Abstract
A free-ranging juvenile male black bear (Ursus americanus), found dead in Alberta, Canada, had severe nonsuppurative encephalitis. Lesions in the brain were most severe in the gray matter of the cerebral cortex, and included perivascular cuffs of lymphocytes and plasma cells, areas of gliosis that disrupted the neuropil, and intralesional protozoan schizonts. The left hindlimb had suppurative myositis associated with Streptococcus halichoeri. Immunohistochemistry and molecular analyses (PCR and sequencing of 4 discriminatory loci: 18S rDNA, ITS-1 rDNA, cox1, rpoB) identified Sarcocystis canis or a very closely related Sarcocystis sp. in the affected muscle and brain tissues. The main lesion described in previously reported cases of fatal sarcocystosis in bears was necrotizing hepatitis. Fatal encephalitis associated with this parasite represents a novel presentation of sarcocystosis in bears. Sarcocystosis should be considered a differential diagnosis for nonsuppurative encephalitis in bears.
Sarcocystis spp. (Sarcocystidae) are apicomplexan protozoan parasites that undergo a typical coccidian life cycle that requires an intermediate and definitive host, usually a carnivore species. 22 Intermediate hosts, usually prey species, become infected by consuming oocysts and/or sarcocysts (encysted form in tissues of intermediate hosts). 22 Asexual life cycle phases occur in intermediate hosts, resulting in sarcocyst formation in striated muscle and less commonly the central nervous system and liver.6,22 Sarcocysts contain thousands of bradyzoites that, when ingested by the carnivorous definitive host, undergo sexual lifecycle stages to produce infective oocysts that pass in the feces, thus completing the life cycle. 22 As is typical for most Sarcocystis spp., knowledge of the life cycle of S. canis, including identification of the definitive host, is incomplete. 22
Infectious diseases represent an important and growing threat to the conservation of free-ranging large carnivore populations, including bears (Ursus spp.). 20 However, most infectious disease studies of bears are limited to serologic surveys with only rare reports of infections associated with disease. 20 Here we describe a case of fatal protozoal encephalitis associated with S. canis (or a closely related organism) and provide molecular characterization of the pathogen in a free-ranging black bear.
In October 2018, a free-ranging juvenile male black bear (Ursus americanus) was found dead ~500 m from a highway in the Eyrie Gap Provincial Recreational Area, which is in the eastern foothills of the Rocky Mountains in Alberta, Canada. The carcass was stored frozen at a Government of Alberta facility until February 2019 when a conservation officer with the Government of Alberta submitted it to the Alberta Region of the Canadian Wildlife Health Cooperative, University of Calgary for autopsy. The University of Calgary Animal Care Committee approved this diagnostic investigation.
The bear weighed 28 kg and was in adequate nutritional condition with moderate retroperitoneal and abdominal fat stores. Subcutaneous tissue and extensor muscles cranial to the left tibia were friable, red-pink, and contained areas of purulent, malodorous material (Suppl. Fig. 1). The overlying skin was intact. The remaining organs and brain were grossly normal.
We fixed samples of the muscle lesion, brain, heart, tongue, salivary gland, lung, liver, trachea, urinary bladder, thyroid glands, stomach, colon, adrenal glands, kidney, testes, and skin in 10% neutral-buffered formalin for 48 h, then processed tissues by routine methods. Veterinary pathologists (J.L. Rothenburger, N.M. Nemeth) examined 4-μm thick sections of paraffin-embedded tissues stained with hematoxylin and eosin. Histologically, there was suppurative cellulitis, fasciitis, and degenerative myositis with mineralization in the soft tissues of the left hindlimb (Fig. 1A). Several myocytes in these areas contained tissue bradyzoites consistent in morphology with Sarcocystis sp. Lesions in the brain were most severe in the cerebral cortex and included perivascular cuffs of lymphocytes and plasma cells, areas of gliosis that disrupted the neuropil, and intralesional protozoan schizonts consistent with severe nonsuppurative encephalitis (Fig. 1B, 1C). There were no microscopic liver abnormalities evident.
