Abstract
A juvenile Taveta golden weaver (Ploceus castaneiceps) bird housed in a zoo aviary developed a sudden onset of neurological signs. The bird demonstrated head tilt, rolling onto its back, and inability to perch. Euthanasia was elected due to lack of response to intensive care, and a necropsy was performed. There were no significant abnormal findings on gross examination. On histopathology, the remarkable findings were localized to the brain and consisted of multifocal cerebral microabscesses and rarefaction. Filamentous rod-shaped bacteria were present within and at the periphery of the necrotic foci, and dispersed throughout the neuroparenchyma and intracellularly in neurons. The bacteria were Gram negative, and Warthin–Starry stain demonstrated characteristic “hay stacking.” Real-time polymerase chain reaction analysis of the brain identified the agent as Clostridium piliforme. An additional microscopic finding was severe crypt enteritis; however, the bacteria were not observed in the intestinal sections.
Clostridium piliforme, the causative agent of Tyzzer’s disease, is a unique species among the Clostridia spp. Clostridium piliforme is obligate intracellular, motile, and Gram variable, mostly reported as Gram negative. Tyzzer’s disease was initially discovered in Japanese Waltzing mice and hence has been extensively studied in laboratory animals. 9 To date, the disease has been reported in a wide variety of animal species, including livestock (calf, sheep 11 ), cervids, 3 foals, 2 wildlife, 16 dogs, 6 cats, 7 and 2 psittacine birds.8,10 In all species, animals susceptible to natural infection appear to be neonates or juveniles in weaning stages. Exposure to conditions that cause immunosuppression, such as poor hygiene, stressors, or corticosteroid therapy has also been associated with clinical disease due to C. piliforme in individuals of all ages. In a previous study, overcrowding was established as the most important factor of high incidence of disease in mice. 9 A similar phenomenon of immunosuppression-related disease has been observed in human beings, as infection with C. piliforme was reported in a patient with Human immunodeficiency virus-1. 12 Additionally, there are no records of immunocompetent human beings symptomatic for Tyzzer’s disease. 12
The primary site of infection by C. piliforme is the distal intestine, specifically the ileum and colon–cecum, from where the organism enters the portal circulation and reaches the liver. The bacteria may disseminate to other organs, predominantly to the myocardium, and the intestine–liver–heart triad is the most typical distribution of lesions associated with the disease. Encephalitis is a rare form of Tyzzer’s disease, and the gerbil is the only animal in which spontaneous necrosuppurative encephalitis due to C. piliforme has been reported. 14 Laboratory studies have reproduced this form of disease by intracerebral inoculation of the agent in mice and in the African white-tailed rat (Mystromys albicaudatus).9,15 The present report describes a case of spontaneous Tyzzer’s disease in a juvenile passerine bird, presenting as encephalitis.
A 2-month-old Taveta golden weaver (Ploceus castaneiceps) bird was found on the ground of its exhibit in a zoo in California. The feathers were wet, the bird was unable to fly or perch, and it demonstrated neurological signs including head tilt, torticollis, and rolling onto its back. Supportive therapy consisted of dexamethasone sodium phosphate a (4 mg/kg intramuscularly [IM]), enrofloxacin b (15 mg/kg IM), and diuresis with crystalloid solution c (20 ml/kg subcutaneously). The bird was placed into a heated oxygen cage with an oxygen flow of 2–5 L/hr. No improvement in clinical signs was noted despite intensive treatment, and it was humanely euthanized due to the poor prognosis. The weaver was submitted to the California Animal Health and Food Safety Laboratory System (CAHFS; Davis, CA) for postmortem examination.
On necropsy, the nutritional body condition of the bird was within normal limits. The only significant observation on gross inspection was the liquid-filled intestinal tract. The internal organs were unremarkable. All tissues were fixed in 10% buffered formalin and 4-µm hematoxylin and eosin–stained tissue sections were prepared for light microscopy. There was no bacterial growth from liver and lung on MacConkey and blood agar plates grown under aerobic conditions. Chlamydophila sp. was not detected by fluorescent antibody test on the liver, air sacs, and spleen. Salmonella was not detected by polymerase chain reaction (PCR) or fecal cultures.
