Abstract
Two 6-month-old raccoon kits, which had been rescued and fostered in preparation for return to the wild, became acutely ill and died 3 weeks before scheduled release. At necropsy, the kits had grossly enlarged livers and spleens, diffusely consolidated lungs, and generalized lymphadenopathy. Histologically, extensive infiltrates of macrophages containing yeast organisms were identified in lung, liver, kidney, spleen, lymph nodes, intestinal tissues, brain, adrenal gland, bone marrow, and thymus of both animals. Histiocytic inflammation with accompanying fibrosis was widespread, with necrotic foci evident in lungs, spleen, and intestinal sections. Fungal organisms were observed on sheep blood agar plates; however, repeated subcultures to fungal media designed to induce conidial structures for fungal identification were unsuccessful. Partial DNA sequencing of the 28S ribosomal RNA gene of the blood agar isolate identified 100% homology with Ajellomyces capsulatus (anamorphic name Histoplasma capsulatum). The kits were rescued and fostered in the San Francisco Bay area and it is likely that the exposure to H. capsulatum occurred in this area. Histoplasma sp. infection in wild mammal species is often used as an indication of spore contamination of a geographic region. Northern California is not known to be an endemic region for H. capsulatum, which is not a reportable disease in this state. The presence of severe, disseminated disease and the need for molecular identification associated with the isolate from a nonendemic region identified in the present report may indicate genetic adaptation and altered characteristics of this agent and may warrant further investigation.
Keywords
Histoplasma capsulatum, the causative agent of histoplasmosis, is a fungal agent that thrives in high-nitrogen soil under humid environmental conditions. Exposure is often associated with excavation or disruption of soil containing large amounts of bird or bat feces in endemic geographic locations. Wild mammal species such as raccoons (Procyon lotor) have been proposed as sentinels for agents of zoonotic disease; however, the most recent published report of H. capsulatum disease in raccoons was published in 1967. 16 Traditionally, H. capsulatum identification requires specialized culture conditions to induce sexual structures needed to differentiate this agent from other pathogenic fungi. The present report describes a case of severe, disseminated H. capsulatum in juvenile raccoons in California that required molecular diagnostics for fungal identification.
Three juvenile raccoons presented to a wildlife rescue and rehabilitation center in northern California in September 2012. The raccoons appeared to be less than 2 months old (not yet weaned) and were placed with a trained and experienced foster volunteer for nursing care in preparation for release back to the wild. They were housed in a large cage within a barn that had been used for previous fostered raccoons. The raccoons were vaccinated with canine distemper–infectious canine hepatitis–canine adenovirus type 2–canine parainfluenza–canine parvovirus a -modified live vaccine and treated with topical parasiticides. b All 3 animals ate well and were growing, and all were scheduled to be released in February 2013. On January 3, 2013, 1 raccoon kit became lethargic, pale, and weak; began vocalizing; and died within 24 hr. No necropsy was performed. The second and third kits demonstrated similar symptoms and died on January 13, 2013.
Raccoons 2 and 3 were submitted for necropsy. Both were in good postmortem condition with mucus visible on the conjunctival mucosa and variable amounts of fecal staining around the perineum. On gross necropsy, the kits had abundant subcutaneous and abdominal fat depots with severe abdominal and thoracic lymphadenopathy. Affected lymph nodes were light tan to gray with fine granular stippling and a firm texture throughout. Additionally, both animals had enlarged, firm livers with enhanced lobular patterns and infrequent pinpoint tan-white foci. The lungs had a diffuse fine tan-red-brown mottled appearance with a marked diffuse consolidated to regionally fleshy texture (Fig. 1). The spleens were also enlarged with widespread pinpoint tan foci, and a single larger discreet, 6–8 mm, tan-yellow fleshy nodule was present at 1 pole of the spleen in raccoon 3. Gastrointestinal tracts were essentially empty. Remaining organs were grossly unremarkable.
