Abstract
Twenty-six 5-month-old Holstein calves were accidentally exposed to discarded branches of yew bushes (Taxus sp.). Several calves were found dead approximately 24 hr after exposure; however, a few calves died several days after exposure. One calf died 18 days after the initial exposure to Taxus sp. and was examined on the farm via necropsy. Gross lesions included ascites, and dilated and flaccid myocardial ventricles. Sections of formalin-fixed heart were submitted to the Indiana Animal Disease Diagnostic Laboratory for histopathologic examination; fresh rumen contents were submitted for toxicologic testing. Histologically, large areas of myocardium were replaced by fibrous connective tissue, suggesting previous myocardial necrosis. Taxus alkaloids were identified in the rumen contents using gas chromatography–mass spectrometry. Based on the clinical history, the gross and histologic lesions, the identification of Taxus alkaloids in the rumen contents, and lack of exposure to other known cardiotoxic agents, yew toxicity was considered the cause of death in this calf. Ingestion of taxines is known to cause acute and subacute toxicity in human beings and animals; however, a chronic clinical course and severe histologic lesions have not been previously associated with yew toxicity. Although only 1 calf was examined, this case suggests that yew toxicity can result in a prolonged clinical course in cattle and can cause histologic myocardial lesions.
Twenty-six 5-month-old Holstein calves were accidentally exposed to discarded clippings from yew bushes in July 2008. Several calves died within 24 hr of exposure; however, 1 or 2 calves died every day or every other day for the following 18 days until 22 of the original 26 animals were dead. The last calf with known exposure to yew, a heifer, died 18 days after removal of the plant material from the pasture. A field necropsy of the heifer was performed. Gross lesions included ascites and flaccid, dilated cardiac ventricles, suggesting heart failure.
