Abstract
A young-of-the year, female raccoon was presented with clinical signs of weakness and tremors. The raccoon was euthanized, and autopsy findings included poor body condition, diffuse lymphadenopathy, and pale, firm kidneys with petechial hemorrhages throughout the renal cortex. Histologic lesions included systemic fibrinoid vascular necrosis and severe renal lesions, including lymphoplasmacytic interstitial nephritis and fibrinosuppurative glomerulonephritis. Inflammatory vascular lesions were also present within the uvea, heart, lymph nodes, and the lamina propria of the gastric wall. Ancillary testing was negative for Borrelia burgdorferi, Leptospira sp., Aleutian disease virus, canine distemper virus, feline coronavirus, porcine circovirus 2, and rabies virus. Transmission electron microscopy revealed large numbers of ~1.3 × 0.35 µm bacterial rods surrounded by a trilaminar cell wall located within the glomeruli and associated with aggregates of fibrin and vascular damage. Analysis of partial citrate synthase gene and 16S-23S ribosomal RNA intergenic spacer region sequences from kidney tissue confirmed that the organism was a Bartonella spp. that was related to numerous Bartonella spp. from shrews in Europe. This group formed a sister clade to the genetically diverse Bartonella taylorii group that has been reported from a wide range of Eurasian rodent and flea species.
Keywords
In the fall of 2012, a young-of-the-year, female raccoon (Procyon lotor) was found weak and experiencing tremors on private property on Prince Edward Island (PEI), Canada [46.3368324, -63.0612442]. It was euthanized and submitted to the Canadian Wildlife Health Cooperative (CWHC) for autopsy. The raccoon was in poor body condition with generalized lymphadenopathy and had diffusely pale, firm kidneys with petechial hemorrhages and 1–2 mm pale cortical foci. The heart had multiple linear, tan streaks throughout the myocardium. Representative tissue samples were fixed in 10% neutral-buffered formalin, processed routinely, and sections stained with hematoxylin and eosin.
A female Ixodes muris tick removed from the left medial canthus was examined by PCR for Borrelia burgdorferi and Anaplasma phagocytophilum at the National Microbiology Laboratory (Winnipeg, MB, Canada). DNA was extracted (DNeasy 96 tissue kits; Qiagen). 6 A previously described duplex real-time PCR (rtPCR) assay for Borrelia spp. (23S rRNA target) and A. phagocytophilum (msp2 target) was used to screen the tick DNA extract. 4
Immunohistochemical (IHC) testing for Bartonella was performed on 4-µm sections of formalin-fixed, paraffin-embedded kidney sections using a 1:100 dilution of Bartonella henselae mouse monoclonal antibody (Biocare Medical), biotinylated anti-mouse antibody (Vector Laboratories), streptavidin conjugated to horseradish peroxidase (Biocare Medical), and diaminobenzidine (Agilent) as chromogen. Slides were counterstained with Gill II hematoxylin (Leica).
Routine aerobic bacterial culture of liver and kidney samples was performed using standard microbiologic procedures. Samples were plated on Columbia agar (Oxoid), with 5% defibrinated sheep blood, and MacConkey agar (Alere). 18 A PCR for Aleutian mink disease virus (AMDV; Carnivore amdoparvovirus 1) was performed by Diagnostic Services at the Atlantic Veterinary College (Charlottetown, PEI,
Canada) using a modified traditional PCR procedure with extraction of tissues (DNA mini kit; Qiagen) and amplification using primers and thermocycling parameters described previously. 12 PCR tests for Leptospira spp. and feline coronavirus (FCoV; Alphacoronavirus 1) were performed by the Animal Health Centre (Abbotsford, BC, Canada). The FCoV PCR is a conventional PCR with in-house primers targeting open-reading frame (ORF) 3c with an expected amplicon size of 901 bp. The Leptospira spp. PCR is a rtPCR based on a published protocol. 17 The limit of detection was determined as 3 copies of the Leptospira spp. genome. Conventional PCR assays for canine distemper virus (CDV; Canine morbillivirus) and porcine circovirus 2 (PCV-2) were performed using standard protocols by the University of Georgia Diagnostic (Athens, GA) and Infectious Disease (Athens, GA) Laboratories, respectively. IHC staining for Leptospira spp. and PCV-2 was conducted at Prairie Diagnostic Services (Saskatoon, SK, Canada) using a technique adapted for an automated slide stainer (Code-On histomatic stainer; Fisher Scientific) as described previously. 8 Protease XIV (MilliporeSigma) digestion was used for epitope retrieval, and the polyclonal primary antibodies (Rabbit anti-PCV2, Dr. G. Allan, Belfast, N. Ireland; rabbit anti-Leptospira [multivalent], NVSL, Ames IA) were incubated overnight at 4°C. Binding of the primary antibodies was detected using biotinylated goat anti-rabbit immunoglobulins (Vector Laboratories) and an avidin-biotin immunoperoxidase complex reagent, with 3,3’-diaminobenzidine tetrahydrochloride (DAB; Electron Microscopy Science) as the chromogen. Testing for rabies virus (RABV; Rabies lyssavirus) was performed by the CWHC via the direct rapid IHC test on a brain impression smear (Centers for Disease Control and Prevention, Atlanta, GA).
