Abstract
Traumatic injury, including bone fracture, is, to date, one of the leading causes of koala mortality in the South East Queensland region of Australia. Further, the specialist diet of koalas, which is restricted to certain Eucalyptus spp., may impact their normal bone physiology. Considering the dramatic koala population decline and high incidence of trauma, a greater understanding of koala bone physiology may support conservation. We retrieved from GenBank the protein sequences of parathyroid hormone (PTH), osteocalcin (OCN), and tissue-nonspecific alkaline phosphatase (TNALP) in human, dog, cattle, horse, koala, and gray short-tailed opossum. After homology was determined, plasma samples from 13 koalas were analyzed with human PTH, OCN, and bone-specific ALP (BALP) assay kits. Although koala PTH exhibited relatively low sequence homology with placental mammals, high sequence homology between humans and koalas was observed for both OCN and TNALP, and successful cross-reactivity was achieved using human enzyme immunoassay kits for detection of OCN and BALP biomarkers in koala plasma. However, we identified no correlation between OCN and BALP concentrations of healthy and trauma-affected koalas (p = 0.66 and p = 0.79, respectively). Further analysis of OCN and BALP in healthy and diseased koalas will allow a better understanding of bone physiology in this unique marsupial.
Introduction
Koalas in South East Queensland (SEQ) are facing a devastating population decrease and are an active conservation focus. 38 Previous research has identified trauma with bone fractures as a common cause of death in SEQ koalas. 13 Although metabolic bone disease, as well as serum vitamin D deficiencies, have been reported both in wild and captive koalas, marsupial bone research is difficult to conduct without assays that quantify specific bone biomarkers.34,35 Metabolic bone disease in koalas is characterized radiographically, but unlike other mammals, blood calcium and phosphorus concentrations are reportedly unreliable indicators in the koala. 35 A study concluded that chimpanzees with rickets were hypocalcemic and hypophosphatemic; whereas koalas with metabolic bone diseases were only hyperphosphatemic, which overlapped with koalas that were identified as normal.18,35 To date, no commercial bone biomarker assays target marsupial species.
Amino acid sequence alignments of a protein biomarker across a variety of species are commonly used to identify active and non-active sites in the protein that interact with biomarker-detection kits. 1 The antibodies of assay kits recognize specific epitopes, thus aligning protein sequences can identify regions of homology, and indicate the likelihood of a biomarker kit reacting with samples from a non-target species. 7 Generally, commercial third-generation enzyme immunoassay (EIA) kits utilize 2 antibodies binding to different epitopes of the targeted protein. For example, the parathyroid hormone (PTH) sandwich assay uses a signal antibody that only reacts to the amino acids of the N-terminal region; the capture antibody binds to the C-terminal fragments. 17 Given that the antibodies vary for each biomarker assay kit, the homology between 2 protein sequences is considered likely to be cross-reactive at ≥ 80%. 31
We selected bone biomarkers, including PTH, osteocalcin (OCN), and tissue-nonspecific alkaline phosphatase (TNALP), for analysis based on their importance in bone physiology, as well as the availability of commercial detection kits. PTH is an 84 amino acid protein that acts to increase ionized calcium levels in the blood. 41 During periods of low ionized serum calcium levels, PTH secreted by the parathyroid glands reduces renal phosphate reabsorption, which increases urine phosphate excretion and enhances vitamin D conversion to its most active form, 1,25-dihydroxycholecalciferol (vitamin D3), which increases urine and intestinal calcium resorption, intestinal resorption of phosphorus, and mobilization of calcium from bones, thus increasing blood calcium levels.5,41 PTH increases bone formation by activating osteoblastic activities, whereas bone resorption is stimulated by increasing osteoclast maturation.4,20 In healthy humans, high PTH levels promote bone turnover; however, given that many factors affect bone turnover, multiple bone biomarkers should be considered simultaneously for an accurate representation of bone physiology.4,8 Nonetheless, because PTH is an indicator of bone turnover rate, it can provide information in cases of metabolic bone disease. 21
OCN is 49 amino acid protein produced exclusively by osteoblasts and megakaryocytes, making it one of the most predominant non-collagenous proteins in bone.1,25 A rise in serum OCN suggests accelerated osteoblastic activity, thus increased bone formation rates.25,37 A rise in uncarboxylated OCN concentration has been associated with increased hip fracture risk and low bone mineral density in humans. 40 However, the relevance of this finding to koalas is unknown given that studies have outlined that most animal species have fully carboxylated OCN, whereas uncarboxylated OCN in humans is observed predominantly from variations in vitamin K consumption.30,33 Although a variety of commercial radioimmunoassays and ELISAs are available, they are manufactured specifically for humans, dogs, rats, and mice because only a few species demonstrate cross-reactivity. 1
Alkaline phosphatase (ALP) is a glycoprotein enzyme that is secreted by cells in various tissues; the number of ALP variants differs among species. 32 For example, there are 4 isoenzymes of ALP in humans, whereas sheep have only 2. 12 Total ALP is widely used as a bone formation and turnover marker, and elevation of total ALP can indicate osteoporosis or other bone diseases.4,5,10,24,39 But given that ALP is nonspecific for bone metabolism, bone-specific ALP (BALP) is considered the gold standard marker when available. 10
We aimed to identify the homology between protein alignments of placental mammals (human, dog, cattle, horse) and marsupials (koala, gray short-tailed opossum) for core bone biomarkers PTH, OCN, and TNALP. Thereafter, we tested human commercial detection kits for PTH, OCN, and BALP in koala blood samples to determine assay validity. Preliminary analysis was conducted on a small number of healthy and abnormal koalas. We aimed to identify and validate biomarker assays for future research into physiologic and pathologic bone metabolism in the koala, a unique and threatened species.