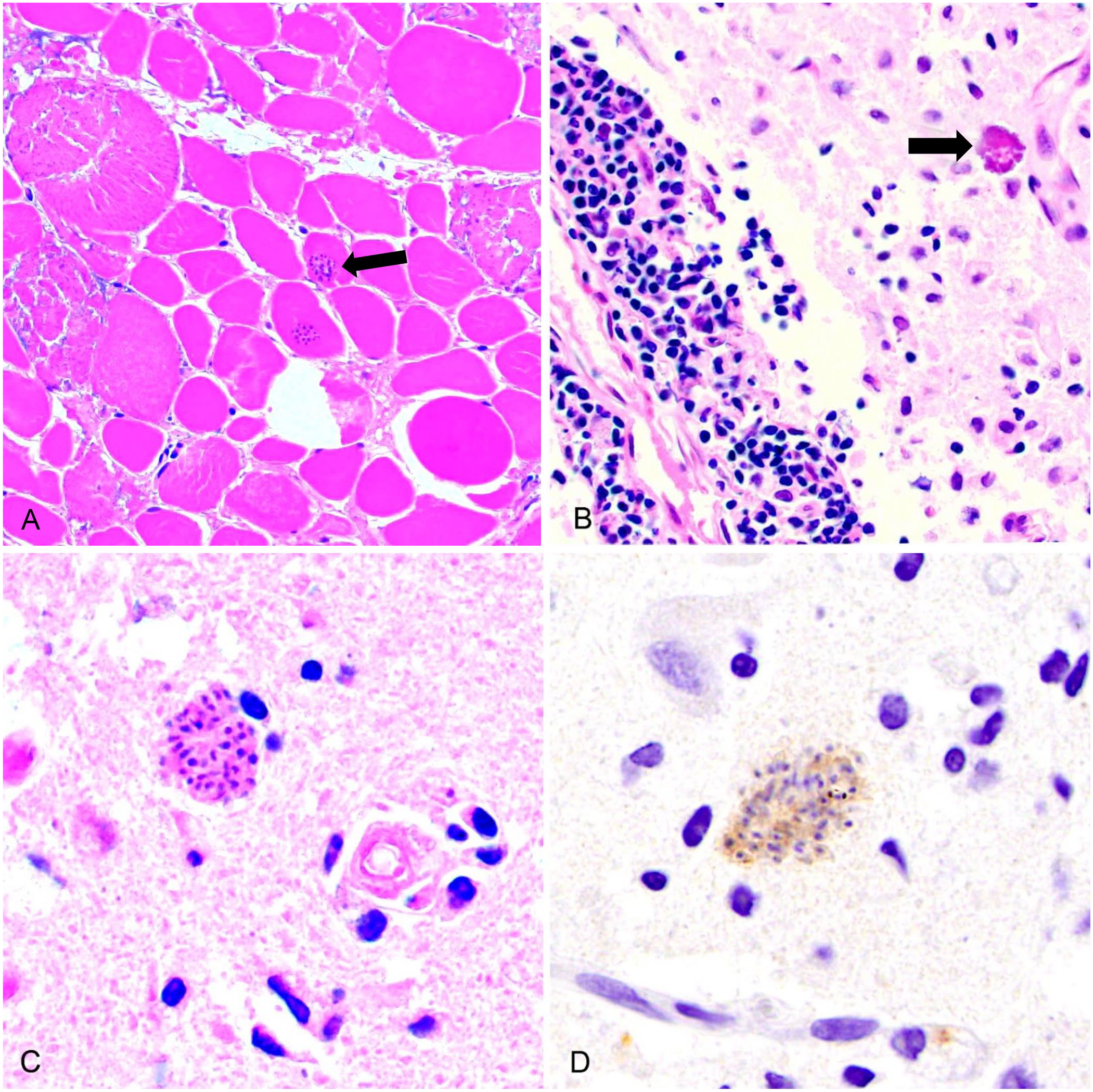
Sarcocystosis in a free-ranging black bear (Ursus americanus).
