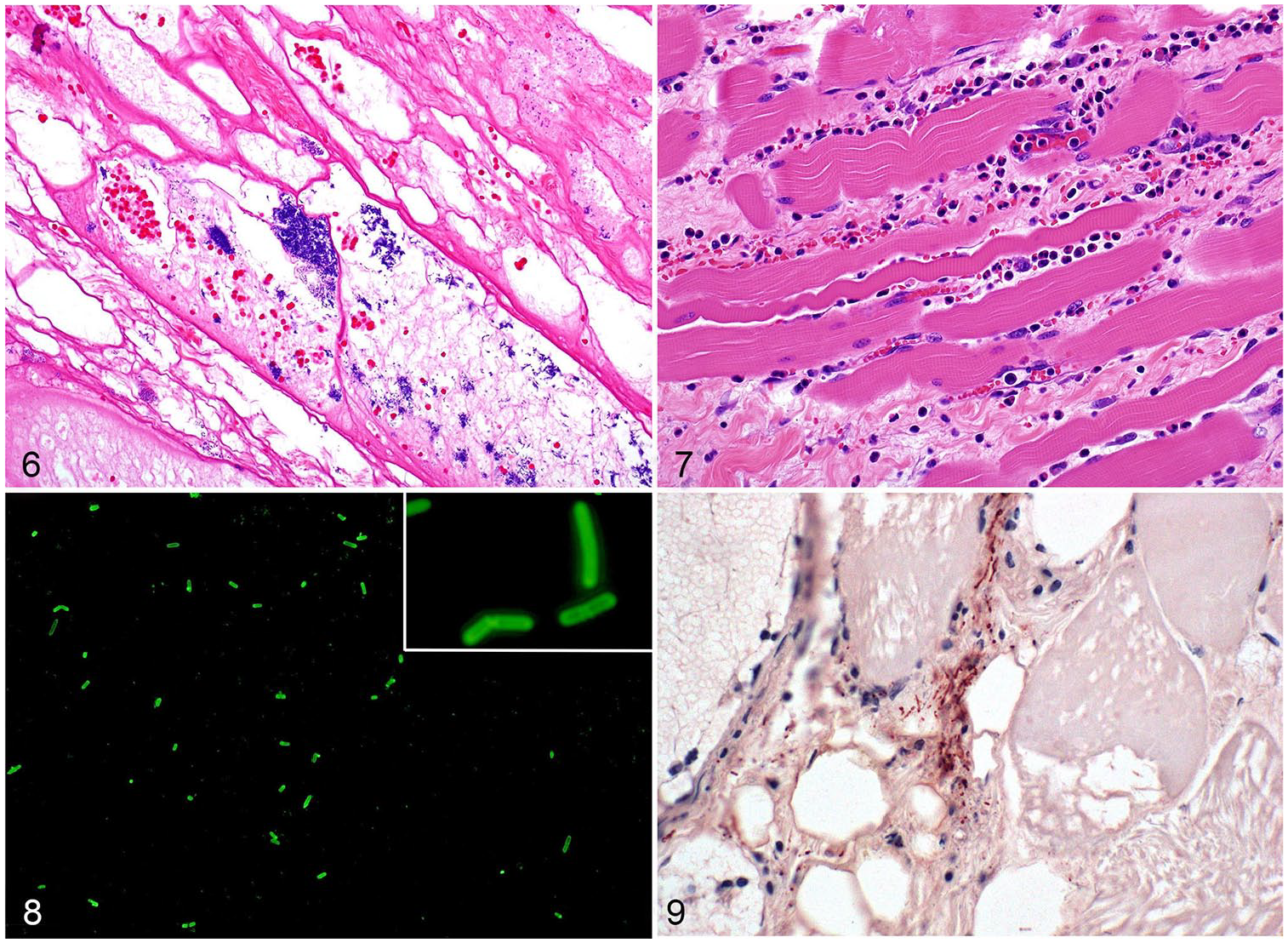
The subcutaneous tissue has severe proteinaceous edema and hemorrhage (Fig. 6). Vasculitis with fibrinoid necrosis and thrombosis is a very frequent finding.10,18 The skin has diffuse congestion, hemorrhage, and emphysema, and in later stages, may show coagulative necrosis. 61 Skeletal myofibers are swollen, hypereosinophilic, and have a loss of striations, vacuolation, and hypercontraction bands (Fig. 7).23,44,61,65 Severe interstitial hemorrhage and edema can be observed. 56 All affected tissues may have minimal-to-severe leukocyte infiltration composed mostly by neutrophils, many of them degenerate (Fig. 7). Abundant gram-positive bacilli with mostly sub-terminal spores, single or in clusters, are present within areas of edema or hemorrhage and rarely within degenerate muscle fibers (Fig. 6).4,43,61 Congestion and hemorrhage of liver and lungs, and medullary hemorrhage and necrosis of thymus and regional lymph nodes, may also be seen.18,36,43
Gas gangrene produced by Clostridium septicum in a heifer.
