Abstract
A lethargic, wild, juvenile raccoon died and was subsequently presented for necropsy. The lungs had widely disseminated, finely granular, yellow–tan foci with moderate pulmonary, as well as, thoracic lymphadenopathy. Histopathology revealed numerous cross-sections of larval trematodes morphologically consistent with diplostomid mesocercariae and metacercariae as well as moderate alveolointerstitial pneumonia with a mixed population of eosinophils, macrophages, lymphocytes, and plasma cells. The presence of larval trematodes within lymphatics in multiple organs indicates a route of fluke migration. Live specimens of mesocercariae and metacercariae recovered from refrigerated lung tissue after 7 days were identified as Pharyngostomoides sp., an intestinal diplostomid parasite (trematode) of raccoons and other mammals that is similar in appearance and life cycle to Alaria spp.
In August 2006, a juvenile, sexually intact female raccoon (Procyon lotor) was presented to a humane society for examination after a homeowner in St. Paul, Minnesota, had observed that the individual was lethargic and moving slower than its siblings. Physical examination findings were unremarkable, and the animal was returned to the neighborhood, where it was found dead later that day. The animal was 1 of an original litter of 8 wild raccoons that lived in the backyard of the client. The whereabouts and health status of the 7 littermates is unknown.
The raccoon was submitted to the University of Minnesota Veterinary Diagnostic Laboratory (St. Paul, MN) for necropsy. The animal weighed 0.9 kg and was thin with prominent ribs and pelvic bones. The tracheal lumen contained a moderate amount of pink froth, the lungs were firm, and cut sections of the lung had numerous granular yellow–tan foci, ranging in size from pinpoint to 1 mm in diameter (Fig. 1). Minimal pericardial and perirenal adipose tissue was evident, and the stomach contained 2 ml of dark brown mucoid material. Nearly all of the internal lymph nodes (tracheobronchial, hilar, and cranial sternal in particular) were prominent and dark gray on cut surface. There were no significant macroscopic findings in the other organ systems.
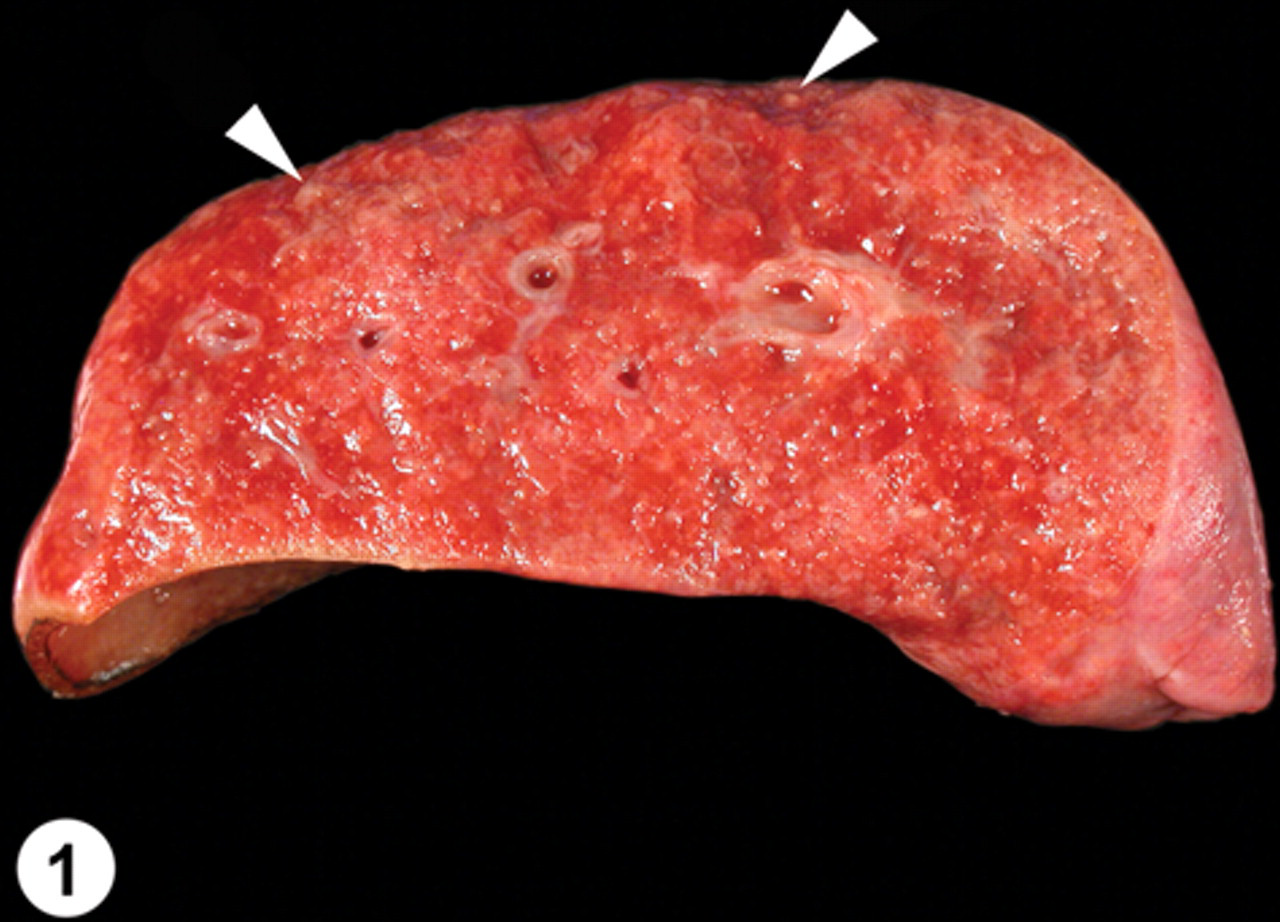
Lung (cross-section); pulmonary trematodosis; raccoon. The lungs were palpably dense with mostly fine granular yellow–tan foci. A few of the larger foci at the periphery of the lung are highlighted (arrowheads).