Additional samples of muscle and brain were stored frozen for further analyses. Routine aerobic culture of frozen samples of brain, skeletal muscle from the left hindlimb, and spleen, by Prairie Diagnostic Services (PDS; Saskatoon, Saskatchewan, Canada), identified heavy growth (4+) of Streptococcus halichoeri in the skeletal muscle tissues; mixed bacteria (Enterococcus faecalis, Escherichia coli), deemed most likely fecal contamination and nonsignificant, were present in the brain and spleen. Immunohistochemical staining, conducted by PDS, for canine distemper and rabies viruses were negative.
The Athens Veterinary Diagnostic Laboratory, University of Georgia (Athens, GA, USA) performed immunohistochemical staining for Sarcocystis neurona (considered cross-reactive for other Sarcocystis spp., Howerth EW, pers. comm., 2019 May 05). Antigen retrieval was achieved using protease 3 (Ventana) for 7 min. Slides were incubated at room temperature with diluted (1:500) rabbit primary antibody (courtesy of David Lindsay, VA Tech) and subsequently with diluted (1:100) biotinylated goat anti-rabbit secondary antibody (Vector) for 10 min. Sections were stained with DAB (3,3′-diaminobenzidine) as the chromogen (Biocare Medical) for 12 min following counterstaining with hematoxylin. Slides were stained on an automated stainer (Nemesis 3,600; Biocare Medical). The positive control was horse brain. Protozoan cysts in the brain and muscle lesions exhibited moderately strong immunostaining for S. neurona (Fig. 1D).
DNA was extracted from both brain and muscle tissue (DNeasy blood & tissue kit; Qiagen). Discriminatory loci were identified from the literature and from the wealth of Sarcocystis spp. sequence data available in GenBank: 18S rDNA, ITS-1 rDNA, cox1 (mtDNA), and rpbB (RNA polymerase B gene of the apicoplast genome). Previously designed primers were used for the amplification of all 4 loci (Table 1) from both muscle and brain using the high-fidelity polymerase Kapa HiFi HotStart (Roche) that minimizes the introduction of polymerase-induced errors. PCR thermocycling conditions were optimized to yield a single band on agarose gel following electrophoresis; 3 min denaturation at 95°C followed by 40 cycles of 98°C for 20 s 12 s at the annealing temperature (TA), and 90 s at 72°C, followed by a final 15-min extension time at 72°C. Amplicons were cloned (CloneJET blunt PCR cloning kit; Thermo Fisher), and 1–3 clones were selected per ligation reaction. Each clone was subsequently Sanger sequenced in both orientations to create a consensus sequence. Primers were removed, and sequences were aligned against all available Sarcocystis spp. sequence data in GenBank (accessed June 2021). Phylogenetic relationships were examined with neighbor-joining trees rooted to the distantly related species, Toxoplasma gondii.
Results of genetic comparison between the Sarcocystis sp. responsible for protozoal encephalitis in a free-ranging black bear (Ursus americanus) compared to its most closely related species, the S. canis infection of polar bears (Ursus maritimus), including the origin of the primers for PCR amplification of the 4 separate loci and GenBank accessions.