Materials and methods
Protein alignments using GenBank, MEGA7, and BioEdit
We retrieved from GenBank the protein sequences of PTH, OCN, and TNALP in human (Homo sapiens), dog (Canis lupus familiaris), cattle (Bos taurus), horse (Equus caballus), koala (Phascolarctos cinereus), and gray short-tailed opossum (Monodelphis domestica). 6 The gray short-tailed opossum, a Brazilian marsupial, was chosen because it is one of the few marsupials with genome sequencing available, thus was theorized to have higher homology with koalas than placental mammals. 11 TNALP is one of the isoenzymes of ALP and is secreted by liver, bone, and kidney. Sequences for BALP were not available in GenBank and, therefore, TNALP was compared as the closest available protein to BALP. The retrieved sequences were aligned by ClustalW, using default parameters in the MEGA7 software. 23 Upon completion of the protein alignments, the file was saved as a FASTA file so that further edits could be done using the BioEdit software. 14
Source of koala blood samples
We obtained 13 koala blood samples for the study, consisting of 7 adult females and 6 adult males. Of the 13 koala samples, 6 were from healthy non-pregnant and non-lactating female captive koalas obtained between September 2009 and November 2010. The remaining 7 koala samples were from injured or terminally ill animals collected immediately after euthanasia from the Moggill Koala Rehabilitation Centre in Queensland from March 2018 to November 2018. Five of the 7 samples of diseased animals came from koalas with acute or healed fractures (Suppl. Table 1).
Blood collection and storage
For each koala, blood was collected into EDTA and lithium heparin pediatric vials, under ethical review and approval of the Queensland Government National Parks and Wildlife Service (permits WISP13247813 and WA0010579) and The University of Queensland Research Integrity Unit, Animal Ethics Committee for Wild Animals (permit ANRFA/SVS/070/16). Blood samples were transported to a laboratory in an insulated portable carrier with refrigeration by ice packs. EDTA tubes were centrifuged at 1,780 × g, and lithium heparin tubes were centrifuged at 8,100 × g, both for 10 min. Plasma, and the remaining solids, were transferred into separate cryovials (labeled and color-coded) and stored at −20°C until analyzed.
Commercial biomarker assay kits
PTH was analyzed by an immunoradiometric sandwich assay (Immunotech PTH IRMA; Beckman Coulter) in 3 koala plasma samples, to pilot test the cross-reactivity between the assay and the samples, as per the manufacturer’s procedure. Testing was discontinued because of failure of cross-reactivity.
OCN was analyzed by an EIA (MicroVue osteocalcin EIA; Quidel) in all 13 koala plasma samples. The manufacturer’s procedure was followed, except koala plasma was analyzed as a 1:5 dilution using 10× diluted wash buffer provided in the kit, allowing OCN concentrations to be interpreted on the standard curve. The assay was repeated for one koala (wild adult male with Chlamydia infection and poor body condition) as a result of an undetectable absorbance reading that was below the standard curve for one of the replicates. Duplicates of OCN concentration from the 13 koala plasma samples were tabulated against parameters including wild or captive, sex, and abnormal versus healthy (Suppl. Table 1). The OCN levels of the normal captive female koalas served as the reference interval (RI), and values significantly under or above the RI were considered outliers.
BALP was analyzed by an EIA (MicroVue BALP EIA; Quidel) in duplicate samples according to the manufacturer’s procedure. The duplicates of BALP concentration obtained from the 13 koala plasma samples were tabulated, along with the same parameters in the OCN table (Suppl. Table 1). The RI was established using the BALP levels of the captive normal female koalas in our study.
Statistical analysis
Descriptive statistics (mean with 95% confidence interval [CI], median, standard deviation, and minimum and maximum) and boxplots were produced to depict OCN and BALP concentration for healthy koalas and koalas with trauma and/or bone fractures (2 unhealthy koalas without trauma, with unknown disease and chlamydia infection, respectively, were excluded from this analysis to create a trauma cohort). The nonparametric Mann–Whitney U test was used to compare the distribution of OCN and BALP concentration between healthy and trauma-affected koalas, with a significance level set at p ≤ 0.05.
