Abstract
A case of atypical mucosal xanthomas in a 14-year-old Shi-Tzu dog is reported. Grossly, 1–6-mm granular plaques or slightly elevated papillary nodules were found in the oral cavity (dorsal and ventral sides of the tongue, inner upper lip) and in the upper digestive tract (esophagus, stomach). Microscopically, subepithelial aggregates of large foamy cells were found in strong association with papillary epithelial hyperplasia. By immunohistochemistry, the majority of these cells tested positive for lysozyme and ACM1. The cells had cytoplasmic lipid content that stained positively with oil red O. These findings confirmed a monocyte/macrophage lineage. On the basis of macroscopic observations, microscopic changes, and the absence of a clinical metabolic defect, the condition in this dog appears similar to that of humans with oral verruciform xanthoma. The pathogenesis of these xanthomas in the dog remains obscure, although this condition appears to affect people and dogs with an Asian origin.
Xanthomas are nodular granulomatous inflammatory lesions characterized by accumulation of lipoprotein-laden macrophages. 7,9 These rare lesions have been reported in humans, 10,17,19,20 birds, 1 reptiles, 6 cats, 2,21 dogs, 4,21 and horses. 9 Most canine xanthomas are associated with defects in lipid metabolism or metabolic disorders, such as hyperlipidemia, diabetes mellitus, hypothyroidism, or hyperadrenocorticism. 3,8,9 They are mostly observed in the subcutaneous tissue of the face, ears, and ventrum 3,21 and in visceral organs such as the spleen, liver, adrenal glands, stomach, duodenum, and pancreas. 8 Xanthomas also can be found in other tissues, including skin, tendons, and knee and elbow joints. 5 In humans, xanthomas are usually found in association with familial and secondary hyperlipidemia due to diabetes mellitus, hypothyroidism, multiple myeloma, or cholestatic liver disease.
In humans, verruciform xanthoma is a distinct, harmless, and rare clinicopathological entity that occurs in the absence of metabolic lipid disorders. 19,20 Single or multiple nodules occur primarily in the oral cavity and rarely in the esophagus, genital mucosa, and skin. 10 They usually have a sessile or pedunculated base with a papillary or granular surface. The color of the xanthomas may vary from pink to pale to brown. Microscopically, these lesions can be papillary, verrucous, or flat. The histological hallmark of verruciform xanthoma is the presence of aggregates of large foamy vacuolated cells, or xanthoma cells, which ultimately fill the connective tissue papilla between hyperplasic, elongated epithelial rete pegs of relatively uniform depth. The xanthoma cells stain positively for cluster of differentiation 68 (CD68) antigen, indicating a monocyte/macrophage origin. 20 CD68 is a cytoplasmic antigen that is associated with lysosomes. It is expressed with a more intense staining in macrophages than in monocytes but also stains granulocytes and mast cells. 22 The purpose of the current study was to characterize the histological features of the multiple atypical mucosal xanthomas observed in the oral cavity and upper gastrointestinal tract of a dog and to compare these features with those of human verruciform xanthomas.
A 14-year-old, neutered, male Shi-Tzu dog was presented to the Department of Clinical Sciences of the Alfort National Veterinary School (Maisons-Alfort, France) with a cough, decreased appetite, and multiple papillary proliferations on the tongue. Clinical examination revealed a marked heart murmur with cardiac arrhythmia. Cardiac ultrasonography led to a diagnosis of grade 3 mitral endocardiosis, which was managed by enalapril (0.425 mg/kg/day) and spironolactone (2 mg/kg/day). Multiple, yellow, 1–8-mm-diameter papillary or slightly raised nodules were observed on the dorsal and ventral sides of the tongue, as well as on the inner face of the upper lip. Some of these nodules were biopsied and histologically examined.
