Abstract
A 36-y-old white rhinoceros (Ceratotherium simum) was presented with respiratory distress, sanguineous vaginal exudate, and anorexia. The clinical signs progressed over 40 d, and the rhinoceros died. Autopsy revealed significant ascites; a unilateral, 12.5-cm diameter, polypoid mass in the left ovary; a white, firm transmural mass in the right uterine horn; a white, friable mass in the lung; and white-to-tan, friable small nodules in the diaphragm. Histologic examination revealed similar neoplastic cells in the masses in all 4 locations, composed predominantly of epithelial cells proliferating in a tubulopapillary pattern with significant nuclear atypia and numerous atypical mitotic figures (18–42 per 2.37 mm2). Immunohistochemistry for CK7 (cytokeratin 7) and CK20 (cytokeratin 20) suggest that the ovarian, pulmonary, and diaphragmatic lesions were of ovarian origin and that the ovary was the primary tumor site.
Keywords
Tumors originating from female genital tracts have been reported sporadically in many species, including in dogs, cats, sheep, goats, and horses.2,6,12,14 In the family Rhinocerotidae, a few cases of gynecologic neoplasms have been reported, including uterine leiomyoma in Indian (Rhinoceros unicornis) and white (Ceratotherium simum) rhinoceroses,7,8,17 and metastatic uterine adenocarcinoma in a white rhinoceros. 16 Although leiomyoma and other benign tumors of the reproductive system are relatively common in captive female rhinoceroses, 17 malignant neoplasms are thought to be rare in white rhinoceroses. 16 Here, we report a case of metastatic ovarian adenocarcinoma in a white rhinoceros.
A 36-y-old, female white rhinoceros had intermittent mucosanguineous vaginal exudate and anorexia; these clinical signs were first observed and resolved in 2016 and were then observed again in 2019 for a 3-wk duration. Marbofloxacin (3.6 mg/kg, q24h, PO), meloxicam (0.28 mg/kg, q24h, PO), and Clostridium butyricum probiotic supplements (200 mg/kg, q24h, PO) were administered