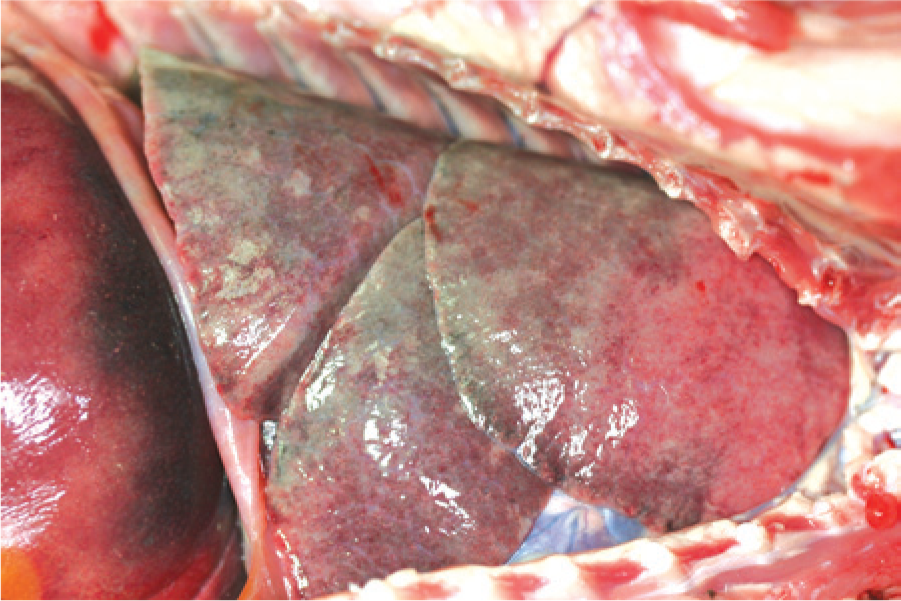
Gross image of the thorax of raccoon 2. The lung lobes are diffusely consolidated and do not collapse, with very fine dimpled appearance to the surface. The lobes were a slightly dark red-pink. The small random pale foci represent small pockets of trapped air within the parenchyma.
Histologically, throughout multiple tissues there were extensive diffuse infiltrates of macrophages containing large numbers of round to oval, 3–5 µm in diameter organisms with an eccentrically placed nucleus and clear cytoplasm. In the liver, histiocytic infiltrates filled with these organisms were present in portal tracts and the perisinusoidal spaces with accompanying moderate to marked bridging portal fibrosis (Fig. 2A). In the lung, diffuse histiocytic infiltrates, with and without detectable intracytoplasmic organisms, accompanied by small numbers of lymphocytes, expanded alveolar septa and filled the alveolar spaces effacing large areas of parenchyma with random multifocal necrosis and variable alveolar septal fibrosis. Multifocally, nodular aggregates of macrophages containing the organisms were adhered to endothelium and partially occluded larger pulmonary vessels. In lymph nodes and the spleen, there were similar prominent diffuse infiltrates of large macrophages, many containing the organisms, expanding the lymph node cortices and medullary and splenic cords, respectively, with mild diffuse interstitial fibrosis and random, variably sized foci of necrosis. The large splenic nodule noted grossly consisted of a large focus of hemorrhage and necrosis. In the intestines, diffuse histiocytic infiltrates, containing myriads of organisms, expanded the small intestinal lamina propria and to a lesser extent the colonic mucosa often with accompanying submucosal fibrosis. Small random focal interstitial histiocytic infiltrates with fibrosis were also seen in the kidneys, primarily in glomeruli with intracapillary monocytes containing the organisms. Aside from these widespread histiocytic infiltrates there also was marked thymic lymphocytic atrophy. Organism-laden macrophages were also present in smaller numbers in brain, adrenal gland, bone marrow, and thymus.
