Abstract
Ulcers of the oral cavity, esophagus, and gastric compartments of South American camelids are uncommon. Multifocal-to-coalescing ulcers were identified in the oral cavity, esophagus, and/or gastric compartments of 5 alpacas submitted for postmortem examination. Fusobacterium necrophorum was isolated from the lesions in all alpacas, in combination with other aerobic and anaerobic bacteria. In 4 of these cases, F. necrophorum–associated lesions were considered secondary to neoplasia or other chronic debilitating conditions; in 1 case, the alimentary ulcers were considered the most significant autopsy finding. It is not known if this agent acted as a primary or opportunistic agent in mucosal membranes previously damaged by a traumatic event, chemical insult, immunodeficiency, or any other debilitating condition of the host.
Fusobacterium necrophorum is a gram-negative, non-motile, non-sporulating, aerotolerant-to-anaerobic, pleomorphic, rod-shaped to filamentous bacterium. 13 This microorganism is a normal inhabitant of the alimentary and urogenital tracts of several animal species and is also present in soil. F. necrophorum does not penetrate a healthy mucosa; a breach in mucosal integrity is required for bacterial infection and disease to occur. 7 F. necrophorum has been associated with multiple conditions, including calf diphtheria, postpartum metritis of cattle, foot root of cattle and sheep, and several other necrosuppurative infections of ruminants. Diseases caused by this microorganism are known generically as necrobacillosis.4,10,14,17,21 In South American camelids, infections by F. necrophorum have been described in the liver, gastric compartments, lung, nervous system, interdigital spaces, footpads, and bones. 6 We report herein a series of 5 cases with ulcerative lesions in the oral cavity, esophagus, and gastric compartments in alpacas, focusing on the clinical, pathologic, and microbiologic findings. In all of these cases, F. necrophorum was isolated, together with other aerobic and anaerobic bacteria.
Between January 2007 and March 2019, 4 alpacas with a history of loss of body condition and 1 alpaca that died during transportation were submitted to the California Animal Health and Food Safety (CAHFS) laboratory system (Davis and San Bernardino branches; n = 4) or to the University of Minnesota Veterinary Diagnostic laboratory (UMNVDL; n = 1; Table 1). An autopsy was performed on each animal, and samples from trachea, lungs, heart, liver, spleen, kidneys, adrenal