Abstract
A 1-mo-old reticulated giraffe had progressive anorexia and died at the Ordos Zoo. Autopsy revealed necrotic stomatitis with severe bilateral necroulcerative lesions at the base of the tongue and of the cheeks near the commissures of the mouth. There was also severe bilateral confluent bronchopneumonia with a pronounced bronchial pattern and multifocal fibrinous pleuritis. Histologically, there was serofibrinous-suppurative bronchopneumonia with necrosuppurative bronchiolitis and necrotic arteritis. Filamentous bacteria with morphology consistent with Fusobacterium necrophorum were observed at the advancing edge of the necrotic tissue in the tongue and cheeks, as well as in the affected alveolar spaces and bronchioles. Aggregates of slender, gram-negative, rod-like or filamentous bacteria were identified in the lung impression smear. PCR results of 16S rDNA of the tongue and lung lesions had 100% homology to the F. necrophorum subsp. funduliforme B35 sequence (EF447425.1). The gross, histologic, Gram stain, and PCR product sequencing features in our case were consistent with oral and pulmonary necrobacillosis in ruminants, a rare disease of giraffes.
A 1-mo-old reticulated giraffe (Giraffa camelopardalis reticulata) at the Ordos Zoo in Ordos (Inner Mongolia, China) was anorectic for 9 d. A week later, the giraffe showed clinical signs of depression, abdominal breathing, a rectal temperature of 40.3°C, and was standing unsteadily. Treatment with raceanisodamine tablets (an anticholinergic; Minsheng Pharma), probiotics, and digestive enzymes by the attending veterinarian did not result in clinical improvement. On the ninth afternoon after onset, the giraffe was ataxic, hypopneic, had a damp hair coat during mechanical capture, and died within minutes of being captured. Antimicrobials were not administered to this juvenile giraffe during the course of the disease.
The dead juvenile giraffe was the first giraffe to be born at Ordos Zoo. After birth, the juvenile giraffe was with her dam and housed on bedding of Chinese wheatgrass (Leymus chinensis). The bedding was replaced monthly.
