Abstract
Fowl adenovirus serotype 4 (FAdV4), the causative agent of hepatitis-hydropericardium syndrome (HPS), has caused major economic losses to the poultry industry worldwide. Although inactivated vaccines have been deployed widely against FAdV4, a DIVA (differentiating infected from vaccinated animals) test specific for FAdV4 has not been available. We synthesized an immunogenic peptide, corresponding to regions 66–88 aa of the 22K nonstructural protein of FAdV4, and used the peptide as coating antigen to develop an indirect ELISA for a DIVA test specific to FAdV4. Specificity analysis showed that the ELISA only reacted with sera against FAdV4, and not with sera against other pathogens tested. Moreover, the ELISA could effectively differentiate FAdV4–infected chickens from vaccinated chickens. In a test of sera from experimentally infected chickens, the ELISA had 95% and 85% concordance with an indirect immunofluorescence assay (indirect IFA) and a commercial ELISA, respectively, and the concordance was 80.5% between the ELISA and the indirect IFA in detecting clinical infection samples. Our peptide-based ELISA provides an efficient DIVA test for FAdV4 in clinical samples.
Fowl adenovirus (FAdV; Adenoviridae, Aviadenovirus) is classified into 5 species (A–E) with 12 serotypes (1–7, 8a, 8b, 9–11) based on its genomic sequence and antigenic characteristics.6,8,10 Chickens infected with FAdV may develop inclusion body hepatitis (IBH), hepatitis-hydropericardium syndrome (HPS), or gizzard erosion and ulceration (GEU).5,8-11 Of the 12 FAdV serotypes, FAdV4, which belongs to species FAdVC, is thought to be the causative pathogen for HPS, and it has caused major economic losses to the poultry industry worldwide.3,4,7-11,13-16,19,20 Inactivated FAdV4 vaccines have been deployed widely to control the disease. 13
Testing the level of antibody or neutralizing antibody against FAdV4 is an efficient way to evaluate the effectiveness of vaccination or the status of infection. Several methods for detection of antibodies specific to FAdV4 have been developed, mainly based on the detection of antibodies against the structural protein fiber.3,4,7,14-16 Given that flocks either vaccinated or infected with FAdV4 can efficiently generate antibodies against fiber, these fiber-based methods cannot distinguish vaccinated chickens from clinically infected chickens. Although serologic methods for differentiating infected from vaccinated animals (DIVA) for FAdV have been reported based on nonstructural protein (NSP) 33K or 100K of FAdV1, it remains to be determined whether these approaches can be used in the detection of FAdV4. 18 Given that FAdV4 is the dominant FAdV serotype in China, a DIVA test specific for FAdV4 is needed urgently. 2 The NSP 22K of FAdV4 is multifunctional in the regulation of late gene expression, encapsidation, and infectivity of the virus.1,12,17 Therefore, the 22K protein could be used for developing a DIVA test specific to FAdV4.
To generate a rapid and specific ELISA for a DIVA test for FAdV4 antibody, we analyzed the potential antigenic epitopes of NSP 22K of FAdV4 (strain SD15, isolated as described previously 20 ) through Protean DNASTAR (data not shown), and 6 antigenic peptides were synthesized (Synpeptide; Table 1). These peptides were coated on the ELISA plate individually to react with known positive and negative sera to test their reactive profile. Among them, the peptide 22K-4P_66-88aa–based ELISA showed the greatest positive-to-negative (P/N) ratio (Fig. 1). Therefore, we selected peptide 22K-4P_66-88aa as coating antigen in our ELISA. Based on checkboard titrations, we determined optimal concentrations of all required ELISA components. We used the peptide 22K-4P_66-88aa–based ELISA in the final protocol.
The sequences of synthetic peptides derived from the 22K protein of fowl adenovirus serotype 4.