glands, tongue, gingiva, esophagus, gastric compartments, small intestine, cecum, colon, and/or brain were collected and fixed by immersion in 10% neutral-buffered formalin, pH 7.2, for 24–72 h. Tissues were processed routinely and slides stained with hematoxylin and eosin; selected sections were also stained with Brown and Brenn, periodic acid–Schiff, and Giemsa.
Signalment, main gross findings, and microbiologic data of 5 alpacas with ulcers in the alimentary tract.
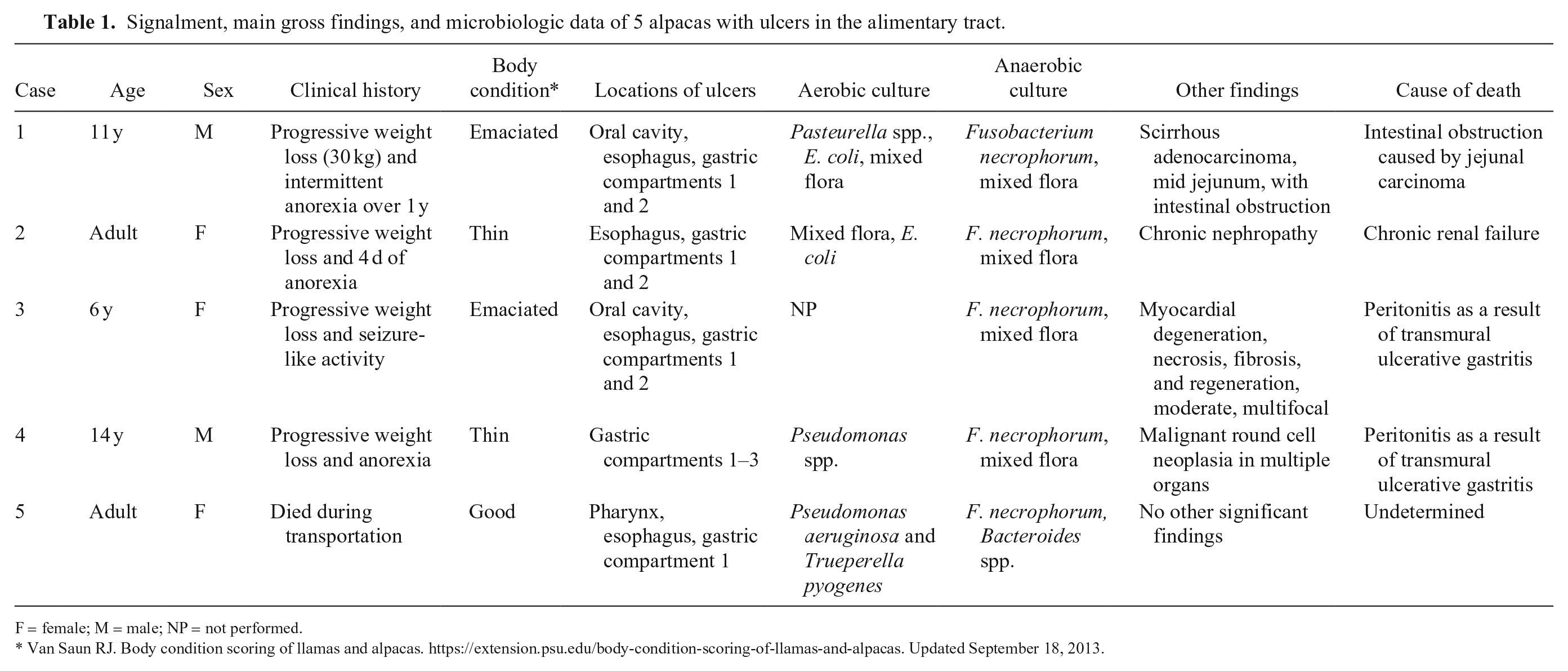
F = female; M = male; NP = not performed.
Van Saun RJ. Body condition scoring of llamas and alpacas. https://extension.psu.edu/body-condition-scoring-of-llamas-and-alpacas. Updated September 18, 2013.
All tests were performed following CAHFS or UMNVDL standard operating procedures. Samples of liver and lung, and swabs from the oral, esophageal, and/or gastric compartments were collected aseptically and processed for aerobic, microaerophilic, and anaerobic bacterial cultures. Briefly, these specimens were streaked onto chocolate, Columbia blood, and MacConkey agar plates (Hardy Diagnostics, Santa Maria, CA) and incubated at 37°C aerobically and/or under microaerophilic conditions. The plates were examined after 24 and 48 h. Aerobic culture of the ulcerative lesions of alpaca 3 was not performed. For anaerobic cultures, samples were included on pre-reduced anaerobically sterilized Brucella plates (Anaerobe Systems, Morgan Hills, CA) and cultured for up to 5 d at 37°C in an anaerobic environment.
A real-time PCR to detect a fragment of the Salmonella-specific invA gene was performed on intestinal content as described previously. 3 Salmonella culture was performed using tetrathionate or selenite enrichment broth and selective plate media from bile and colon pool and intestinal content.
The spleens from cases 1–4 were processed by PCR for bovine viral diarrhea virus (BVDV-1 and -2; Pestivirus A and B), malignant catarrhal fever virus (MCFV; Ovine gammaherpesvirus 2), bluetongue virus (BTV), and epizootic hemorrhagic disease virus (EHDV). The spleen from case 5 was tested for BVDV only. Feces were processed for parasitology by a flotation method. Liver was analyzed for heavy metals (lead, manganese, iron, mercury, arsenic, molybdenum, zinc, copper, and cadmium) by inductively coupled argon plasma–optical emission spectrometry. The selenium concentration in the liver was determined by inductively coupled plasma–mass spectrometry using hydride generation.
