Abstract
A juvenile raccoon was euthanized because of severe neurologic signs. At postmortem examination, no significant gross lesions were present. Histologic evaluation demonstrated nonsuppurative encephalitis in thalamus, brainstem, and hippocampus, cerebellar Purkinje cell loss, as well as poliomyelitis and demyelination of the spinal cord. Parvovirus antigen–specific immunohistochemistry revealed immunopositive neurons in the brainstem, cerebral cortex, and hippocampus. A few Purkinje cells were also immunopositive. DNA extracted from formalin-fixed, paraffin-embedded brain tissue (thalamus, hippocampus, cerebral cortex) yielded a positive signal using PCR targeting both feline and canine parvovirus. Sequencing analyses from a fragment of the NS1 gene and a portion of the VP2 gene confirmed the presence of DNA of a recent canine parvovirus variant (CPV-2a–like virus) in the cerebellum. Our case provides evidence that a recent canine parvovirus (CPV) strain (Carnivore protoparvovirus 1) can infect cerebral and diencephalic neurons and cause encephalitis in an otherwise healthy raccoon. Parvovirus-induced encephalitis is a differential diagnosis of rabies and canine distemper in raccoons with neurologic signs.
On the East Coast of the United States, rabies virus (RV; Rabies lyssavirus) infection and canine distemper virus (CDV; Canine morbillivirus) infection are the top differential causes of viral encephalitis in raccoons. 7 Herein, we report a juvenile raccoon (Procyon lotor) infected with a recent canine parvovirus (CPV; Carnivore protoparvovirus 1) strain in the brain and manifesting encephalitis.
A male juvenile raccoon, estimated to be 4–5 mo old, based on size and body weight (1.5 kg), was admitted to the Wildlife Rehabilitation Center of Minnesota (Roseville, MN) because of head shaking and ataxia. After the admission examination, the animal was euthanized with the top differential diagnoses of CDV infection or cerebellar hypoplasia, based on the experience of the clinicians. At postmortem examination, the animal was in a good nutritional state. Significant gross lesions were not detected, but a few ascarids (presumably Baylisascaris procyonis) were present in the small intestine.
At histologic evaluation, lesions were present in the brain and spinal cord. Numerous capillaries, mostly in the gray matter, were surrounded by lymphocytes and fewer plasma cells and macrophages (Figs. 1, 2). The ventral thalamus, hippocampus, and parahippocampal cortex were particularly affected. The hippocampus had extensive loss of pyramidal neurons, including the CA2 and CA3 segments; glial proliferation and hypertrophy and scattered infiltration of lymphocytes were evident (Figs. 1, 2). A few necrotic neurons were present in the hippocampus and nuclei of the reticular formation of the mesencephalon, pons, and cerebellum (Fig. 3). The cerebellar vermis had segmental loss of Purkinje cells with mild Bergmann gliosis and individual scattered necrotic Purkinje cells. There was moderate gliosis and a few necrotic neurons in the cerebellar nuclei. There was satellitosis of glial cells and inflammatory cells around neurons in the brain cortex, hippocampus, thalamus, mesencephalon, and pons (Fig. 3). Neuronophagia was present in these glial nodules. Throughout the brain, affecting mostly the telencephalic “U” fibers, the pyramids, the reticular formation, and cerebellar peduncles, there were numerous swollen axons. Macrophages were present in dilated degenerate myelin sheaths, undertaking myelinophagia and axonophagia. In the thoracic segment of the spinal cord, there was marked demyelination with gliosis in lateral and ventral funiculi; the integrity of the dorsal funiculi was preserved (Figs. 4–6). A few degenerate axons and numerous macrophages with gitter cell morphology were present within degenerated myelin sheaths (Fig. 6). Perivascular lymphocytic infiltration, microglial nodules, and neuronal satellitosis and neuronophagia were present in the spinal cord gray matter. Significant lesions were not detected in other organs, including the small intestine and bone marrow.