Histopathology revealed extensive lesions in the brain sections, predominantly in the cerebral cortex, consisting of ill-defined, regionally extensive areas of hypercellularity and rarefaction (Fig. 1). Large numbers of infiltrating heterophils disrupted the neuroparenchyma and formed multiple microabscesses with smaller numbers of macrophages. There was widespread neuronal cell death in the malacic regions. The capillaries were congested and often had expanded clear perivascular spaces. There was widespread extravasation of erythrocytes in the affected areas. The cytoplasm of many neurons within and in the periphery of the microabscesses was packed with faintly visible long, rod-shaped bacteria. Gram stain revealed the bacteria as Gram negative. Warthin–Starry stain highlighted the stacks of argyrophilic slender rods compatible with C. piliforme (Fig. 2). Immunohistochemistry was negative for both Listeria spp. and West Nile virus. Other microscopic changes were in the intestines and consisted of crypt enterocolitis, in which multiple crypts were severely ectatic, lined by attenuated epithelium, and contained abundant luminal necrotic and degenerate cell debris. The liver sections were unremarkable. Silver stains of the intestinal sections as well as the liver did not reveal any bacteria morphologically similar to C. piliforme.
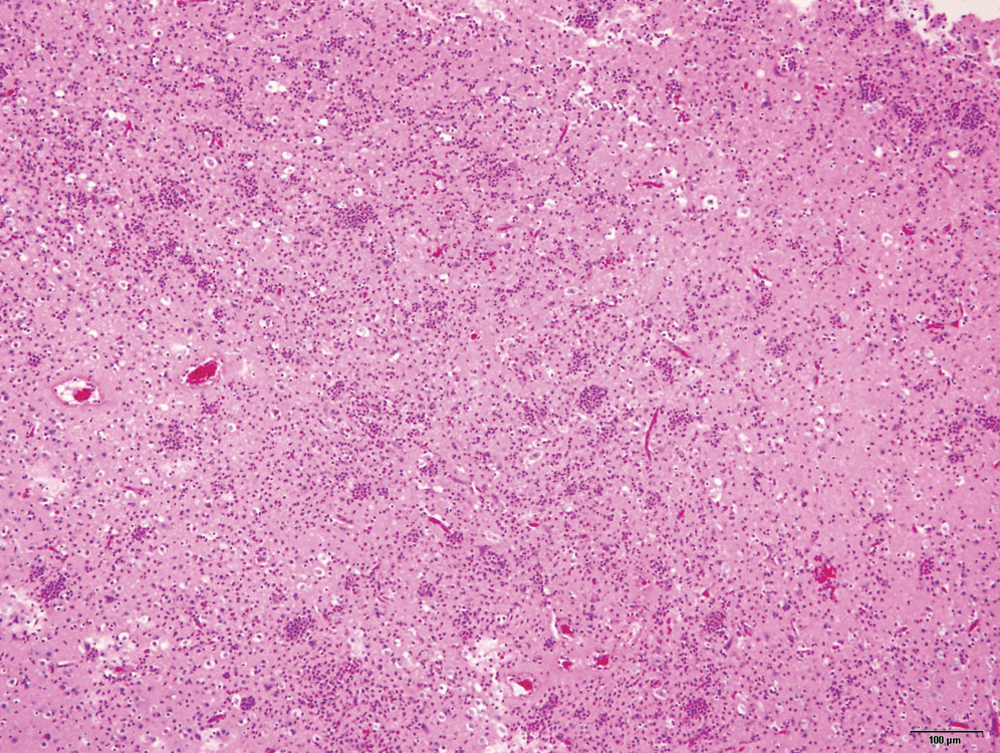
A lower magnification illustration of the extensive encephalitis with microabscesses, rarefaction, and prominent capillaries. Hematoxylin and eosin stain. Bar = 100 µm.
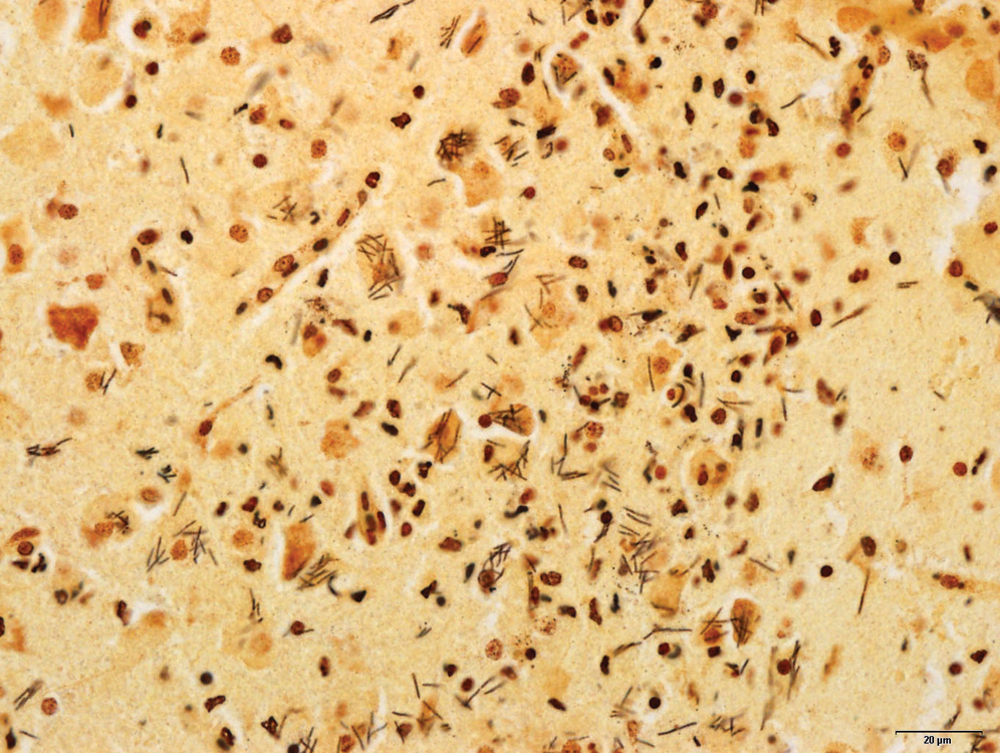
Silver stain (Steiner) section of the brain lesions, highlighting the characteristic curvilinear bacteria in stacks, often in neuronal cell bodies. Bar = 20 µm.
