Abstract
A 7-y-old, intact male Alaskan Malamute was presented with a 3-mo history of stertor and epistaxis. Computed tomography of the skull revealed generalized loss of gas throughout both nasal passages with replacement by a soft tissue mass that traversed the cribriform plate. Histopathology revealed neoplastic neuroblast cells arranged in anastomosing cords, as well as separately located aggregates of ganglion cells. Both neoplastic cell populations demonstrated immunoreactivity to MAP-2, TuJ-1, and synaptophysin. Neuroblastic cells additionally exhibited punctate immunoreactivity to MCK and CK8/18. We document here both the positive neural immunohistochemical markers for this neoplasm, as well as propose possible histomorphologic variants.
Neuroblastic tumors (NTs; neuroblastoma, ganglioneuroblastoma [GNB], and ganglioneuroma) are rare neoplasms that have been reported in humans and a variety of veterinary species.1,3,10,19 NTs exist on a continuum with neuroblastomas being the most primitive; ganglioneuromas are the most differentiated. GNBs comprise a moderately differentiated group of NTs that vary in their composition and degree of cellular maturation. The hallmark histopathologic features of GNBs are the presence of both ganglion cells associated with neuronal processes (ganglioneuromatous component) and an undifferentiated neuroblastic component. 15
NTs have been identified in association with sympathetic nervous tissue, within the central nervous system, and within the superior nasal cavity of humans and veterinary species.3,4,7-9 Peripheral NTs are derived from neural crest cells that form the sympathetic nervous system, whereas olfactory NTs (ONTs) arise from basal progenitor cells of the olfactory epithelium that line the superior nasal cavity and cover the ethmoid cribriform plate.7,10 Within the human literature, evidence supporting ONTs as nosologically unrelated to peripheral NTs include gene expression profiling similarities between normal olfactory mucosa and olfactory neuroblastoma cells, and ONTs can be diagnosed in all age groups, compared to peripheral neuroblastomas, which are usually diagnosed in patients < 5 y old.1,5,7 Additionally, there is tangential laboratory evidence to support that ONTs possess a different cellular ontogeny than peripheral NTs. A study found that olfactory marker protein-SV40T oncogene transgenic mice developed NTs associated with the adrenal medulla and sympathetic ganglia as well as metastatic lesions; however, no neoplastic lesions were observed within the nasal cavity. 14 These findings suggest that ONTs and peripheral NTs might arise through separate tumorigenic mechanisms. The cell of origin for NTs within the CNS has not been well defined. In sum, olfactory NTs are thought to arise from basal progenitor cells of the olfactory epithelium and are considered to be neuroectodermal tumors.
Multiple canine ONT case reports and case series have been published; there have been no documented cases of canine olfactory ganglioneuromas, to our knowledge.2,3,16 A single case report has documented an olfactory ganglioneuroblastoma (OGNB) in a dog 12 in which postmortem nasal passage dissection revealed that the neoplasm was confined to the nasal cavity (i.e., the cribriform plate was intact). Immunohistochemistry (IHC) performed on the neoplasm revealed that the ganglion cells exhibited immunoreactivity for neuron-specific enolase (NSE) and neurofilament protein (NF), with rare spindle cells exhibiting immunoreactivity for glial fibrillary acidic protein (GFAP). These cells exhibited no immunoreactivity for chromogranin A, synaptophysin, or S100. Neuroblastic cells demonstrated immunoreactivity for cytokeratin, but the immunoreactivity to the remaining aforementioned markers were not discussed. No diagnostic imaging or autopsy was performed.
Here, we report a second case of an OGNB in a dog, in which both neoplastic cell populations demonstrated immunoreactivity for microtubule-associated protein-2 (MAP-2), class III beta-tubulin (TuJ-1), and synaptophysin, and paired with computed tomography (CT) of the skull. Multicytokeratin (MCK) and cytokeratin combination 8 and 18 (CK8/18) immunopositivity were also demonstrated in neuroblasts.
A 7-y-old, intact male Alaskan Malamute was presented to a private animal hospital because of a 3-mo history of stertor and epistaxis. On physical examination, there was decreased air flow through the right nasal passage and absent air flow through the left nasal passage. The remainder of the physical examination, as well as clinical pathology results, were within normal limits. CT of the skull revealed generalized loss of gas throughout both nasal passages with replacement by a soft tissue mass (up to 4.0 × 4.9 × 6.0 cm). The mass invaded the right nasal passage, dorsocaudal oral cavity, and traversed the cribriform plate and presphenoidal sinuses to infiltrate the olfactory bulb ventrally (Suppl. Fig. 4). No abnormal regional lymph nodes were detected. After the definitive diagnosis was made based on IHC, the patient did not receive interventional therapies, and was euthanized 2 mo thereafter. A postmortem examination was not performed.