for 2 mo. The clinical signs resolved, and the general appearance of the rhinoceros improved; medications were discontinued. However, 6 mo after termination of therapy, sanguineous vaginal exudate was observed again. Cytologic examination of the vaginal discharge revealed erythrocytes, macrophages, neutrophils, and clusters of epithelial cells. The epithelial cells displayed pleomorphism, anisokaryosis, and coarse, ropy chromatin (Fig. 1); neoplastic disease of the lower genital tract was suspected. Thereafter, the same therapy was administered for 30 d.
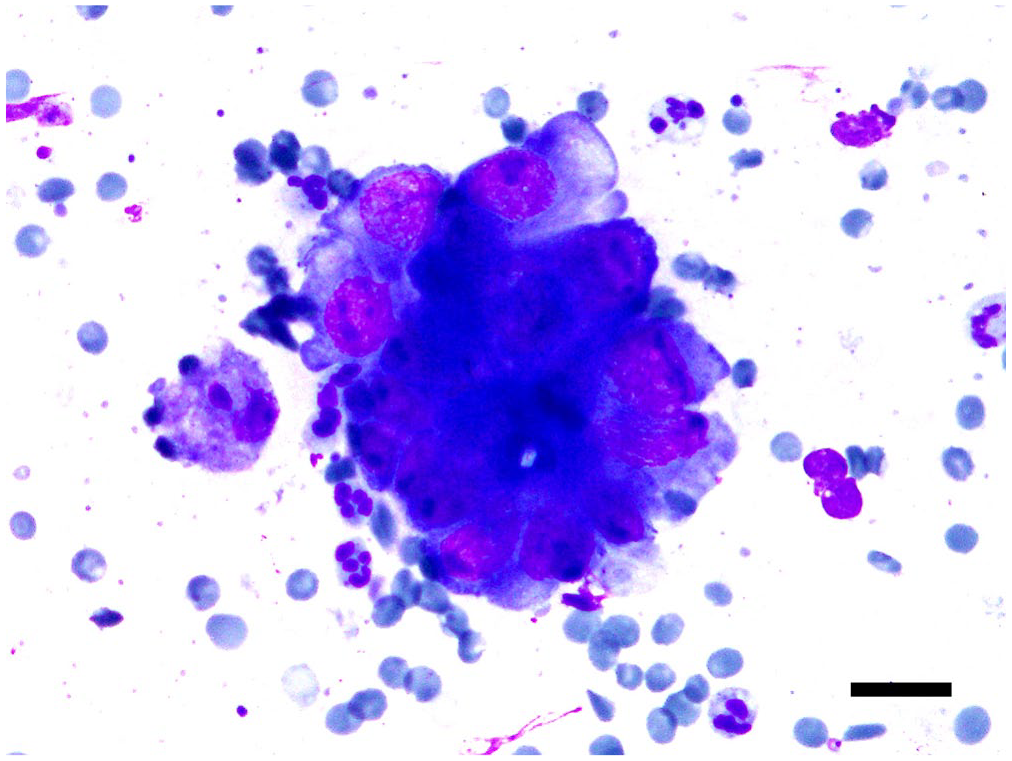
Cytology of vaginal exudate in an adult female captive white rhinoceros. Cluster of epithelial cells with marked anisokaryosis contains coarse and ropy chromatin, with distinct nucleoli. Neutrophils are scattered around the neoplastic epithelial cells. Diff-Quik stain. Bar = 20 µm.
On day 31 after the cytological examination, the rhinoceros displayed anorexia, lethargy, vaginal bleeding, and respiratory distress. Doxycycline (1.5 mg/kg, q24h, PO) and ephedrine (0.08 mg/kg, q24h, PO) were administered on day 32, but clinical signs did not improve. Dexamethasone (150 mg, q24h, IM) and ampicillin sodium (10 g, q24h, SC) were additionally injected on days 39 and 40; however, the rhinoceros died 41 d after the cytologic examination.
At autopsy, ~100 L of brown ascitic fluid filled the abdominal cavity. The right uterine horn contained 8–10 L of serosanguineous exudate. A white-to-tan, 12.5-cm diameter friable mass replaced the left ovarian parenchyma, and a white, solid, hard, 7.5-cm diameter transmural mass was in the middle part of the right uterine horn. A white, 5-cm diameter friable mass was in the right craniomedial lung lobe, and 30–40 white-to-tan, 0.2–3-cm diameter, friable nodules were distributed throughout the diaphragm (Fig. 2A–C). The right ovary and left uterine horn did not have macroscopic changes. Samples of ovaries, uterine horns, diaphragm, and lung were fixed in 10% neutral-buffered formalin for histologic examination.