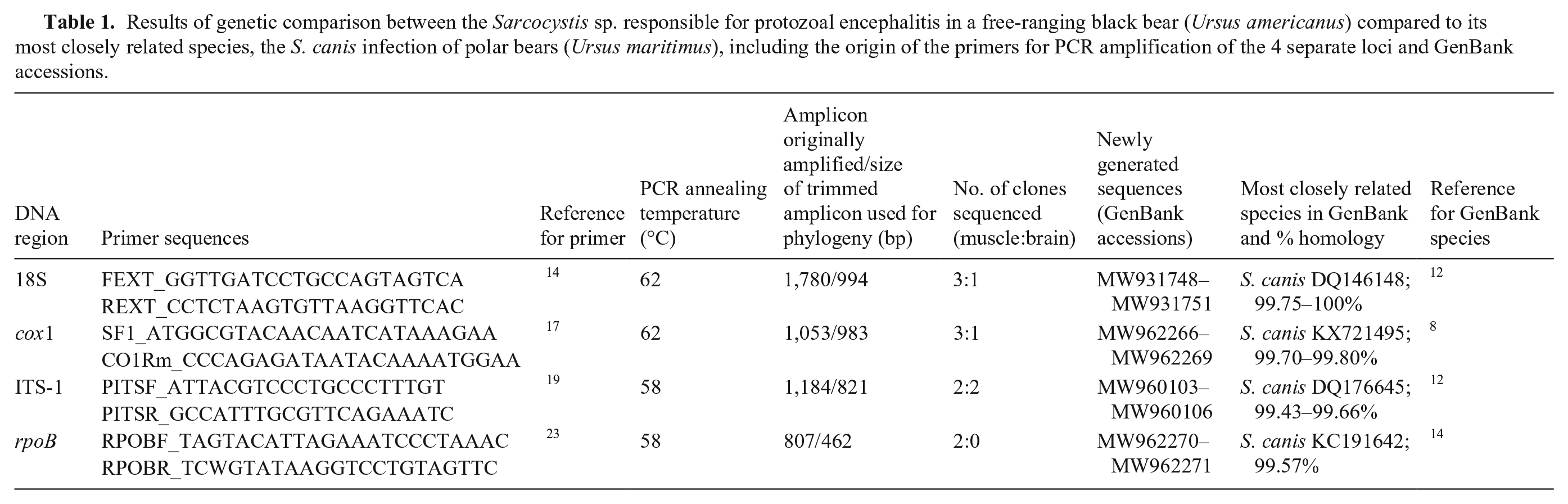
Phylogeny based on a 994-bp partial region of the 18S rDNA gene revealed 100% identity (MW931750), or near 100% identity (MW931748, MW931749, MW93175; 99.85–99.90%), to the hepatic schizonts of S. canis in polar bear (DQ146148),12,16 and the sarcocysts of S. arctosi in grizzly bear (EF564590). 11 The S. canis found in polar bears have subsequently undergone further genetic characterization with the generation of sequence data for a number of more variable loci and ultimately more discriminatory loci, namely ITS-1 rDNA (DQ176645), 12 cox1 (KX721495), 8 and rpbB (KC191642). 14 For all 3 of these additional loci, separate, phylogenetic analysis of our generated sequences revealed that our sequences are most closely related to this S. canis from polar bears: our ITS-1 sequences MW960103–MW960106, our cox1 sequences MW962266–MW962269, and our rpoB sequences MW962270–MW962271 were 99.7–99.8%, 99.43–99.66%, and 99.57% homologous to S. canis in polar bear, respectively (Table 1; Suppl. Fig. 2).
Based on the histologic lesions and molecular characterization, we concluded that this bear died of encephalitis caused by a S. canis species or a very closely related Sarcocystis species. In general, pathogenic Sarcocystis spp. cause disease in the intermediate host and are subclinical in the definitive host. 22 Intermediate hosts of pathogenic Sarcocystis spp. may experience acute or chronic disease manifestations, depending on the stage of infection. Disease severity is thought to depend on both the dose and immune status of the infected host. 22 We suspect that a penetrating wound introduced bacteria into the hindlimb soft tissues, leading to suppurative myositis. This infection may have led to stress and altered immunologic responses that may have indirectly contributed to the development of encephalitis. In addition, consumption of a sufficiently large dose of infective sarcocysts (in tissues from a heterospecific or conspecific intermediate host) or environmental oocysts may have facilitated clinical disease development. Additional immunosuppressive factors such as stress from intraspecific interactions, substandard habitat quality, anthropogenic disturbance, adverse weather events, and other sources are possible contributory factors that are challenging to objectively assess in a single case.
Sarcocystosis in bears appears to affect the liver most commonly, causing multifocal, necrotizing hepatitis, and has been described in 5 cases, encompassing all North American bear species (Table 2). Liver lesions were not evident in our case. Interestingly, gliosis was also present in the brain of 3 of the 4 cases in which the brain was examined histologically, although visible schizonts were not identified.6,10,16 In one of these cases with gliosis but no visible schizonts, brain tissue was PCR positive for Sarcocystis spp. 10 The brain of a third case had neither lesions nor schizonts, but was also PCR positive. 6 These previous reports suggest that the brain may be a predilection site for Sarcocystis spp. in bears, but infection does not always manifest as encephalitis. The Sarcocystis-associated encephalitis in the bear in our case is a different presentation compared to other reports of this parasite in the literature.