Two clinical examinations of the animal were carried out at 2-month intervals. They revealed moderate weight loss associated with a moderate increase in liver enzyme activity (alkaline phosphatase activity: 988 UI/l, reference interval: 0–50 UI/l; alanine transaminase activity: 162 UI/l, reference interval: 0–30 UI/l). Urinalysis failed to detect glucosuria or bilirubinuria. One week after the last examination, the dog was presented with prostration, anorexia, and acute abdominal pain. A laparotomy was conducted and revealed a nodular hemorrhagic, 4-cm-diameter tumor of the left adrenal gland in association with hemoperitoneum. The left adrenal gland and neoplasm were totally removed. The tumor subsequently was submitted for histological examination. The dog died a few hours after surgery.
A complete necropsy revealed generalized subcutaneous edema with abdominal (400 ml) and thoracic (20 ml) effusion. The effusion fluid had a serohemorrhagic appearance and was associated with cardiac and hepatic lesions. In the heart, moderate to slight nodular endocardiosis was noted on the mitral and tricuspid valves, respectively. The left atrium was moderately dilated and had a focus of subendocardial fibrosis (so-called “jet lesion”). The liver was moderately enlarged, friable, and had an enhanced lobular pattern with a few hyperplasic hepatocellular nodules. The oral cavity contained multifocal, yellow to brown mucosal nodules, and similar nodules were also found in the esophagus and the stomach.
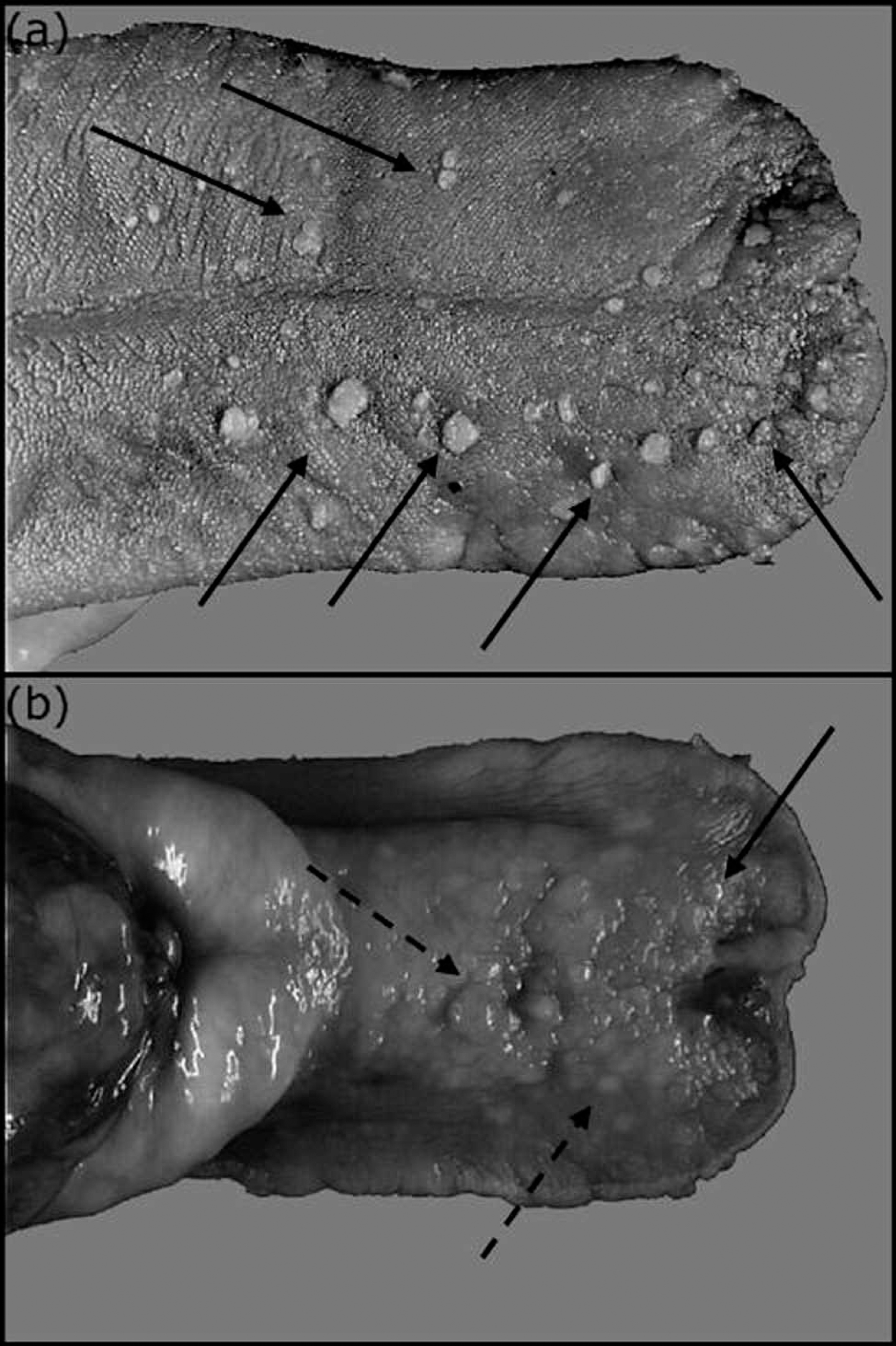
Macroscopic features of the mucosal nodules in a Shih-Tzu with multiple xanthomas.
On the dorsal side of the tongue (Fig. 1a) and on the inner upper lip, yellow, papillary, cauliflower-like nodules were present. These nodules were numerous (about 30 on the dorsal side of the tongue), well-delineated, and measured 4–8 mm in diameter. On the ventral side of the tongue (Fig. 