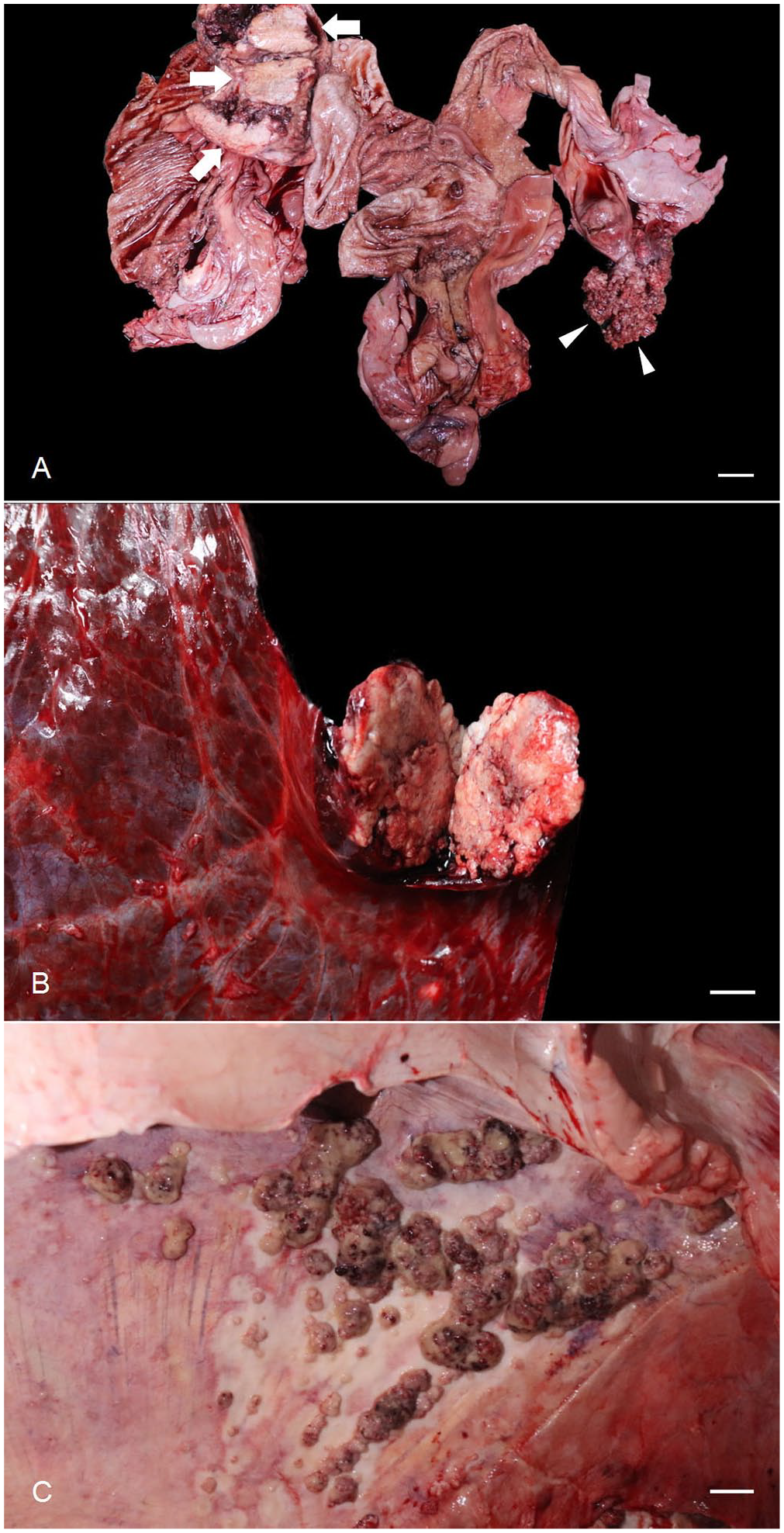
Macroscopic findings in the autopsy of a white rhinoceros.
Histologic lesions were similar in the ovary, uterus, lung, and diaphragm. In the ovary, neoplastic epithelial cells formed a tubulopapillary pattern and normal ovarian structures were effaced (Fig. 3A). Neoplastic epithelial cells appeared columnar and had marked nuclear atypia, including anisokaryosis, distinct nucleoli, and numerous atypical mitotic figures (42 per 2.37 mm2). Prominent accumulation of mucinous material was found within the cytoplasm of the neoplastic cells and inside the tubules (Fig. 3B). In the uterine lesion, the normal lining of the endometrium was not detected, and malignant epithelial cells created tubulopapillary structures. Neoplastic epithelial cells invaded the myometrium and had significant nuclear atypia and atypical mitotic figures (18 per 2.37 mm2; Fig. 3C, 3D). There was prominent vascular and lymphatic development adjacent to the lesion; however, invasion of blood or lymphatic vessels by neoplastic cells was not observed. Mucinous infiltration in the tubules was also detected, but only within a small proportion of cells compared to the ovarian lesion. In the diaphragm, an exophytic papillary neoplasm containing mucinous material was identified involving the diaphragmatic muscle (Suppl. Fig. 1A). Neoplastic cells had invaded deeper regions of the diaphragmatic muscle, and invasion of lymphatic vessels was evident. Again, severe nuclear atypia and large numbers of atypical mitotic figures (39 per 2.37 mm2) were present. Foci of osseous metaplasia were also observed in the diaphragmatic lesion, adjacent to neoplastic cells. Metastatic epithelial cells were observed in the pulmonary parenchyma, in solid and cribriform patterns (Suppl. Fig. 1B). These cells showed moderate nuclear pleomorphism and several atypical mitotic figures (18 per 2.37 mm2). There was focal aggregation of lymphocytes around the tumor.