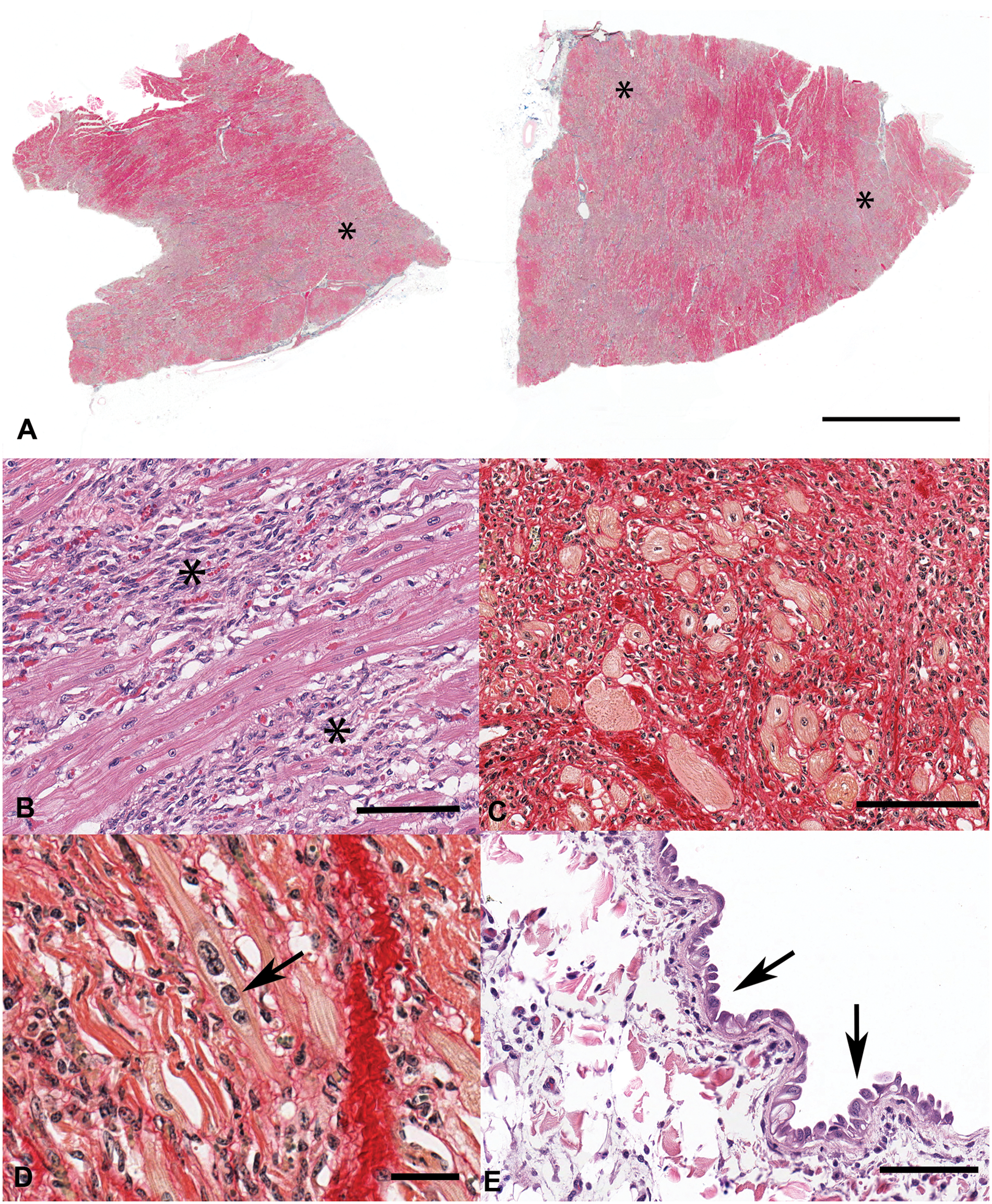
Sections of myocardium were preserved in 10% neutral buffered formalin and submitted to the Indiana Animal Disease Diagnostic Laboratory (ADDL; West Lafayette, Indiana) for histologic examination. Histologically, large areas of myocardium extending from the epicardium to the endocardium were replaced by elongate, spindle-shaped cells with vesicular, ovoid nuclei, consistent with fibroblasts (Fig. 1). In the submitted sections, approximately 80% of the myocardium was replaced by fibrosis, which was well-demarcated from adjacent, viable cardiac myocytes (Fig. 1). Collagen fibrils were interspersed between fibroblasts, and were best visualized using specific histochemical stains for collagen such as Masson trichrome and Picrosirius red, in which collagen fibers stain blue or red, respectively (Fig. 1). Areas of fibrosis were highly cellular, with many fibroblasts and varying amounts of collagen fibers, suggesting a fairly immature lesion. Many areas contained cardiac myocytes that were thin and wavy, indicative of myofiber atrophy. Rare myocytes, characterized by rowing of nuclei and suggestive of attempted myofiber regeneration, were observed (Fig. 1). Few mononuclear leukocytes, including macrophages and rare lymphocytes were scattered throughout areas of myocardial fibrosis. Additionally, the epicardium was multifocally infiltrated by lymphocytes and macrophages and lined by activated mesothelial cells, suggesting pericardial effusion (Fig. 1). Myocardial fibrosis was severe and widespread throughout the submitted sections of heart; thus, heart failure secondary to myocardial fibrosis was considered the cause of death in this heifer. Although direct evidence of cardiac myonecrosis was not evident in the examined sections, putative cardiac myofiber regeneration, extensive myofiber loss, and interstitial fibrosis were considered indirect evidence of a previous, likely monophasic, episode of cardiac myonecrosis.
Holstein calf.