1b), the lesions were of 2 types. A few white, smooth, papillary nodules were sharply delineated, firm, and measured up to 8 mm in diameter. Also present were numerous multifocal to coalescing, slightly granular, raised, white to yellow plaques. These latter lesions were soft and measured up to 6 mm in length and 4 mm in width. Esophageal and gastric lesions consisted of numerous, slightly granular, raised plaques with a few smooth papillary nodules (data not shown). They were soft on palpation and ranged from a few millimeters to 1 cm in diameter.
Biopsies of the tongue and inner upper lip and necropsy samples of the esophagus, gastric nodules, and left adrenal gland were examined histologically. Tissues were fixed in 10% neutral buffered formalin for 48 hr, processed routinely, and embedded in paraffin. Sections (3 μm thick) were cut and stained with hematoxylin–eosin–saffron. Immunohistochemical reactions were performed on unstained tissue sections. These slides were placed in citrate buffer (pH 6) and heated in a water bath for 40 min at 90°C to promote antigen retrieval. After blocking with 2.5% horse serum, a the tissue sections were incubated with primary antibody (lysozyme b 1:300 dilution, MAC387 b 1:300 dilution, ACM1 c ready to use, and CD68 d 1:300 dilution) for 1 hr at room temperature. The slides were then incubated with a peroxidase reagent a (anti-mouse/rabbit Ig) for 30 min and then with NovaRED chromogen. e A negative control was included for each section. The negative control was treated following the same immunohistochemical protocols, except for addition of the primary antibody. Known positive control sections were derived from canine specimens with granulomatous lymphadenitis and splenic histiocytic sarcoma. Frozen sections of OCT (optimum cutting temperature)-embedded specimens of the oral nodules and upper gastrointestinal tract plaques were prepared and stained with oil red O.