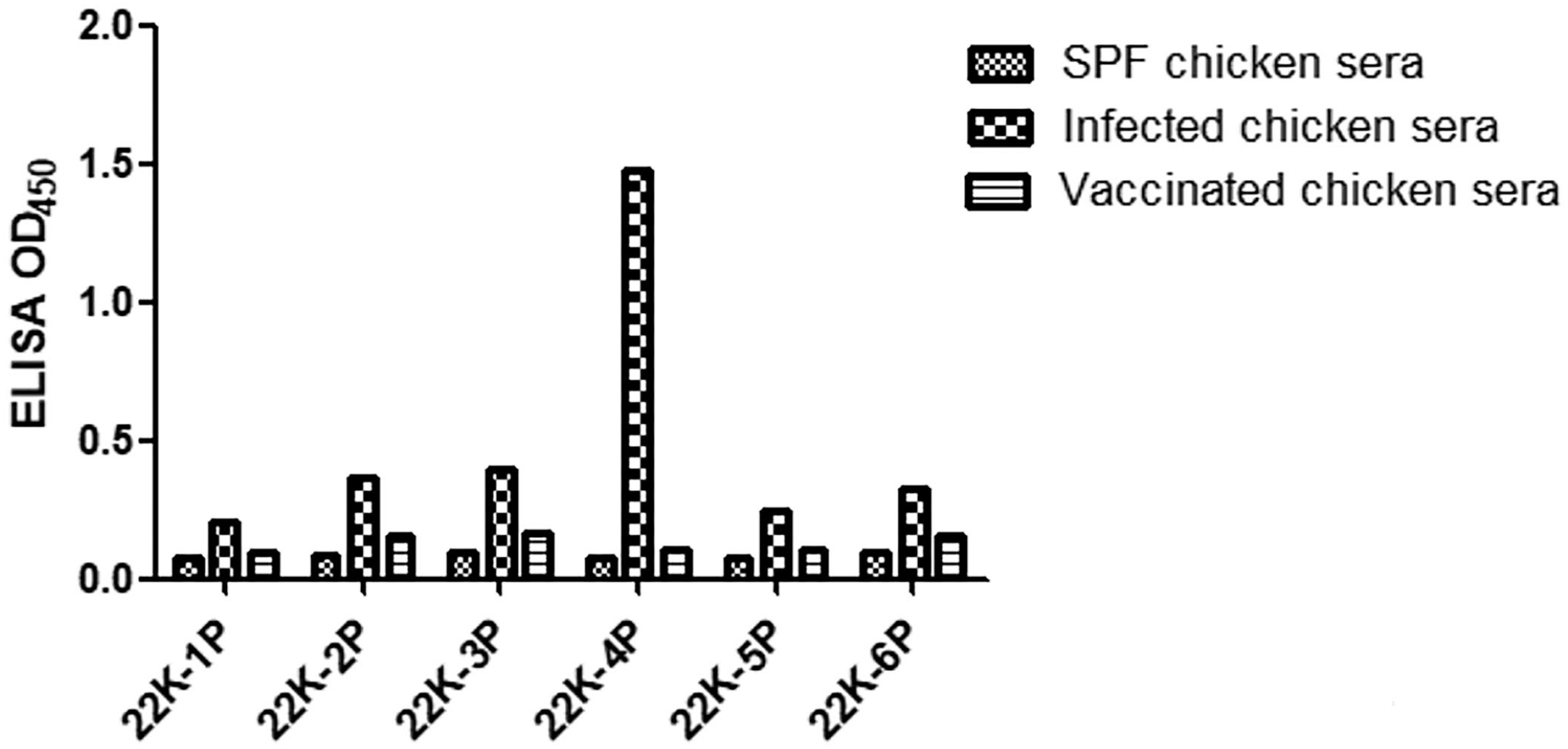
Peptide selection analysis. Peptides 22K-1P, -2P, -3P, -4P, -5P, and -6P were coated onto an ELISA plate individually to test SPF chicken sera, infected chicken sera, and vaccinated chicken sera.