All cases had multifocal-to-coalescing, variably sized (0.4–30 cm diameter), depressed or slightly raised, roughly circular ulcers in different locations from the oral cavity and the gastric compartment 3 (C3), with a thick fibrous ulcer bed that was frequently covered with a tan or green pseudomembrane (Fig. 1; Table 1). In the oral cavity (n = 3), esophagus (n = 4), and C2 (n = 4), the ulcers were 0.4–2 cm diameter. In C1 (n = 5), the ulcers were up to 30 cm long by 6 cm wide (Fig. 2). In C3 (n = 1), the ulcers were 0.5–2 cm diameter. The ulcers were mostly shallow, except for those in C2 of cases 3 and 4, which were transmural, and associated with fibrinous peritonitis. Other significant gross findings were a jejunal adenocarcinoma with stenosis, obstruction, and subsequent dilation of the prestenotic jejunum of case 1; irregular, pale, and firm kidneys in case 2; and fibrinous peritonitis associated with transmural ulcers of C2 in cases 3 and 4 (Table 1). No other significant gross findings were detected in any of the animals examined.
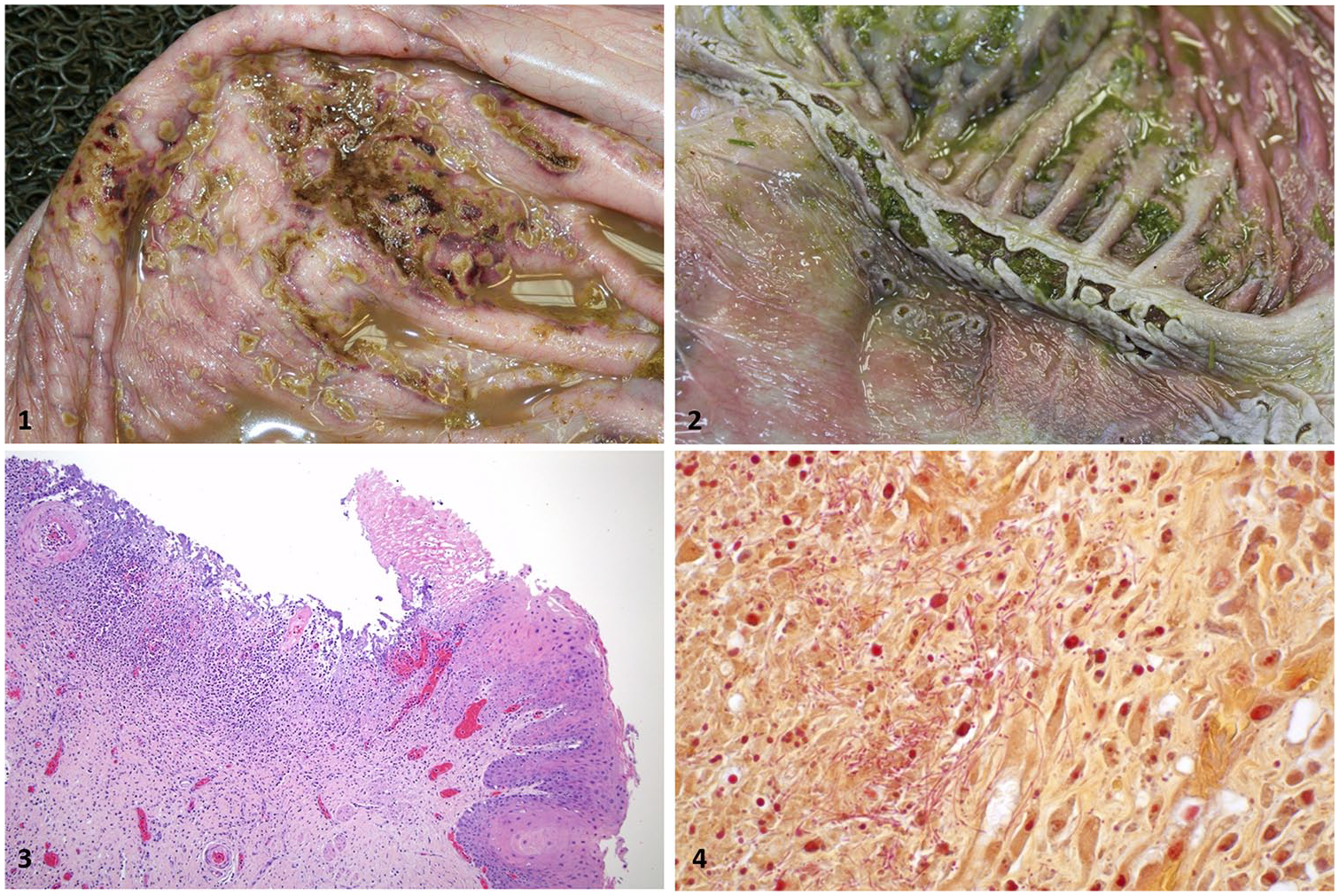
Ulcers in gastric compartments (C) in alpacas.
