Abstract
A 14-y-old spayed female Labrador Retriever was presented with an 8-mo history of chronic vomiting. Abdominal ultrasound and gastrointestinal endoscopy revealed a mass protruding into the gastric lumen, with cytologic features suggestive of sarcoma. A partial gastrectomy was performed; the gastric body and antrum were thickened, with a cerebriform appearance of the mucosal surface. Histologic examination revealed a submucosal neoplastic proliferation of fusiform cells variably arranged in irregular bundles and scattered whorls. Fusiform cells strongly reacted to antibodies against vimentin, S100, and neuron-specific enolase; glial fibrillary acidic protein was moderately and multifocally expressed. Pancytokeratin, KIT, α–smooth muscle actin, and desmin were nonreactive. Histologic and immunohistochemical findings suggested a diagnosis of gastric sarcoma with features referable to a non-GIST (gastrointestinal stromal tumor), non–smooth muscle NIMT (non-angiogenic, non-lymphogenic intestinal mesenchymal tumor). The overlying gastric mucosa was thickened by elongated and dilated gastric glands, predominantly lined by intensely periodic acid-Schiff–stained mucous cells. This altered mucosal architecture was suggestive of Ménétrier-like disease. Although this disease has been hypothesized to predispose to gastric adenocarcinoma in dogs, an association with gastric sarcoma has not been documented previously in the veterinary literature, to our knowledge.
A 14-y-old spayed female Labrador Retriever was presented to the Veterinary Teaching Hospital, University of Teramo (Italy), with an 8-mo history of chronic vomiting occurring at a variable time (usually 5–6 h) after eating, in association with a recent onset of hyporexia. Physical examination and urinalysis were unremarkable, whereas complete blood count and serum biochemical profile revealed mild microcytic, hypochromic anemia (hematocrit 0.27 L/L, reference interval [RI]: 0.37–0.55 L/L; hemoglobin 81 g/L, RI: 120–180 g/L; red blood cell count 4.65 × 1012/L, RI: 5–8.5 × 1012/L; mean corpuscular volume 58 fL, RI: 60–77 fL; mean corpuscular hemoglobin 17.5 pg, RI: 19.5–24.5 pg; mean corpuscular hemoglobin concentration 303 g/L, RI: 320–380 g/L), as well as decreased iron levels (5.2 μmol/L, RI: 14.3–39.4 μmol/L) and saturation (8%, RI: 28–65%).
Abdominal ultrasound examination showed moderate dilation of the gastric lumen, which was filled with anechoic liquid and gas. A mass was detected originating from the gastric wall and protruding into the lumen, characterized by complete loss of normal wall layers, as well as heterogeneous echogenicity and echotexture (Fig. 1, inset). A gastrointestinal endoscopic examination also confirmed the presence of a proliferative mass showing cerebriform appearance and partially occluding the gastric lumen. During endoscopy, multiple biopsy samples of the gastric mass were taken with grasping forceps, and slides with impression smears of biopsied tissue were submitted for cytologic examination. Because cytologic examination revealed a cellular population composed predominantly of atypical fusiform cells, suggestive of gastric sarcoma, the dog underwent partial gastrectomy, including pylorectomy, and the excised gastric wall was submitted for histologic examination.