Rumen contents were submitted to the ADDL for toxicology, and forwarded to the California Animal Health and Food Safety Laboratory (Davis, California) in order to test for taxines, the toxic alkaloids found in Taxus spp. A 5-g representative sample of rumen contents was extracted with a 100-ml solution of 5% ethanol a in ethyl acetate b (v/v) combined with 1 ml of 10 N sodium hydroxide b and 50 g of sodium sulfate. b Specific Taxus alkaloid standards were not commercially available; therefore, a 1-g sample from the Taxus baccata plant was extracted alongside to serve as a reference standard. After centrifugation, a 40-ml aliquot was extracted with 10 ml of 0.5 N hydrochloric acid b followed by a second extraction of 5 ml after the addition of 100 ml of hexane. The combined aqueous extract was sparged with a gentle stream of nitrogen to remove all traces of organic solvents before adjusting the pH to >10 with 10 N sodium hydroxide. The analytes in this aqueous extract were then adsorbed on a polymeric C18 solid phase extraction column c under slight vacuum. The alkaloids were eluted with 2 ml of ethyl acetate, and sodium sulfate was added to remove any residual water. The extract was evaporated to dryness and derivatized with bis (trimethylsilyl) trifluoroacetamide (BSTFA). d The extract was analyzed qualitatively using gas chromatography–mass spectrometry (GC-MS) and a 15 M × 0.25 mm × 0.25 µM capillary column. e The relative retention time of the prominent peak present in the extract of T. baccata reference plant standard was at 17.4 min, generating a mass spectrum with m/z 492, 136, and 73 (Fig. 2), which was also present in the rumen content sample (Fig. 3) and spiked rumen content (not shown). The mass spectrum match quality in the rumen content sample was 90%, compared to the T. baccata reference plant extract.
