Abstract
Fibroblast growth factor 23 (FGF-23) is an independent monitor of the progression of chronic kidney disease (CKD) in human medicine, and FGF-23 may have value as a biomarker in feline CKD. We evaluated the relationship between serum FGF-23 and CKD stages, and the effect of age on FGF-23 in normal cats. We measured FGF-23 and intact parathyroid hormone (iPTH) concentrations by ELISA, with intra- and inter-assay CVs ≤ 15%. The percentage recovery of FGF-23 and iPTH remained stable for up to 7 d in samples stored at −20°C and −80°C. We measured FGF-23 in 304 cats, among which 196 were diagnosed with CKD. The 108 clinically healthy cats were divided into 5 subgroups based on growth stage (0–2 y, 3–6 y, 7–10 y, 11–14 y, ≥ 15 y). No statistical difference was found in FGF-23 among age groups (p = 0.15) or by sex in healthy subjects. Using the International Renal Interest Society guideline, 34 cats were defined as CKD stage 1, 74 stage 2, 51 stage 3, and 37 stage 4. FGF-23 was higher in cats in all CKD stages than in controls. Higher serum phosphorus was observed in stage 3 (p = 0.04) and 4 (p < 0.01) compared to controls. iPTH increased as CKD progressed. Pearson analysis indicated a positive linear relationship between FGF-23 and iPTH (control: r = 0.70, p < 0.01; CKD: r = 0.46, p = 0.02). FGF-23 may be a useful biomarker of feline CKD and may precede hyperphosphatemia in advanced feline CKD.
Introduction
Fibroblast growth factor 23 (FGF-23) is a small glycoprotein produced in bone that acts on renal proximal tubules. 4 Physiologically, FGF-23 suppresses reabsorption of phosphate in the proximal tubules and intestinal phosphate absorption. 20 Mice overexpressing FGF-23 displayed hypophosphatemia, indicating that FGF-23 participates in phosphate regulation.1,22 FGF-23 is a phosphatonin that participates in the maintenance of mineral homeostasis by regulation of serum phosphate (Pi) and parathyroid hormone (PTH). 21 In human medicine, dietary Pi loading increases FGF-23 production.5,24 Patients with chronic kidney disease (CKD) have been shown to have high levels of circulating FGF-23, which could be a mechanism to compensate for disturbed phosphorus metabolism. 5 Moreover, FGF-23 has been demonstrated to be a novel independent predictor of progression of kidney disease in human patients.11,19 However, only a few studies have evaluated the FGF-23 reference interval in healthy cats and the role of FGF-23 in feline kidney diseases.
We measured the serum FGF-23 concentration in healthy cats; evaluated the effect of age on FGF-23; correlated FGF-23 with serum creatinine (sCr) and intact PTH (iPTH) concentrations for CKD staging; and assessed the relationship between serum FGF-23 and serum Pi concentration to evaluate the hypothesis that FGF-23 is a useful monitor of progressive feline CKD.
Materials and methods
Animals
All experiment procedures and protocols were approved by the Animal Care and Use Committee of China Agricultural University (permit CAU20170505-2). Client-owned cats recruited in this study were from the patient population presented to the China Agricultural University Animal Teaching Hospital between July 2017 and March 2019.
Inclusion and exclusion criteria
Patients with complete medical records were included in this study. Cats classified as clinically healthy did not have clinical signs or medical evidence of previous kidney injury, renal disease, or exposure to nephrotoxic drugs; and had normal serum biochemistry, complete blood count (CBC), and urinalysis results. A diagnosis of CKD was made based on history, CBC findings, biochemistry, imaging tests, and urinalysis. After the patients were stable and well hydrated, CKD grading was performed based on sCr according to the International Renal Interest Society (IRIS) guideline 15 with slight modification. We excluded cats with missing values of FGF-23, iPTH, or laboratory tests before or during the study period.
