Abstract
Beta 2 (β2)-toxigenic Clostridium perfringens type A was recovered in large numbers from the intestine of a neonatal foal with colitis. The foal had been treated with gentamicin. Necropsy revealed marked distension of cecum and colon with watery, rust-colored homogeneous fluid and gastric infarction. Microscopic colonic lesions were superficial necrosis of 50% of the colonic mucosal surface and scattered 1–3-mm ulcers with subjacent neutrophilic infiltration and large Gram-positive bacilli in the necrotic mucosa. Beta-2 toxin was demonstrated in the lesions by immunohistochemical staining.
Both Clostridium perfringens and Clostridium difficile have been associated with enterocolitis in horses. More recently, associations with cpb2 (β2 toxin)-positive C. perfringens 1,5 as well as cpe (enterotoxin)-positive C. perfringens 10 with equine enteritis and colitis have been described. In a previous study of intestinal disease in horses, there was an association between lethal progression of colitis associated with cpb2-positive C. perfringens and antibiotic treatment (gentamicin–penicillin). 5 Subsequently, it was shown that the cpb2 gene in equine isolates was cryptic because of a single nucleotide frame-shift mutation in an area shortly after the gene's start codon. 9 Remarkably, aminoglycoside antibiotics including gentamicin induced β2-toxin production in such isolates, presumably by frame shifting at the level of the ribosome, the target of this antibiotic. 9 This present report describes the gross and microscopic findings in a neonatal foal with colitis apparently caused by β2-toxigenic C. perfringens type A and associated with gentamicin treatment.
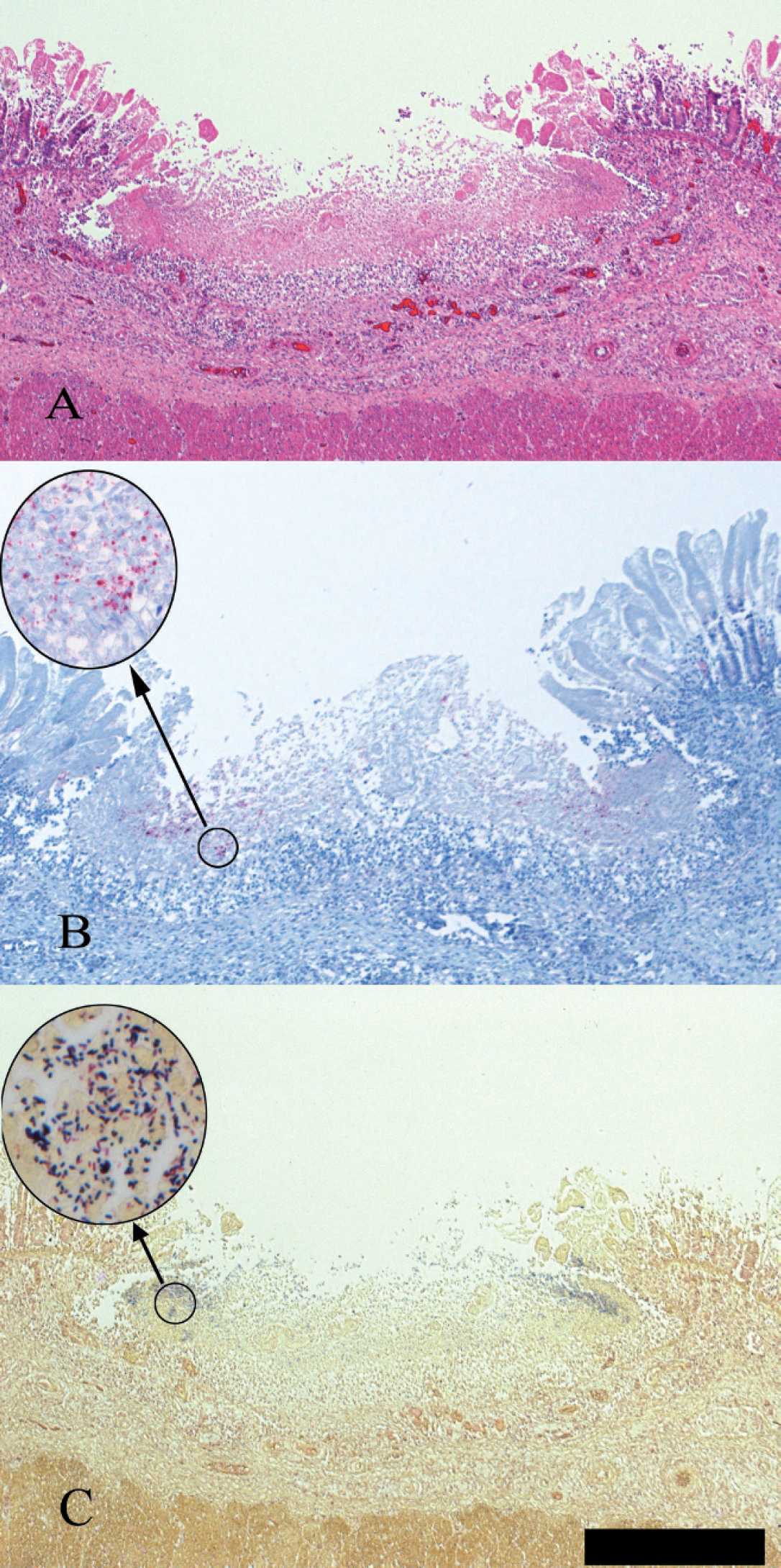
Ulceration of colonic mucosa:
A 3-day-old male foal developed acute diarrhea and a fever of 39.6°C and was treated with oral antacids as well as a single intravenous dose of flunixin a (50 mg) and gentamicin b (100 mg). When examined by the referring veterinarian, the foal was slightly dehydrated but bright and alert, and the owner reported that on the evening after treatment, the foal was alert and eating. It was found dead the following morning. The foal had experienced a normal birth and had a normal immunoglobulin G test performed by the practitioner. It had contact with birds including chickens, geese, and peacocks.