The renal interstitium was expanded by fibrous connective tissue with lymphoplasmacytic and eosinophilic infiltrates. Renal tubules were dilated, lined by attenuated epithelial cells, and filled with eosinophilic fluid, aggregates of neutrophils, and cellular debris. Glomeruli were effaced by fibrin admixed with cellular debris and neutrophils and replaced by Splendore-Hoeppli–like material associated with gram-negative coccobacilli. Basement membranes of Bowman capsules were moderately thickened. Blood vessels were surrounded by concentric layers of fibrillar material within the tunica muscularis; the tunica adventitia was infiltrated heavily by leukocytes and pyknotic nuclear material (leukocytoclasia). Warthin–Starry and Grocott methenamine silver staining of the kidney did not reveal additional organisms. Vascular lesions were observed in the urinary bladder, spleen, lung, uvea, adrenal glands, liver, uterus, mesenteric lymph node, and gastric tunica muscularis. Polyphasic necrosis associated with foci of hemorrhage, mineralization, inflammatory infiltrates, and karyorrhectic debris was present within the myocardium. No organisms were detected in the heart with Warthin–Starry staining. Additional microscopic findings included hepatic bile stasis and hemosiderin-laden macrophages within alveolar spaces.
There was no growth from the liver and kidney after 48 h of aerobic incubation at 35°C. IHC staining did not detect PCV-2, rabies virus, or Leptospira spp. antigen. All other assays for the range of other pathogens detailed above were negative. The results of the duplex PCR assay for B. burgdorferi and A. phagocytophilum on the tick were also negative. No additional ectoparasites had been detected upon clinical examination or autopsy.
IHC staining for B. henselae detected faint positive staining of coccobacilli within glomeruli. Transmission electron microscopy revealed large numbers of 1.3 × 0.35 µm bacteria with trilaminar cell walls within glomeruli (Fig. 1). These organisms appeared to be aggregated at a focal region of damage to the endothelium and were associated with aggregates of fibrin (thin strands of electron-dense fibrillar material) and remnants of damaged endothelial cells.