Molecular analysis was performed on the paraffin-embedded brain sections based on an assay described earlier. 2 Briefly, multiple 25-µm sections were pretreated with 70% ethanol and xylene to remove the paraffin, and the genomic DNA was extracted according to the manufacturer’s directions. The real-time assay conducted was previously established and validated for detecting a specific 162–base pair (bp) product of the 16S ribosomal RNA (rRNA) gene of C. piliforme (GenBank accession no. D14639), with a FAM-TAMRA–labeled probe. d The PCR was highly positive at a threshold cycle value of 28.52, with a baseline of 3–10, and a threshold value of 0.10. The BLAST (http://www.ncbi.nlm.nih.gov/blast/Blast.cgi) results of the PCR product attained in the current study retrieved 100% homology with only C. piliforme as the top 5 hits and the remainder of the hits as unidentified bacterial clones with a low percentage of identity. The 16S rRNA of the sample was further sequenced, and the obtained 614-bp linear DNA product was compared with multiple sequences using an alignment program. e Alignment of this product with the GenBank sequences of C. piliforme (accession nos. DQ352808 and D14639) revealed 98% homology.
Diagnosis of Tyzzer’s disease relies on the combined findings of histopathological lesions and the typical histomorphology, silver stain reactivity, and Gram-negative properties of the intracellular bacteria. In the present case, the aforementioned criteria characterized the agent as C. piliforme and confirmation was by real-time PCR. In recent years, application of PCR has been a useful adjunct to the diagnosis of Tyzzer’s disease with anticipation of providing a means of antemortem diagnosis by fecal testing. 5 While the use of PCR has enhanced the diagnostic approach, caution should be used in interpretation of PCR results used on feces due to the phylogenetic proximity of C. piliforme to other species of Clostridia.1,4 This is especially critical when testing feces or tissues from avian species in which C. colinum may cause similar lesions in the intestine and liver and must be distinguished from C. piliforme. 4 Indeed, a high percentage of homologous regions and a 30-bp hypervariable region (between base pairs 63 and 93 from the 5’ end) have been detected among Clostridia species within the 614-bp long 16S rRNA. Alignment of the sequence in the current study with the C. colinum sequence (GenBank accession no. X76748) revealed homology of only 93%. In addition, C. colinum is Gram positive and morphologically distinct from C. piliforme.
Tyzzer’s disease in avians has previously been reported in 2 psittaciformes.8,10 Enteritis was not a feature of the disease described in either of those cases; however, the authors detected few bacteria in the intestinal wall and in the intestinal macrophages. In the present case, organisms could not be detected in any of the intestinal sections by silver stains, but severe crypt enteritis and the degree of autolysis may have precluded more detailed findings. The primary route of transmission of C. piliforme is fecal–oral by ingestion of spores. Studies in mice have revealed that significant intestinal damage is a prerequisite for infection of organs other than the liver. 13 Due to the lack of liver lesions in the weaver in the current study, it is postulated that the intestine was likely the initial site of infection, with enteritis leading to bacteremia and encephalitis.
The only other avian cases of Tyzzer’s disease are the 2 reports in psittacine birds, which raised the possibility that psittaciformes may have a predisposing susceptibility for the disease. 8 The current case, however, demonstrates that immunocompromised individuals of any species exposed to C. piliforme may develop Tyzzer’s disease. Perhaps the most intriguing aspect of the current case was the presentation of the disease, and C. piliforme should be included as a differential in birds with neurologic signs. In addition, the use of real-time PCR in the diagnostic work-up of these cases for definitive diagnosis is recommended.
Footnotes
Notes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
