Abstract
An embryonal carcinoma was diagnosed in the abdominal cavity of a 55-day-old male calf. Macroscopically, a large volume of ascitic fluid was observed along with white to yellowish-white masses of various sizes densely located on the abdominal wall and the surface of abdominal organs. There was an absence of testes in the scrotum. Histologically, the tumor cells were polygonal, and the tumor was mostly composed of sheets of densely packed solid patterns with occasional papillary and tubular structures. Cell nuclei were variable in size, and cellular mitotic rate was high. Immunohistochemically, tumor cells were positive for alpha-fetoprotein, placental alkaline phosphatase, cytokeratin, and carcinoembryonic antigen. Ultrastructurally, the tumor cells had large nuclei, extensive rough endoplasmic reticulum, and small numbers of mitochondria. Microvillus-like structures and desmosomes were occasionally observed. From lectin histochemical examination, the tumor cells were positive for concanavalin A, wheat germ agglutinin, peanut agglutinin, Dolichos biflorus agglutinin, soybean agglutinin, Griffonia simplicifolia I, and Bauhinia purpurea, and negative for Ulex europaeus agglutinin I. Results of histopathological, immunohistochemical, and ultrastructural examinations of the tumor were similar to those obtained for human embryonal carcinoma.
Embryonal carcinoma is a rare germ cell tumor in human beings that occurs in the ovaries, 21 testes, 4 and mediastinum. 12 When this tumor is derived in testes, it is dominantly expressed. In animals, embryonal carcinoma is also a rarely occurring tumor with reports limited to the tumor derived in testes. 7,10,20 To date, embryonal carcinoma has not been reported in cattle. The purpose of the current report is to describe the histopathologic, immunohistochemical, ultrastructural, and lectin histochemical characteristics of embryonal carcinoma in the abdominal cavity of a male calf.
A 55-day-old male crossbred (Holstein–Friesian/Japanese Black) calf was clinically diagnosed with emaciation and abdominal distension. Because of severe emaciation, the animal was euthanized, and a necropsy was performed. At postmortem examination, a large volume of ascitic fluid was observed as well as numerous, closely spaced, white to yellowish-white masses of various sizes on the abdominal wall and the surface of abdominal organs. The largest mass, the diameter of which was approximately 20 cm, was present on the abomasal serosa, though macroscopically visible masses were not present at the lumen of the abomasum. Each mass was fragile with a villous appearance on cut surface (Fig. 1). Numerous masses were also present on the surface of the liver and spleen with adherence to the diaphragm. Red fluid had accumulated in the rumen, and masses of various sizes weakly adhered to the luminal surface. There were no testes in the scrotum, or in the pelvic or abdominal cavities. Tissue samples collected from masses on the serosal surface of the abomasum and intestinal tract, hepatic lymph node, liver, and spleen were all fixed in 10% neutral formalin solution.