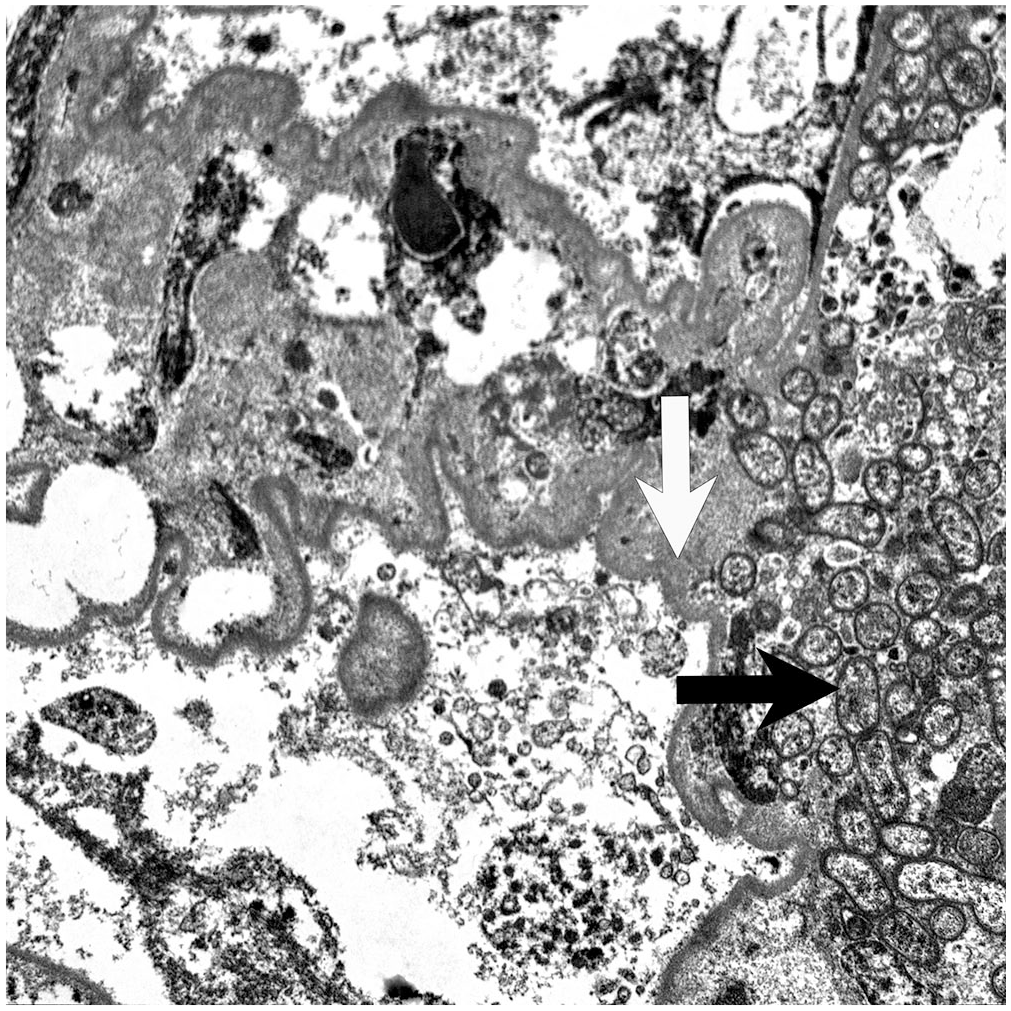
Transmission electron microscope image of a capillary in the kidney of a raccoon from Prince Edward Island with aggregates of bacteria (interpreted to be Bartonella taylorii) with trilaminar cell walls (black arrow) associated with disorganized remnants of endothelial cells and deposition of fibrin as thin strands of electron-dense fibrillar material associated with a thickened basement membrane (white arrow).
Bartonella DNA was detected from renal tissue by amplification of the partial citrate synthase (glta) gene (727 bp) using primers CS-443F and CS1210r as described previously.3,16 The partial 16S-23S ribosomal RNA intergenic spacer region (ISR; 710 bp) was amplified using primers QHVE-1 and QHVE-3. 11 Amplicons were bidirectionally sequenced at the University of Georgia Genomics Facility (Athens, GA). Chromatograms were analyzed using Geneious (Biomatters), and consensus sequences were aligned in MEGA v.7.0.26 14 with related sequences in GenBank. The glta sequence had 95% identity to Bartonella spp. from shrews (Sorex spp.) from Europe and 93–95% identity to numerous B. taylorii strains from voles (Microtus and Myodes [Clethrionomys] spp.) and field mice (Apodemus spp.) from Europe and Asia.
A phylogenetic neighbor-joining analysis using a Jukes–Cantor model showed that the raccoon Bartonella sp. glta gene sequence was present within a clade with Bartonella from Sorex shrews, with this group clustering with B. taylorii strains 3 (Suppl. Fig. 1). The partial 16S-23S ribosomal RNA ISR sequence had 79–80% identity to several B. taylorii strains, and phylogenetic analysis supported the glta analysis (Suppl. Fig. 2). Additional sampling of shrews, voles, and rodents is needed to better understand the diversity of Bartonella spp. in this group as well as host-specificity and distribution, especially because the Sorex- and raccoon-infecting Bartonella sequences clustered separately from the vole- and rodent-infecting Bartonella sequences.