Numerous incisional biopsies from the left nasal passage were collected, on 2 separate occasions, and submitted to the Colorado State University Veterinary Diagnostic Laboratory (Fort Collins, CO, USA). The samples were formalin-fixed, paraffin-embedded, processed routinely, and stained with hematoxylin and eosin. IHC for GFAP, synaptophysin, and MCK (PA0026, PA0299, PA0909, respectively; Leica) was performed on the samples using an automated staining system (Bond-Max; Leica). MAP-2 and TuJ-1 IHC were performed as described previously; CK8/18 was additionally performed at the University of California–Davis.3,6
Histologically, biopsy specimens varied significantly by region. A few tissue sections were regionally infiltrated by primitive polygonal-to-elongate neoplastic cells arranged in anastomosing cords, and occasionally formed disorganized radial arrangements around clear central spaces, surrounded by scant-to-moderate amounts of fibrovascular stroma (Fig. 1). These cells contained scant light-basophilic cytoplasm with variably distinct borders, ovoid-to-elongate to irregularly shaped nuclei with finely stippled chromatin, and 1 or 2 variably prominent basophilic nucleoli. Six mitotic figures were observed in two 400× high power fields. These cells were rarely individualized and were scattered throughout the surrounding submucosa. Neuronal soma embedded within wispy eosinophilic stroma regionally formed small, loosely arranged, and disorganized aggregates within other sections (Fig. 2). These neurons contained abundant cytoplasm with Nissl substance and a central to slightly eccentric round nucleus with one prominent nucleolus. Anisocytosis and