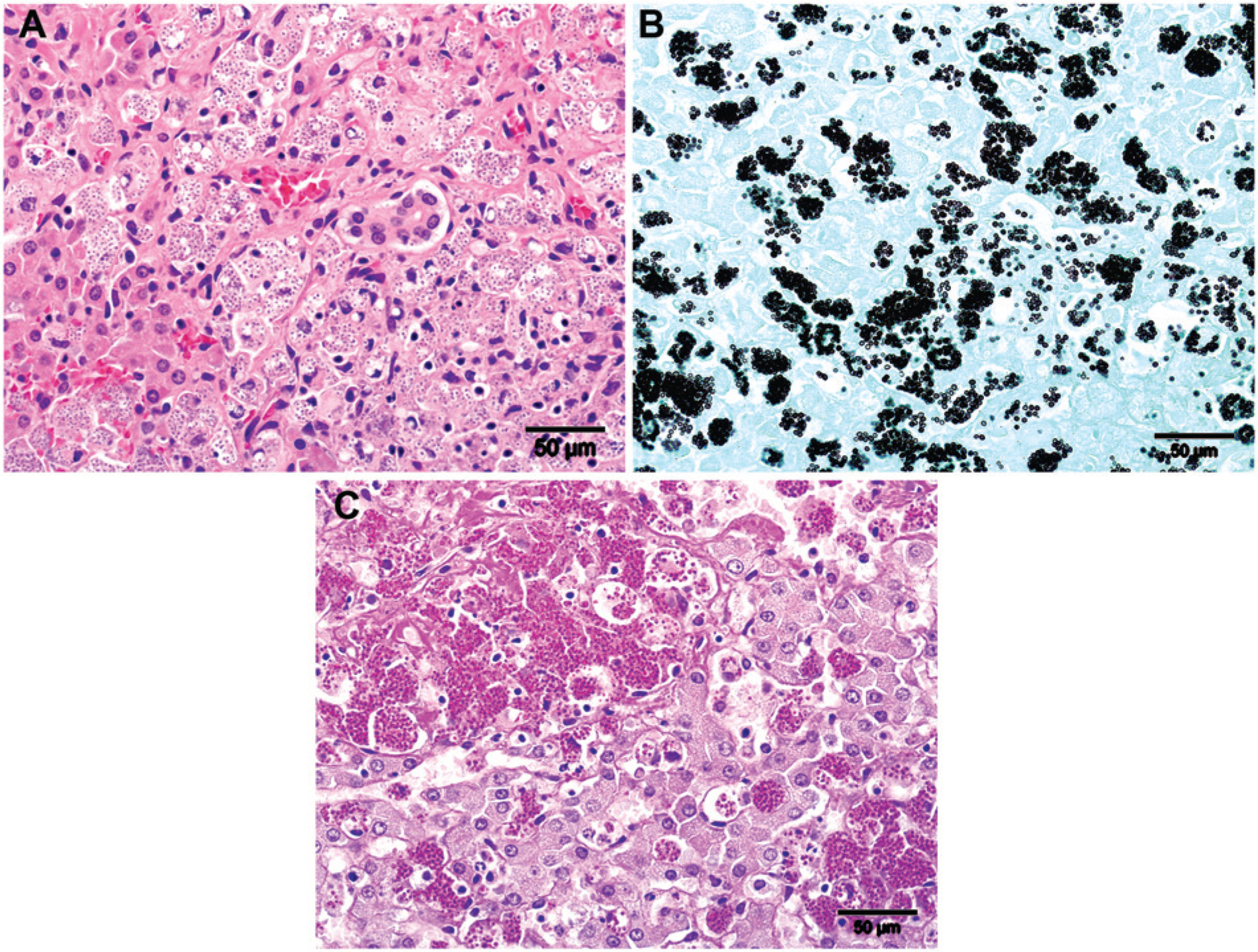
Raccoon tissues were negative for rabies by fluorescent antibody (FA) staining and Canine distemper virus by immunohistochemical staining (IHC). On special stains, the organisms were consistent with a yeast species staining positive with both Gomori methenamine silver (GMS; Fig. 2B) and periodic acid–Schiff (PAS; Fig. 2C) stains and only very faintly with Giemsa stain. On Romanowsky-type stains of lymph node impression smears, the organisms were seen as negatively staining, clear round structures either free or within degenerate macrophage cytoplasm.
Fungal cultures were performed on the gastric lymph node, lung, and spleen tissues from raccoon 2, and gastric lymph node, thoracic lymph node, and liver tissues from raccoon 3. Tissues were plated onto 5% sheep blood agar (SBA), c Sabouraud dextrose agar with antibiotics d (SabDex), and potato flake agar (PFA) plates incubated at both 25°C and 37°C. No fungal growth was detected after 28 days of incubation on SabDex or PFA plates from any of the tissues tested. Fungal colonies were visible on SBA plate cultures incubated at 37°C from lung and spleen tissues from raccoon 2 after 9 days of incubation. The SBA plates from raccoon 3 lung, gastric lymph node, and thoracic lymph node samples demonstrated growth of Aeromonas sp. and no fungal colonies were identified; however, fungal growth was detected from the liver sample on the SBA plate incubated at 37°C after 8 days of incubation. No growth was detected on any of the media plates incubated at 25°C. Lactophenol cotton blue staining on all fungal colonies were performed daily for 10 days and demonstrated only fungal hyphae without conidia. Subcultures of these colonies to SBA, SabDex, and PFA failed to grow at either 25°C or 37°C.