Microscopically in all cases, the ulcerated mucosa was replaced by abundant necrotic debris and chronic active inflammation with infiltration of viable and degenerate neutrophils, macrophages, fewer lymphocytes and plasma cells, edema, hemorrhage, and fibrin (Fig. 3). Granulation tissue and a few fibrin thrombi were present in the submucosa at the margins of the lesions. Large numbers of gram-positive and gram-negative bacteria colonized the surface of the ulcers. Deep within the ulcers, numerous gram-negative, pleomorphic filamentous bacteria were present (Fig. 4). Other microscopic findings included scirrhous adenocarcinoma in the jejunum of alpaca 1, which was responsible for the intestinal stricture seen grossly; chronic glomerulonephropathy and tubulointerstitial nephritis in alpaca 2; fibrinous peritonitis and mild cardiomyopathy characterized by multifocal myocardial degeneration, necrosis, and fibrosis of undetermined cause in alpaca 3; and malignant round cell neoplasia in the liver, spleen, kidneys, and gastric compartments in alpaca 4. The cell of origin of this neoplasm was not identified. No additional findings were observed in any of the animals.
The anaerobic culture of the wall of the gastric compartments in cases 1–4, and the esophagus of case 5, yielded moderate-to-large numbers of F. necrophorum, together with mixed aerobic and/or anaerobic bacteria (Table 1). Mixed flora, Escherichia coli, Pasteurella spp., Pseudomonas aeruginosa, and/or Trueperella pyogenes were isolated from these tissues in alpacas 1, 2, 4, and 5 (Table 1). PCR for BVDV, BTV, EHDV, and MCFV was negative on samples from alpacas 1–4. Alpaca 5 was negative for BVDV by PCR. In all cases, hepatic levels of heavy metals and selenium were within normal ranges; no parasite eggs were identified in feces, and Salmonella culture and PCR were negative.
In our series of cases, it is not known if F. necrophorum acted as a primary agent or as an opportunistic infection after previous mucosal injuries. In cases 1–4, F. necrophorum–associated lesions were considered secondary findings but were deemed responsible for the death of the animals in the 2 cases with fibrinous peritonitis (cases 3 and 4). In case 5, however, these lesions were considered the most significant finding at autopsy. Although it is not possible to confirm F. necrophorum as the causal agent of these ulcerative lesions in alpacas, several other infectious agents were ruled out with histochemical and molecular techniques.
F. necrophorum is considered a normal inhabitant of the alimentary tract of alpacas.8,11 Two subspecies of this microorganism have been identified: F. necrophorum ssp. necrophorum (biotype A) and F. necrophorum ssp. funduliforme (biotype B), the former being more pathogenic than the latter. 12 F. necrophorum major virulence factor is a leukotoxin, which induces apoptosis of ruminant leukocytes. Other important virulence attributes of this microorganism are the production of high levels of endotoxin, hemagglutinin, and a collagenolytic cell wall component that is responsible for dermotoxic activity.12,13 In a study performed with F. necrophorum isolates obtained from different necrotic lesions in alpacas and llamas, leukotoxin was demonstrated in the culture supernatant in all isolates, suggesting that this toxin may play a role in the pathogenesis of necrotic lesions in these animal species. 11
In ruminants, most of the ulcerative lesions in the upper digestive system that are associated with F. necrophorum occur after physical damage to tissues (e.g., trauma, ischemia, and parasitic or viral infections), which provides an anaerobic environment.12,23,25 Once established in the tissue, F. necrophorum proliferates, causing extensive foci of coagulative necrosis. 23 In wild ruminants, other predisposing factors have been associated with F. necrophorum infection, such as exposure to wet soil contaminated with feces of domestic ruminants, eruption of teeth, vitamin A deficiency, and intensive farming conditions.4,5,12 In pigs, oral and esophageal necrobacillosis have been associated with physical trauma (e.g., improper snout snare), previous caustic chemical damage, vesicular diseases, and exposure to mycotoxins. 16