Histologic examination revealed a malignant pheochromocytoma of the left adrenal gland and characterized the nature of the mucosal nodules and plaques. The oral lesions included papillary nodules or raised plaques. The papillary nodules (Fig. 2a) were characterized by large exophytic finger-like projections composed of hyperplasic, parakeratotic, squamous epithelium with elongated rete ridges that covered the lamina propria. The lamina propria contained closely packed, foamy cells. The flat plaques (Fig. 2b) consisted of hyperplasic epithelium with deep rete ridges that enclosed foamy cells in the lamina propria. Multifocal, poorly delineated accumulations of foamy cells were observed in the lamina propria, muscularis mucosa, submucosa, and tunica muscularis of the esophagus and stomach (data not shown). In all mucosal nodules, foamy cells filled the lamina propria between elongated epithelial rete pegs of relatively uniform depth (Fig. 2c). These foamy cells were large (25–50 μm in diameter) and round with distinct cell borders, finely vacuolated cytoplasm, and an eccentrically placed nucleus. Their overall morphology was similar to that of macrophages. Extracellular acicular cholesterol clefts and pigmentary incontinence were sometimes observed in the connective tissue around these lesions.
The nature of the foamy cells filling the superficial lamina propria was examined with the use of special staining. These cells had red cytoplasmic globules after staining by oil red O, confirming the presence of lipid (data not shown).
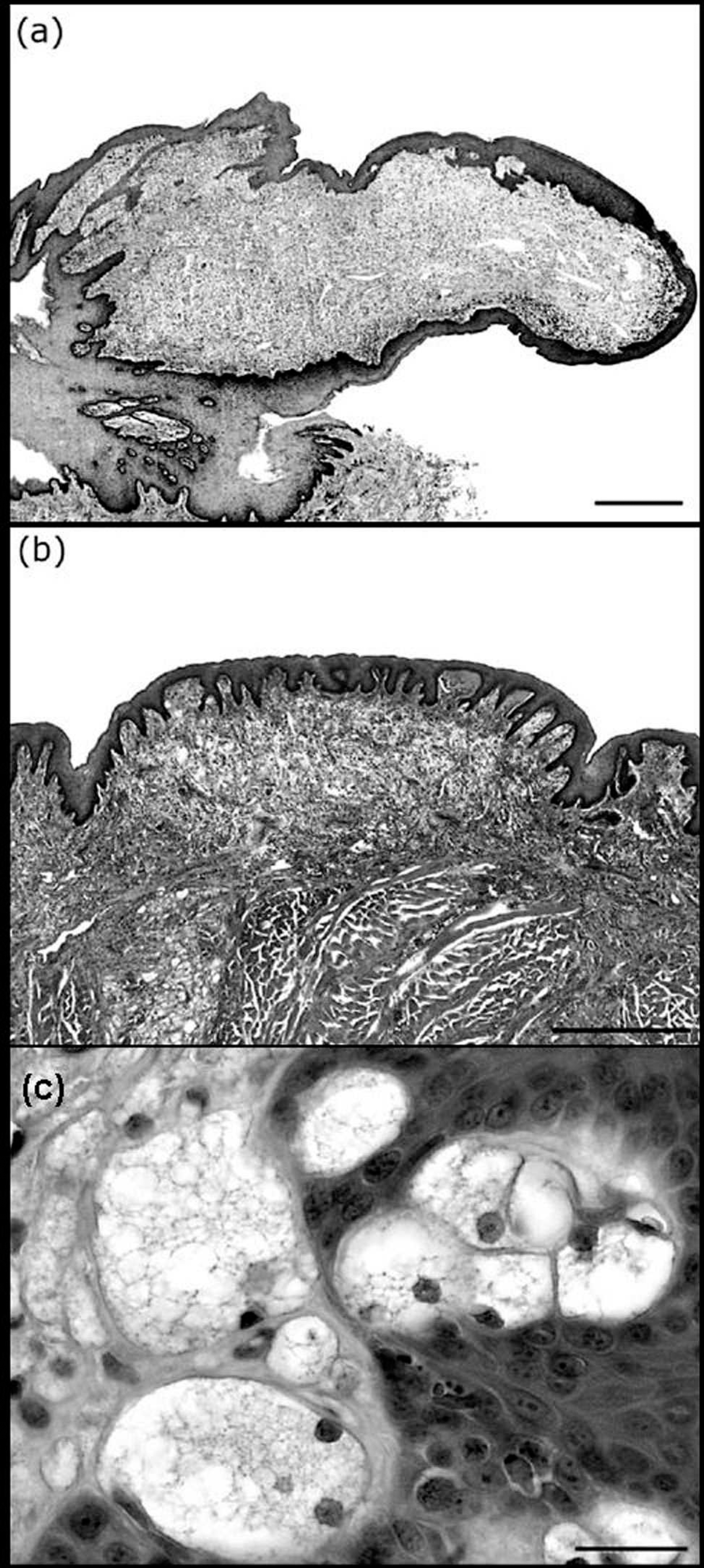
Microscopic features of the mucosal xanthomas.