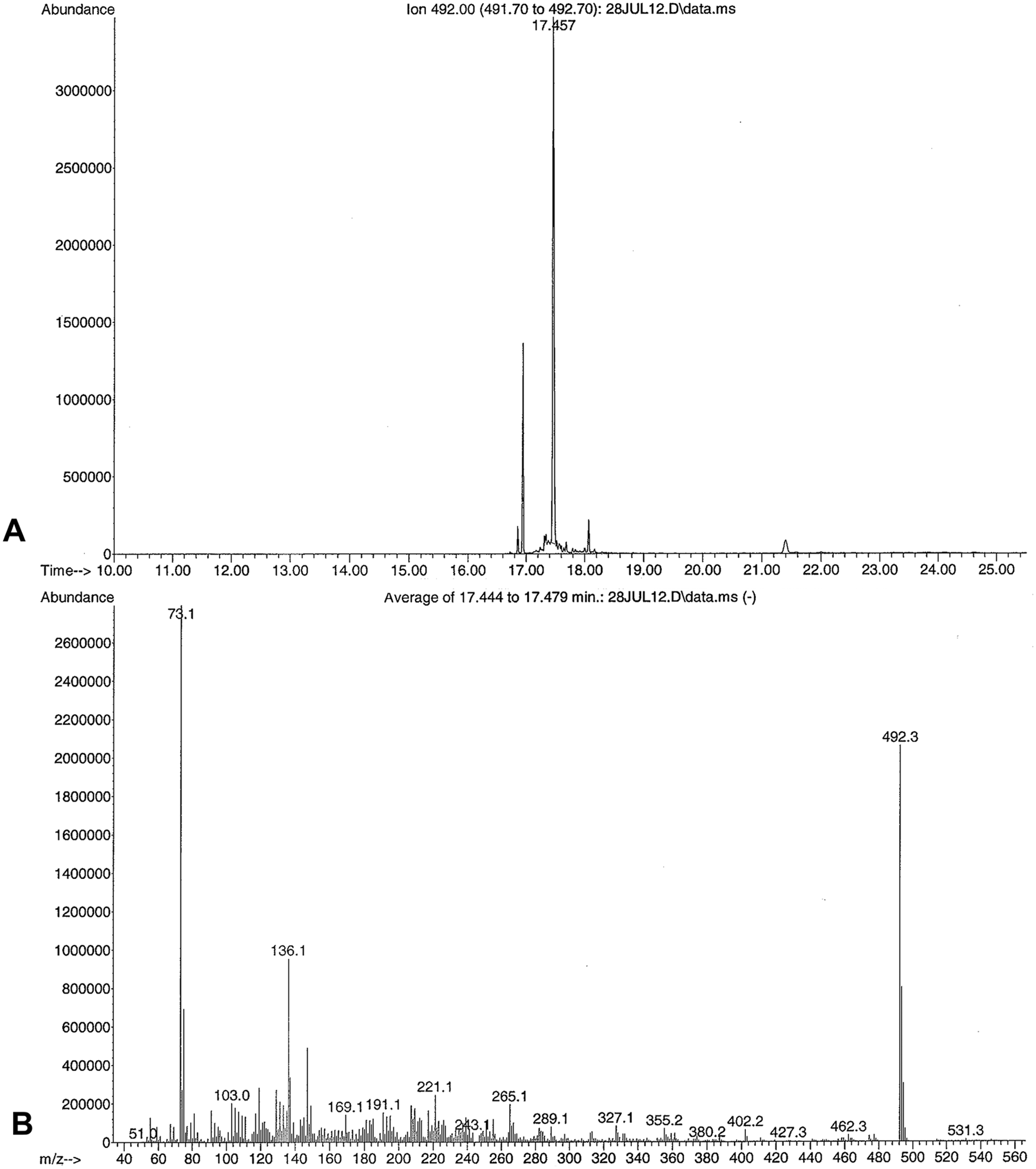
Gas chromatography–mass spectrometry ion chromatogram of a Taxus baccata extract.
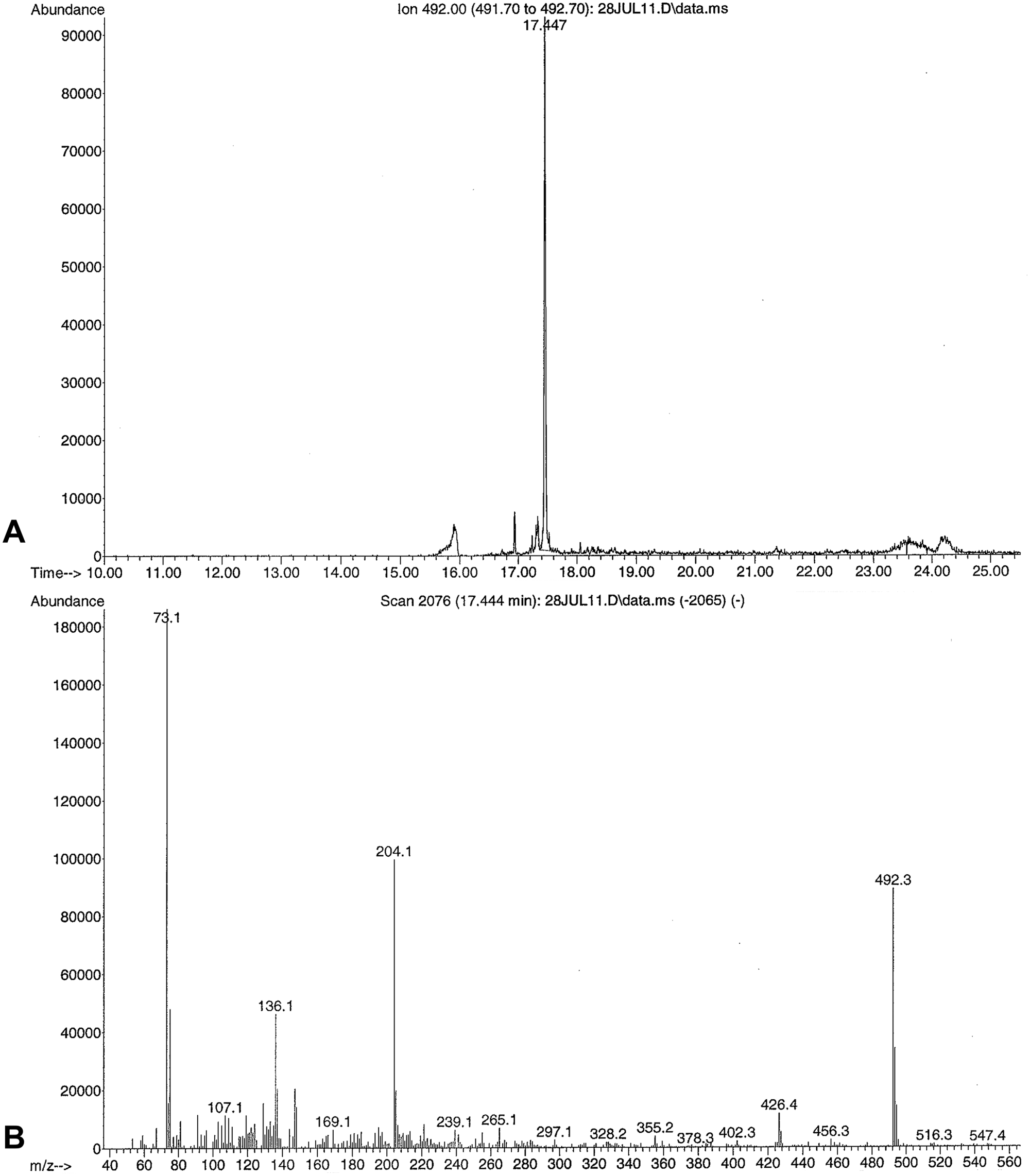
Gas chromatography–mass spectrometry ion chromatogram of the Holstein calf rumen content extract.
Thus, although evidence of yew branches or needles was not found in the rumen during the necropsy, Taxus alkaloids were identified via GC-MS. More likely causes of myocardial necrosis in ruminants were excluded from consideration via anamnesis. The heifer’s diet included 1.8 kg per day of a grain mixture balanced by a professional nutritionist and was devoid of ionophores; thus, vitamin E and/or selenium deficiency and ionophore toxicity were not considered likely causes of the observed lesions. The heifer grazed an orchard grass and clover pasture; known native cardiotoxic plants in Indiana, including white snakeroot (Ageratina altissima), were not identified on the pasture where the animal was kept. In addition to Taxus alkaloids, the rumen contents were also tested and found to be negative for the following alkaloids: coniine, nicotine, anabasine, sparteine, atropine, scopolamine, and deltaline using GC-MS. Based on the clinical history, the gross and histologic lesions, and the identification of taxines in the rumen contents, chronic yew toxicity was considered the probable cause of myocardial necrosis, subsequent fibrosis, and eventual death in this heifer.