Summary of fatal cases of sarcocystosis in North American bears (Ursus spp.).
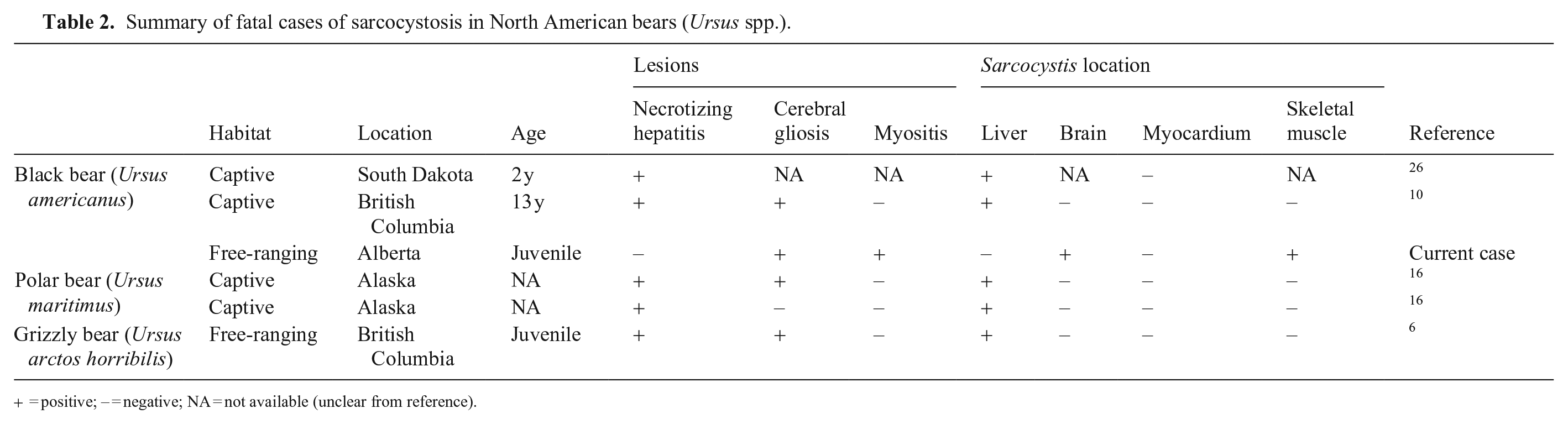
= positive; − = negative; NA = not available (unclear from reference).
Based on our molecular analyses, we conclude that the Sarcocystis spp. from this case is genetically similar to previous reports of S. canis–associated sarcocystosis in bears.12,16 Previous cases described in the literature used 100% similarity of partial but discriminatory regions of the 18S gene (DQ146148).6,10 The 100% homology of 994 bp of the 18S gene of the S. canis–like parasite in polar bear (DQ146148) to sarcocysts in grizzly bears in Alaska currently described as S. arctosi (EF564590) suggests these may be the same etiologic agent. 11 Although S. canis was originally described from parasitic schizonts associated with fatal hepatitis in dogs, no DNA has yet been derived from infected dogs. Similar cases have been identified in a surprisingly diverse range of host species, including sea lion (Zalophus californianus), chinchilla (Chinchilla lanigera), and bears. 12 In addition to S. canis, that has now been implicated in a number of cases in bears, a new species, Sarcocystis ursusi, has recently been discovered in black bears. 13 The lack of molecular data for this proposed new species precludes a comprehensive comparative analysis here.