Results
Sequence homology of PTH, OCN, and TNALP among koalas, placental mammals, and gray short-tailed opossum
Human PTH showed good similarity to other placental mammals, such as the dog, cattle, and horse, but low homology with the gray short-tailed opossum or koala (Fig. 1). In placental mammals, PTH has a length of 84 amino acids whereas marsupial PTH has 94 amino acids, with additional amino acids in the middle of the polypeptide. The lowest homology noted was between human and koala at 31.9%; the highest homology was between human and dog at 88.1% (Table 1).
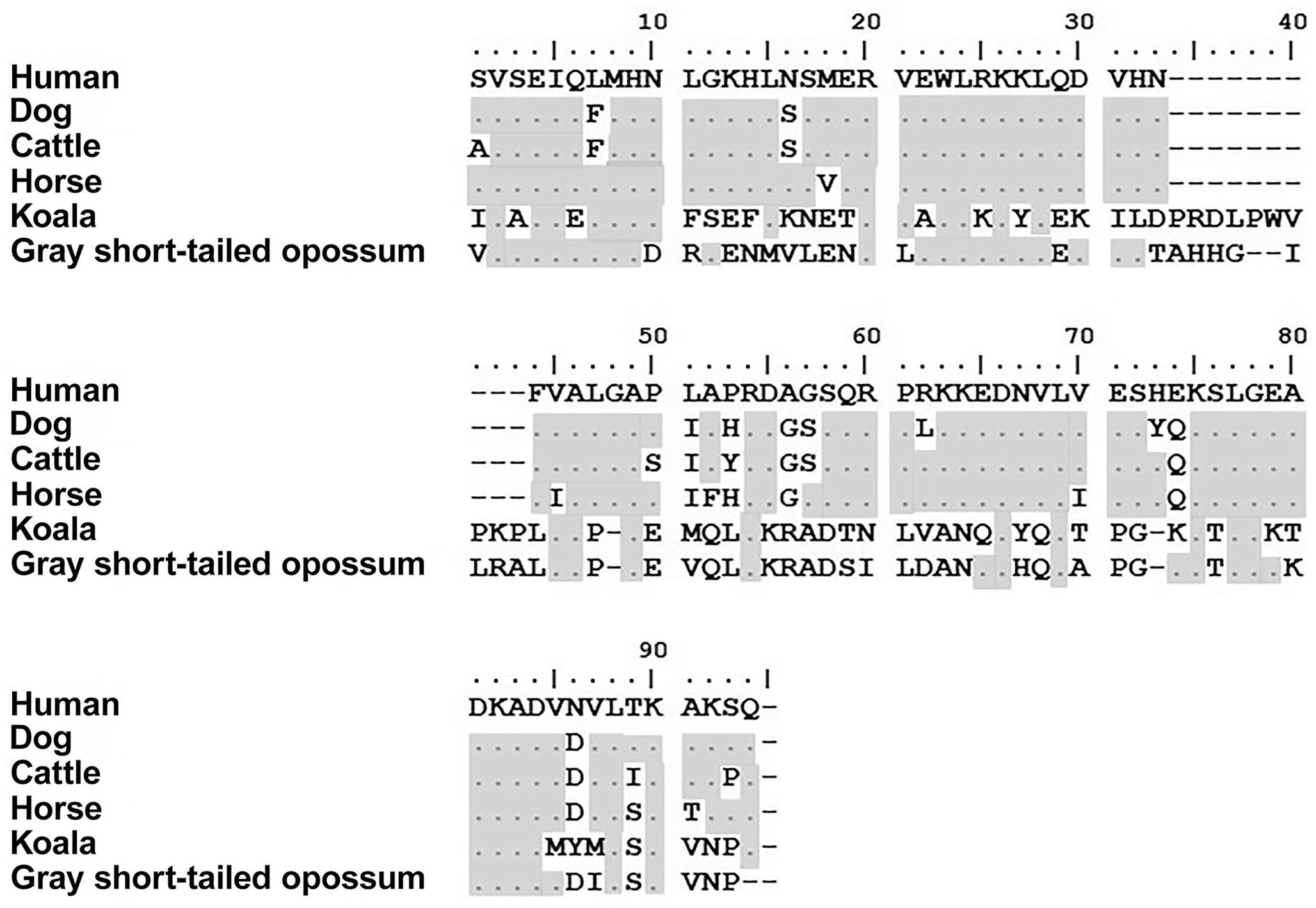
Parathyroid hormone protein alignments of human, dog, cattle, horse, koala, and gray short-tailed opossum. Changes in amino acids are shown relative to the human sequence. The dot (.) and shading indicate that the amino acid is identical to the one in the human sequence. The dash (-) represents a gap in the sequence after alignment by ClustalW on MEGA7.
