Abstract
A 5-y-old, male degu (
Degus (
A 5-y-old, male degu weighing 215 g, with no previous medical history, was presented to the animal hospital with the chief complaint of a cervical mass. The 1.3 × 1.0 × 0.7-cm subcutaneous mass was located in the ventral cervical region. No other significant abnormalities were noted on physical examination, and the mass was removed surgically. At surgery, the mass was localized in the midline of the cervical region, just caudal to the thyroid glands. Following removal, the mass was fixed in 10% neutral-buffered formalin and submitted to the Laboratory of Veterinary Pathology, Nihon University (Kanagawa, Japan), for histologic examination.
On section, the formalin-fixed mass was solid and tan-to-white (Fig. 1). After trimming, representative tissues were processed routinely, and 5-µm thick sections were stained with H&E. Immunohistochemistry was performed on tumor sections for keratin/cytokeratin AE1/AE3 (monoclonal mouse antibody; Nichirei Biosciences), vimentin (monoclonal mouse antibody; Nichirei Biosciences), CD3 (monoclonal mouse antibody diluted 1:50; Dako), CD20 (polyclonal rabbit antibody diluted 1:800; Funakoshi), and Pax5 (polyclonal rabbit antibody diluted 1:1,000; GeneTex). After deparaffinization, antigen retrieval was achieved using target retrieval solution, pH 9 (Dako), or target retrieval solution 10× concentrate (Dako) at 121°C for 20 min for all antibodies. Labeling was visualized with 3,3′-diaminobenzidine substrate (Fujifilm Wako Chemicals), and sections were counterstained with Mayer hematoxylin. Positive immunohistochemical controls consisting of normal thymus from the degu were included.
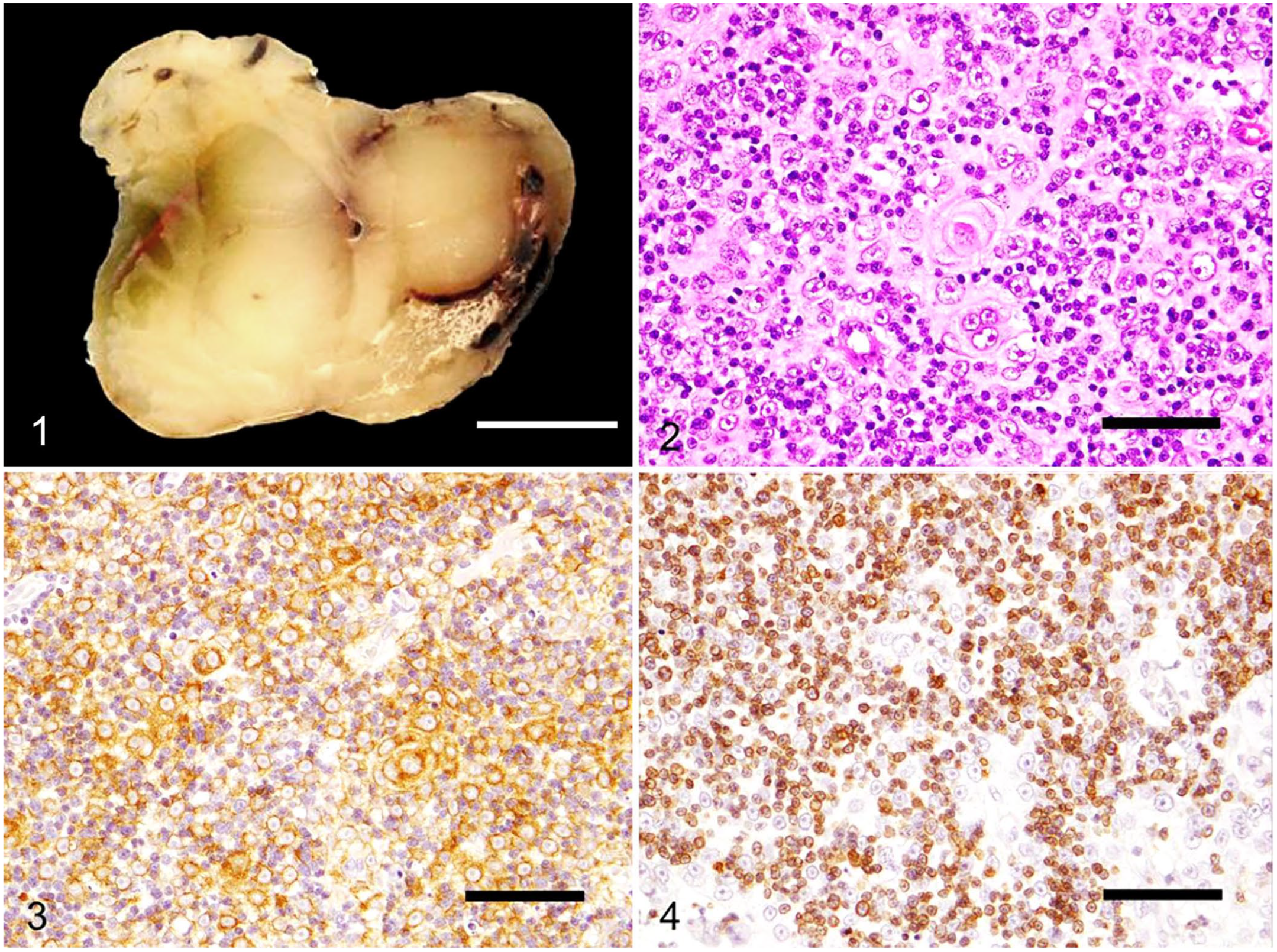
A cervical thymoma in a degu.