Sections of all major organs, including: lung, trachea, heart, liver, pancreas, stomach, intestines, kidneys, adrenal glands, thyroid glands, lymph nodes, and brain were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin. Sections of lung were stained with Gomori trichrome and Prussian blue stains. Transmission electron microscopic examination of intestinal contents was negative for viral particles, and the feces were negative for protozoan cysts and trophozoites, and helminth eggs and larvae, by centrifugal flotation in ZnSO4. The brain was negative for Rabies virus by indirect fluorescent antibody test at the Minnesota Department of Health. A tissue homogenate (lung, lymph node, spleen, kidney, and intestine) was negative for virus isolation after 2 passes on Madin–Darby canine kidney cells and on Crandell feline kidney cells.
Histopathology of the lung revealed a compact, alveolointerstitial pneumonia with a pattern of alternating alveolar atelectasis and emphysema (predominantly contiguous alveoli with ruptured walls). Dilated bronchoalveolar spaces were either empty or contained numerous nondegenerate larval trematodes. The alveolar walls were in close apposition, and the alveolar septa and pulmonary interstitium were thickened by infiltrates of eosinophils, lymphocytes, macrophages, and fewer plasma cells admixed with patchy interstitial fibrosis and multifocal hemorrhages. The alveolar spaces contained many individual and clustered foamy macrophages, hemosiderin-laden macrophages, free erythrocytes, pale eosinophilic and finely fibrillar proteinaceous material (edema), and mucus. Expanding many of the terminal bronchiolar airways and alveolar spaces as well as within bronchiolar lumina, there were sections of numerous larval trematodes (mesocercariae and metacercariae), ranging in size from 100 to 300 µm in diameter (Figs. 2, 3, 4). These parasites were determined to be trematodes based on the presence of a digestive tract and suckers and the absence of a body cavity. The absence of eggs and the presence of the flukes migrating through tissue were also consistent with these being immature larval trematodes. In tissue sections, it can be difficult to identify individual larval trematodes as either mesocercariae or metacercariae. Histomorphological examination of 3 sections of lung yielded an estimate of the parasite density, with a mean value of 1.71 parasites per mm2 of lung parenchyma.