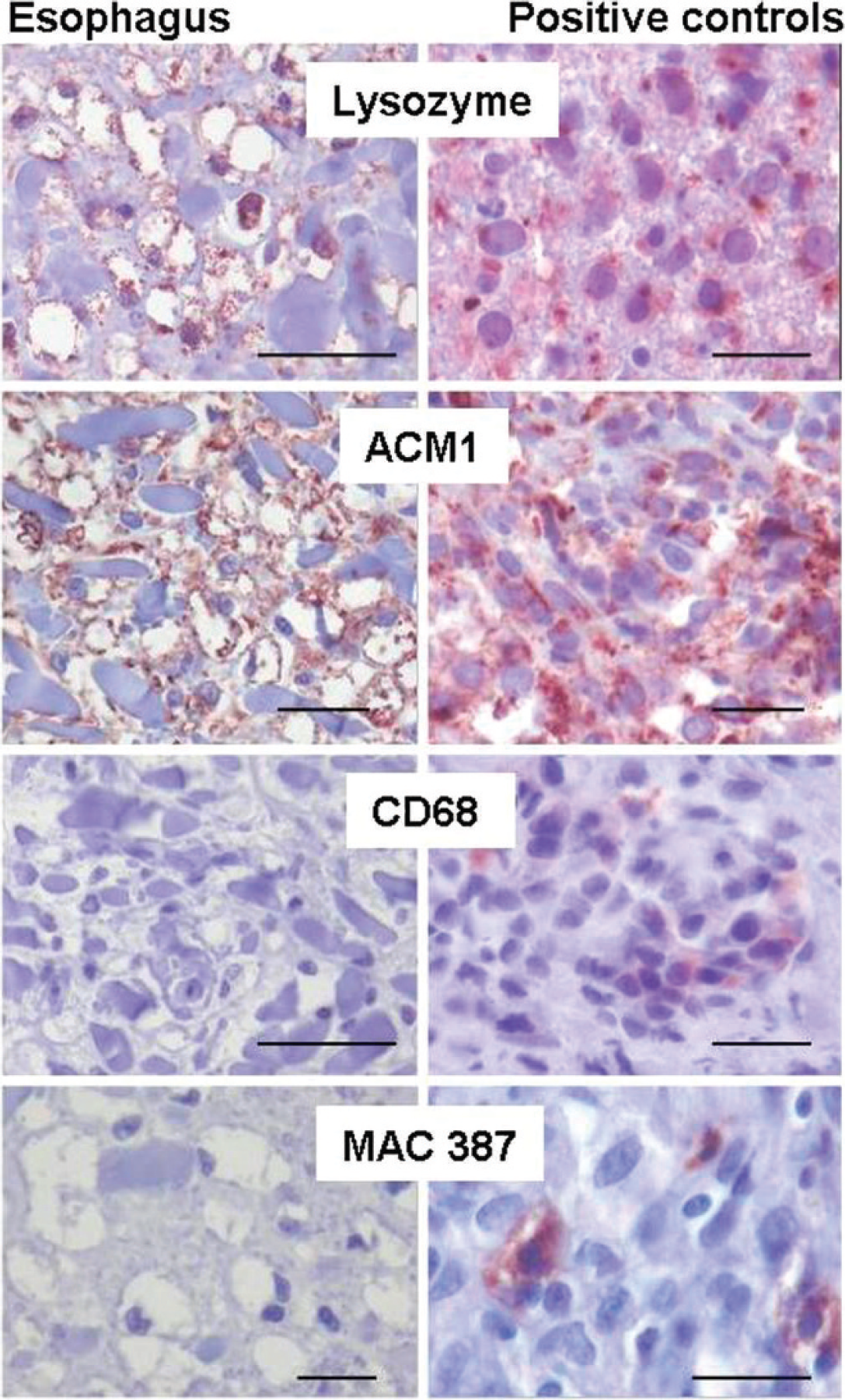
Immunohistochemical characterization of the foamy cells in the lamina propria of the esophageal mucosa.