Polymerase chain reaction (PCR) testing was utilized on the fungal isolates to determine if a definitive identification could be established. Genomic DNA was extracted using a commercial kit. e A portion of the variable region of the 28S ribosomal gene was amplified using the forward primer 5′-GGTCCGTGTTTCAAGACG-3′ and reverse primer 5′-GCATATCAATAAGCGGAGG-3′. Polymerase chain reaction was performed in a 50-µl reaction mixture containing 2 µl of DNA, 1× buffer containing 1.5 mM of MgCl2, 150 µM of each deoxyribonucleotide triphosphate, 0.2 mM of each primer, and 1 U of DNA polymerase. f The PCR set up included initial denaturation at 94°C for 30 sec, annealing at 50°C for 1 min, extension 72°C for 2 min (50 cycles), and a final extension step at 72°C for 7 min. The PCR products were electrophoresed on 3% agarose gel in Tris–borate–ethylenediamine tetra-acetic acid–ethidium bromide buffer (pH 8.4) and photographed with an imaging system. g Amplicon was purified h and sequenced with the same primers used for PCR, which yielded a 523-bp amplicon.
DNA sequencing and a BLAST analysis using GenBank (www.ncbi.nlm.nih.gov/genbank/) identified the PCR amplicon sequence to be a 100% identity match to Ajellomyces capsulatus. Ajellomyces capsulatus is the telomorphic or holomorphic name representing all forms of the fungi, while Histoplasma capsulatum is used to describe the anamorphic or yeast form of the organism. 21
As a dimorphic fungus, H. capsulatum exists as mycelia in the environment and in the yeast form at body temperature.1,17 Culture conditions in the laboratory are designed to mimic the switch from yeast (37°C) to mycelia (25°C) and the induction of sexual structures that permit fungal identification. 17 The difficulties with culture and identification of the organism described in the current report, including the lack of growth on any fungal media at 25°C, the need for extended incubation on growth media for fungal recovery, and the inability to detect fungal organisms in the presence of bacterial contaminants highlight the difficulties associated with confirmation of this pathogenic agent. Additionally, only hyphal elements with no characteristic tuberculate macroconidia or pyriform microconidia were identified on wet mount preparations examined over a 10-day period. Partial ribosomal gene sequencing, which has been used for fungal identification from formalin-fixed tissues when culture could not be performed, was required for H. capsulatum confirmation in the current case. 21
Histoplasma capsulatum is found in temperate and tropical environments worldwide that are favorable to a variety of fungi, including decomposing plant material, caves, mines, water troughs, and basements.1,13 At least 8 clades of H. capsulatum have been identified with various geographic predispositions. 13 Survival of this organism is favored by soil containing high concentrations of nitrogen, which promote sporulation.1,6 Consequently, environments in which bird and bat guano have accumulated are notorious for harboring large quantities of H. capsulatum micro- and macroconidia.1,6,7,9 Disruption of contaminated soil results in aerosolization of fungal filaments and spores, increasing the risk of human and animal exposure.14,15,17,18
Inhaled microconidia (2–4 µm) enter terminal bronchioles and alveolar spaces, convert to the yeast form, and are rapidly phagocytized by alveolar macrophages.1,13,20,26 While the innate immune response is generally effective against other yeast organisms such as Candida sp. and Saccharomyces cerevisiae, it is insufficient to clear H. capsulatum infections.19,26 This fungal agent upregulates the production of superoxide dismutase (SOD3), which is secreted out of the cell and into the phagosome; organisms without the SOD3 gene cannot resist phagocyte killing. 26 Infected macrophages can then traffic to other organs, providing a mechanism for dissemination of the fungus to distant sites.15,18,19
In the United States, H. capsulatum is endemic in the Midwest most notably in the Mississippi, Missouri, and Ohio River valleys.3,6,14,21,26 Most published cases of histoplasmosis are identified in human beings and domestic animals that live or have spent time in these regions.2,3,8,25 However, evidence of infection in nonendemic regions of H. capsulatum has been reported.3,11,12,24 A retrospective cohort study of persons >65 years of age determined that 11% of histoplasmosis patients surveyed had no exposure to endemic geographic regions for H. capsulatum prior to disease development. 3 A microendemic region for H. capsulatum was identified in El Paso, Texas, and systemic histoplasmosis was diagnosed in 2 indoor cats from central California, both of which are geographic regions with hot, dry conditions unfavorable for survival of this fungal agent.11,12