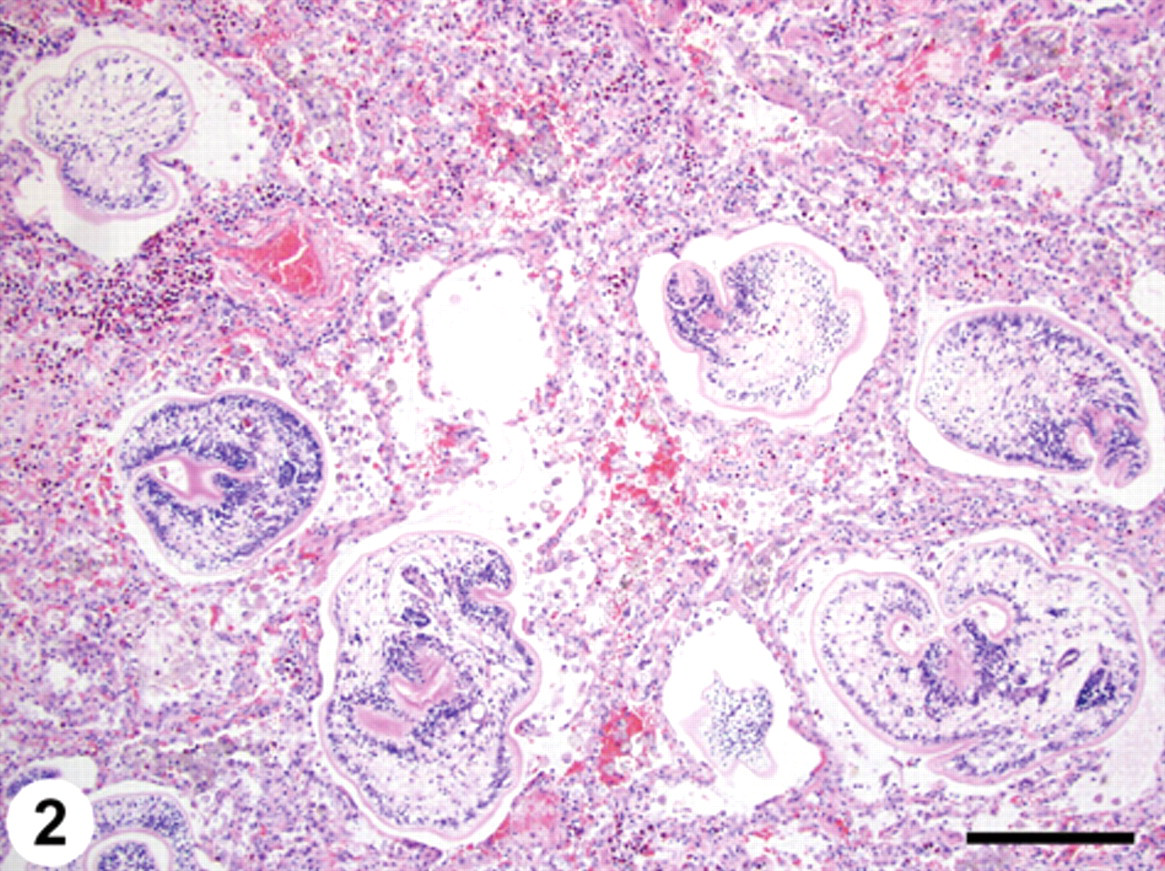
Lung; pulmonary trematodosis; raccoon. Multiple dilated bronchoalveolar spaces contain sections of trematode metacercariae. The adjacent pulmonary parenchyma is hypercellular with thickened alveolar walls in close apposition. The pulmonary interstitium is expanded by eosinophils, lymphocytes, and macrophages admixed with erythrocytes. Hematoxylin and eosin. Bar = 200 µm.
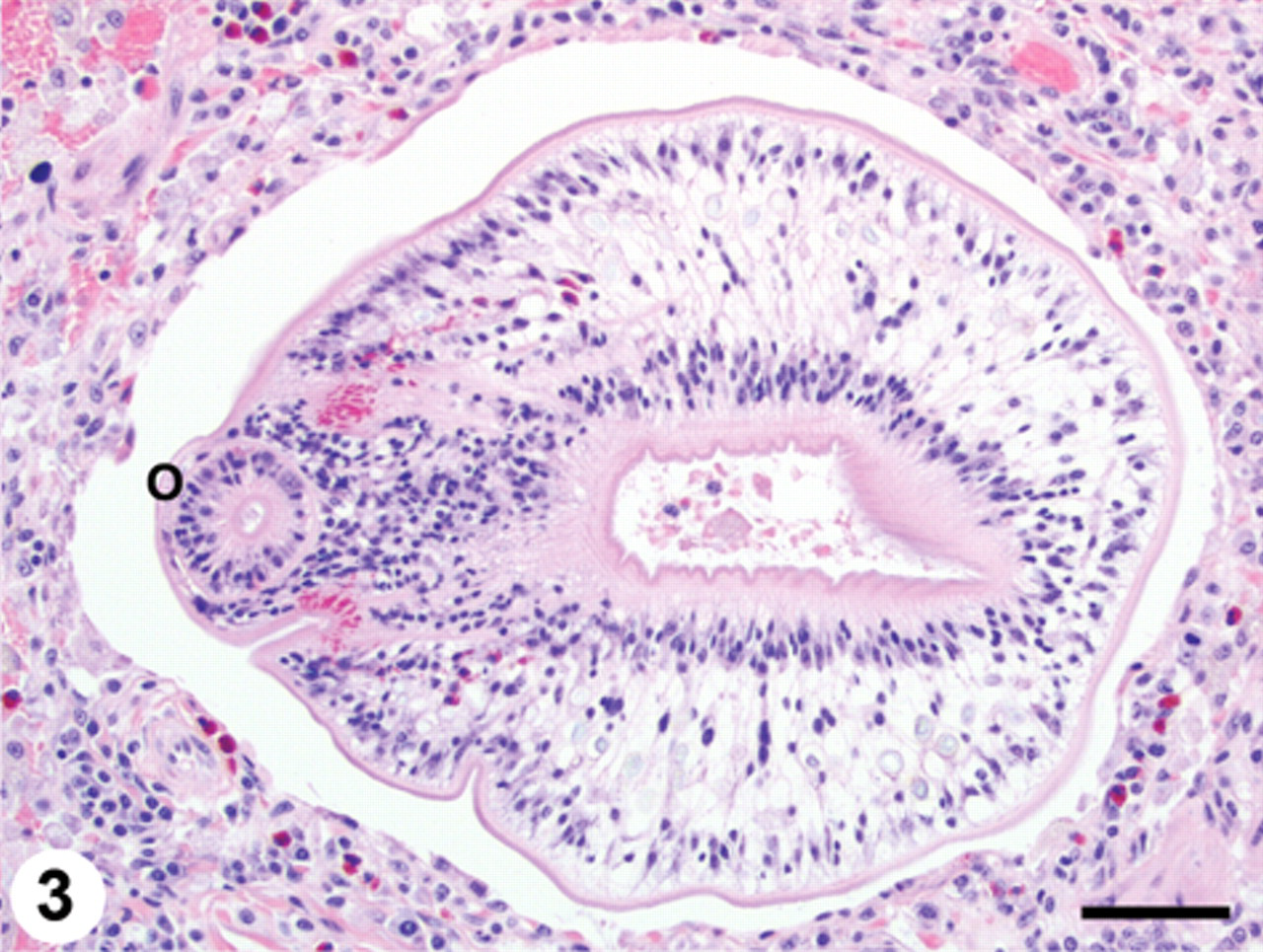
Lung; pulmonary trematodosis; raccoon. Larval trematode (metacercaria) within a markedly dilated bronchoalveolar space. Note