Similarity of protein sequences of human parathyroid hormone (PTH), osteocalcin (OCN), and tissue-nonspecific alkaline phosphatase (TNALP) compared to dog, cattle, horse, koala, and gray short-tailed opossum (%, amino acids).

Protein sequences were retrieved from GenBank.
OCN is a relatively short polypeptide of 49 amino acids for all species investigated (Fig. 2). Overall, there was high homology between species, with the greatest similarity between human and horse at 93.9%, a difference of only 3 amino acids (Table 1). There was high similarity in OCN between the 2 marsupials that we evaluated (91.8%), and between the 2 marsupials and human (79.6%), with conserved regions in the mid and C-terminal (residues 11–47).
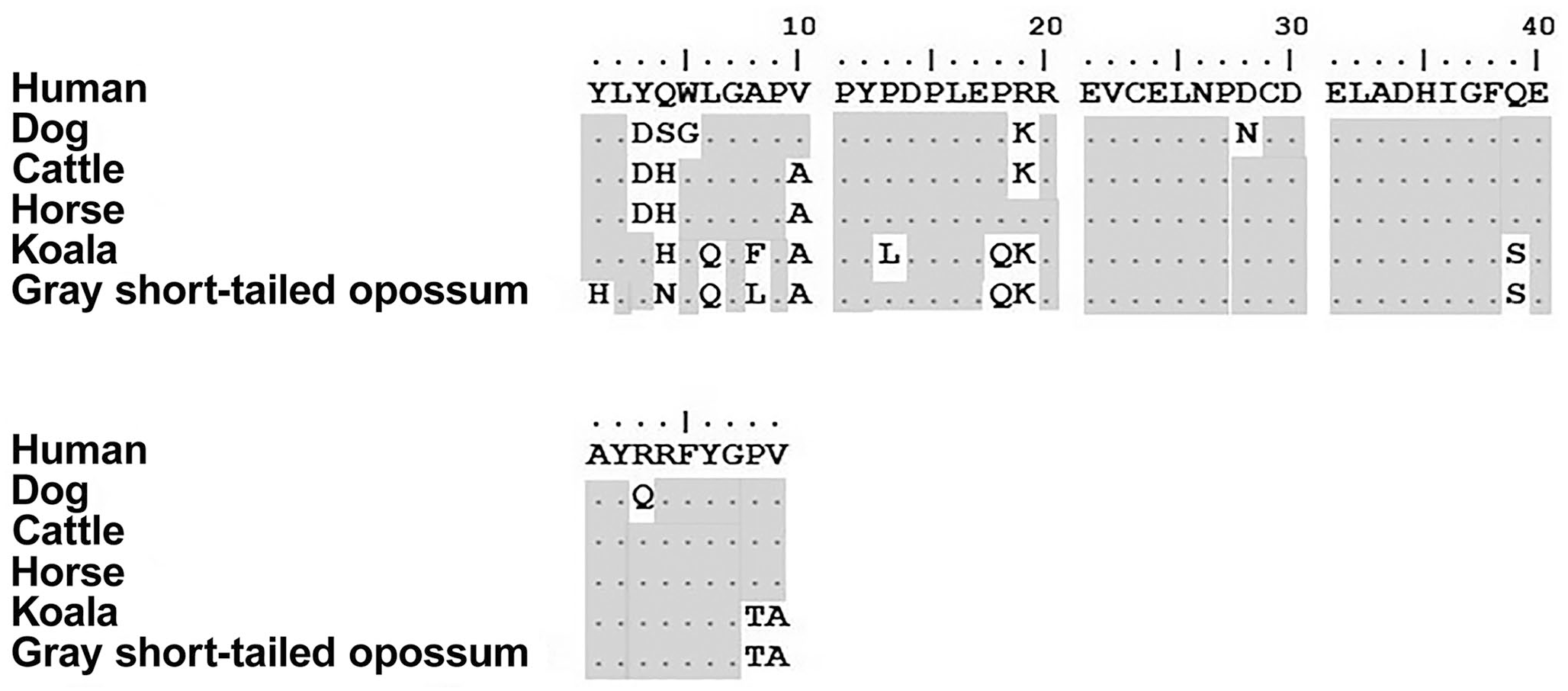
Osteocalcin protein alignments of human, dog, cattle, horse, koala, and gray short-tailed opossum. Changes in amino acids are shown relative to the human sequence. The dot (.) and shading indicate that the amino acid is identical to the one in the human sequence. The sequences were aligned using ClustalW on MEGA7.
TNALP comprised a protein of 526 amino acids in all species (Fig. 3). Marsupials had lower homology to the placental mammals, but there was still significant alignment to humans, with the koala sharing 81.9% and the gray short-tailed opossum at 80.2% alignment (Table 1). Within placental mammals, there was ≥ 90% homology across human, horse, dog, and cattle.