In brief, 5 μg/mL of peptide 22K-4P_66-88aa in 0.1 M carbonate–bicarbonate buffer (pH 9.6) was coated into a 96-well ELISA plate at 100 μL per well at 4°C overnight, and then blocked with 380 µL of blocking buffer (2% bovine serum albumin [BSA] in polysorbate 20 in phosphate-buffered saline [PBST]) for 1 h at 37°C. After the plates were washed twice with PBST, 100 µL of positive and negative sera (diluted 1:100 in PBST containing 2% BSA) were added at 100 μL per well and incubated at 37°C for 45 min, followed by 3 washes with PBST. Then, 100 μL per well of rabbit anti-chicken antibody labeled with horseradish peroxidase (diluted 1:15,000 in PBST containing 1% skim milk) was incubated at 37°C for 1 h, followed by 3 washes with PBST. Color development was performed by adding 100 μL per well of 3,3′,5,5′-tetramethylbenzidine (TMB) single-component substrate solution (Solarbio) in the dark for 10 min, and the reaction was stopped by addition of 50 μL of 2 M H2SO4. The absorbance values were measured at 450 nm (optical density at 450 nm [OD450]) with an ELISA reader. The ELISA cutoff was set at 0.209 by calculating the arithmetic mean plus 3 SDs of 40 specific pathogen–free [SPF] sera. OD450 values ≥0.209 were regarded as positive.
To evaluate the specificity of our ELISA, we tested the ELISA with polyvalent sera against FAdV4 and FAdV8a; monovalent sera against FAdV serotypes 1–7, 8a, 8b, 9–11, influenza A(H9N2) virus (AIV), chicken anemia virus (CAV), Marek disease virus (MDV; Mardivirus), avian reticuloendotheliosis virus (REV; Reticuloendotheliosis virus), infectious bronchitis virus (IBV; Avian coronavirus), and egg-drop syndrome virus (EDSV; Duck atadenovirus A), and sera from SPF chickens. The above sera were either preserved in our laboratory or kindly provided by Dr. Junping Li (China Institute of Veterinary Drugs Control). The ELISA reacted only with the polyvalent and monovalent sera against FAdV4, not with sera against other pathogens tested, demonstrating the specificity of our ELISA for FAdV4 (Fig. 2).

Specificity assay of the 22K-4P_66-88aa–based ELISA. Polyvalent sera against FAdV4 and FAdV8a, and monovalent sera against FAdV serotypes 1–7, 8a, 8b, 9–11, influenza A(H9N2) virus (AIV), chicken anemia virus (CAV), Marek disease virus (MDV), avian reticuloendotheliosis virus (REV), infectious bronchitis virus (IBV), and egg-drop syndrome virus (EDSV), and sera from the SPF chicken were used to evaluate the specificity of the ELISA. The OD450 value of the polyvalent and monovalent chicken sera against FAdV4 was > 0.6; the values were <0.209 against other viruses tested. Bars = SEM.
To test the sensitivity of our ELISA, we simultaneously tested 4 positive sera against FAdV4 (sera 1–4) with serial dilutions (1:100, 1:200, 1:400, 1:800, 1:1,600, 1:3,200, 1:6,400, 1:12,800) by ELISA and indirect immunofluorescence assay (indirect IFA). The minimum titers of positive sera 1–4 for detection by ELISA were 1:3,200, 1:800, 1:3,200, 1:3,200, respectively, whereas those by indirect IFA were 1:1,600, 1:1,600, 1:3,200, 1:3,200, respectively (Table 2). The result demonstrated that our ELISA had sensitivity comparable to indirect IFA. Notably, the OD450 values from positive sera showed an efficient dose-dependent effect, further highlighting the specificity of our ELISA.
Sensitivity of the 22K-4P_66-88aa–based ELISA for detection of antibody against fowl adenovirus serotype 4 in 4 chickens (1–4).

Results are presented as positive (+) or negative (–). Numbers in parentheses are OD450 values. The cutoff value is 0.209.