the prominent oral sucker (O) at the left of the image. There are scattered eosinophils within the adjacent pulmonary parenchyma. Hematoxylin and eosin. Bar = 50 µm.
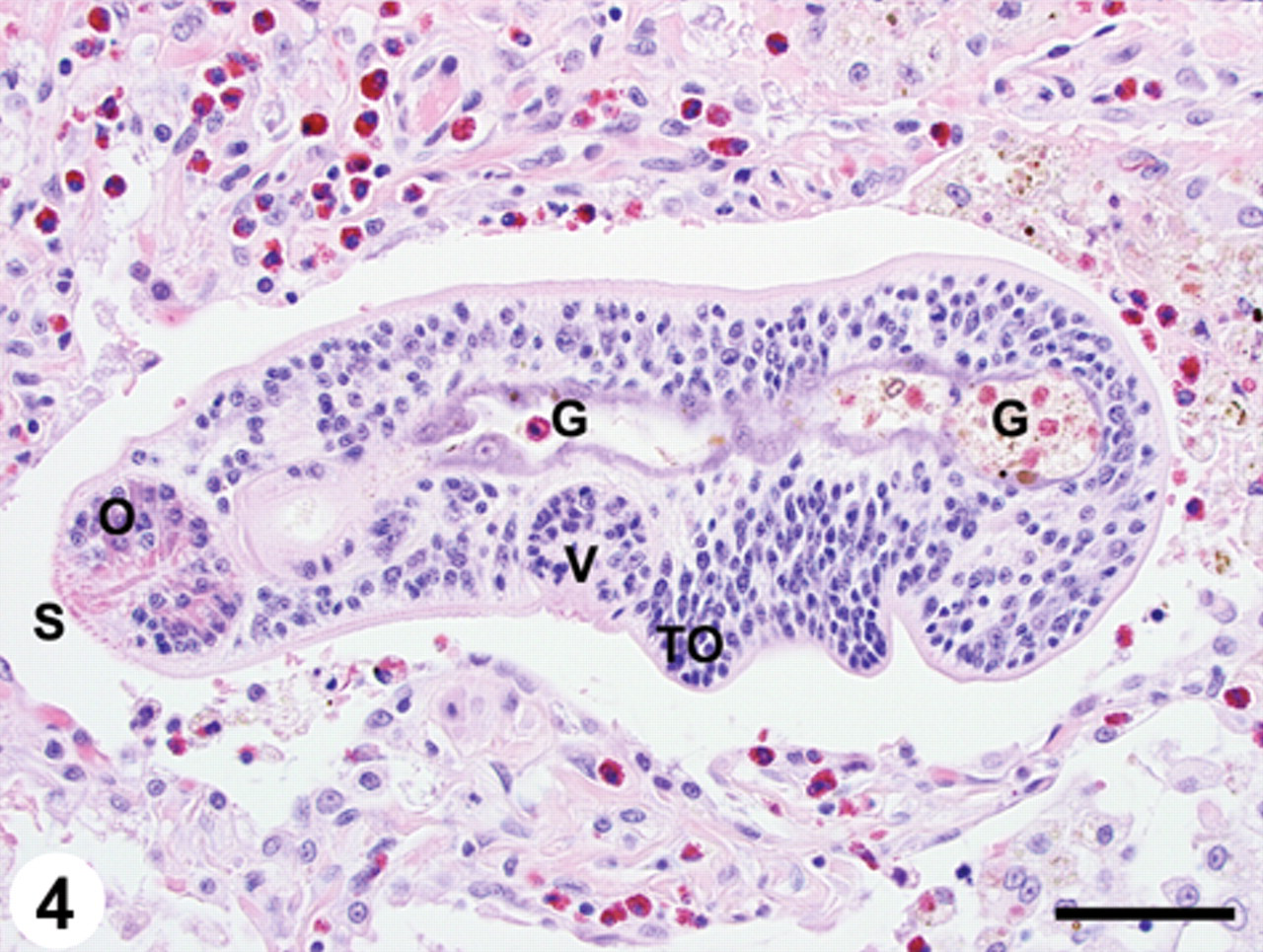
Lung; pulmonary trematodosis; raccoon. Larval trematode (mesocercaria) within a dilated bronchoalveolar space. Note the tegumental spines (S), prominent oral sucker (O) and ventral sucker (V), as well as the developing tribocytic organ (TO) and gut (G). Moderate numbers of eosinophils are within the adjacent pulmonary parenchyma. Hematoxylin and eosin. Bar = 50 µm.