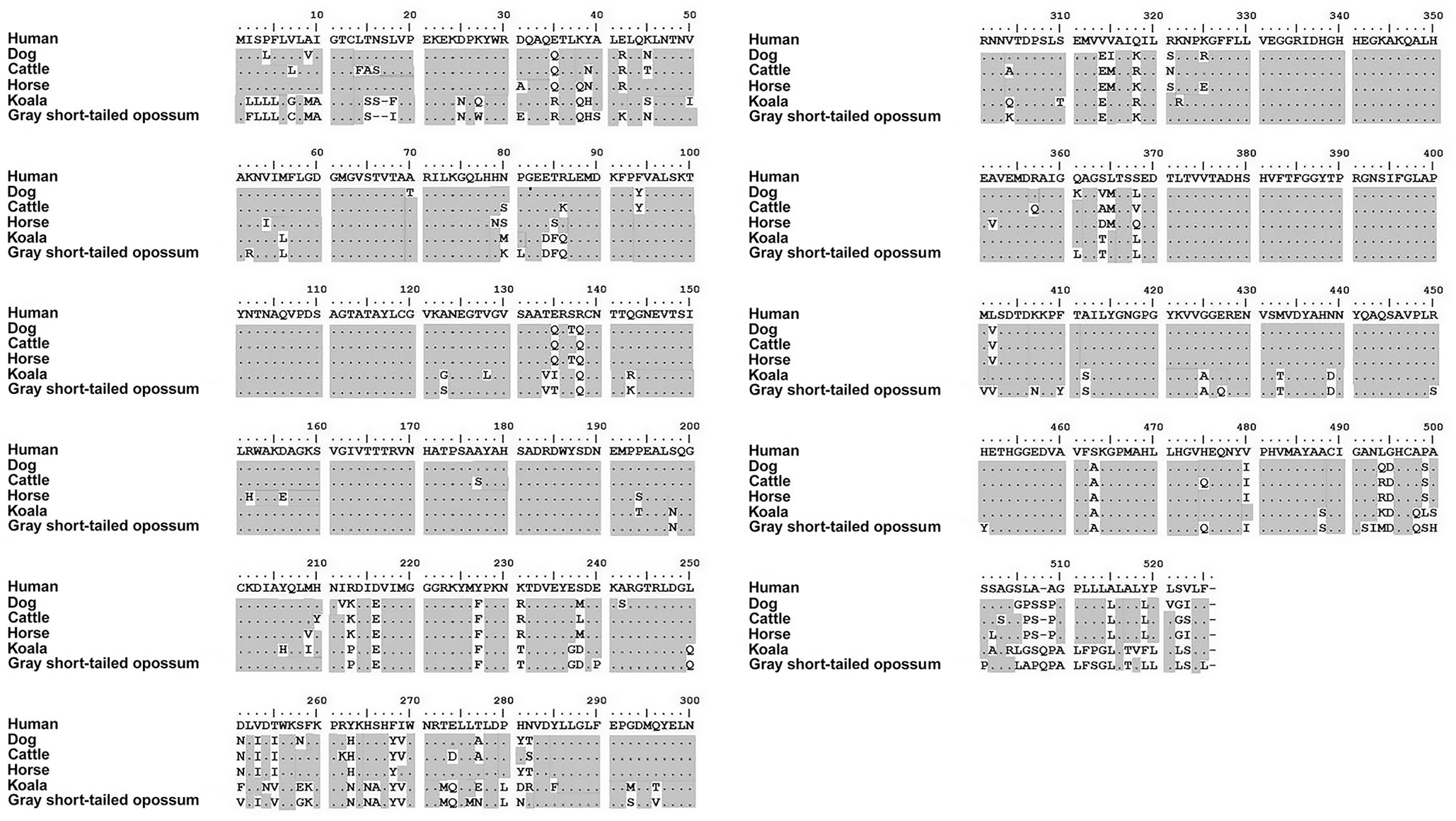
Tissue-nonspecific alkaline phosphatase protein alignments of human, dog, cattle, horse, koala, and gray short-tailed opossum. Changes in amino acids are shown relative to the human sequence. The dot (.) and shading indicate that the amino acid is identical to the one in the human sequence. The sequences were aligned using ClustalW on MEGA7.
Quantification of koala PTH, OCN, and BALP using commercial assay kits
Koala plasma samples from healthy and abnormal koalas were tested in PTH, OCN, and BALP human bone biomarker detection kits. Human OCN and BALP assays successfully cross-reacted with koala plasma, as predicted by the relatively high sequence homology in these markers. However, no cross-reactivity of koala samples was observed with the human PTH IRMA kit after testing 3 pilot samples, so analysis was discontinued.