In post-parturient gas gangrene, multifocal necrosis and ulcers are observed in vulvar, vaginal, and uterine mucosae. Perivulvar and systemic tissues have lesions similar to those described above for other cases of gas gangrene.36,50
In cases of clostridial omphalophlebitis, arteritis is observed in umbilical arteries, and the urachus has transmural edema, hemorrhage, and mixed inflammatory cell infiltrates. Diffuse hemorrhagic and gelatinous edema and congestion are seen in all tissues around the navel.44,51
Absence of significant inflammatory response is a characteristic of cases of human C. perfringens–associated gas gangrene. 15 This is the result, in part, of impaired neutrophil mobility caused by the action of alpha-toxin of C. perfringens. 15 A similar lack of inflammatory infiltrate has been described in some, but not all, cases of animal gangrene, produced by several clostridial species,44,50 and it is possible that other clostridial toxins have a similar leukocytoclastic effect to that seen with C. perfringens alpha-toxin, but this has not been proved.
Diagnosis
A presumptive antemortem diagnosis is based on history and clinical signs. Confirmation of the diagnosis in the live animal may be achieved by extraction of subcutaneous exudate, which can be examined by Gram, FA test (Fig. 7), or when the course of the disease allows it, bacterial culture, and/or PCR.26,52 When outbreaks occur, the diagnosis is usually based on postmortem examination and diagnostic workup on one or more affected animals.52,70
A presumptive postmortem diagnosis of gas gangrene can be established based on clinical history, including clinical signs, and gross and microscopic changes (Figs. 6, 7). 70 Laboratory tests are, however, essential for confirmatory diagnosis, which should be based on identification of the clostridia involved. Muscle, subcutaneous tissue, and/or fluid from grossly affected areas of recently dead animals should be submitted to the laboratory. These samples should be collected in sterile containers and submitted fresh, refrigerated, or frozen (Table 2), for anaerobic culture and/or PCR.17,23,26,62,65,66 Rapid identification of several clostridia involved in gas gangrene may be achieved by targeting the flagellin gene (fliC) of C. novyi type A, C. septicum, and C. chauvoei by direct PCR on clostridial isolates or DNA extracted from affected tissues. 66 However, failure to amplify certain virulence genes should be interpreted with caution because some of these genes are encoded in plasmids that may be lost upon subculture (e.g., tcsL and tcsH genes of C. sordellii).21,83 Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry is also useful in identification of isolated clostridia. 40 Air-dried smears of grossly affected tissues should be submitted for a FA test (Fig. 8). 70 Fluorescent antibodies have been used, albeit with variable efficacy, to identify several clostridial species in formalin-fixed (FF) tissues. 79 The low sensitivity for FF tissue is a result of the fact that the routinely used indirect fluorescent technique provides minimal amplification of the signal on tissues. 75 IHC is also useful in the identification of several clostridial species in FF tissues. This technique has the advantage of correlating the presence of the microorganisms with microscopic lesions (Fig. 9), and, because most modern IHC systems amplify the signal significantly, the sensitivity of these tests is usually much higher than that of the FA test routinely used for clostridial disease diagnosis.8,60
Main detection techniques for gas gangrene in mammals.