Scattered lymphatics within the submucosa, muscularis mucosa, and muscular tunics of the stomach wall contained larval trematodes (Fig. 5). A few lymphatics in the muscular layers of the small intestine contained individual larval trematodes as described above. The mucosa of the small intestine had a moderate, diffuse infiltrate of eosinophils admixed with lymphocytes and plasma cells. There were multiple larval trematodes in capsular lymphatics of the mesenteric lymph node, and the hilar connective tissue was infiltrated by moderate numbers of eosinophils. Also, 1 lymphatic within the perithyroid connective tissue contained a larval trematode. There were no other significant microscopic lesions.
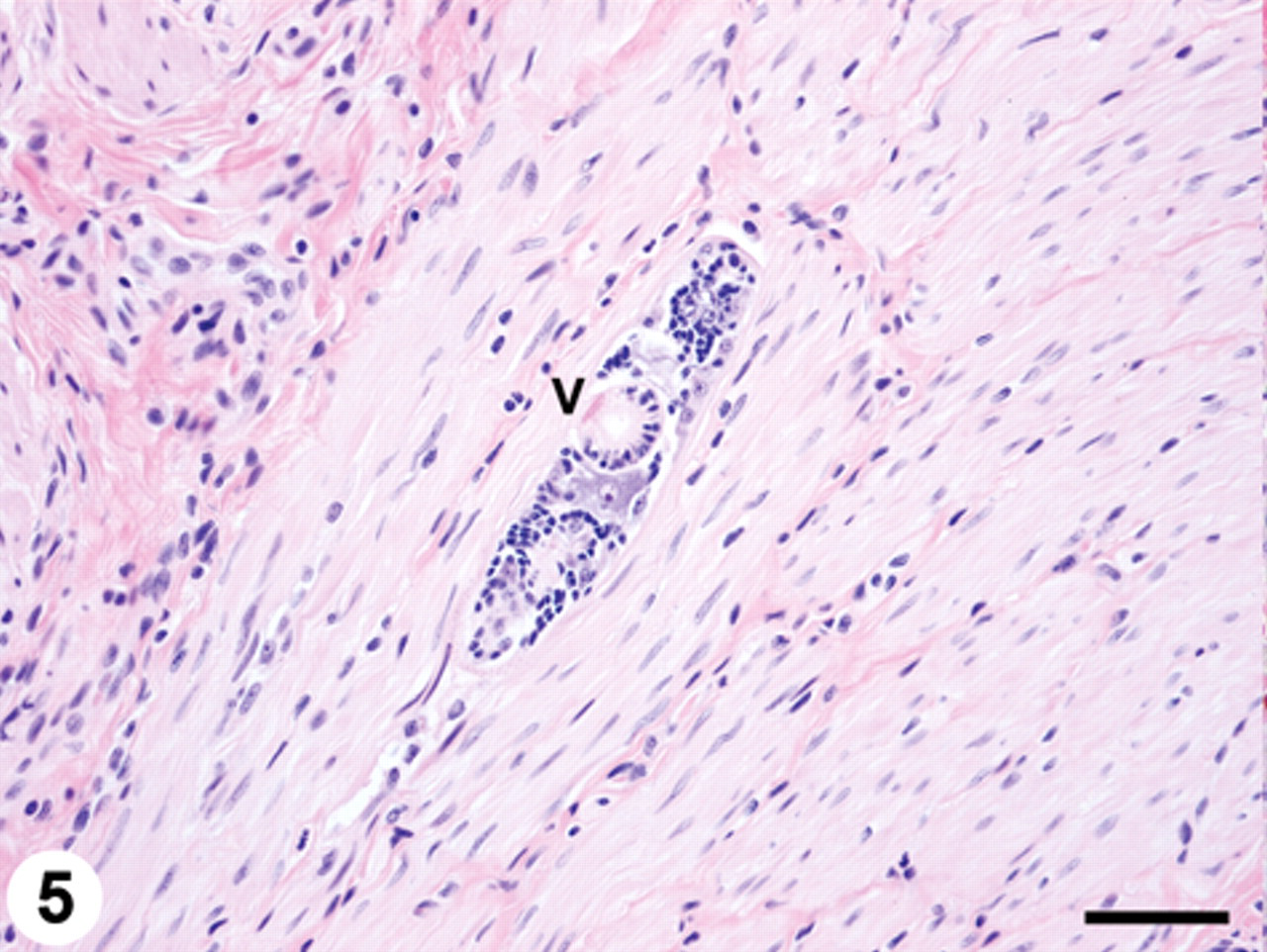
Stomach; trematodosis; raccoon. Larval trematode (mesocercaria) within a lymphatic in the muscular tunic. Note the prominent ventral sucker (V). Hematoxylin and eosin. Bar = 50 µm.