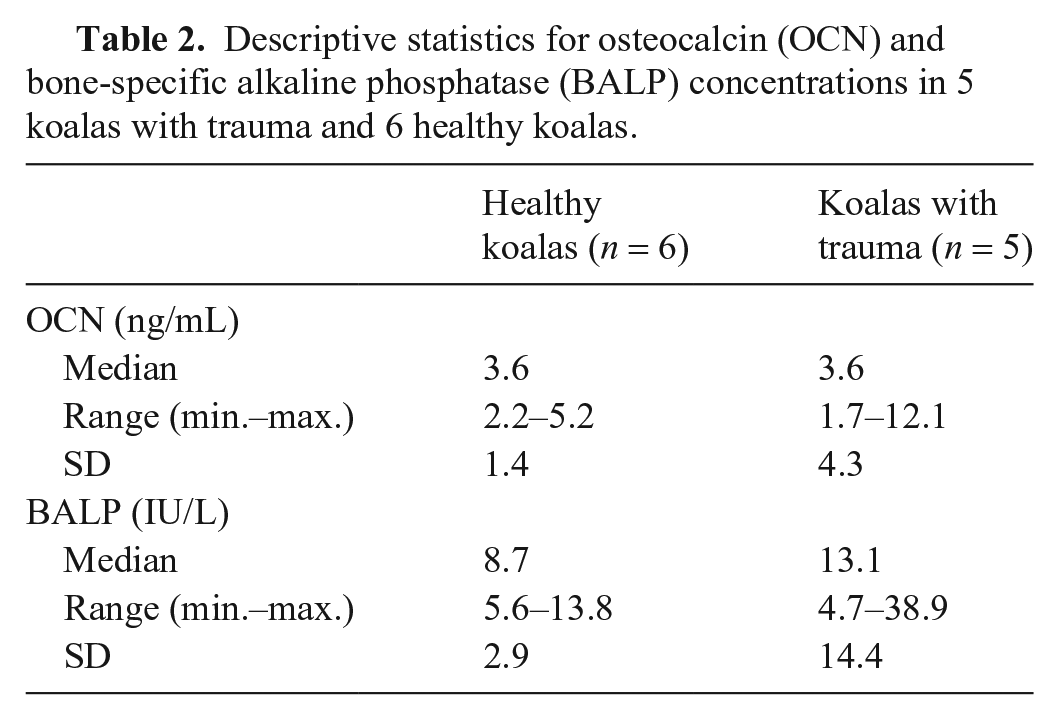
Overall, there were no statistically significant differences in plasma OCN or BALP concentrations between healthy koalas and those with trauma, although there was greater variation for both bone biomarkers in koalas with trauma (Fig. 4). Substantial differences between the descriptive statistics of plasma OCN and BALP concentrations for both groups were observed (Table 2). Mean plasma OCN concentration of healthy koalas was 3.7 ng/mL (95% CI: 2.7–5.1) compared to 4.7 ng/mL (95% CI: 0–9.9) for koalas with trauma (p = 0.66). Notably, a young male koala with trauma from a vehicle collision and good body condition score had a very high plasma OCN concentration of 60.4 ng/mL. Mean plasma BALP concentrations were not significantly different in koalas with trauma (9.4 IU/L; 95% CI: 6.3–12.4) compared to healthy koalas (17.3 IU/L; 95% CI: 0–35.1; p = 0.79). Of koalas with trauma, 1 had acute dog attack trauma, but evidence of old healed tibia and fibula injury, 1 had an old fracture but was debilitated from neoplasia, 3 had acute trauma from tree fall, dog, and car, respectively, but the bone injury was only well defined in the car victim, which had an acute comminuted coxofemoral joint fracture. Given study limitations, further medical imaging and histopathology were not undertaken.