Clinically healthy group
This group of cats was divided into 5 subgroups (immature = 0–2 y, prime = 3–6 y, mature = 7–10 y, senior = 11–14 y, geriatric = ≥ 15 y), with the immature life stage including kittens and juniors. 31
CKD group
Cats with signs of CKD, such as inadequate urinary concentrating ability without identifiable non-renal cause, radiographic changes of kidneys, and/or a persistent increase in sCr ( < 140 μmol/L) in the past 3 mo (reference interval [RI]: 45–135 μmol/L), were defined as CKD stage 1. Renal azotemia 12 was defined as sCr > 140 μmol/L with a concurrent urine specific gravity (USG) < 1.035, or persistent azotemia on 2 consecutive occasions without evidence of a non-renal cause. Renal azotemic cats were grouped as stage 2 (sCr 140–250 μmol/L), stage 3 (sCr 251–440 μmol/L), or stage 4 (sCr > 440 μmol/L).
Sample collection and laboratory methods
Blood samples for CBC were collected in EDTA-anticoagulant tubes (1.5 mL; Kangjian). Blood samples collected in non-additive tubes (5 mL; Yongkang) were kept at room temperature (22–25°C) for ≥ 30 min and then centrifuged at 3,170 × g for 5 min (HT175R; Xiangyi Centrifuge) to separate serum. Serum was submitted to the laboratory of the China Agricultural University Veterinary Teaching Hospital for biochemical analysis on the day the sample was obtained. Residual serum samples were stored at −80°C for batch analysis of FGF-23 and iPTH within 7 d. FGF-23 and iPTH were measured in batches using commercial feline ELISA kits (Feline fibroblast growth factor-23 [FGF-23] ELISA kit, Feline intact parathyroid hormone [iPTH] ELISA kit; Deyihengda Biotech).
Verification of FGF-23 and iPTH assay performance
Precision and reproducibility were assessed by measuring intra- and inter-assay coefficients of variation (CV) in feline samples with low, medium, and high FGF-23 and iPTH concentrations. We measured 6 samples of each FGF-23 value 5 times, and measured 5 samples of each iPTH value 3 times, each on the same day, for intra-assay CV. We evaluated 5 each of low-, medium-, and high-concentration samples, which were different from those in the intra-assay CV, measured daily for 4 (FGF-23) or 3 d (iPTH). Temperature stability of feline FGF-23 and iPTH was examined by storing 3 samples (baseline values measured in advance) each at 4°C, −20°C, and −80°C; we then measured each sample 3 times after freezing for 3 h and freezing for 7 d, compared to the baseline.
Data analysis
Data analyses were performed (Statistics 22.0, SPSS; Prism 8.0, GraphPad). Descriptive statistics were used to characterize demographic data, including sex, neuter status, and breed. For CKD cats, CKD stage was determined and the prevalence of each stage or sex was reported as a ratio to the total number of recruited cats. The quantitative data (FGF-23, iPTH, creatinine, and Pi concentrations) were assessed for normal distribution via the Kolmogorov–Smirnov test (n > 50) and Shapiro–Wilk test (n < 50), followed by the homogeneity of variance test. For normally distributed data, the differences between 2 groups were analyzed using an unpaired t-test, and differences involving ≥ 2 groups were analyzed using one-way analysis of variance (ANOVA) and the Dunnett test to evaluate for multiple testing. Results were expressed as the mean ± SD. For data not normally distributed, the Welch ANOVA was used to assess the differences and expressed as median with range (min.–max.). Association between parameters was analyzed with Pearson correlation analysis and regression analysis. The level of significance was set at p ≤ 0.05.
Results
The FGF-23 and iPTH assays of feline serum were run in duplicate. The intra-assay CVs of FGF-23 and iPTH were < 10%, and the inter-assay CVs were ≤ 15% (Table 1). No significant changes were found in the recovery of FGF-23 or iPTH for 3 samples stored for up to 7 d at −20°C (p = 0.37, p = 0.29, respectively) or at −80°C (p = 0.42, p = 0.45). But there was a significant decrease in the recovery of FGF-23 after 7 d for 3 samples stored at 4°C (p = 0.02).
Verification of the performance of a feline serum fibroblast growth factor 23 (FGF-23) ELISA.