A small sample (approximately 5 cm3) of lung tissue that had been refrigerated for 7 days was manually teased apart in saline, yielding hundreds of motile larval trematodes. These parasites were fixed in AFA (alcohol–formalin–acetic acid) and stained in Semichon carmine for detailed morphological analysis and identification because histological analysis in tissue sections was inadequate to classify these parasites beyond the level of trematodes. Based on morphology, the trematodes recovered from the lungs were determined to be a mixture of mesocercariae and diplostomulum metacercariae.
The mesocercariae (n = 5) ranged in size from 200–340 µm × 70–160 µm (mean = 241 µm × 101 µm) and had fine tegumental spines adjacent to the oral sucker. The only discernible organ development in the mesocercariae consisted of oral and ventral suckers, and a gut (Fig. 4). In some of the mesocercariae, the tribocytic organ could be seen in the early stages of development (Fig. 4).
The body of the metacercariae had a spatulate forebody and a cylindrical hindbody, both located in a single plane. The lateral margins of the forebody were folded ventrally giving it a scoop or “cone” shape (Fig. 6). Metacercariae at the most advanced stage of development (n = 7) were 430–586 µm × 269–313 µm (mean = 506 µm × 298 µm). The metacercariae had oral and ventral suckers, 2 pseudosuckers, a tribocytic organ, and varied in the stage of development with the most mature having a well-defined ovary located just posterior to the oval tribocytic organ and anterior to the testes (Fig. 6). The 2 testes of the metacercariae were arranged in an opposite fashion (i.e., side by side). Organ measurements were as follows: oral sucker 52–57 µm × 52–62 µm (mean = 55 µm × 58 µm), pharynx 38–57 µm × 26–45 µm (mean = 48 µm × 32 µm), ventral sucker 43–52 µm × 50–69 µm (mean = 47 µm × 60 µm), tribocytic organ 121–162 µm × 90–131 µm (mean = 134 µm × 103 µm), ovary 38–52 µm × 40–55 µm (mean = 45 µm × 49 µm), right testes 43–88 µm × 29–60 µm (mean = 65 µm × 45 µm), and left testes 48–98 µm × 38–67 µm (mean = 63 µm × 44 µm). The observed morphology of the specimens recovered was consistent with an identification of Pharyngostomoides sp. 1
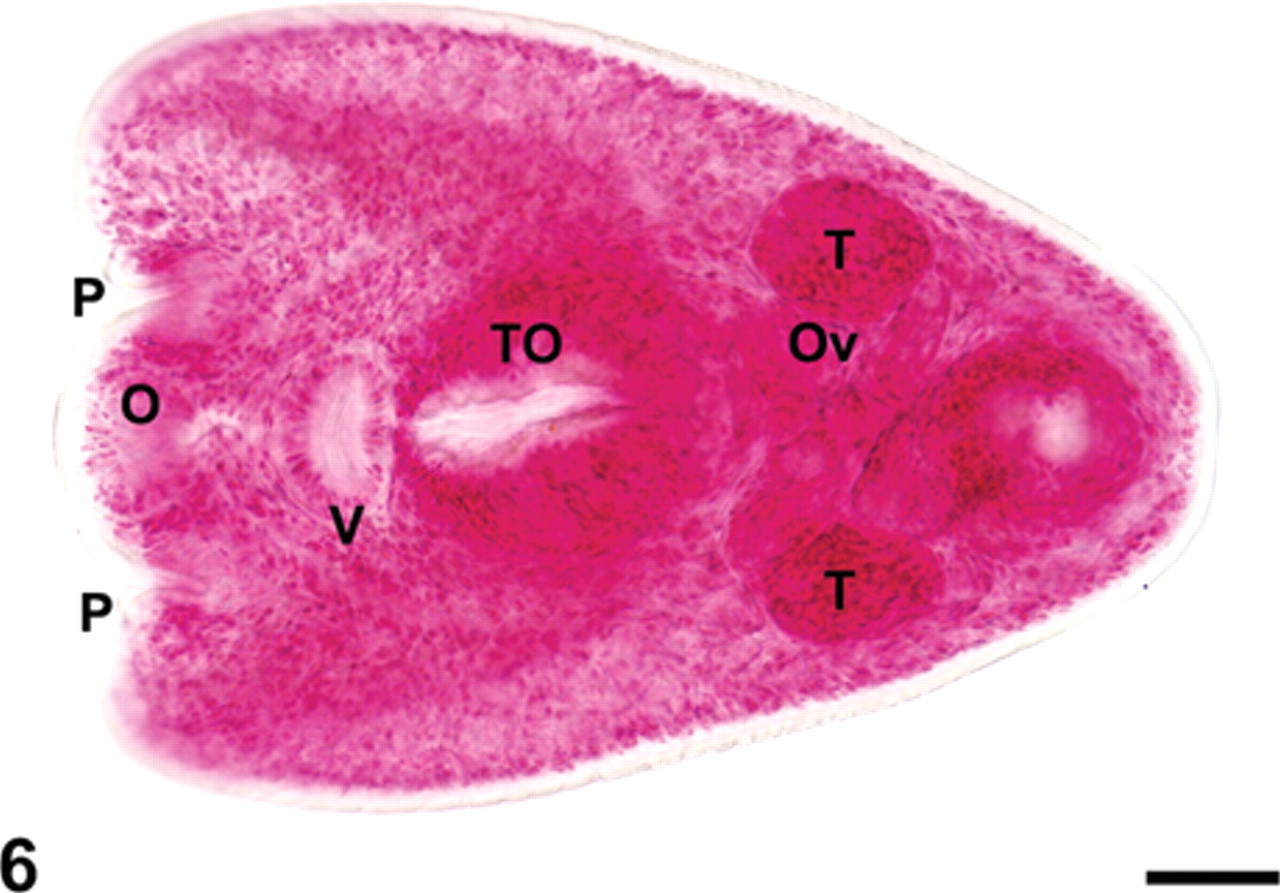