anisokaryosis were moderate. Additional findings included mucosal ulceration and mixed inflammation. No neoplastic cells were observed within the first nasal biopsy specimens submitted, only abundant necrosis, histiosuppurative inflammation, and early granulation tissue.
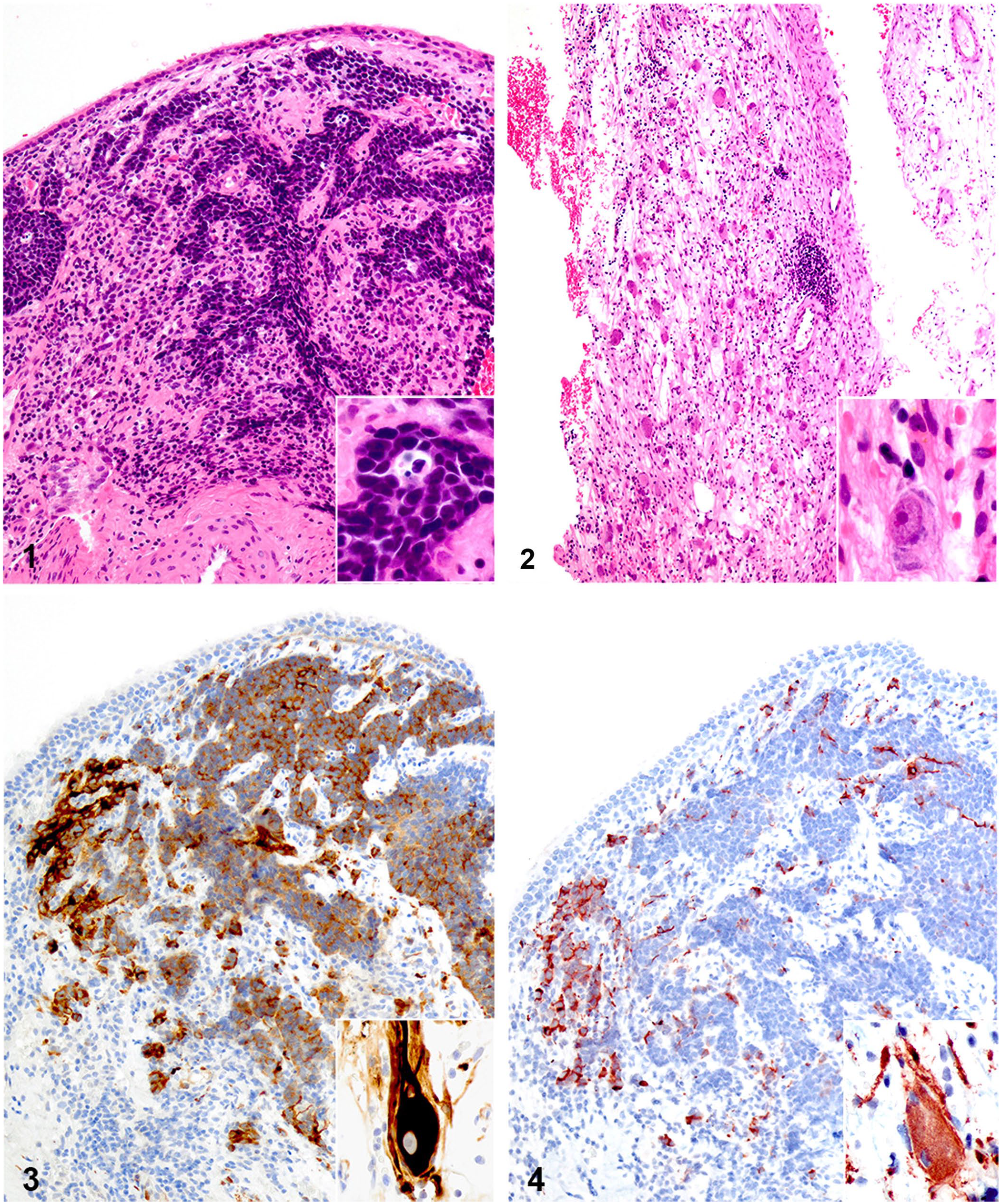
Olfactory ganglioneuroblastoma in the nasal cavity of a dog.
Neuroblasts exhibited positive cytoplasmic immunoreactivity to MAP-2 (Fig. 3). Fewer than 40% of neuroblasts also demonstrated positive cytoplasmic immunoreactivity to TuJ-1 (Fig. 4) and synaptophysin (Suppl. Fig. 1). Neuroblastic cells exhibited punctate cytoplasmic immunoreactivity for MCK and CK8/18 (Suppl. Figs. 2, 3). Ganglia exhibited positive cytoplasmic immunoreactivity for MAP-2 (Fig. 3, inset), TuJ-1 (Fig. 4, inset), synaptophysin (Suppl. Fig. 5), and GFAP. Neuroblasts were diffusely negative for GFAP; ganglia were diffusely negative for MCK.
Intranasal tumors are rare neoplasms in dogs, comprising ~1% of all canine neoplasms, and ONTs are an exceedingly rare subgroup of intranasal tumors. 3 No data exist on the incidence of canine ONTs, but olfactory neuroblastomas in humans comprises ~3% of all nasal cavity and paranasal sinus tumors. 7 Both neuroblastoma and GNB subtypes have been documented within the nasal cavity of humans and dogs.
Canine ONTs are reported to demonstrate positive immunoreactivity to NSE and S100, and less consistent immunoreactivity to NF, GFAP, synaptophysin, chromogranin A, TuJ-1, MAP-2, and MCK.2,3 NSE and S100 were not performed on our case given their lack of specificity for neural-derived cells, and alternative IHC markers were chosen instead.