One value was missing because of sample mishandling.
A total of 1,879 cats visited the hospital over the study period; 426 cats with complete medical records qualified for our study. We dropped 125 cats from the study because of lost data, inability to follow-up visit, or any problems that might have negative effects on the study. Eventually, 304 subjects were included, of which 196 were diagnosed with CKD based on clinical and laboratory findings. In the CKD group, 114 of 196 (58.2%; 56.1% castrated) were male and 82 of 196 (41.8%; 48.8% spayed) were female; cats were classified as stage 1 (n = 34), stage 2 (n = 74), stage 3 (n = 51), or stage 4 (n = 37). Median age was 11 y (range: 5 mo–23 y).
The remaining 108 of the 304 subject cats were considered clinically healthy; we found no differences in this group in serum FGF-23 concentration among the 5 age categories (p = 0.15; Fig. 1). There was no statistical difference in serum FGF-23 between sex or neuter status (Table 2), but FGF-23 in healthy male cats (194 ± 16.7 pg/mL) was somewhat higher than in females (163 ± 19.8 pg/mL, p = 0.08). The RI of serum FGF-23 in our study was 0–336 pg/mL (95% CI). The median age of the control group was 3 y (range: 4 mo–20 y), and breeds included American Shorthair (n = 31), Domestic Shorthair (n = 26), British Shorthair (n = 18), Domestic Longhair (n = 14), Scottish Fold (n = 6), Munchkin (n = 5), Birman (n = 4), and Leopard cat (n = 4). No difference was found among breeds (p = 0.58; Table 2). No difference was found in FGF-23 between healthy geriatric cats and those ≤ 11 y old (p = 0.78).
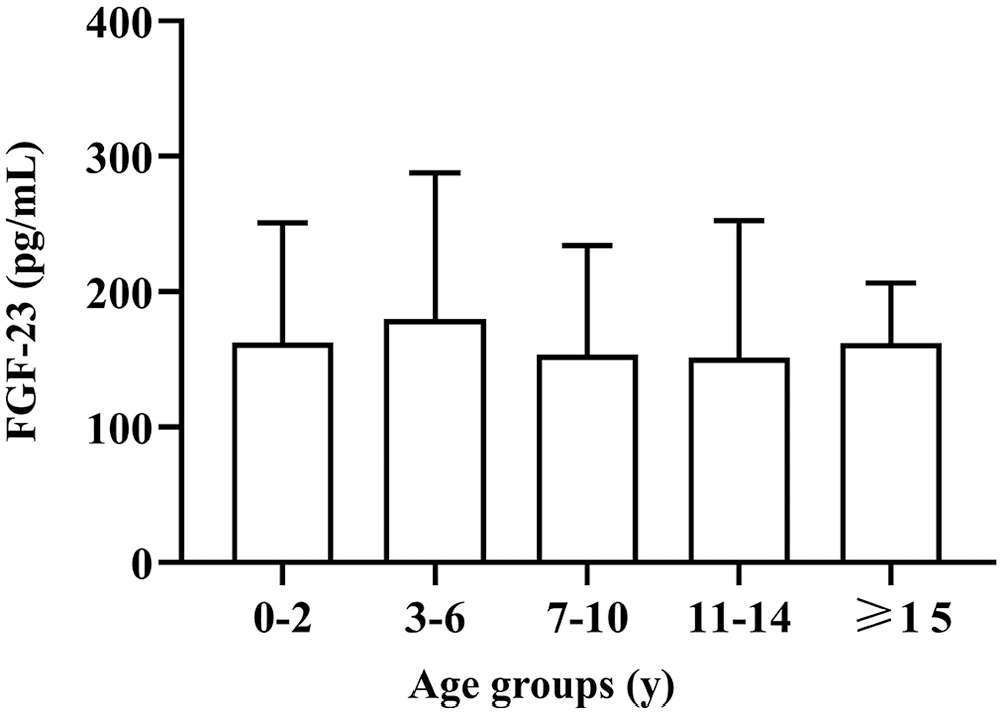
Serum fibroblast growth factor 23 (FGF-23) concentration in 108 healthy control cats. Cats were grouped by age (group 1: 0–2 y; group 2: 3–6 y; group 3: 7–10 y; group 4: 11–14 y; group 5: ≥ 15 y). Bar on each column is 1 SD.