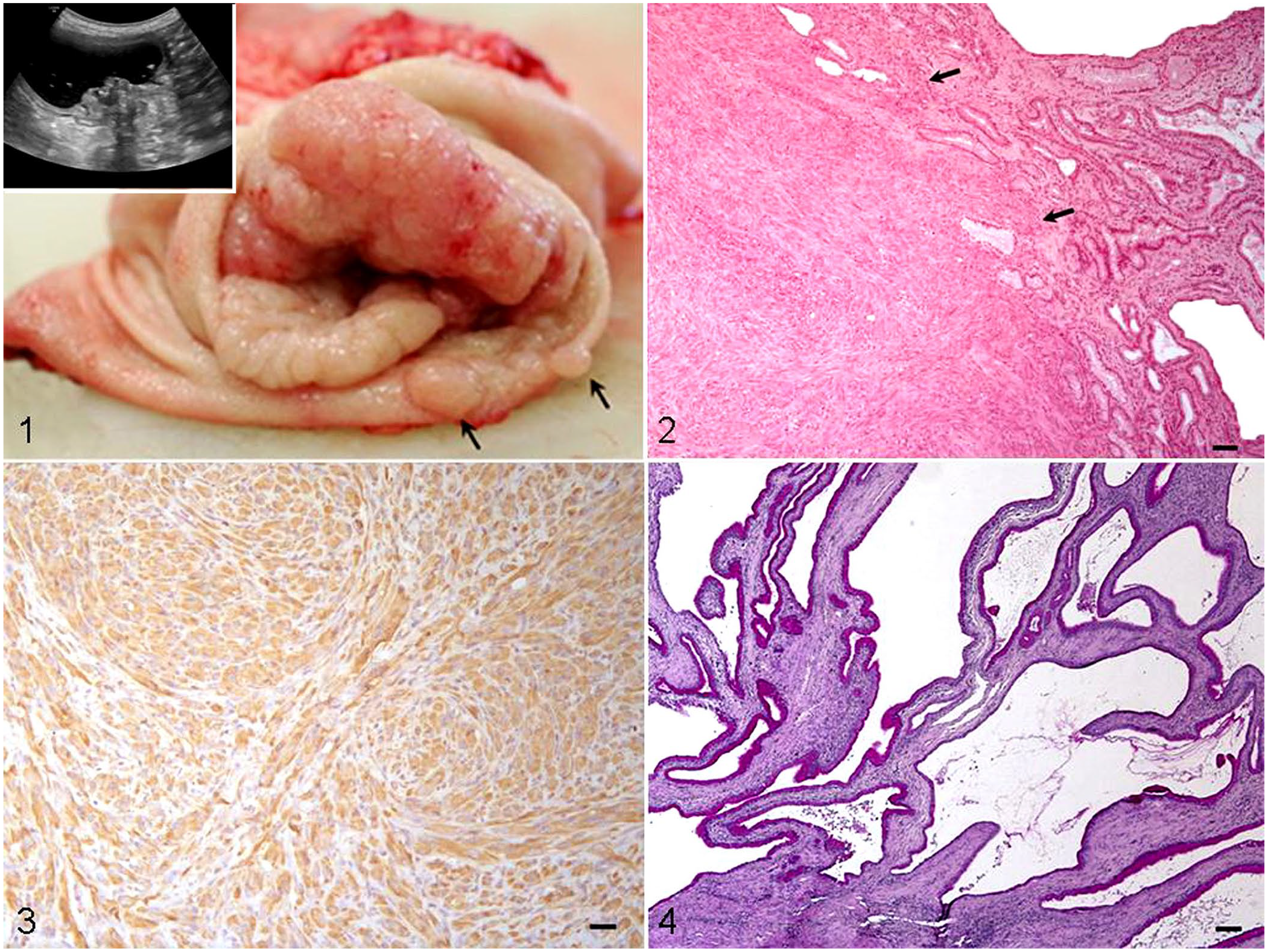
Hypertrophic gastropathy associated with gastric sarcoma in a dog.
During recovery from surgery, the patient died suddenly after an acute onset of respiratory distress and the owner declined postmortem examination. Although the cause of the death remained unclear, pulmonary thromboembolism was suspected.
The surgically excised gastric wall, including part of the body, antrum, and pylorus, had a wide, mass-like thickening (up to 5 cm thick), encircling the gastric body and antrum, characterized by a cerebriform appearance of the mucosa, which was multifocally ulcerated. Nodular thickenings (0.3–1 cm diameter) of the adjacent gastric mucosa were also observed (Fig. 1).
Samples of the thickened gastric wall, as well as samples of the surrounding, grossly normal wall, were fixed in 10% neutral-buffered formalin, processed routinely, and sections were stained with hematoxylin and eosin, as well as periodic acid–Schiff (PAS). Additional sections were also subjected to immunohistochemical staining (IHC) using primary antibodies directed against vimentin (1 in 100 dilution, V9, mouse monoclonal; Dako), pancytokeratin (CK; 1 in 100 dilution, cytokeratin AE1/AE3, mouse monoclonal; Dako), α–smooth muscle actin (α-SMA; 1 in 200 dilution, 1A4, mouse monoclonal; Dako), desmin (1 in 50 dilution, D33, mouse monoclonal; Dako), KIT (1 in 500 dilution, polyclonal rabbit; Dako), glial fibrillary acidic protein (GFAP; 1 in 100 dilution, rabbit polyclonal; Chemicon), S100 protein (1 in 100 dilution, rabbit polyclonal; Dako), and neuron-specific enolase (NSE; 1 in 200 dilution, BBS/NC/VI-H14, mouse monoclonal; Dako). Immune complexes were treated with secondary biotinylated goat anti-mouse or anti-rabbit antibody (1:200 dilution; Vector) and subsequently detected using an avidin–biotin complex (ABC) method (Vectastain ABC kit; Vector). Peroxidase activity was detected using 0.1% hydrogen peroxide in 3,3’-diaminobenzidine solution (MilliporeSigma). Sections were counterstained with Mayer hematoxylin (Merck).
Histologic examination of the gastric mass and adjacent nodules revealed a predominantly submucosal, moderately to highly cellular neoplastic proliferation composed of wavy ovoid-to-plump fusiform cells arranged in irregular bundles and scattered whorls. Neoplastic cells had moderate-to-large amounts of eosinophilic cytoplasm with indistinct cell borders, and highly pleomorphic, rounded-to-ovoid nuclei with 1 or 2 prominent nucleoli and a high mitotic count (>20 mitotic cells/2.37 mm2) with several atypical features. Transmural invasion, characterized by infiltration of the lamina propria or muscularis, was also observed (Fig. 2). Fusiform cells reacted strongly to antibodies against vimentin (Fig. 3, Suppl. Fig. 1), S100 and NSE, and showed moderate, multifocal positivity for GFAP; the cells did not express CK, KIT, α-SMA, or desmin.