Immunohistochemically, the foamy cells were negative for CD68 and MAC387 antigens but were positive for lysozyme and ACM1 reactivity (Fig. 3). The myeloid/histiocyte antigen (MAC387) is a calcium-binding molecule expressed by circulating neutrophils, monocytes, and some tissue macrophages. CD68 is an intracellular glycoprotein associated with cytoplasmic granules and, to a lesser extent, the cell membranes of macrophages, monocytes, neutrophils, and large lymphocytes. ACM1 is an antigen that has not been characterized biochemically. It is similar to CD68 reactivity and identifies cells of histiocytic lineage. 13,14 Lysozyme is associated with lysosomes of granulocytes and monocytes/macrophages. 16 The reactivity appears as small brown precipitates in the cytoplasm at the sites of antibody binding. 13 All of these markers can be used to confirm the monocyte/macrophage origin of cells, but none of these markers are very specific and sensitive in canine tissues. For example, absence of lysozyme antigen reactivity in suspected histiocytic proliferations do not exclude the possibility of histiocytic origin. 15 Conclusions after immunohistochemical staining have to be correlated with cellular morphology and special staining. In this case, the foamy cells had the morphology of macrophages, contained oil red O–stained cytoplasmic globules, and stained positively for lysozyme and ACM1. On the basis of these findings, the foamy cells could be characterized as macrophages (Fig. 3). Gross and histologic examinations suggested that these multifocal mucosal xanthomas, arising in the lamina propria of the oral cavity and upper gastrointestinal tract, closely resembled human verruciform xanthoma.
The distribution of xanthomas in the mucous membranes of the dog in the current study mimics typical verruciform xanthoma in humans. Indeed, human verruciform xan-thoma is observed in many tissues and organs that are lined by stratified squamous epithelium, including the oral cavity (gingiva, soft and hard palate, tongue, lips, buccal mucosa), skin, and upper digestive tract. 10,20 The most common site for xanthoma formation is in the oral mucosa.
The present case of multiple, atypical, mucosa-associated xanthomas in a dog has many particularities compared with previously described xanthomatous lesions. In dogs, xanthomas can be associated with hyperlipidemia, diabetes mellitus, hypothyroidism, and hyperadrenocorticism. Despite the absence of a complete laboratory testing in this study, the dog did not show any indications of hyperlipidemia (lacked plasma turbidity), glycosuria, or bilirubinuria (excluded by urinalysis). Hypothyroidism and hyperadrenocorticism were excluded because of an absence of cutaneous lesions, polyuria, polydipsia, glucosuria, and muscular wasting. Xanthomatous lesions in dogs with metabolic disorders also might involve visceral organs such as liver, adrenal gland, eye, and skin. Thus, the resemblance of these multiple, atypical, mucosa-associated lesions in the dog of this report are more similar to human verruciform xanthoma.