Characteristic of serum fibroblast growth factor 23 (FGF-23) in 108 healthy control cats and 196 cats with chronic kidney disease (CKD).

Data are presented as mean ± SD. Breeds including ≤ 6 samples include * Munchkin, Birman, and Leopard cat in the control group, and Exotic Shorthair, Cornish Rex, Maine Coon, Persian cat, Ragdoll, Siamese, Sphynx, Leopard cat, and Abyssinian in the CKD group.
The prevalence of CKD was significantly higher in females (11.33% in female vs. 9.87% in male; p < 0.01). Breeds observed in CKD cats included Domestic Shorthair (n = 77), Domestic Longhair (n = 32), American Shorthair (n = 26), British Shorthair (n = 14), Scottish Fold (n = 8), Exotic Shorthair (n = 5), Ragdoll (n = 5), Persian (n = 5), Siamese (n = 5), Cornish Rex (n = 4), Maine Coon (n = 4), Sphynx (n = 4), Munchkin (n = 3), Leopard (n = 3), and Abyssinian (n = 1). The prevalence of CKD increased with age; 1.9%, 11.3%, 22.5%, 38.5%, and 48.8% in the 5 age groups, respectively.
Pearson correlation analysis revealed positive correlation of FGF-23 with serum Pi (control group: r = 0.33, p = 0.03; CKD group: r = 0.34, p = 0.03) and iPTH (control group: r = 0.70, p < 0.01; CKD group: r = 0.46, p = 0.02). In cats with CKD, serum FGF-23 was significantly higher in all CKD stages (stage 1: p = 0.04; stages 2–4: p < 0.01, respectively; Fig. 2) compared to the controls (Table 3). There were significant differences in serum Pi and iPTH levels between the control group and CKD stage 3 and 4 (p = 0.03, p < 0.01, respectively; Fig. 2).
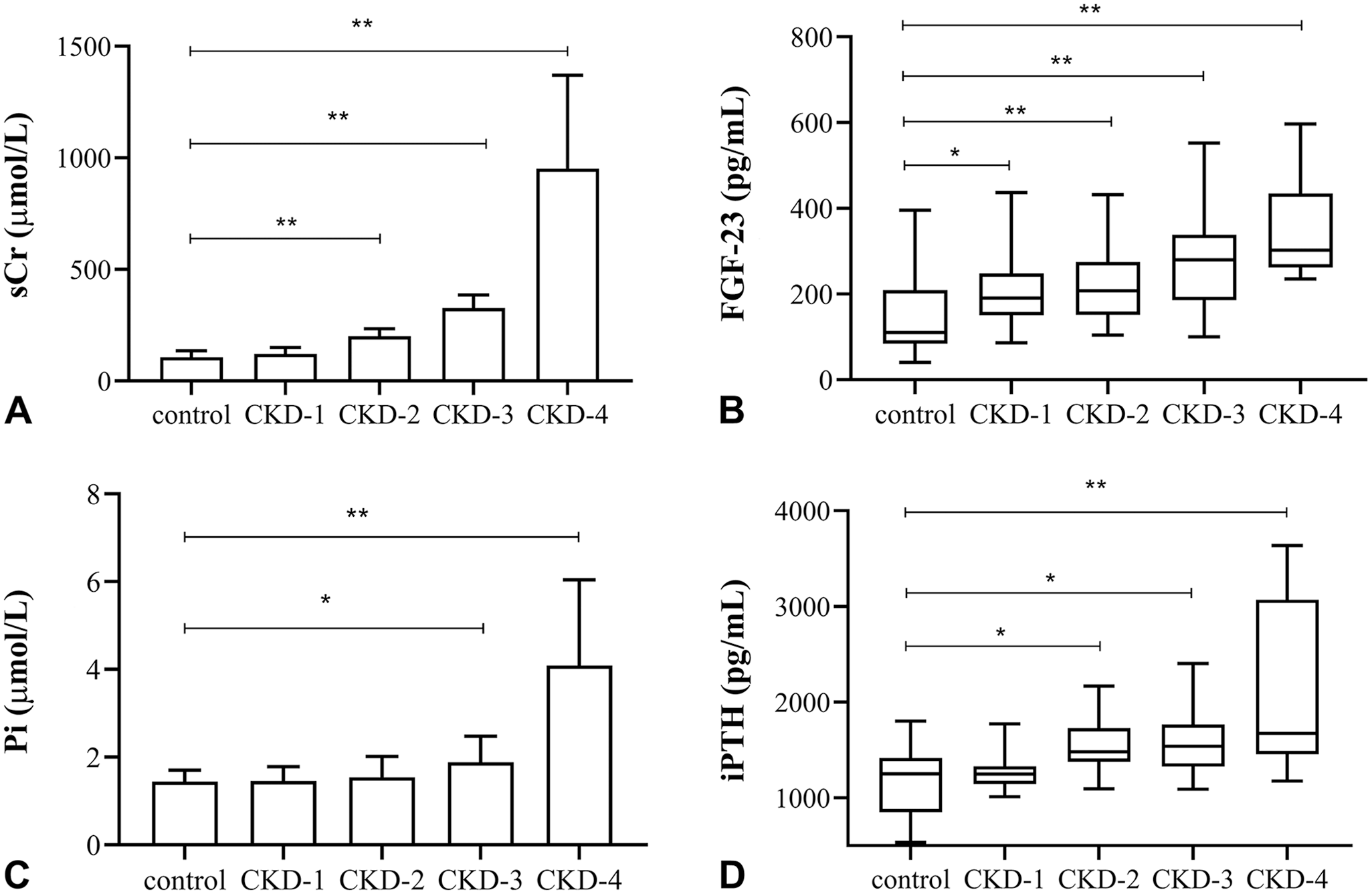
Biochemistry results for 304 cats.
Median (range) serum chemistry, fibroblast growth factor 23 (FGF-23), and intact parathyroid hormone in 304 cats.
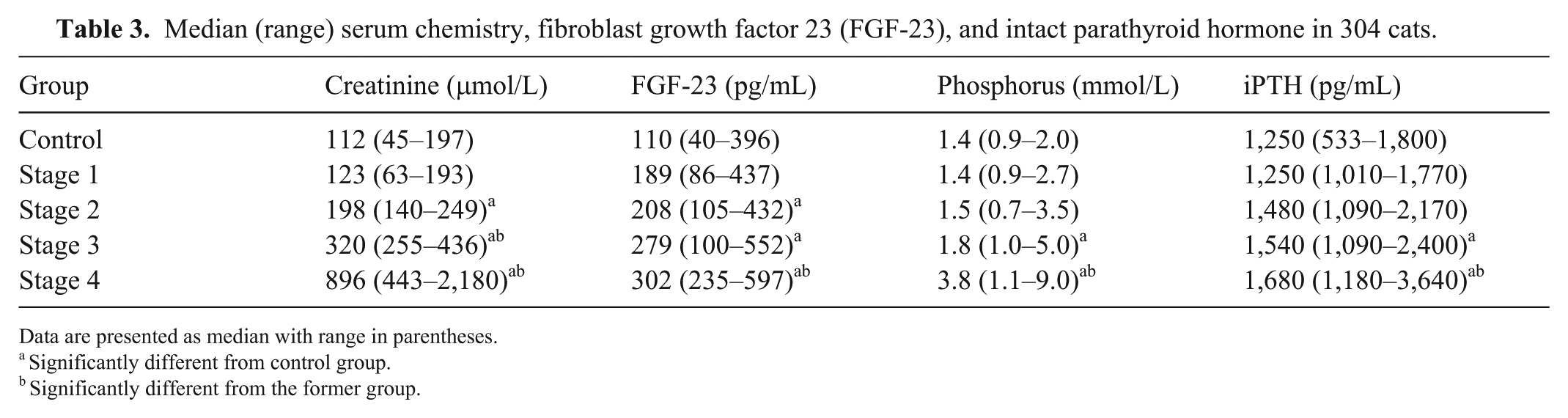
Data are presented as median with range in parentheses.