A postmortem examination was performed within 1 h of the animal’s death, and severe bilateral necroulcerative lesions at the base of the tongue (Fig. 1) and on the cheeks near the commissures of the mouth were observed. The bilateral necroulcerative lesions in the tongue were well-demarcated and surrounded by a 1 × 3 cm hyperemic area. Dark-red consolidation of large areas of the cranial and caudal lobes of the lungs had a pronounced bronchial pattern caused by bronchiolitis. Individual or dense clusters of small yellow-white nodules were widely scattered on the pleural surfaces (Fig. 2), and, on cut section, similar nodules were present within the parenchyma (Fig. 3). Multifocal thin layers of organized fibrin were attached along the pleural surface overlying areas of consolidation. The trachea contained a small amount of frothy white foam. The giraffe was emaciated, and there were small amounts of milk curd and 500 g of 2–8 cm long Chinese wheatgrass in the rumen. No additional remarkable gross findings were observed.
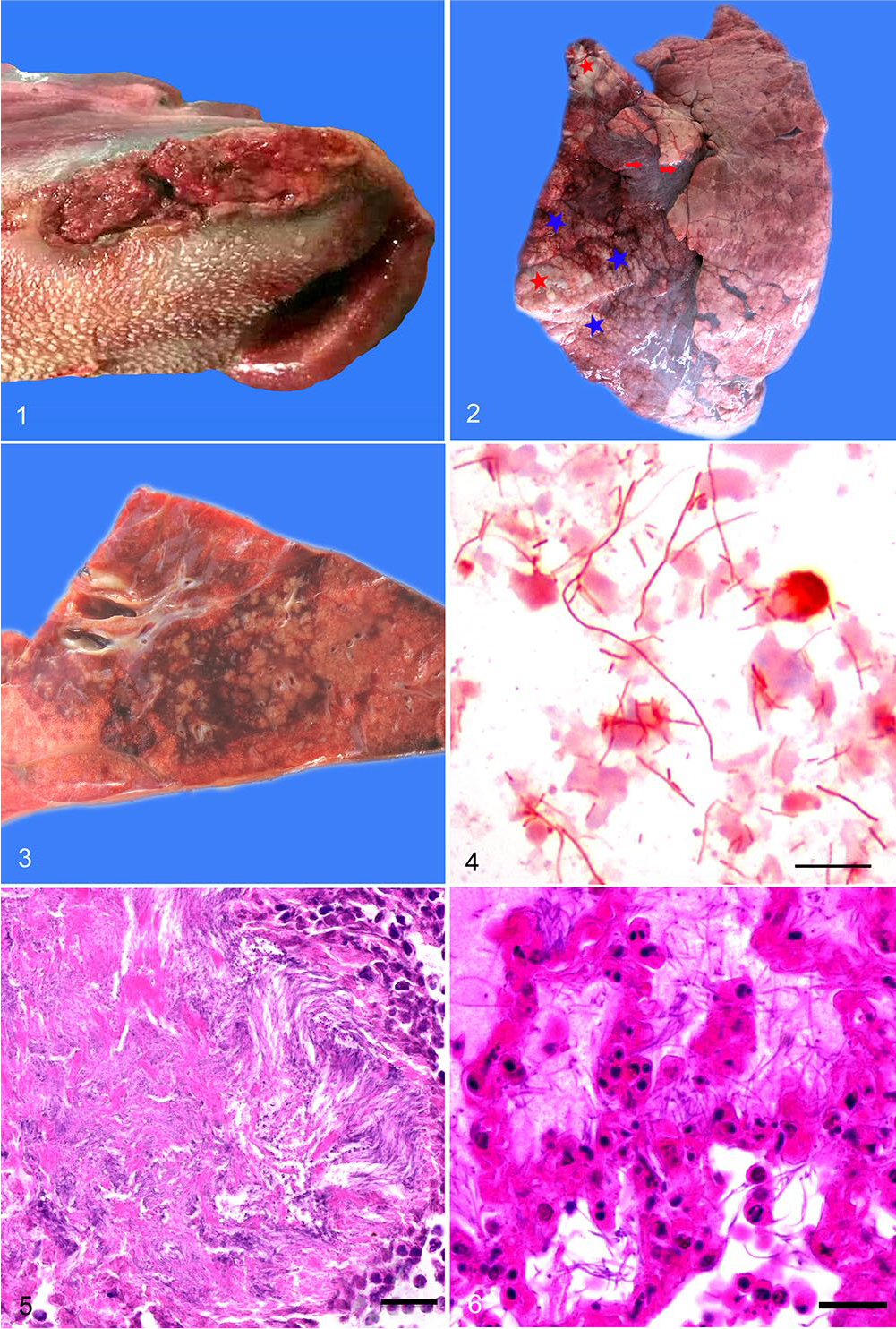
Pathologic and etiologic findings of necrobacillosis in a juvenile giraffe.
Impression smears were performed on the cut surface of the consolidated lung, and Gram staining was performed after air-drying. Tongue lesions, lung, spleen, heart, liver, kidney, stomach, tracheobronchial lymph node, and intestine collected during autopsy were fixed in 10% neutral-buffered formalin, processed routinely, and sections stained with hematoxylin and eosin. Aggregates of slender, gram-negative, rod-like or filamentous bacteria were observed in lung impression smears (Fig. 4). Histologically, necrotic tissues in the tongue and cheeks of the giraffe were surrounded by a dense, narrow rim of neutrophils and encapsulating granulation tissue. Numerous filamentous bacteria with morphology consistent with that of Fusobacterium necrophorum were observed at the advancing edge of the necrotic tissue (Fig. 5). The interlobular septa of the lung were markedly widened by serous fluid, fibrin, and neutrophils. Within the parenchyma there were numerous variably sized and often confluent inflammatory foci that were centered on and extended out from bronchioles. Affected bronchiolar walls were focally invaded by numerous filamentous bacteria and were often necrotic. Severe fibrinous necrotizing pneumonia with numerous necrotic and disintegrating neutrophils and numerous intra-alveolar filamentous bacteria extended into the adjacent alveolar spaces (Fig. 6). There was necrotic arteritis with neutrophils and thrombosis within affected areas. In more peripheral areas, alveolar spaces were filled with proteinaceous fluid, neutrophils, and a few filamentous bacteria.