The raccoons described in the present report were found in the San Francisco Bay Area; while the source of their exposure cannot be established, it is reasonable to assume that due to their young age at capture they were exposed within that vicinity. Contact may have occurred before rescue in which case these animals may have seeded the housing unit with contagious fungal spores, or contact might have originated in the soil within the housing unit although no previously raised raccoons showed signs of illness. A 2013 published study indicated that fungal infections in human beings may be expanding beyond previously described borders. 5 Wildlife species in close contact with a caretaker as described in the current report may present an additional zoonotic exposure risk to this fungal agent.
A review of 10 National Center for Biotechnology Information–deposited sequences with the highest homology to the DNA region examined in this isolate revealed that these strains originated from a variety of countries (United States, Argentina, China, and Brazil) and most were from clinical cases of histoplasmosis in human beings. Multilocus sequence typing performed on 5 tissues and 1 isolate from 6 cats previously diagnosed with H. capsulatum from nonendemic regions of the United States determined that these isolates grouped most closely to but outside of the North American-1 clade. 4 Further molecular analysis may provide insight into phylogeographic patterns of and exposure risks to H. capsulatum from geographic locations not thought to support long-term survival of this agent.
Clinical histoplasmosis in human beings is associated with immunosuppressive conditions that result in incomplete destruction of the fungus or recrudescence of a latent infection.8,10,15,18,22,25 While immunosuppressive agents have been associated with animal cases of H. capsulatum, clinical histoplasmosis has been identified in dogs and cats with no detectible immune deficiencies.6,11,21,23 Liver, spleen, and lymph nodes are primary targets of dissemination, although fungus has been found in other organs, including bone, eye, and skin.14,23 Inhalation is the primary mode of entry, although respiratory symptoms are not always present, particularly in cats.2,23 As in human beings, animal hosts that are immunocompromised or received a large dose of infectious fungal elements appear to be at a greater risk for developing infection and disseminated disease.14,21,23 In the wild, raccoons give birth in early spring and wean their kits in autumn at 16–20 weeks of age. The reasons for the unusually late birth of these kits is unknown; however, it is plausible that a combination of their age, stress from separation from their dam and capture, and exposure to fungal spores resulted in an insufficient adaptive immune response and disseminated histoplasmosis.
The present report describes a massive H. capsulatum infection in juvenile raccoons from a nonendemic region of the United States utilizing molecular methods. Histoplasmosis should be included in the list of differential diagnoses for systemic granulomatous disease in wildlife species such as raccoons even in nonendemic regions. Molecular methods should be pursued for agents that cannot be identified by routine culture procedures to enhance surveillance for this agent and to further characterize its movement into environments previously deemed to be unable to support H. capsulatum survival.
Footnotes
a.
Duramune Max 5, Boehringer Ingleheim Vetmedica, St. Louis, MO.
b.
Revolution, Zoetis Inc., Florham Park, NJ.
c.
Remel, Thermo Fisher Scientific, Waltham, MA.
d.
Difco, BD Diagnostic Systems, Sparks, MD.
e.
Phase-lock gel, Eppendorf AG, Hamburg, Germany.
f.
Applied Biosystems Amplitaq gold polymerase, Life Technologies, Grand Island, NY.
g.
FluorChem HD2 imaging system, Alpha Innotech, Santa Clara, CA.
h.
Amicon Ultra-0.5 centrifugal filter device, EMD Millipore Corp., Billerica, MA.
Declaration of conflicting interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