Significantly different from control group.
Significantly different from the former group.
Discussion
Age, sex, and neuter status did not have significant effects on the serum FGF-23 levels of healthy cats in our study. Males had slightly higher (non-significant) FGF-23 levels than females, which is similar to the results in human studies.17,25,30 Given that the estrogen receptor is located in proximal tubules and is important in regulating the structure and function of kidneys, as well as in Pi excretion, 28 it has been suggested that female hormones could inhibit the action of FGF-23. A previous study 9 reported that the RI for FGF-23 in geriatric cats (median age: 13 y old) was 56–700 pg/mL, which was higher than we observed (0–336 pg/mL). The reason for this marked difference could be regional and age variation, or assay-specific. We used a feline-specific FGF-23 ELISA kit and therefore it is uncertain that the results obtained by a human FGF-23 ELISA are comparable to those in our study. Moreover, a prior study 11 indicated that FGF-23 increased in geriatric cats with non-azotemic CKD. Unfortunately, we did not include sufficient data from non-azotemic cats or healthy controls to build a RI. Thus, further large-scale studies are still needed.
Serum FGF-23 increased with the progression of CKD, consistent with findings published for humans, cats, and dogs.6,8,12,14,27 In our study, serum FGF-23 concentrations became significantly higher in CKD stage 1 compared with the control group (p = 0.04), indicating that FGF-23 was more sensitive than sCr as an indicator of renal dysfunction. Thus, FGF-23 may be a practical biomarker for the early detection of CKD. The FGF-23 concentration increased earlier than the increase in serum Pi. In humans, FGF-23 works as a Pi-regulating hormone in response to hypophosphatemia and hyperphosphatemia.7,18,29 Hence, a phosphorus metabolism disorder may appear before CKD-related hyperphosphatemia is apparent. Measurement of serum FGF-23 may be a useful tool for monitoring Pi homeostasis and hypophosphatemic or hyperphosphatemic disorders. High levels of FGF-23 and serum Pi were also found to be independent risk factors of unfavorable mortality in human dialysis patients with end-stage renal disease.13,26 Data in companion animals are not yet available.
Renal secondary hyperparathyroidism (RHPT) and hyperphosphatemia are common comorbidities in advanced CKD. We found a positive linear relationship between FGF-23 and Pi concentration in cats with or without CKD. Increased serum FGF-23 in cats with early-stage CKD (stage 1 and 2) may be a response to changes in serum Pi to maintain the Pi concentration within the normal range, as occurs in humans. 16 In vivo and in vitro experiments showed that FGF-23 in rats suppressed both PTH secretion and PTH gene expression. 2 This finding indicated that FGF-23 acts directly on the parathyroid gland to decrease serum PTH. We observed a strong positive linear correlation between FGF-23 and iPTH levels, suggesting lack of suppression of PTH by FGF-23 in cats with progressive CKD (Fig. 2B, D). A calcium and phosphorus metabolic disturbance may be responsible for the increase of PTH in RHPT. The observation that FGF-23 was correlated with iPTH in our study supported this point of view. In human medicine, increasing FGF-23 is a strong predictor of mortality in dialysis patients. Increased FGF-23 develops with decreasing estimated glomerular filtration rate and manifests earlier than increased serum Pi or PTH. 16 Cats in CKD stage 1 and 2 develop RHPT before hypocalcemia and hyperphosphatemia are evident. 10 Positive correlation between FGF-23 and PTH concentrations was also documented in dogs and human patients,3,14,16,23 indicating that an increase of FGF-23 may also be an indicator of mineral metabolism disorder in feline CKD.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by a research project grant awarded by the China Agricultural University (2016YFD0501010).