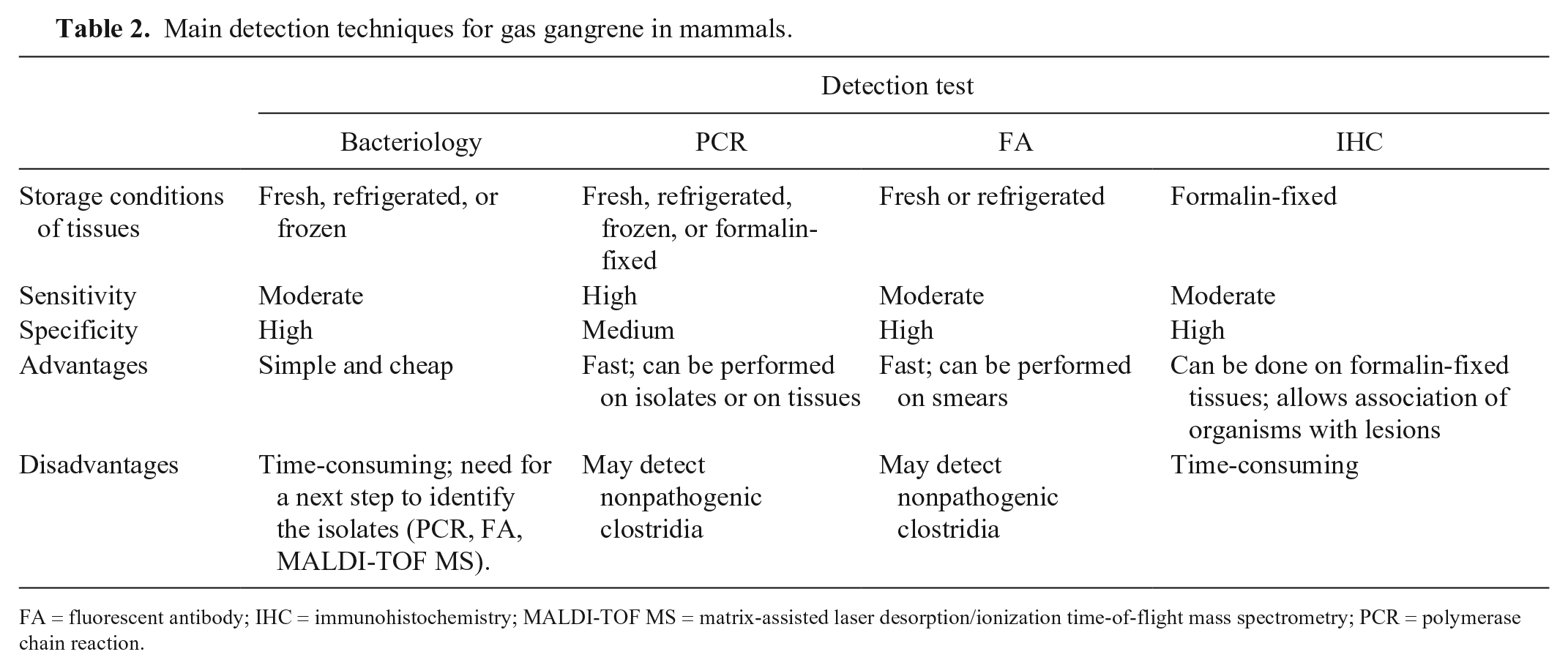
FA = fluorescent antibody; IHC = immunohistochemistry; MALDI-TOF MS = matrix-assisted laser desorption/ionization time-of-flight mass spectrometry; PCR = polymerase chain reaction.
The collection of samples for microbial analysis must be performed as soon as possible after death because several clostridial species that are normally present in the intestine invade tissues soon before or after death, which can generate false-positive results. 12
Control and prophylaxis
Gas gangrene is usually an acute disease and therefore there is usually no time for treatment. 38 This is particularly true for livestock under extensive rearing conditions and in other species that are not watched closely. 52 However, in animals that are watched closely (e.g., horses), and in which an early diagnosis is established, treatment can be attempted. When possible, affected animals should receive high doses of antibiotics, preferably beta-lactams, such as a narrow-spectrum penicillin with high activity against the etiologic agents of gas gangrene and with minor effects on the indigenous microbiota. 52 Sodium or potassium penicillin (IV formulations) are preferred when possible because of the frequency and volume that is needed, particularly when considering the irritating nature of procaine penicillin. The effectiveness of this treatment in ruminants is very limited. 19
In horses, in addition to high doses of antibiotics, anti-inflammatory drugs, fluid therapy, and fasciotomy or myotomy in the first 24 h of hospitalization have been recommended. 53 In 37 cases of equine gas gangrene, 32 animals were treated with fasciotomy or myotomy, and the survival rate was 78% versus 40% for the animals that did not undergo this treatment. 53 There are few data on the effectiveness of treatment against gas gangrene in wild animals.42,62
Clostridial species are widespread, and their spores are extremely resistant to environmental conditions, therefore eradication of the disease is practically impossible. 68 Immunization of animals and strict hygienic measures are critical for the prevention of the disease. To mitigate contamination with clostridia, proper handling of carcasses and aseptic practices during procedures that generate wounds are required.12,38
Systematic vaccination of ruminants with a combination of bacterins and toxoids against the clostridial species involved in gas gangrene is one of the most important control measures for this disease, and it results in a marked reduction of the incidence and mortality of gas gangrene. 13 Vaccination should start early in the life of animals, followed by re-vaccination 4–6 wk later and, at least, an annual booster.38,52
Conclusion
Gas gangrene is still a common disease in domestic species that can affect mammals individually or in outbreaks. This review of the information about this disease should help veterinarians to recognize the risk factors and adopt preventive measures to reduce the impact of this disease in livestock.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