To evaluate the feasibility of the established ELISA for DIVA test, sixty 2-wk-old SPF chickens were divided into 3 groups. Chickens in the infection group (n = 20) were inoculated intramuscularly with 0.2 mL of F12/DMEM (Gibco) containing 1 × 104 TCID50 FAdV4 (strain SD15) virus per chicken. Chickens in the vaccinated group (n = 20) were immunized intramuscularly with an oil-emulsion, inactivated FAdV4 vaccine candidate using whole virus FAdV4 from China. The formaldehyde-inactivated FAdV4 antigen solution was emulsified with oil adjuvant at a ratio of 25:75 (w/w); the final dose of the vaccine was 5 × 106 TCID50 per chicken. Chickens in the control group were inoculated with 0.2 mL of F12/DMEM. The blood of all groups was collected at 0, 7, 14, 21, 28 d post-inoculation (dpi). The SPF chickens and vaccine candidate used above were from Sinopharm Yangzhou Vac Biological Engineering. All animal experiments were under the guidance of the Animal Committee at Yangzhou University in China, approval protocol SYXY-19.
The sera from 5 chickens infected with FAdV4 at different dpi were detected by our ELISA (Fig. 3). The antibodies against 22K of FAdV4 could be detected as early as 7 dpi, peaked at 14 dpi, and then declined gradually, indicating the ability of the ELISA to detect early infection of FAdV4. Of the 60 experimental sera (20 from chickens vaccinated with an inactivated FAdV4 vaccine candidate, 20 from chickens infected with FAdV4 at 14 dpi, and 20 from SPF chickens) tested in the ELISA, only the 20 sera from chickens infected with FAdV4 were positive in the ELISA, whereas the other 40 sera tested were all negative (Fig. 4). To confirm the data from our peptide-based ELISA, these 60 experimental sera samples were also tested by indirect IFA and a commercial ELISA (BioChek). In the indirect IFA, the LMH (leghorn male hepatoma) cells infected with FAdV4 were used as an antigen. In the commercial ELISA, the purified FAdV1 viruses were used as coating antigen. In 20 positive sera in the peptide-based ELISA, 19 of 20 (95%) and 17 of 20 (85%) samples were positive in the indirect IFA and the commercial ELISA, respectively. These negative sera in our peptide-based ELISA, which were from the chickens vaccinated with inactivated FAdV4, were all positive by indirect IFA, whereas the commercial ELISA detected 16 of 20 (80%) positive (Suppl. Table 1). Our peptide-based ELISA can thus be used to perform a DIVA test for FAdV4 antibody.
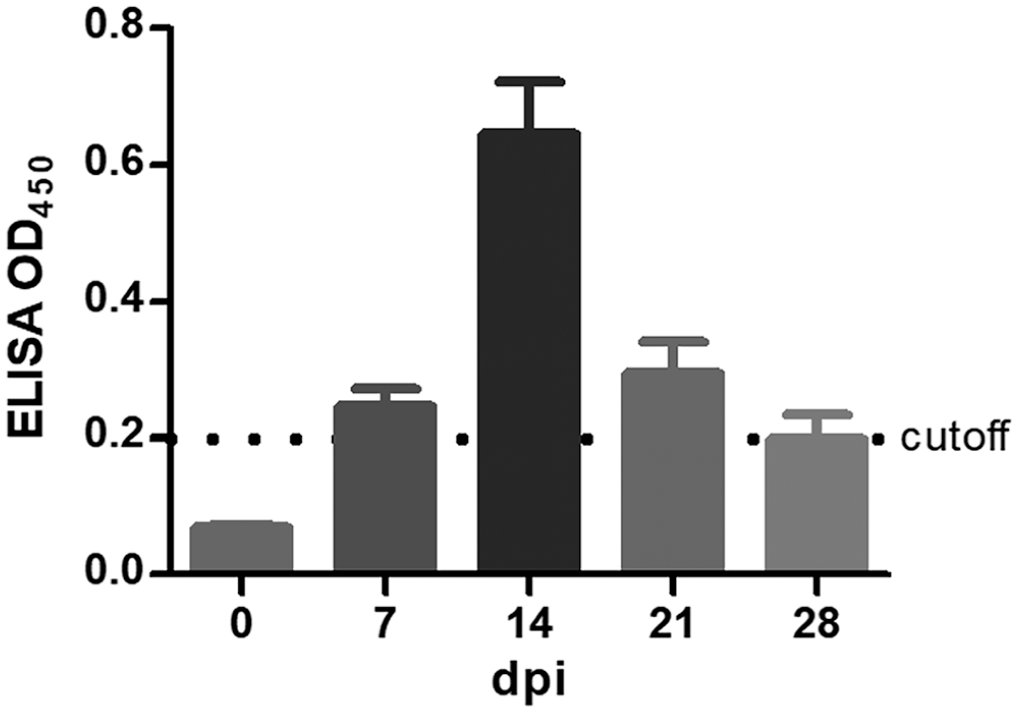
ELISA detection of antibody at 0, 7, 14, 21, and 28 d post-infection (dpi) from chickens (n = 5) infected experimentally with FAdV4. Sera collected at 7, 14, 21, and 28 dpi were all positive in the ELISA; sera collected at 0 dpi remained negative. Bars = SEM.