Ingestion of yew is known to cause acute and subacute toxicity in human beings and in a range of animal species, including cattle.1,3,7,8,15 Toxicity usually results in sudden death, likely as a result of cardiac arrhythmia, atrioventricular conduction block, and diastolic arrest secondary to cardiac myocyte calcium and sodium channel antagonism. 15 In published accounts of cattle poisoned with yew, most animals die within minutes to hours after ingestion of the plant; however, subacute toxicity, in which cattle die between 2 and 3 days after ingestion, is well recognized.1,7,8 In these instances, cattle exhibit lethargy, dyspnea, bradycardia, ataxia, jugular distension, gastroenteritis, and death.1,7,8 Interestingly, in these and other cases, all cattle exposed to yew clippings or bushes do not die, and at least a few animals survive exposure.1,8,12 In comparison, many human cases of yew toxicity result in fatalities.5,10,15 However, there are also documented instances of patients that have life-threatening symptoms due to yew ingestion, but survive as a result of the heroic effort of physicians, or have moderate symptoms and survive the initial toxic insult.5,9,11 Thus, some precedent exists for the observation that yew ingestion is not always, or immediately, fatal. The cattle that survived the initial insult in the current case were treated with atropine after exposure to yew was confirmed. Physicians and veterinarians use atropine, as well as lidocaine, to treat cardiovascular disturbances secondary to yew intoxication, with questionable efficacy.8,9,11 Whether atropine prolonged the clinical course in the present case is unknown.