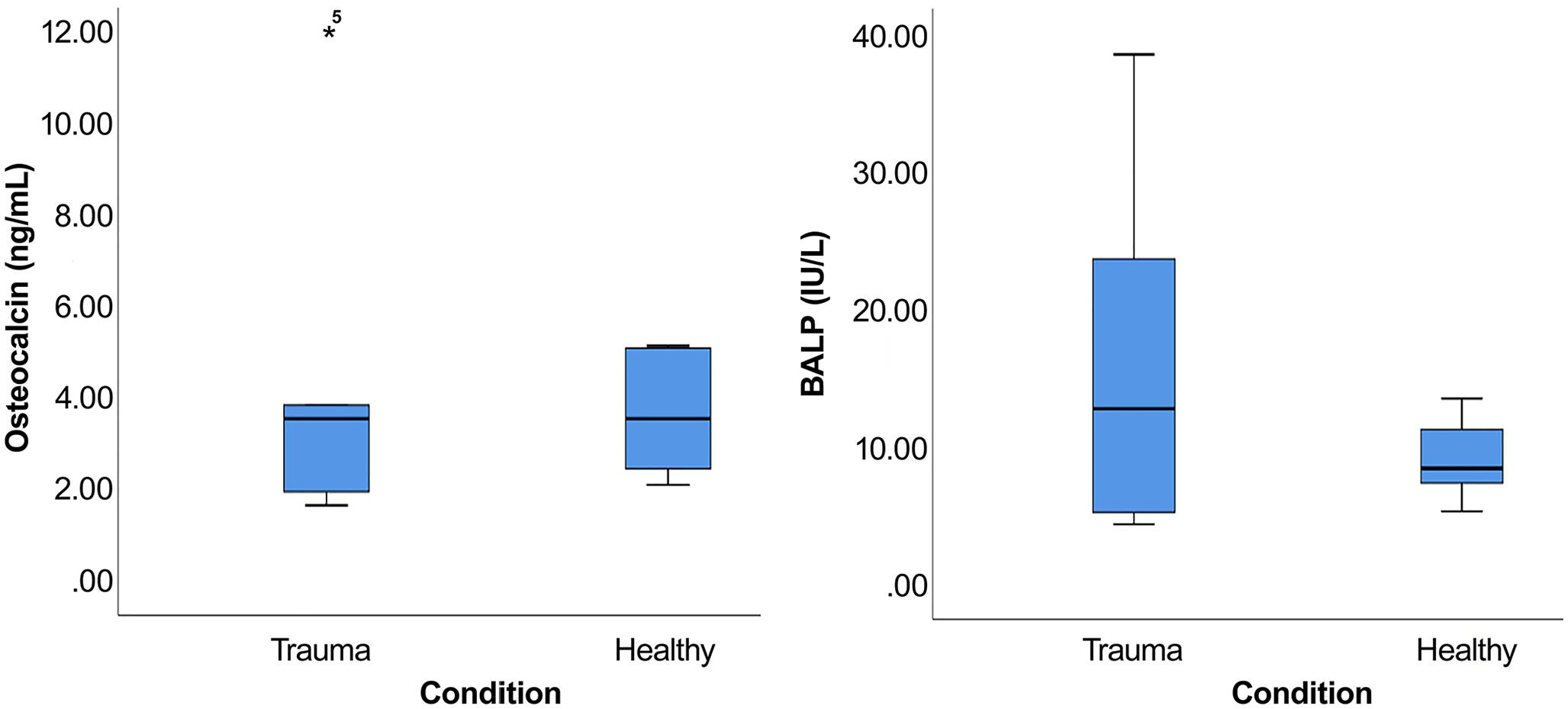
Box plot of the distribution of osteocalcin and bone-specific alkaline phosphatase (BALP) concentration in 5 koalas with trauma and 6 healthy koalas.
Descriptive statistics for osteocalcin (OCN) and bone-specific alkaline phosphatase (BALP) concentrations in 5 koalas with trauma and 6 healthy koalas.
Discussion
Koalas showed variable homology to humans and other placental mammals for the 3 bone biomarkers (PTH, OCN, and TNALP) assessed in our study. Protein alignments using BLAST (http://blast.ncbi.nlm.nih.gov/Blast.cgi) were a good predictor of the commercial bone biomarker assay kit success. Cross-reactivity of koala OCN and TNALP was observed with human EIA kits, providing preliminary data for healthy koalas and those with trauma. Substantial variability was observed with the small cohort of koalas with trauma.
PTH homology between human and koala was poor at 32%. Alignments with < 80% similarity were unlikely to exhibit cross-reactivity in any assay, and koala plasma samples did not cross-react with the human IRMA kit. This assay kit has antibodies targeting both ends of the polypeptide. There was little homology at the N-terminal (1–34) and even less at the C-terminal (35–94). Additionally, the length of PTH in marsupials differs from placental mammals. Thus, the lack of N- and C-terminal homology, along with the different protein length, may explain why the PTH kit demonstrated no signal.
The homology of OCN between placental mammals and within the marsupials was high overall. The human EIA kit detected koala OCN successfully. The high OCN homology between the 2 species of marsupials suggests that this OCN assay may work in other marsupials. The variability in plasma OCN concentrations in the abnormal koalas was high, probably reflecting the different etiologies, stage of healing, and severity of the conditions in this cohort. Further OCN might be inherently variable in koalas as a species, and additional sample testing would be required to define species RIs. Mean plasma OCN was lower in abnormal koalas, although both groups had the same median value of 3.6 ng/mL. The elevated serum OCN level in a young male that suffered from trauma (close to 3-fold increase from the highest normal koala) may reflect a rapid physiologic response to the bone trauma suffered by this animal, although the period between injury and death is unknown. However, the higher plasma concentrations in this animal might be more related to active bone growth; this koala was a 2.5-y-old adolescent. In contrast, a debilitated elderly male animal had the lowest OCN (6.2 ng/mL). Apart from age, OCN also differs between sex, suggesting that female humans with metabolic syndrome have increased uncarboxylated OCN and reduced carboxylated OCN.25,26 Thus, OCN concentration is greatly dependent on age and sex. Recent studies have supported that OCN is a negative regulator of bone formation, with evidence acknowledging OCN as an endocrine hormone, controlling energy metabolism, male fertility, and brain development. 25 Similarly, global alteration of bone metabolism is also reflected in blood concentrations of ALP, as shown in patients with low ALP levels without hypophosphatasia.10,27 Therefore, all possible factors affecting the rise and fall of these biomarkers should be considered. Further analyses of koalas with various disease and injury states, as well as sex and age distribution, would be required to confirm if serum OCN concentration is correlated with growth, bone healing, or general debilitation in koalas.