A necropsy was performed at the Animal Health Laboratory of the University of Guelph (Guelph, Ontario, Canada) on the same day as the foal's death, with a postmortem interval of approximately 10–14 hr. External examination revealed questionable hydration and staining of the perineal region with watery fecal material. The stomach contained clotted milk with dark black flecks of blood, and there was mucosal congestion. The small intestine contained small amounts of soft, creamy, golden-brown content. The cecum and large colon were distended with a watery, rust-colored, homogeneous fluid with hyperemia of the mucosa. There was mild pulmonary congestion.
Tissue sections were prepared from samples fixed for 36 hr in 10% buffered formalin. Examined tissues were brain, liver, lung, spleen, stomach, kidney, adrenal, colon, heart, thymus, thyroid, and skeletal muscle. Tissues were paraffin embedded, cut at 4 μm, and stained with hematoxylin and eosin (all tissues) or by Gram stain (colon). In addition, immunohistochemical (IHC) stains for C. perfringens β2 toxin using diluted (1:8,000) rabbit polyclonal antibody prepared against His-tagged Cpb2 antigen with detection using a goat anti-rabbit alkaline phosphatase–linked polymer detection system c were performed on selected sections of colon. Colonic content and stomach were subjected to bacterial culture and toxin testing, and colon content was tested for type A rotavirus with a commercial latex agglutination test. d
Histology preparations revealed gastric and both small and large intestinal mucosal congestion. There was mild edema of lamina propria and submucosa. The colon had scattered 1–3-mm foci of mucosal ulceration, approximately 400–500-μm deep into the lamina propria, with a moderate subjacent neutrophil infiltrate (Fig. 1). Immunohistochemistry for β2 toxin showed strong staining in the same areas of mucosal ulceration as those in which large numbers of large Gram-positive bacilli compatible with C. perfringens were seen (Fig. 1). The bacteria themselves stained prominently. A small fibrin thrombus was present in the submucosa beneath one of these foci. Shallow ulceration of approximately 50% of the remaining mucosa was present with a very mild neutrophil infiltrate in lamina propria in these areas and only occasional Gram-positive bacilli. There was gastric mucosal congestion but no ulceration in the section examined. The lung was congested and edematous with some aspiration of squamous epithelium. The liver, meninges, thymus, and adrenal cortex were also congested. The spleen had multiple foci of acute hemorrhage in capsule, with small numbers of lytic lymphocytes in follicles.
Aerobic and anaerobic cultures were performed on stomach and colon contents to detect the presence of bacterial pathogens. For aerobic culture, MacConkey and Hektoen enteric agars were used, whereas for anaerobic culture, a blood agar containing phenylethyl alcohol and Brucella agar were set up. To exclude the presence of Campylobacter spp., charcoal-containing plates were incubated under microaerophilic conditions. In addition, Salmonella enrichment was attempted by using tetrathionate broth. Clostridium difficile culture was performed as described previously. 8
No Salmonella spp. were detected either on direct culture or on enrichment. Selective media for Campylobacter spp. did not yield this pathogen, and anaerobic culture for C. difficile was negative. Furthermore, no C. difficile A and B toxins were detected using a commercial enzyme-linked immunosorbent assay kit e directly on the fecal sample. The rotavirus latex agglutination test was also negative. Large numbers of C. perfringens were isolated from stomach and colon. These C. perfringens isolates were examined by multiplex polymerase chain reaction (PCR) for the presence of α-, β-, ε-, ι-, and β2 toxin (cpb2) and enterotoxin (cpe) genes. 2,6 Briefly, a few colonies from overnight blood agar plate cultures were resuspended into 200 μl of InstaGene Matrix. f This suspension was incubated at 56°C for 20 min, 100°C for 10 min, and on ice for 5 min. The samples were then centrifuged at 16,000 X g for 5 min. Two microliters of the supernatant was used as a template for the PCR assay. The PCR was performed using a commercial multiplex PCR. g The PCR analysis established the presence of genes for α toxin (cpa) and β2 toxin (cpb2) and the absence of genes for cpb, etx, itx, or cpe toxins.