Fresh tissues were not available for culture; formalin-fixed, paraffin-embedded tissues (tongue lesions, lung, spleen, liver, kidney, stomach, and intestine) of the animal were examined for F. necrophorum by using 16S rRNA gene sequencing. Genomic DNA was extracted (E.Z.N.A. FFPE-DNA kit; Omega), according to the manufacturer’s instructions. The 16S rDNA was amplified using 25 μL of 2× Taq master mix (Vazyme Biotech), 2 μL of template DNA, and 2 μL of each primer (10Fx: 5′-AGAGTTTGATCCTGGCTCAG-3′; 1509R: 5′-GTTACCTTGTTACGACTTCAC-3′) 6 in a final volume of 50 μL. The PCR conditions were: 5 min of initial denaturation at 94°C followed by 35 cycles of denaturation (30 s at 94°C), annealing (30 s at 52°C), and extension (1.5 min at 72°C), and a final extension for 7 min at 72°C. Amplified PCR products were analyzed by using electrophoresis on 1.5% agarose gels and sent for purification and sequencing (Sangon Biotech, Shanghai, China). DNA sequences of 1,414 bp (GenBank accession MH236113) were recovered from the tongue lesion and lung, but not from the spleen, liver, kidney, stomach, or intestine. The sequences from the tongue lesion and lung were identical and had 98.6% nucleotide homology with the GenBank F. necrophorum ATCC 25286 sequence (accession AJ867039.1). The highest nucleotide homology (100%) was with the F. necrophorum subsp. funduliforme B35 sequence (EF447425.1).
Necrobacillosis, which is caused by the anaerobic gram-negative bacterium, F. necrophorum, is a multi-syndrome disease that predominantly affects domestic ruminants, 7 macropods,1,3,8,10 wild ruminants,4–6,11 and rarely human beings. 2 F. necrophorum has been isolated from the normal flora in the oral cavity, gastrointestinal tract, and genitourinary tract of humans and animals,9,13 and may be present in environments contaminated by feces. Cattle and sheep are the livestock most susceptible to F. necrophorum. Necrobacillosis in wild ruminants has been reported in wild pronghorns (Antilocapra americana), 4 tundra reindeer (Rangifer tarandus tarandus), 6 black-tailed deer (Odocoileus hemionus columbianus), 5 and blue duikers (Philantomba monticola). 11 The predominant disease manifestations in ruminants include necrotic stomatitis, pododermatitis, balanoposthitis, rumenitis, hepatic abscessation, phlebitis, bronchopneumonia, and sepsis in some cases.
The best-known form of necrobacillary stomatitis is calf diphtheria, an acute necrotizing ulcerative inflammation of the mucosa of the cheeks, pharynx, and larynx (necrotic laryngitis). 14 Oral necrobacillosis can also manifest as facial and mandibular abscesses.4,12 Occasionally, oral lesions extend to affect large areas of the palate and gums. 4 The primary routes of transmission for oral necrobacillosis are traumatic injuries to the oral mucosa caused by punctures, molar exfoliation, dental calculus, feed contaminated with feces, adverse environmental conditions, and cold weather.3,8,10 The juvenile giraffe described herein had typical necrobacillary stomatitis with bilateral ulcerated lesions at the base of the tongue and on the cheeks near the commissures of the mouth.
Occasionally, fusobacterial pneumonia has extended to cause pleuritis in wild-caught pronghorns. 4 In our case, the juvenile giraffe had serofibrinous-suppurative confluent bronchopneumonia with necrosuppurative bronchiolitis, necrotic arteritis, and numerous filamentous bacteria.
The juvenile giraffes lived on bedding of Chinese wheatgrass, which they could chew at any time. The proposed pathogenesis in our case involved trauma to the oral cavity through consumption of hard stems of false wheatgrass, resulting in opportunistic infection of submucosal tissues by F. necrophorum. F. necrophorum in the environment (probably bacteria on dried forage) invaded the damaged area of the mucosa of the tongue and the cheeks and multiplied, causing more necrosis. Tissue with bacteria shed from the necrotic foci were then aspirated into the lungs and caused necrosuppurative bronchiolitis, which gradually involved alveoli around the bronchioles and developed inflammatory foci centered on bronchioles. The juvenile giraffe ultimately succumbed to severe confluent bronchopneumonia.
To prevent such cases, we recommend using soft herbage or bedquilt as bedding, replacing bedding frequently, isolated feeding after nursing, and disinfection of the nursery 3 times a week while giraffe calves are still nursing. Anti-microbial treatment of necrobacillosis may be successful if started early in the course of disease.
Footnotes
Acknowledgements
We thank Peng Wang at the Ordos Zoo for his help with the autopsy.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was financially supported by the Inner Mongolia Grassland Innovative Talent Team Project (20151031) and the Scientific Research Project of Colleges and Universities in the Inner Mongolia Autonomous Region (NJZZ19041).