ONTs arise from olfactory neuroepithelium, and can, therefore, exhibit positive immunoreactivity to neural and epithelial IHC markers. Two small-scale studies have documented that TuJ-1 and MAP-2 exhibit more consistent and higher specificity, respectively, for canine ONBs compared to other IHC markers.3,4 TuJ-1 is a neuron-specific class III beta-tubulin cytoskeletal protein that can be used as a marker for immature neurons; MAP-2 is a neuron-specific cytoskeletal protein that is used as a marker for mature neurons. Although neuroblasts and ganglia in our case respectively exhibited patchy and diffuse immunoreactivity to TuJ-1, both cell populations exhibited diffuse immunoreactivity to MAP-2. Moreover, both cell populations demonstrated immunoreactivity to synaptophysin, and neuroblasts also exhibited punctate immunoreactivity to MCK and CK8/18. MCK detects all cytokeratin intermediate filaments except CK18; cytokeratins 8 and 18 are usually complexed and expressed in simple epithelial cells. Up to one-third of human ONBs exhibit positive immunolabeling for cytokeratin, and one case report has specifically documented neuroblast immunopositivity to CK8 and CK18.7,11 A perinuclear and dot-like cytoplasmic cytokeratin immunopositivity expression pattern, as seen in our case, is a commonly reported feature in a variety of human neuroendocrine tumors, and has been reported in a human ONT.11,18
The arrangement of neuroblasts and ganglion cells in our case represented a micronodular histomorphology that was not co-located within the tissue sections. Even with IHC performed, this neoplasm could have been diagnosed incorrectly as an olfactory ganglioneuroma if multiple tissue sections had not been evaluated. Given the incisional nature of the specimens, distinction between a differentiating ONB (i.e., ONB with ganglioneuroblastic differentiation) and an OGNB remains speculative. However, a diagnosis of GNB was favored, given the well-differentiated ganglia histomorphology and preponderance of axonal processes. Other possible sinonasal neoplasms (e.g., ganglioneuroma, neuroendocrine carcinoma, undifferentiated carcinoma) were excluded as differentials based on the presence of both neuroblasts and ganglion cells. Rosettes (Homer Wright, Flexner–Wintersteiner) are a possible microscopic feature of neural-associated tumors, but these structures are not always present.7,10 Although we did not observe rosettes explicitly in our case, neuroblasts occasionally demonstrated attempts at Flexner–Wintersteiner rosette formation.
Not only are ONTs unrelated to peripheral NTs, but histologic distinction between NT subtypes may also appear as a diagnostic conundrum, especially when discerning differentiating NB from GNB subtypes. It has been well documented that peripheral NTs exist along a morphologic continuum, and human peripheral NBs can spontaneously mature into GNBs. 15 As such, the distinction between a peripheral differentiating NB and a GNB may be arbitrary, and a similar tumor maturation phenomenon may exist within the nasal cavity. Moreover, in human medicine, peripheral GNBs can exhibit classical nodular, atypical nodular variants, and intermixed patterns. 8 The incisional biopsy nature of our case prevents complete characterization of this neoplasm, but a similar spectrum of neuroblast and ganglia arrangements may occur in OGNBs. Future studies will be required to assess this observation because reported human and veterinary OGNBs are rare and lack a large sample size to characterize their spectrum of histomorphology. Until then, if a small sinonasal biopsy specimen is obtained for histopathology, veterinary pathologists should be aware that histomorphologic variants of OGNB might exist.
The most important prognostic factors for human ONBs are the Hyams and/or Morita modified grade and Kadish stage; however, these assessment schemes might not align with canine ONB behavior.7,16 These schemes only reference neuroblastomas, which precludes assigning a human-based grade and stage to our OGNB. Likewise, human peripheral GNB prognostic information has not been applied to human OGNBs. Although we cannot ascribe a grade to our case, the biologic behavior of our OGNB was documented via CT of the skull. CT is considered to be the ideal modality for NT assessment, given that it allows determination of the neoplasm dimensions, identification of the organ of origin, and assessment of invasion into adjacent tissues and vessels. 1 Neuroimaging of our case revealed neoplasm-associated turbinate bone destruction that extended through the skull base. ONTs are known to invade the cribriform plate, cranial vault, and brain, and similar locally aggressive biologic behavior was demonstrated in our case. 20
OGNBs are reported rarely in the human and veterinary literature, and few human cases of differentiating ONB have been documented.12,13,17 We have documented here the second case of a canine OGNB, with documentation of both neoplastic cell populations demonstrating positive immunoreactivity for neuronal and neuroendocrine IHC markers. Our case additionally documents punctate immunoreactivity of MCK and CK8/18 in a neuroectodermal tumor. The neoplasm histomorphology and positive IHC profile, in combination with the regional tumor bony lysis and invasion detected on CT, supports that our case is an OGNB with locally aggressive biologic behavior. Once the collective utilization of CT, histopathology, and IHC become standard tools in the veterinary armamentarium, future studies can be developed to evaluate possible OGNB histomorphologic patterns and to characterize the full range of its IHC profile and biologic behavior.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387211022864 – Supplemental material for Olfactory ganglioneuroblastoma in a dog: case report and literature review
Supplemental material, sj-pdf-1-vdi-10.1177_10406387211022864 for Olfactory ganglioneuroblastoma in a dog: case report and literature review by Ashley M. Romano and Chad B. Frank in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Dr. Gavin Olsen for obtaining the biopsy specimens; Drs. Andrew Miller and Kevin Woolard for their pathology expertise; and Maddi Funk and the CSU-VDL histopathology laboratory staff for their technical assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