TNALP had high sequence homology among species, and the human EIA kit cross-reacted successfully with koala plasma. Similar to plasma OCN, high variability in BALP concentrations were noted in unhealthy koalas. No significant difference in the diseased koala distribution was observed (Fig. 4). Interestingly, the highest plasma BALP concentration (38.9 IU/L) belonged to the young male koala with car strike trauma and good body condition with high plasma OCN. Again, further testing of other koalas would be required to determine if the elevated levels of BALP in this animal are physiologic as a result of young age and growth, or in response to early traumatic fracture healing. Apart from age, bone growth, sex, and previous fracture healing, other factors that could affect BALP concentrations include nutrition and calcium supplementation, which are possible causes of the wide variation in BALP levels of healthy koalas.22,28
RIs of both plasma OCN and BALP biomarkers in koalas cannot be determined from our study because of the limited sample size, and lack of variation in the age and sex of koalas analyzed. Nevertheless, results from our koala study are similar to the RIs reported in healthy adult humans: human OCN, males: 9.2–32.5 ng/mL, females: 7.6–31.9 ng/mL; human BALP, males: 7.4–27.0 ng/mL, females: 6.0–22.7 ng/mL.15,29
The limitations of our study include the small number of koala blood samples that were available for the validation and analysis of the bone biomarkers. A particular limitation was that healthy koala samples were restricted to only non-lactating, non-pregnant captive female koalas. Whether captivity and associated environmental factors affect the concentrations of bone biomarkers in koalas requires further analysis of larger cohorts. There was also variability in the storage time between samples given the necessary limitation of having access to the captive population, as well as the opportunistic sampling of abnormal animals that required euthanasia after veterinary assessment. Although the stored samples showed strong and consistent results with limited variability, some degradation of the sample as a result of the prolonged storage time cannot be excluded. Studies have investigated the use of fibroblast growth factor 23 (FGF23) and PTH-related protein (PTHrP) as potential biomarkers for various diseases in humans and animals.2,16 FGF23 is one of the key regulators of PTH and vitamin D synthesis, and of calcium–phosphorus balance; whereas PTHrP stimulates bone resorption and formation similar to PTH.3,4,36 Studies have concluded that abnormal elevations or dips of either FGF23 or PTHrP concentrations could indicate bone, kidney, or cardiovascular diseases in humans and animals.3,9,19 Unfortunately it was out of the scope of our study to consider FGF23 and PTHrP.
Further, it would have been highly beneficial to have diagnostic imaging (radiographic, computer tomography [CT], or microCT) evaluations, coupled with site-targeted histopathology, to establish the chronicity and stage of healing, and quality of healing from both control and diseased populations in our study. Unfortunately, this was not possible for logistical reasons, with blood sampling from the healthy females being retrospective and opportunistic, and lack of funding for the more detailed imaging studies in the deceased diseased and traumatized koalas. The sampling from the diseased and traumatized koalas was also opportunistic, and unfortunately, only limited information was provided in the medical histories. Further, koalas had been presented with a complicated pattern of acute-on-chronic illness and injury when diseased. After the successful validation of BALP and OCN in koalas, a much broader sample of age- and sex-matched healthy and unwell animals with various conditions is needed to get a better picture of the behavior of these biomarkers in this species.
We demonstrated that commercial BALP and OCN EIA kits cross-react with koala plasma and can be used in future studies of koala bone physiology. Given the homology between koalas and gray short-tailed opossum as a comparable marsupial, it is likely that these biomarker kits would be suitable for analysis of other Australian and new world marsupial species.
Supplemental Material
Supplemental_material – Supplemental material for Bone biomarkers in koalas: validation of assays and preliminary analyses
Supplemental material, Supplemental_material for Bone biomarkers in koalas: validation of assays and preliminary analyses by Chien-Jung Chen, Stephen T. Anderson, Natasha Steiger, Allan McKinnon, Joerg Henning and Rachel Allavena in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Kenneth Lopez-Loo and Peter Theilemann for technical assistance, Viviana Gonzalez-Astudillo for assisting in the collection of some samples, and Drs. Lisa Kidd and Karen Jackson for scientific comments and expertise in the planning and analysis stage of the project.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding for this project was provided by The School of Veterinary Science, The University of Queensland, and the Annandale bequest for Wildlife Research.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