One of the initial differential diagnoses in the present case was salmonellosis, particularly given the history of close association with poultry. Although treatment of salmonellosis with antimicrobials is controversial, at least one recent publication advocates the use of parenteral gentamicin for treatment of horses with salmonellosis or idiopathic enterocolitis. 3 Because the foal was treated with gentamicin and then showed improvement, it was not possible to completely exclude salmonellosis as the cause of the initial diarrhea in this case, since the use of gentamicin can interfere with Salmonella spp. recovery. Similarly, C. perfringens could not be excluded as a cause of initial diarrhea as no pretreatment culture was performed. The role of β2 toxin–producing C. perfringens in diarrheal illness in animals is not well defined, although there is evidence for a role in several species. For example, in human beings, antibiotic-associated C. perfringens diarrhea has been associated with strains carrying plasmids encoding both the enterotoxin gene (cpe) as well as cpb2 gene. 4 Immunohistochemistry was recently used to demonstrate the presence of β2 toxin in association with intestinal lesions in a Hamadryas baboon (Papio hamadryas) with enteritis. 7 Although the β2-toxin antibody used in the present study is of unknown specificity, the staining pattern matches the pattern seen in the previously mentioned study, 7 and no staining was seen in control sections when substituting 1:8,000 diluted normal rabbit serum for the anti–β2-toxin antibody. In addition, staining was seen in the same areas that the large Gram-positive bacilli were seen in the histology sections (Fig. 1). No antibody was available to check for other C. perfringens toxins by IHC.
An association has been previously noted between gentamicin treatment and the development of lethal typhlocolitis in horses. 5 Interestingly, many equine isolates of C. perfringens carry a “consensus” cpb2 gene, which is out of frame because of the absence of an adenine residue in the poly A tract immediately downstream of the start codon of the cpb2 gene, which results in a stop codon 19 bases downstream of the start of the gene. 9 Because gentamicin and other aminoglycoside antibiotics target the ribosome, treatment with these antibiotics allow translation of the cpb2 messenger RNA into an intact protein through induction of a ribosomal frame shift. 9 Unfortunately, the isolate was discarded, and it was therefore impossible to sequence the gene to determine whether it was in frame. The presence of large numbers of cpb2-positive C. perfringens in the colon and the evidence from IHC that the Gram-positive bacteria found closely associated with the mucosal ulcerations were β2 toxin positive suggest that this was a case of β2 toxin–associated C. perfringens type A colitis. One notable feature of the IHC staining, also observed in a previous study, 1 was the presence of the stain around individual bacteria. No other recognized enteric pathogens were identified by routine laboratory diagnostic procedures.
The present case is unusual because of the very young age of the animal. The disease had a rapid onset, since the foal died overnight after the owner had observed it in the evening as alert and eating. There were signs of generalized toxemia, as evidence by generalized congestion and the histopathological changes of hemorrhage and some intra-vascular coagulation in organs. Whether gentamicin treatment induces increased production of β2 toxin would be worth investigating. To the authors' knowledge, gentamicin-associated β2 toxin–associated C. perfringens type A colitis has not previously been reported in a foal. The case suggests that veterinarians might want to exercise caution in the use of gentamicin as a treatment for enteritis in foals and in horses more generally.
Acknowledgements.
The authors thank Dr J. Glenn Songer for the gift of an Escherichia coli–carrying Histagged consensus cpb2 gene, from which the antiserum to rCpb2 was made; the Ontario Ministry of Agriculture, Food and Rural Affairs–funded Animal Health Laboratory's Animal Health Strategic Investment for funding research on cpb2; and the Natural Sciences and Engineering Research Council for funding research on C. perfringens in animal disease. Thanks also to Dr. Martyn Potter of Newmarket Animal Hospital, Ontario, for providing additional clinical information, and Dr. Josepha DeLay and Ms. Susan Lapos for technical assistance.
Footnotes
a.
Banamine, Schering-Plough Animal Health, Pointe Claire, Quebec, Canada.
b.
Gentamax 100, Phoenix Pharmaceutical Inc., St. Josepha, MO.
c.
Ventana Medical Systems Inc., Tucson, Arizona, AZ.
d.
Rotascreen, Microgen Bioproducts Ltd., Camberley, United Kingdom.
e.
C. difficile tox A/B II, TechLab Inc., Blacksburg, VA.
f.
Bio-Rad Laboratories, Mississauga, Ontario, Canada.
g.
Qiagen Inc., Mississauga, Ontario, Canada.