Bartonella spp. have been identified in raccoons,10,11 but, to our knowledge, lesions have not been described. The Bartonella spp. detected in our case was related genetically to Bartonella spp. from shrews and rodents, specifically the B. taylorii complex associated with Eurasian fleas and rodents 15 as well as stray dogs in Thailand. 1 Although the origin of this Bartonella is unknown, there are numerous potential hosts endemic to PEI, including Sorex cinereus, S. fumeus, Blarina brevicauda, Microtus pennsylvanicus, and Myodes (Clethrionomys) gapperi, 9 none of which have been tested for Bartonella on PEI.
Bartonella spp. are fastidious hemotropic, gram-negative, aerobic bacilli in the class Alphaproteobacteria. 18 Acute and chronic renal lesions, consistent with a longstanding infection with possible acute multi-organ failure was associated with this bacterium in our case. However, antemortem blood chemistry was not available to confirm acute renal failure. The widespread vascular lesions may be a result of bacterial injury of endothelium, associated with acute renal failure, or represent immune complex deposition. The ultrastructural morphology of the bacterium is consistent with previous reports in human and veterinary patients infected with B. henselae, including bacterial aggregation along the vascular endothelium.5,13 Acute renal failure and membranoproliferative glomerulonephritis caused by immune complex deposition have been reported in cases of bartonellosis in humans, often with concurrent endocarditis. 7 Although our raccoon had concurrent myocarditis and microscopic evidence of heart failure, the relationship to the renal lesions is unknown.
Diagnosis of bartonellosis can be challenging given that Bartonella spp. are fastidious, and specialized techniques (e.g., molecular) are often necessary for detection. 19 Potentially novel rodent-associated Bartonella spp. have been associated with disease in human cases. In one report, B. henselae infections in a dog owner and her dogs were associated with the presence of raccoons on the property. 2 Our case was a clinical form of Bartonella-associated disease in a free-ranging raccoon on PEI. Thorough ancillary testing for a number of coinfections or differential diagnoses (AMDV, FCoV, CDV, PCV-2, RABV, as well as Borrelia burgdorferi, Leptospira spp., and routine bacteria) was attempted without any positive results. Concurrent immunosuppression associated with the young age of the animal or previous infection (e.g., parvovirus) cannot be ruled out as potential cofactors associated with morbidity in our case.
Previous detections of the B. taylorii complex bacteria have been outside of North America. Given the ubiquitous nature of raccoons in urban environments, veterinary and human health professionals should be aware of this finding. Because the bacterium is difficult to detect with routine bacterial culture, considerations for additional molecular testing could be considered in future cases of raccoons or other species with systemic vascular lesions without an obvious etiology. Given the tropism of Bartonella for endothelium, blood culture in enriched media in live patients is performed in human medical cases of suspected bartonellosis, 7 and could be considered in veterinary cases with a similar presentation (i.e., unexplained multisystemic illness).
Supplemental Material
sj-pdf-1-vdi-10.1177_1040638720988515 – Supplemental material for Lesions associated with Bartonella taylorii–like bacterium infection in a free-ranging, young-of-the-year raccoon from Prince Edward Island, Canada
Supplemental material, sj-pdf-1-vdi-10.1177_1040638720988515 for Lesions associated with Bartonella taylorii–like bacterium infection in a free-ranging, young-of-the-year raccoon from Prince Edward Island, Canada by Heather Fenton, Scott McBurney, Elizabeth J. Elsmo, Christopher A. Cleveland and Michael J. Yabsley in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Robbin Lindsay, Darlene Weeks, Marion Desmarchelier, Mary Ard, and David Dowling, Animal Health Centre, Abbotsford, BC, Canada; the Public Health Agency of Canada; Forests, Fish, and Wildlife Prince Edward Island; and the histotechnology laboratories at the Atlantic Veterinary College and the University of Georgia.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