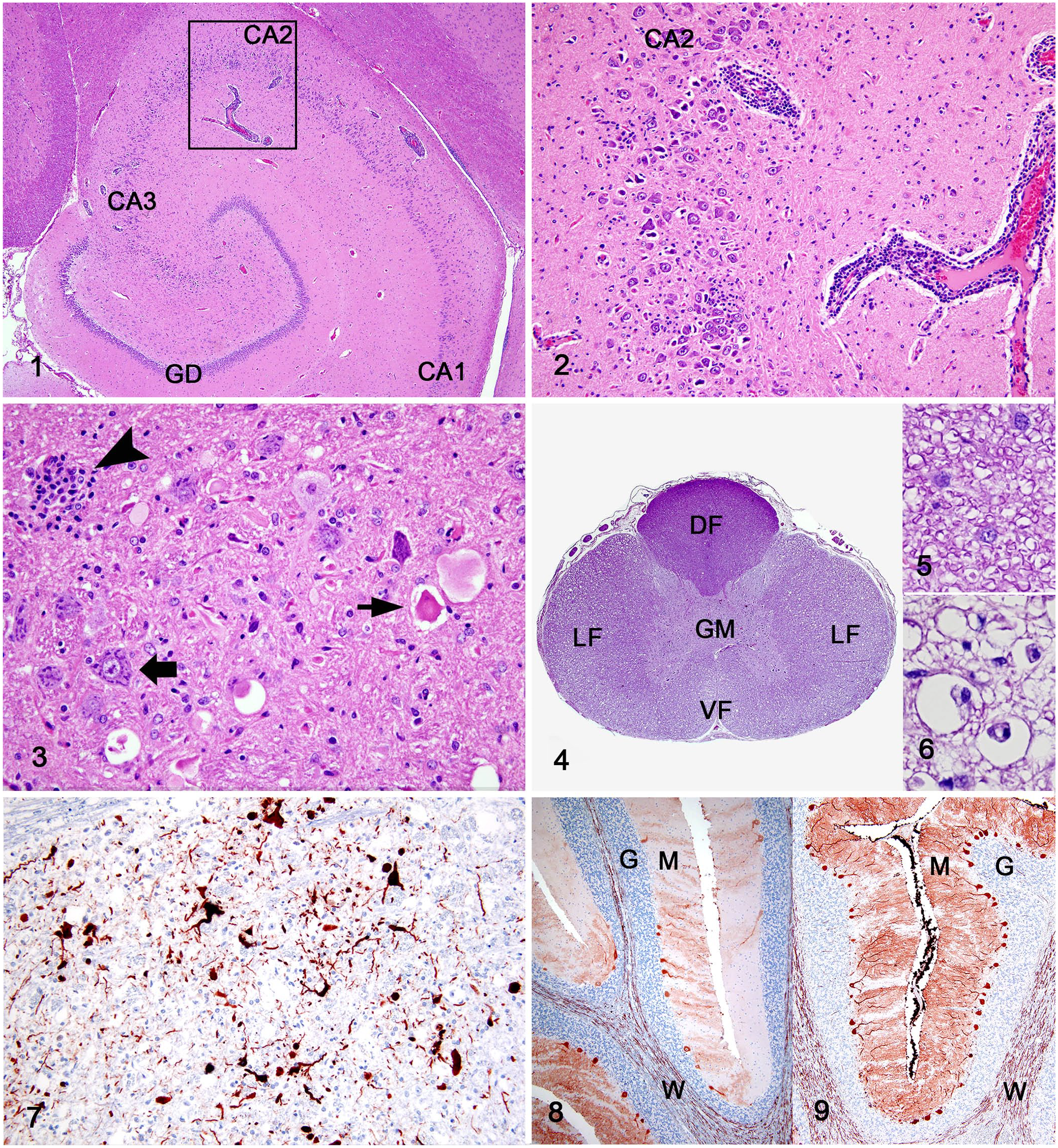
Parvovirus-induced encephalitis in a juvenile raccoon.
Immunohistochemistry using markers specific for CPV antigen, CDV antigen, RV antigen, and calcium-binding protein (calbindin) was performed with a peroxidase polymer system (Suppl. Table 1). Calbindin immunoreactivity is specific for Purkinje cells in the cerebellum.
8
An automated slide stainer (Dako) was utilized with a peroxidase-labeled polymer conjugate system (Dako) as a secondary antibody (EnVision+ goat anti-mouse). A moderate number of CPV antigen–positive neurons and cell processes was present in the hippocampus (1–5/high-power field [hpf]) and reticular formation nuclei of the mesencephalon and pons (up to 10/hpf; Fig. 7). A few Purkinje cells (
Formalin-fixed (~ 24 h of fixation in 10% neutral-buffered formalin), paraffin-embedded cerebellar tissue was examined for parvoviral DNA. The sample was handled and processed inside a biosafety cabinet to avoid cross-contamination. The extraction process was repeated twice, resulting in the generation of duplicate samples that were further processed in 2 independent experiments to address reproducibility. DNA was extracted (E.Z.N.A. tissue DNA kit; Omega Bio-tek) as per the manufacturer’s instructions and quantified (> 10 ng/µL; NanoDrop 2000/2000c spectrophotometer; Thermo Scientific). The isolated DNA was used as template for PCR (Q5 High-Fidelity DNA polymerase; New England BioLabs) under the conditions recommended by the manufacturer (30 cycles). The following set of primers, specific for nonstructural protein 1 (NS1) and viral capsid protein 2 (VP2) parvoviral genes was used: 5′-ATAAAAGACAAACCATAGACCGTTACTGAC-3′ and 5′-ACTCGCTTGCACGTCTTT-3′ (for NS1 gene, ~ 2,000 base amplicon), and 5′-CAGCCTGCTGTCAGAAATGA-3′ and 5′-CTTAACATATTCTAAGGGCAAACCAACCAA-3′ (for VP2 gene, ~ 2,000 base amplicon). The PCR amplifications showed positive results for both of the NS1 and VP2 genes, confirming the presence of parvoviral DNA; negative controls had no DNA amplification. Amplification products were purified with 0.45 × volume AMPure XP beads (Beckman Coulter).