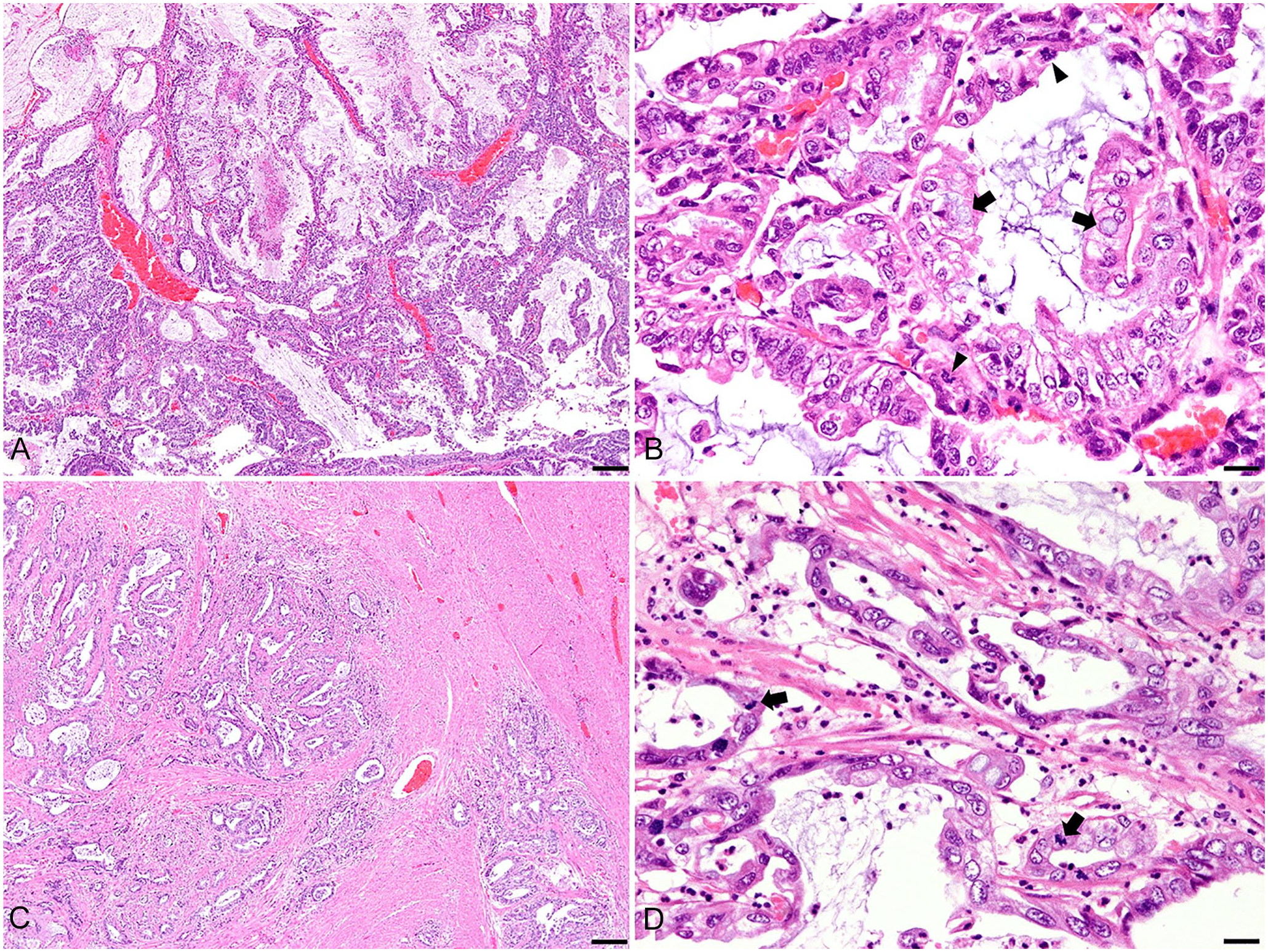
Microscopic findings in the ovary and uterus of a white rhinoceros.