Trematode metacercaria recovered from the raccoon lung. Note oral sucker (O) flanked by a pseudosucker (P) on either side, ventral sucker (V), tribocytic organ (TO), ovary (Ov), and paired testes (T) arranged in an opposite fashion (i.e., side by side). Semichon carmine stain; mounted in Canada Balsam. Bar = 50 µm.
Fluke morphology, size, tissue location, and host in the present case were consistent with an identification of mesocercariae and metacercariae of Pharyngostomoides sp. 1 Other diplostomids reported from raccoons include Alaria mustelae, Alaria marcianae, Fibricola cratera, Fibricola texensis, Parallelorchis diglossus, and Procyotrema marsupiformis. 2,4,5,12,13,16,17 These trematodes were ruled out in the present case on the basis of morphology and tissue location. The testes are tandem in Alaria; Fibricola lacks pseudosuckers, and there is no mesocercarial stage in its developmental cycle; Parallelorchis differs in morphology of the tribocytic organ; and Procyotrema is not found in the lungs and has an elongated, linguiform body. 13,21,23
Two species of Pharyngostomoides have been reported in raccoons (Pharyngostomoides procyonis and Pharyngostomoides adenocephala) and infections often consist of both species. Adults can be differentiated based on body shape (P. procyonis with a forebody and hindbody in a single plane; P. adenocephala with a dorsal flexure at the junction of the forebody and hindbody), glands associated with the pseudosuckers (P. procyonis with weakly developed glands; P. adenocephala with prominent glands), and the presence of a muscular ejaculatory pouch (present in P. procyonis; absent in P. adenocephala). 1 A third species of Pharyngostomoides, Pharyngostomoides dasyuri, has been reported from the southeastern quoll (Dasyurus viverrinus) and the spotted-tail quoll (Dasyurus maculatus), 2 dasyurid marsupials in Tasmania. 8
There are no published descriptions comparing the morphology of the metacercariae of P. procyonis and P. adenocephala and therefore, it is presently unknown whether the 2 species can be differentiated at this stage of development. Although many of the metacercariae recovered in the present case showed advanced organ development, the extent to which adult fluke morphological characteristics could be used to identify metacercariae is unknown. The metacercarial body shape in the specimens recovered from the lungs of the raccoon in the present study was most consistent with that described for adults of P. procyonis.