Sarcocystis spp. have been associated with encephalitis in a wide variety of terrestrial and marine mammalian and avian species.3,22,23,25 In addition to encephalitis, sarcocysts in skeletal and cardiac muscle incite variable inflammatory responses, ranging from none to severe inflammation. 25 The level of inflammation incited by the sarcocysts in skeletal muscle of the bear in our case is uncertain given the concurrent bacterial infection. However, there were sections of relatively unaffected muscle that had sarcocysts with no associated inflammation. In raccoons (Procyon lotor) and skunks (Mephitis mephitis) that are coinfected with canine distemper virus (Canine morbillivirus) and Sarcocystis spp., immunosuppressive effects of the morbillivirus are suspected to be critical to the development of severe necrotizing encephalitis. 18 We ruled out canine distemper virus infection in our case.
A number of pathogens are associated with encephalitis in captive bears (Ursus spp.), including West Nile virus and herpesviruses (equine herpesvirus 1 [Equid alphaherpesvirus 1], equine herpesvirus 9 [Equid alphaherpesvirus 9], suid herpesvirus 1 [Suid alphaherpesvirus 1]).15,24 Reports of agents of infectious encephalitis in free-ranging bears include rabies virus [Rabies lyssavirus], canine distemper virus, and potentially ursid gammaherpesvirus 1.5,9,15
In free-ranging bears, there is serologic evidence of prior infection with pathogens that have the potential to cause encephalitis such as several morbilliviruses, T. gondii, Saint Louis encephalitis virus, and western equine encephalitis virus, although these studies were conducted on apparently healthy bears and thus infection was not associated with clinical disease.4,7,15 Metagenomic investigation of free-ranging black bears with idiopathic encephalitis has identified a range of novel viruses, including Ursus americanus circovirus. 2 It is unclear if any of the aforementioned pathogens cause encephalitis or other clinical illness in free-ranging bears. There is also a single case of noninfectious encephalitis described in a captive polar bear caused by antibodies to NMDA (N-methyl-D-aspartate) receptors. 21 It is unknown if anti-NMDA encephalitis occurs in free-ranging bears.
The isolation of Streptococcus halichoeri in association with suppurative myositis of the hindlimb represents a new host species for this bacterium. First isolated from gray seals (Halichoerus grypus) in the United Kingdom, S. halichoeri is considered an emerging pathogen with a growing species range that includes people, domestic dogs, and mink. 1
Sarcocystis sp. and other infectious diseases may be an important and underappreciated cause of wild bear mortality. 20 Our case demonstrates that in addition to fatal hepatitis, Sarcocystis sp. infection may cause fatal encephalitis in free-ranging bears, and may be considered a differential diagnosis for idiopathic nonsuppurative encephalitis in these species. 2 The health of free-ranging black bear populations is affected by multiple factors, including habitat loss and human–wildlife conflict. There is growing interest in the impacts of these and other cumulative stressors such as climate change on wildlife, including bears; however, the impacts of infectious disease require further investigation. 15
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387211038389 – Supplemental material for Molecular characterization of Sarcocystis spp. as a cause of protozoal encephalitis in a free-ranging black bear
Supplemental material, sj-pdf-1-vdi-10.1177_10406387211038389 for Molecular characterization of Sarcocystis spp. as a cause of protozoal encephalitis in a free-ranging black bear by Jordan B. Greenfield, Madison V. Anderson, Emily A. Dorey, Elizabeth Redman, John S. Gilleard, Nicole M. Nemeth and Jamie L. Rothenburger in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
The area where the bear originated and the University of Calgary, where this work was completed, are located on the traditional territories of the people of the Treaty 7 region in southern Alberta and is also home to the Métis Nation of Alberta (Region 3). Thanks to Nick Dykshoorn, Conservation Officer, Alberta Environment and Parks, Government of Alberta, for submitting this case. We thank the University of Calgary Diagnostic Services Unit staff for invaluable technical support (Susan Calder-Lodge, Jim Carlsen, Jennifer Larios, Melencio Nicolas, Betty Pollock). We thank Musangu Ngeleka of Prairie Diagnostic Services for assistance with bacteriology. This case was presented in abstract form at the 2019 annual meeting of the American College of Veterinary Pathologists.
Declaration of conflicting interests
Our authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our work was supported by the University of Calgary Faculty of Veterinary Medicine, the Canadian Wildlife Health Cooperative, and NSERC Discovery Grant (RGPIN-2015-03976).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