Antibody detection by the 22K-4P_66-88aa–based ELISA performed on experimental samples. Sera from 20 FAdV4-infected chickens at 14 dpi, 20 SPF chickens, and 20 vaccinated chickens were tested by the ELISA. The FAdV4-infected chicken sera were all positive in the ELISA; sera from SPF and vaccinated chickens remained negative. The horizontal line in the box represents the median value, and the low and upper edge of the box mark the 25th and 75th percentiles, respectively. The whiskers extending from the box show the highest and lowest values.
To evaluate the practical applicability of our ELISA in clinical samples, sera (n = 266) were collected from commercial chicken flocks (Jiangsu Province, China) and tested. Sera were randomly collected from chickens on farms naturally infected with FAdV4 (n = 159) and from chickens on farms vaccinated with the inactivated FAdV4 vaccine candidates (n = 107). One hundred twenty-one of 159 sera from the infected farms and 3 of 107 sera from the vaccinated farms were positive in our ELISA (Table 3; Fig. 5). We identified as positive by our ELISA and indirect IFA 121 of 159 (76.1%) and 137 of 159 (86.2%) of clinical infection sera and 3 of 107 (2.8%) and 97 of 107 (90.7%) of clinical vaccination sera, respectively (Table 3). Our ELISA and indirect IFA agreed on the result as “positive” in 113 of 159 (71.1%) or “negative” in 15 of 159 (9.4%) clinical infection sera. Thus, their concordance is P0 = 71.1 + 9.4% = 80.5%. Our ELISA only detected antibodies in samples collected from infected farms, but rarely from vaccinated farms. Given that the indirect IFA using FAdV4 as antigen could react with antibodies against all 12 serotypes of FAdV, the clinical infection sera that were positive in indirect IFA but negative in our ELISA may have carried antibodies against other serotypes of FAdV. The high correlation for the positive sera further highlights the specificity of our ELISA as a DIVA test.
Comparison of the 22K-4P_66-88aa–based ELISA and indirect IFA for detecting fowl adenovirus serotype 4 clinical samples.

NA = not applicable. Results are presented as positive (+) or negative (–).