Inherent differences in species susceptibility may be the underlying reason for subacute toxicity and/or the prolonged clinical course of yew toxicity in cattle. Monogastric species, such as the horse, seem to be more susceptible to yew toxicity than ruminants, and do not show clinical signs other than death.4,15 The ruminal environment, which can be around pH 7.0 in grazing animals, may have provided the neutral to slightly alkaline environment needed to degrade taxines and prevent further absorption of the toxic alkaloids in this and other cases. 15 Indeed, white-tailed deer (Odocoileus virginianus) are known to be relatively resistant to yew toxicity as compared to cattle, partly because deer ruminal fluid reduces taxine concentrations better than bovine ruminal fluid. 14
In addition to the relatively chronic clinical course, the current case is notable due to the severe histologic lesions that were observed. Neither fulminant myocardial necrosis nor resultant fibrosis has been previously associated with yew toxicity in cattle, or any species, to the authors’ knowledge. In a case of yew intoxication in a horse, mild contraction band necrosis and a mild inflammatory infiltrate consisting of neutrophils and lymphocytes were identified histologically within the myocardium. 13 Thus, at least 1 report suggests that acute myocardial necrosis can occur with yew toxicity. In the vast majority of reported cases, no gross or histologic lesions are documented. 15 In some cases, myocardial hemorrhages or pulmonary congestion have been observed grossly.1,8 Described microscopic lesions are either related to pulmonary edema and congestion secondary to heart failure, or consist of mild interstitial myocarditis.7,8
The suspected initial lesion in the present case, myocardial necrosis, could have resulted from 1 of 2 potential mechanisms. Firstly, antagonism of calcium and sodium channels results in an increase in cytoplasmic calcium within cardiac myocytes. 15 Increases in cytosolic calcium are detrimental to cells generally, via activation of phospholipases, endonucleases, proteases, and ATPases. 6 Increased cytosolic calcium also opens the mitochondrial permeability transition pore, which results in decreased adenosine triphosphate generation. 6 Thus, calcium-induced cell death of cardiac myocytes could occur with antagonism of important membrane ion channels by taxines. Secondly, taxines cause both negative ionotropic and chronotropic effects in ex vivo models, and severe hypotension in clinical cases.2,9,15 Thus, myocardial hypoxia due to general alterations in blood flow and pressure could result in ischemic necrosis in the heart, as well as other organs. No other organs were examined histologically in the present case, so this potential mechanism could not be verified. As supportive evidence for yew-induced myocardial necrosis in human beings, the patient described in a report of severe Taxus toxicity showed increased serum levels of cardiac troponins, known biomarkers of myocardial necrosis. 9
Because the clinical history suggests that exposure to other known causes of myocardial necrosis did not occur, and Taxus alkaloids were demonstrated in the rumen contents, the cause of putative myocardial necrosis and resultant fibrosis in the current case was considered to be ingestion of yew. While vitamin E and/or selenium status and exposure to ionophores were not directly tested in the present case, the authors consider the clinical history and demonstrated exposure to taxines strongly suggestive of previous yew-induced myocardial necrosis. Well-controlled experiments will be needed to confirm these observations and definitively link taxine exposure and myocardial necrosis. However, the current case suggests that yew toxicity can result in a prolonged clinical course and can cause severe histologic myocardial lesions.
Footnotes
Acknowledgements
The authors would like to thank the Indiana Animal Disease Diagnostic Laboratory Histopathology Section for processing and sectioning slides. This work was presented in partial format as a poster at the 52nd Annual Meeting of the American Association of Veterinary Laboratory Diagnosticians, October 7–14, 2009, San Diego, CA.
a.
Gold Shield Distributors, Hayward, CA.
b.
Thermo Fisher Scientific Inc., Waltham, MA.
c.
Act-1, Transgenomic Inc., Omaha, NE.
d.
Pierce Biotechnology Inc., Rockford, IL.
e.
HP-1, Agilent Technologies Inc., Santa Clara, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