Histologic and IHC results suggested a diagnosis of gastric sarcoma with features referable to a non-GIST (gastrointestinal stromal tumor), non–smooth muscle NIMT (non-angiogenic, non-lymphogenic intestinal mesenchymal tumor), of possible neurogenic origin. 9 In non-GIST, non–smooth muscle NIMTs, neoplastic cells usually lack SMA and KIT expression, thus allowing exclusion of diagnoses of leiomyosarcoma and GIST, respectively. However, these tumor cells may show immunolabeling for S100, GFAP, and vimentin. 9
In addition, the overlying gastric mucosa was thickened by elongated gastric glands, predominantly lined by intensely PAS-stained mucous cells with severe cystic dilation within the base, in association with multifocal, moderate interstitial fibrosis with scattered, mild lymphoplasmacytic inflammation (Fig. 4). This altered mucosal architecture was suggestive of hypertrophic gastropathy with features referable to Ménétrier-like disease.1,10 However, the surrounding, grossly normal gastric mucosa did not show significant histologic alterations. Thus, mucosal changes consistent with Ménétrier-like disease were not observed in areas not associated with the gastric mass.
Hypertrophic gastropathy, previously called “giant hypertrophic gastritis” or “hyperplastic gastropathy,” describes a diffuse or localized thickening of the gastric wall because of epithelial hyperplasia, conferring a cerebriform appearance to the mucosal surface.8,10 Hypertrophic gastropathy can be also subdivided according to the hyperplastic cell type. 10 In this respect, Ménétrier disease is a rare idiopathic condition of the stomach in humans as a result of hyperplasia of the mucous cells, which is thought to carry an increased risk of gastric cancer, although the magnitude of this risk is uncertain.3,10
Ménétrier-like disease (Ménétrier disease-type hypertrophic gastropathy) has been reported in dogs of various breeds,7,8,11,13,14 also in association with gastric carcinoma,7,8 suggesting that, as in humans, this condition predisposes to neoplasia in dogs. 10 A case of hyperplastic and fibrosing gastropathy resembling Ménétrier disease has also been described in a cat. 1 Based on the information in scientific literature databases (i.e., Medline, Scopus, Web of Science), an association of Ménétrier-like disease with gastric sarcoma has not been reported in the veterinary literature.
Ménétrier disease is a hyperproliferative disorder of the stomach caused by a dysregulated receptor tyrosine kinase, which is attributed to overexpression of transforming growth factor–alpha (TGF-α) with excessive stimulation of gastric epidermal growth factor receptors in humans. 3 Although most cases are considered to be idiopathic, rare inherited or familial forms of this disease have been reported in people2,6 and have been suspected in 3 Cairn Terrier littermates. 8
Mucosal changes characteristic of Ménétrier disease have also been linked to various lesions, both infectious and malignant, although the precise association remains uncertain. Although Ménétrier disease is known to predispose to the development of gastric adenocarcinoma in humans and also probably in dogs,8,10 Ménétrier mucosa has also been found to develop overlying Kaposi sarcoma in human patients. Because Ménétrier mucosa was limited to areas where there was underlying Kaposi sarcoma and was not present in areas characterized by an underlying normal stroma, an association between Ménétrier mucosal changes and gastric Kaposi sarcoma was suggested. 4 Because in our canine case the gastric mucosa adjacent to the mass was grossly and histologically normal, we suspected a similar association between Ménétrier-like disease and gastric sarcoma.
Ménétrier disease may cause abdominal pain, vomiting, and weight loss, and clinical signs may be present for years prior to diagnosis,8,12 although it can also be asymptomatic. 10 Hypoproteinemia and anemia because of protein and blood leakage through the altered gastric mucosa have been reported in people with Ménétrier disease2,3,5,12 and dogs with Ménétrier-like disease, alone or in association with gastric carcinoma.7,11,13 Although our case was not hypoproteinemic, the dog had mild anemia associated with iron deficiency. The 3 Cairn Terrier littermates with Ménétrier-like disease and gastric adenocarcinoma were not hypoproteinemic or anemic. 8
Supplemental Material
Supplemental_material – Supplemental material for Hypertrophic gastropathy associated with gastric sarcoma in a dog
Supplemental material, Supplemental_material for Hypertrophic gastropathy associated with gastric sarcoma in a dog by Mariarita Romanucci, Paolo E. Crisi, Maria Veronica Giordano, Morena Di Tommaso, Francesco Simeoni and Leonardo Della Salda in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Dr. Marina Baffoni for her technical assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