The nucleotide sequences, determined using Sanger sequencing, included nucleotides 350–1,190 (NS1 gene) and 2,995–3,715 (VP2 gene). Sequences determined were aligned to a CPV reference sequence (CPV-2; GenBank M38425.1) and to references of feline parvovirus (FPV; M38246.1) and CPV-2a (GenBank MN451663.1) using Geneious Prime v.2019.0.4. 9 The analysis showed 4 polymorphic sites in the NS1 gene that were synonymous; the sequences were more closely matched with CPV than FPV. The VP2 capsid gene sequence had the characteristics of CPV-2a–derived. 14 Sequences of VP2 capsid protein showed substitutions of amino acids at residues 80, 87, 93, 101, and 103 (Suppl. Table 2). These substitutions distinguished the virus from both FPV and the original CPV-2 viral strain. The sequence of the parvovirus from this raccoon is consistent with sequences found in contemporary dogs and raccoons in a study of natural host samples, 1 and is identical in the examined gene segments to a CPV that was associated with cerebellar hypoplasia in a juvenile raccoon in Minnesota in 2018. 17
Both feline and canine parvovirus are readily capable of infecting raccoons. 2 Transmission of introduced CPVs occurs among raccoons; raccoon-adapted variants have been detected.1,2,4 The most common manifestation of carnivore protoparvovirus infection in raccoons is parvoviral enteritis. 12 The raccoon of our case did not have any evidence of an enteric parvoviral infection based on the absence of gross and histologic lesions, the immune-negativity of intestinal tissue for parvoviral antigen, and the negative electron microscopy result for the intestinal contents. Instead, the parvoviral infection was manifest as encephalitis. The detection of viral DNA by PCR and sequencing, and of viral protein antigen expression by IHC, is evidence of active replication of CPV-2 in the brain. The lack of detection of an alternative cause of nonsuppurative encephalitis in raccoons, such as RV, CDV, or protozoa, supports our conclusion that parvovirus was the cause of the encephalitis. 7 Although CDV is a common cause of encephalitis in raccoons in Minnesota, RV infection is extremely uncommon in Minnesota’s raccoon population.3,11
To our knowledge, encephalitis caused by parvoviral infection has not been described in raccoons. 7 Parvoviral infections of the central nervous system, particularly of neurons, are thought to be rare given that parvovirus requires mitotically active cells for replication. It is unknown why a viral infection that is usually limited to the replicating cells of the alimentary and hematopoietic system resulted in a productive infection of neurons in this raccoon. Subpopulations of neurons in the mature feline brain are suspected to retain the potential for re-entering the cell cycle based on the neuronal expression of the cell cycle proteins PCNA and Ki67. 6 Furthermore, FPV has been shown to proceed far into its replication cycle in post-mitotic Purkinje cells of cats using mechanisms still to be elucidated. 13 A 2019 study described an 8-wk-old and a 5-mo-old cat diagnosed with a productive and clinically manifest parvoviral encephalitis with widespread neuronal and glial cell infection and neuronal necrosis. 10 Furthermore, encephalopathies, encephalitis, and meningoencephalitis have been reported in immunocompromised and immunocompetent humans with parvovirus B19 infection.5,16 However, the diagnoses in the human patients were established based on clinical signs and PCR analysis of cerebrospinal fluid; central nervous system lesions were not described.
It is unknown how the parvovirus gained access to the brain parenchyma and spread within the brain of this raccoon. Principally, viruses gain access to the brain parenchyma via ascending infections of nerves or hematogenously. 15 The latter route requires either transport of virus from endothelial cells across the blood–brain barrier to pericapillary astrocytes, entry at points of reduced blood–brain barrier efficiency, such as choroid plexus or circumventricular organs, or the virus passes the blood–brain barrier within leukocytes. 15 The relatively young age of the raccoon suggests that the animal was infected intrauterine or shortly after birth. Analogous to cats, the infection of a juvenile raccoon with a parvovirus, identical to the one detected in our case in the examined gene segments, has been linked to cerebellar hypoplasia and dysplasia. 17 In contrast to cases of parvoviral cerebellar hypoplasia and dysplasia, the lack of any significant disorganization of the cerebellar cortical architecture suggests that the infection occurred after the stratification process was accomplished. Nevertheless, the multifocal lack of Purkinje cells in the face of a few parvovirus antigen–positive Purkinje cells may suggest that the infection directly or indirectly (e.g., as a result of excitotoxicity) induced insidious degeneration and death of this cell population in the absence of significant cerebellar inflammation.
Supplemental Material
Supplemental_material – Supplemental material for Parvovirus-induced encephalitis in a juvenile raccoon
Supplemental material, Supplemental_material for Parvovirus-induced encephalitis in a juvenile raccoon by Arno Wünschmann, Robert Lopez-Astacio, Aníbal G. G. Armién, Leslie Reed and Colin R. Parrish in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This project was funded by the University of Minnesota Veterinary Diagnostic Laboratory.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