Ulcerative lesions of the upper alimentary tract are uncommon in South American camelids, and the few cases described have been associated with multiple causes. In the oral cavity, ulcers are most commonly associated with trauma, bacterial infections (e.g., actinobacillosis) and, rarely, viral infections (e.g., camelpox, papillomavirus, and BTV). 1 Esophageal ulcers have been associated with the ingestion of caustic plants, such as rhododendron, laurel, and Labrador tea. 6 Within the gastric compartments, ulcers are usually limited to C1 and C2, and the main risk factors are ingestion of caustic or abrasive feed, bezoars, tumors, and infectious agents. 1 Erosions and ulcers of C3 of South American camelids have been described in 5–20% of all autopsy cases in a series of 87 llama cases 20 and were associated with a high grain diet, non-steroidal or glucocorticoid anti-inflammatory drugs, stress, and chronic conditions that may increase the acidity of ingesta or decrease mucosal protection.15,20 Studies have reported reflux of duodenal contents, including bile acids, into the third compartment of camelids with ulcers in C3, similar to what is seen in camelids with poor intestinal emptying, suggesting a relationship between these findings. 2 Most reported cases are associated with chronic diseases, and it is very difficult to establish when the ulcer developed and what role it played in the presentation of clinical signs.1,9 These lesions can be complicated by infection with F. necrophorum.
Ulcerative lesions of the alimentary tract in alpacas are very difficult to recognize premortem without conducting a battery of laboratory tests. 1 One factor that has been incriminated in gastric compartment damage is gastric acidosis, which has been described in South American camelids after the ingestion of highly fermentable carbohydrates and is characterized by atony, lethargy, ataxia, diarrhea, and tachycardia. 1 The pathophysiology of forestomach acidosis in camelids was believed to be similar to ruminal acidosis of ruminants and it was first postulated that the amount of grain required to induce acidosis in camelids may be less than for ruminants.1,24 However, camelids are not very susceptible to gastric acidosis because of their feeding behavior and forestomach buffering capacity.19,24 Studies have shown that abrupt feeding of grains to alpacas does elicit a pH decrease response, but not to the extent seen in true ruminants. The buffering capacity and rapid absorption of volatile fatty acids along with other undetermined factors may be responsible for this response. 18 If gastric compartment acidosis occurred, affected animals may survive with treatment, but a small proportion become chronic poor-doers with hypoproteinemia and weight loss, most likely as a result of deep non-healing ulceration of the gastric mucosa. 1 The presence of ulcers in the gastric compartments is similar to our cases. Unfortunately, the pH of the contents of C1 and C2 was not assessed in any of our 5 cases, thus whether ulcerative lesions associated with F. necrophorum coexist with abnormally low pH levels in the gastric compartments remains to be addressed in camelids. In ruminants, it is frequent to find intramucosal pustules in the ruminal mucosa as evidence of chemical damage caused by acidosis. 23 Such lesions were not present in the mucosa of C1 or C2 of our cases.
In case 5, F. necrophorum was isolated with aerobic bacteria, including T. pyogenes and P. aeruginosa. T. pyogenes and F. necrophorum are normally present on mucous membranes of the alimentary tract of camelids.8,11 Pseudomonas spp. have been implicated in opportunistic infections and linked to environmental sources, particularly aqueous systems. 22 It is postulated that primary traumatic damage occurred, which allowed F. necrophorum to invade and proliferate. Subsequent T. pyogenes invasion acted synergistically with F. necrophorum, which produced locally acting toxins and proteases that destroy tissue, trigger inflammation, and modulate the immune response.4,12,14 A similar association was described for P. aeruginosa and F. necrophorum in an immunocompromised heifer with ulcers of the tongue, esophagus, and omasum. 22 In case 5, no evidence of immunosuppression was noted; therefore, the interaction between the bacteria and their association with the death of this alpaca remains unknown.
Footnotes
Acknowledgements
We thank Ms. Sue-Ellen J. Uzal for the editing of this manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