In North America, Pharyngostomoides spp. have been rarely reported in hosts other than raccoons (i.e., a black bear [Ursus americanus] in the southeastern United States). 7 Infection in raccoons appears to be most common in the southeastern United States, but has also been reported in Michigan and parts of Canada (Ontario and Saskatchewan). 3–6,11,14,16,22 In the authors’ laboratory, from 2001 to spring 2010, there are records of 107 raccoon necropsies, including 98 with histopathology of the lung. In 3 raccoons, 1 or 2 trematodes were found in the lung; these parasites were not available for identification. To the authors’ knowledge, Pharyngostomoides has not previously been reported in Minnesota. The flukes are acquired as mesocercariae from the ingestion of intermediate hosts (branchiobdellid annelids associated with crayfish). The mesocercariae migrate through various tissues, some developing into metacercariae in the lungs and some staying in the mesocercarial stage residing in various tissues for prolonged periods of time. Metacercaria are coughed up and swallowed, passing into the small intestine where they mature to adult flukes. Raccoons can be infected for life from a single exposure due to the tissue reservoir of mesocercariae (infections maintained up to 14 years without re-exposure). 20 In addition, it has been demonstrated that Pharyngostomoides spp. can be acquired through transmammary transmission in the raccoon. 15,19
The presence of numerous larval trematodes (mesocercariae and metacercariae) in the lungs of this raccoon is consistent with exposure to a massive number of mesocercariae over a relatively short period of time. Published reports of parasite surveys of wild raccoons (especially those from the southern United States) have identified P. procyonis in the small intestine. Pharyngostomoides spp. are often reported to be the most common or second most common trematode encountered in wild raccoons and are thought to have a nearly ubiquitous presence in the southeastern United States. Intestinal worm burdens can number in the thousands to as high as 50,000 adult flukes in naturally infected raccoons. 4,11 The high prevalence of P. procyonis may be attributed to the longevity of infections in raccoons from a single exposure (essentially for life) and the efficiency of transmammary transmission of mesocercariae to nursing kits. 20
The recovery of live mesocercariae and metacercariae from refrigerated lung tissue 1 week after the death of the animal was unexpected, although similar observations have been reported for the mesocercariae and metacercariae of the closely related diplostomids, Alaria mustelae 2 and Alaria americana. 10 Mesocercariae have been isolated from the mammary glands and milk as well as skeletal muscles of the abdominal wall of raccoons, and it is thought that a hormonal factor, possibly during pregnancy, may stimulate the migration of mesocercariae to the mammary glands. 15,19 Mesocercariae and metacercariae of P. procyonis have been found in the lungs of raccoons that harbored adult flukes in the lumen of the small intestine, 14,19 and it is thought that the mesocercariae undergo a prolonged somatic migration, periodically moving to the lungs and then to the digestive tract, where they mature. 20 However, the larval trematodes identified in the gastrointestinal tract of the raccoon of the present report were restricted to lymphatics. The presence of moderate numbers of larval trematodes in lymphatics at various anatomic sites in the present case, including the gastrointestinal tract, mesenteric lymph node, and thyroid gland implies that the lymphatic system serves as a route of fluke migration throughout the host as suggested by a previous study for the closely related trematode, A. americana. 9
Given the host range and large populations of raccoons in the United States, it is surprising that others have not documented pneumonia-associated mortality with Pharyngostomoides spp. A fatal systemic infection of A. americana in a 24-year-old backpacker in Canada involving lung migration of several thousand mesocercariae has been reported. 9,10 The presence of inflammatory tracts in the stomach wall suggests that the gastrointestinal tract may have been the portal of entry. Investigation into amphibians and reptiles on the patient’s family farm and immediate vicinity yielded many (7,500) mesocercariae of A. americana. 10 The authors hypothesized that the patient was likely exposed to inadequately cooked frog legs and that the parasites probably spread by direct passage through the tissues and by the lymphatics. 9 There have been at least 7 human infections with mesocercariae in North America and these have historically involved handling or eating infected intermediate or paratenic hosts. 18
To the authors’ knowledge, the present study is the first to document Pharyngostomoides sp. in the raccoon in Minnesota; is believed to be the first report to discuss pulmonary disease linked to migration of this trematode in raccoons; and clearly demonstrates lymphatics as a route of migration. The high density of larval trematodes in the lungs is believed to be responsible for the death of this raccoon; no concurrent bacterial or viral disease was identified and no morphologic evidence of an immunosuppressive disease process was found; however, an underlying defect in immunity cannot be definitively excluded. The authors speculate that extensive lung migration of this parasite has the potential to cause some level of attrition in populations of wild raccoons. The present case also highlights the importance of performing a complete necropsy on raccoons, including thorough examination of the lungs, and stresses the value of saving fresh tissues for diagnostic procedures including parasite recovery and identification.
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