Histologically, the encapsulated, highly cellular, expansile mass compressed normal thymic tissue to the periphery. The mass was composed of polygonal cells arranged in dense sheets, mixed with fewer small lymphocytes scattered throughout the neoplasm (Fig. 2). Polygonal cells had distinct cell borders and moderate amounts of eosinophilic cytoplasm. Nuclei were medium-to-large, round, with coarsely stippled chromatin and distinct nucleoli. Anisocytosis and anisokaryosis were mild. Polygonal cells occasionally formed concentric whorls with a central area of keratinization, consistent with Hassall bodies (Fig. 2). A total of 16 mitoses were seen in 2.37 mm2 (equivalent to ten 40× fields). Small lymphocytes were well-differentiated with no obvious atypia. Immunohistochemically, the polygonal cells had diffuse cytoplasmic immunoreactivity to keratin/cytokeratin AE1/AE3 antibody (Fig. 3) and were negative for vimentin antibody. Small lymphocytes were immunoreactive to CD3 antibody (Fig. 4) and negative for CD20 and Pax5 antibodies. The positive controls showed appropriate reactions for all antibodies. The histologic and immunohistochemical findings of the cervical mass are consistent with a thymoma.
The degu died as a result of respiratory failure 8 mo after the surgery. A cosmetic autopsy was performed by a clinician. A mass on the thoracic wall, a nodular lesion in the lung, and a thoracic effusion were noted. Representative internal organs were submitted for histology. The significant histologic lesions included myxosarcoma of the right thoracic wall with metastasis to lung, myocardial hypertrophy, pulmonary congestion and edema, and thromboembolism in multiple blood vessels, which may have contributed to respiratory failure and circulatory disturbances, culminating in death. The myxosarcoma was composed of interlacing bundles of spindle cells, within abundant basophilic mucinous matrix. The neoplastic cells had indistinct cell borders and small amounts of eosinophilic cytoplasm. Nuclei were medium-sized and elongated, with coarsely stippled chromatin and small nucleoli. Anisocytosis and anisokaryosis were moderate. A total of 1–2 mitoses were seen in 2.37 mm2 (equivalent to ten 40× fields). In addition, islet amyloidosis, membranous nephropathy, lymphoplasmacytic interstitial nephritis, and fibrosis of the adrenal glands were noted as minor or incidental lesions. No recurrence and/or metastasis of thymoma was observed.
Thymomas usually form tuberous tumors in the mediastinum and are composed of neoplastic cells derived from thymic epithelial cells accompanied by variable numbers of non-neoplastic lymphocytes. 24 In veterinary medicine, the World Health Organization (WHO) classification scheme for thymic epithelial tumors has been applied. 25 According to this classification, thymomas are subdivided into types A, B1, B2, B3, AB, micronodular thymoma, combined thymoma, thymoma with anaplasia, and thymic carcinoma. 25 We classified our case as type B3 thymoma, which is composed of sheets of well-differentiated round-to-polygonal neoplastic epithelial cells admixed with a minor component of non-neoplastic lymphocytes. Thymomas must be differentiated from thymic carcinomas, and, by the WHO classification system for thymic epithelial tumors, thymic carcinomas are distinguished from thymomas by the lack of encapsulation and/or invasion into the surrounding tissues.20,24 Thymomas are encapsulated, with well-defined borders. 20 In our case, the neoplasm was encapsulated with no invasion into surrounding tissue. Dogs or cats with thymomas commonly develop myasthenia gravis and exfoliative dermatitis as paraneoplastic syndromes, 24 but there was no evidence of paraneoplastic syndromes in our case.
Given the anatomic location of the mass, the differential diagnoses for the present case included, as well as thymic carcinoma, integumental tumor, carotid body tumor, lymphoma, and thyroid tumors such as thyroid adenoma, thyroid carcinoma, and C-cell carcinoma. These tumors were excluded based on histopathologic features. Degus have both a cervical and a mediastinal component of the thymus. The mediastinal component is multilobed, amorphous, and morphologically similar to the typical thymus in other rodent species. On the other hand, the cervical component is bilobed, large, and remains active for life. 26 Mice, rats, and guinea pigs have a cervical thymus similar to degus.3,8,27 In goats, both cervical and mediastinal thymomas appear to be common. Goats with mediastinal thymomas were likely to have severe respiratory signs such as tachypnea and cardiovascular impairment; clinical signs were not observed except for a palpable mass in goats with cervical thymomas. 5 The prognosis for goats with cervical thymomas was favorable following surgical excision, whereas goats with mediastinal thymomas had a guarded prognosis. 5 Among animals without a cervical thymus, an ectopic thymoma in the cervical area with increased respiratory sounds and lateral deviation of the trachea has been reported in cats. 11
We found no previous report of a thymoma that originated from a cervical component of the thymus in rodents, including degus. In rodents, thymomas have been reported in mice, rats, hamsters, and gerbils.4,15,19,27 Under laboratory conditions, Wistar Hannover rats and several inbred strains of rats such as BUF/Mna, WAB, and W/Nhg are strains known to be predisposed to thymomas.6,16,17,23 In addition, some transgenic mice strains, including SV40T and E2F2, have a high incidence of thymic tumors.12,19 Interestingly, no cervical thymomas have been detected in thoracic thymoma–prone BUF rats. 27
Myxosarcoma has rarely been reported in degus. 22 Our case with 2 concurrent neoplasms was unusual because degus are known to have a low incidence of neoplastic diseases. 22 When a mass occurs in the cervical area in animals with cervical thymuses, thymoma should be included in the differential diagnosis.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interests with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