Gross and histological aspects of the canine mucosal xanthomas in the present case are similar to those reported in humans. These lesions can be solitary to multiple and appear as papillary nodules or raised plaques. The papillary nodules are usually less than 2 cm in diameter and are characterized microscopically by the presence of clusters of large, vacuolated, lipid-laden macrophages covered by a hyperplasic, hyperkeratotic, squamous epithelium. 17,19,20 As in the human cases, no other types of inflammatory cells are present.
In human verruciform xanthoma, the monocyte/macrophage origin of the foamy cells has been demonstrated by positive CD68 immunohistochemical staining. 20 In the present canine case, the xanthoma cells stained positively for lysozyme and ACM1, confirming their macrophage origin. 13,14 The lipid overload in the cytoplasm of the foamy cells might explain the lack of staining for MAC387, which is considered a marker of activated macrophages. 18
Despite numerous features common to human verruciform xanthoma, the present canine case showed distinct differences. First, the number of xanthomatous lesions was quite high, whereas multiple lesions are rarely described in humans. Second, the gastric mucosa was also involved, which has not been reported in humans.
The etiology of the human verruciform xanthoma remains uncertain. Although it appears similar to a virus-induced papilloma, these lesions have proved to be nonviral in most cases. 20 Attempts to link verruciform xanthoma to a systemic disease, primarily to a defect in lipid metabolism, have been unsuccessful. Thus, etiology and pathogenesis of oral verruciform xanthoma are incompletely understood. 20 Alternatively, moderate repetitive trauma to the oral mucosa has been suggested as a potential etiology. Lipid accumulation in macrophages could have diverse origins, such as lipid components of damaged connective tissue 11 or from damaged keratinocytes. 10,20
The differential diagnosis for large foamy cells includes balloon cell melanocytic tumors, 15 granulomatous inflammation, and xanthoma secondary to a metabolic defect. 7 Granulomatous inflammation and balloon cell melanocytic tumors could be excluded on the basis of the benign accumulation of macrophages, the absence of any other inflammatory cell types associated with the lesions, and the occurrence of lesions in multiple sites. Standard biochemical analyses failed to demonstrate any metabolic defect; however, hyperlipidemia was not specifically investigated.
The malignant pheochromocytoma of the left adrenal gland is an uncommon tumor of dogs, which can cause edema, tachycardia, paroxysmal hypertension, and cardiac hypertrophy. 12 In this case, the arrhythmia probably was the result of catecholamine production by the neoplastic cells. Hemoperitoneum probably was a sequela to tumor rupture or necrosis in the presence of hypertension. The etiological link, if any, between hemoperitoneum and the development of xanthomatous lesions was not established.
The pathogenesis of this atypical xanthoma in the dog remains obscure. Interestingly, this rare pathological entity is seen in people of Asian heritage 20 and it is interesting that the dog of this report also is of Asian origin.
Acknowledgements. The authors gratefully acknowledge P. Wattier for technical assistance; Dr. N. Towanou for macroscopic illustrations; S. Balleydier (Veterinary School of Lyon, France) for her excellent technical assistance concerning immunohistochemistry staining; Dr. T. Marchal (National Veterinary School of Lyon, France) for the kind gift of ACM1, lysozyme, and MAC387 antibodies; Dr. N. Cordonnier-Lefort for necropsy assistance; and J.-J. Fontaine, Dr. G. Aubin-Houzelstein, and Dr. M. Chodkiewicz for critical review of the manuscript. The authors are indebted to Dr. A. L. Bauchet for very helpful discussions.
Footnotes
a.
ImmPRESS™ Universal Antibody (anti-mouse immunoglobulin [Ig]/anti-rabbit Ig, peroxidase) Kit, Vector Laboratories Inc., Burlingame, CA.
b.
Dako Denmark A/S, Glostrup, Denmark.
c.
Provided by Dr. T. Marchal, Veterinary School of Lyon, France.
d.
Novocastra Laboratories Ltd., Newcastle upon Tyne, UK.
e.
Vector® NovaRED™, Vector Laboratories Inc., Burlingame, CA.