We performed immunohistochemistry to help determine the tumor origin and to differentiate it from other metastatic mucinous cancers. Formalin-fixed, paraffin-embedded tissue sections were deparaffinized and immersed in 3% hydrogen peroxide to block endogenous peroxidase activity. For antigen retrieval, each section was boiled in a microwave or pressure cooker (30 min in a pressure cooker for cytokeratin 7 [CK7]; 5 min in a microwave for cytokeratin 20 [CK20]). To block nonspecific binding, sections were incubated with 5% normal goat serum for 30 min, followed by incubation with primary antibodies. After washing 3 times in phosphate-buffered saline (PBS), peroxidase-conjugated polymer (EnVision detection system; Dako) was applied and incubated for 40 min at room temperature. Samples were again washed in PBS 3 times and visualized by 3′3-diaminobenzidine solution. Sections were counterstained with Gill hematoxylin, dehydrated in graded ethanol, and coverslipped. Isotype controls were also performed.
In primary or metastatic lesions from the ovary, uterus, diaphragm, and lung, neoplastic cells had weak, diffuse cytoplasmic staining for CK7 (Suppl. Fig. 2A), whereas adjacent fibrous or muscular tissues were negative. For CK20, focal moderate-to-intense cytoplasmic or membranous staining was observed in lesions in the ovary, diaphragm, and lung, but not in the uterus (Suppl. Fig. 2B–D). This differential expression of CK20 in the uterine lesion, compared to the lesions from the other 3 organs, suggests that the cancerous cells in the uterus and other lesions may differ in their tissue of origin. CK7 tends to be constitutively expressed in both the uterine and ovarian epithelial cells.3,10 Meanwhile, focal expression of CK20 is a characteristic of ovarian mucinous adenocarcinoma but not endometrial adenocarcinoma, although its expression pattern can vary according to the subtype of ovarian adenocarcinoma.1,3,4,10,11,13
Although diaphragmatic metastasis is often involved in advanced ovarian cancer, 9 it is rare in endometrial cancer. 5 In our case, massive ascites could be related to diaphragmatic metastasis through obstruction of lymphatic vessels and the subdiaphragmatic lymphatic structure, so-called “stomata,” leading to decreased fluid evacuation. 15 In addition, increased permeability of blood vessels caused by production of growth factor from malignant cells causes accumulation of protein and albumin in the peritoneal space, resulting in a change of Starling forces and ascites. 15
It is possible that the uterine and ovarian cancers are primary cancers arising from each respective organ, and the diaphragmatic and pulmonary lesions are metastatic ovarian adenocarcinoma. However, it is also possible, and more likely, that all neoplastic lesions described here originated from the ovary and are thus ovarian adenocarcinoma. This is because neoplastic cells from the majority of lesions including the ovarian, diaphragmatic, and uterine mass focally expressed CK20, and the typical highly heterogeneous nature of ovarian adenocarcinoma 1 could explain why CK20-expressing cells were not observed in the uterine mass. Case reports and studies utilizing immunohistochemical markers in rhinoceros tissues are scarce, and species-specific bioinformatic data including amino acid sequences for this species are incomplete. Furthermore, a negative control for immunohistochemistry could not be performed because tissues were not collected from other organs. Hence, verification of the reactivity of primary antibodies for this species was challenging, and diagnosis cannot be made with complete certainty regarding the origin of the primary and secondary tumors. Nevertheless, our case demonstrates an aggressive, malignant, and ultimately fatal gynecologic cancer in a white rhinoceros, a species in which malignant tumors are considered rare.
Supplemental Material
sj-pdf-1-vdi-10.1177_1040638720986789 – Supplemental material for Ovarian adenocarcinoma with metastases in a white rhinoceros
Supplemental material, sj-pdf-1-vdi-10.1177_1040638720986789 for Ovarian adenocarcinoma with metastases in a white rhinoceros by Soo-Hyeon Kim, Byung-Joon Seung, Seung-Hee Cho, Ha-Young Lim, Min-Kyung Bae, Kyung-Yeon Eo, Yong-Gu Yeo, Jang-Won Son and Jung-Hyang Sur in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the veterinarians and staff who participated in the autopsy at Seoul Zoo. We also thank Ms. Eun-Mi Yu for preparing the microscope slides.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by the Bio & Medical Technology Development Program of NRF funded by the Korean government, MSIT (2016M3A9B6903437).
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