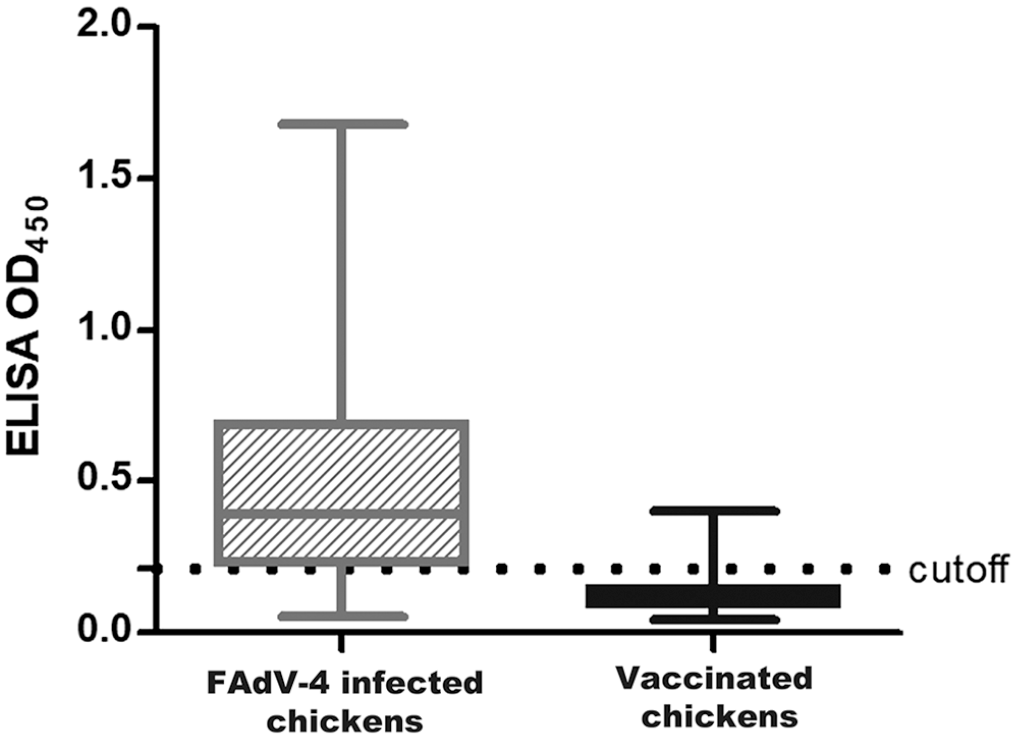
Detection result from the ELISA performed on clinical samples. Sera from 159 chickens naturally infected with FAdV4 or from 107 vaccine candidates that had been vaccinated with the inactivated FAdV4 were tested by the ELISA; 121 of 159 sera from infected farms and 3 of 107 sera from the vaccinated farm were positive by the 22K-4P_66-88aa–based ELISA. The horizontal line in the box represents the median value, and the low and upper edge of the box mark the 25th and 75th percentiles, respectively. The whiskers extending from the box show the highest and lowest values.
The NSP 22K is coded by the late gene 4 (L4) region of FAdV4 and released only during viral replication, playing vital roles in the regulation of late gene expression and encapsidation of the virus.1,12 In addition, the 22K protein may contribute to adenovirus-induced cell death by regulating expression of ADP as an integral component of crucial aspects of infection. 17 Sequence assay revealed that peptide 22K-4P_66-88aa is highly invariant in different FAdV4 strains (data not shown). In the comparison analysis, our peptide-based ELISA showed high compatibility with the indirect IFA and the commercial ELISA for samples from chickens experimentally infected with FAdV4 and from SPF chickens, and had high concordance with the indirect IFA in detecting clinical infection sera. Our peptide-based ELISA could be used as a DIVA test to distinguish FAdV4 infection from an inactivated virus-based vaccination response.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387211005749 – Supplemental material for An efficient peptide-based ELISA for differentiating fowl adenovirus 4–infected chickens from vaccinated chickens
Supplemental material, sj-pdf-1-vdi-10.1177_10406387211005749 for An efficient peptide-based ELISA for differentiating fowl adenovirus 4–infected chickens from vaccinated chickens by Songhua Xie, Qiuping Shen, Wei Zhang, Weikang Wang, Quan Xie, Tuofan Li, Zhimin Wan, Hongxia Shao, Aijian Qin and Jianqiang Ye in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Dr. Jianjun Zhang (Sinopharm Yangzhou VAC Biological Engineering) for kindly assisting us to prepare chicken sera against fowl adenovirus, and Dr. Junping Li (China Institute of Veterinary Drugs Control) for kindly providing the positive sera against FAdV2, 3, 7, 8b, and 11.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our study was supported by Jiangsu Agricultural Science and Technology Innovation Fund (CX(19)3026), the Key Research & Development (R&D) Plan in Yangzhou City (YZ2020052), Key Laboratory of Prevention and Control of Biological Hazard Factors (Animal Origin) for Agrifood Safety and Quality (26116120), and the Priority Academic Program Development (PAPD) of Jiangsu Higher Education Institutions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