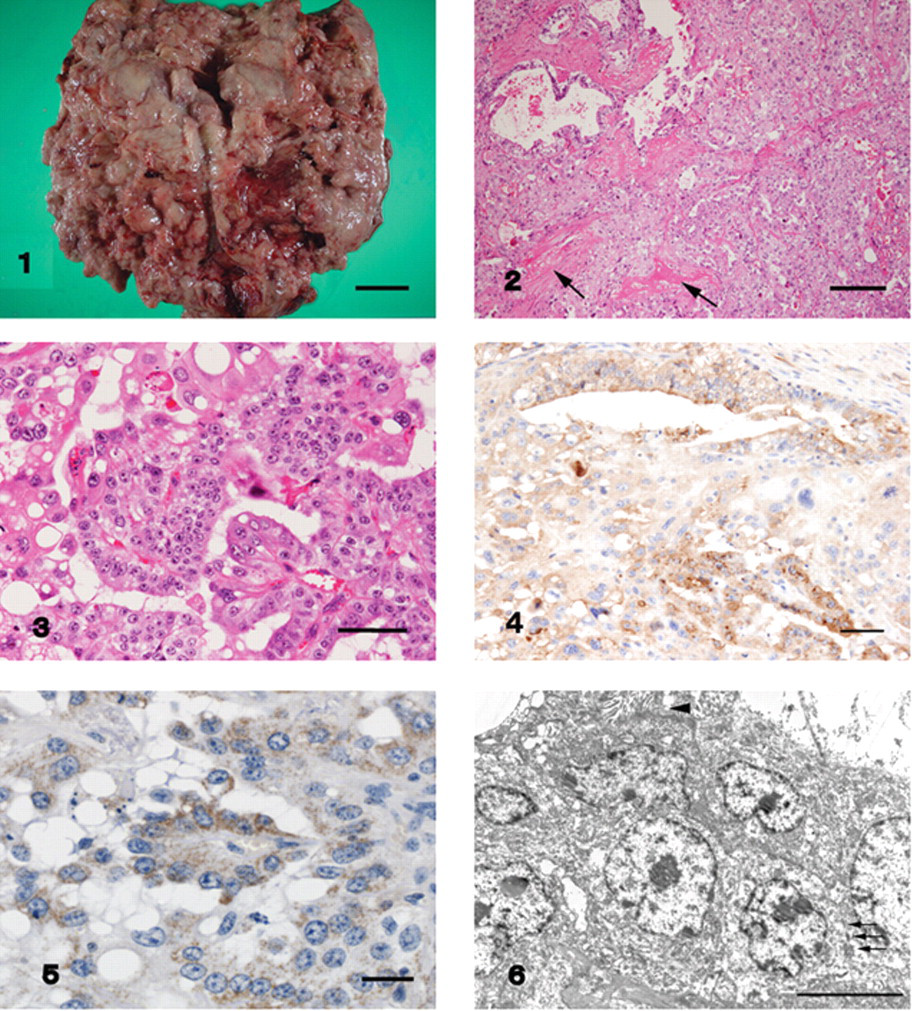
The largest mass was present on the serosal surface of the abomasum. The mass was fragile and villous on cut surface. Bar = 3 cm. Figure 2. The tumor was composed of sheets of cells with acinar and tubular structures. Small foci of woven bone were occasionally present within the stroma (arrows). Hematoxylin and eosin. Bar = 200 µm. Figure 3. The cell cytoplasm was eosinophilic, granular, and sometimes contained large vacuoles. Hematoxylin and eosin. Bar = 50 µm. Figure 4. Positive reactions to alpha-fetoprotein were observed in the cells within papillary and cyst-like structures. Immunohistochemistry, counterstained with hematoxylin. Bar = 50 µm. Figure 5. Positive reactions to placental alkaline phosphatase appeared on the cell surface and the cytoplasm of several small foci of cells. Immunohistochemistry, counterstained with hematoxylin. Bar = 25 µm. Figure 6. Features of the tumor included closely packed discrete cells, extensive rough endoplasmic reticulum, small numbers of mitochondria, and intercellular desmosomes (arrows). Occasional microvillus-like structures were also present (arrowhead). Transmission electron micrograph. Bar = 5 µm.
For microscopic examination, tissue samples were embedded in paraffin, and stained with hematoxylin and eosin, periodic acid–Schiff (PAS), alcian blue, and toluidine blue. All masses were composed of densely packed sheets of cells, as well as occasional papillary, acinar, tubular, and cyst-like structures (Fig. 2). Tumor cell borders were indistinct, especially in solid areas. Tumor cell cytoplasm was eosinophilic and granular, with occasional large vacuoles (Fig. 3). The tumor cells also had eosinophilic granules that were PAS positive, but negative for alcian blue and toluidine blue. Nuclei were anisokaryotic with prominent nuclei. The mitotic rate was high, and atypical mitosis and multinucleated cells were occasionally observed. Tumor invasion into blood vessels and the lymphatics occurred, and tumor thrombi were found in capillaries. The stroma was mainly composed of fibrous connective tissue, and small foci of woven bone were also seen occasionally (Fig. 2). Multifocal lymphocytic foci and areas of necrosis and hemorrhage were also observed. In the intestinal mass, intestinal structure was almost completely effaced by tumor. In the lymph node, tumor emboli were detected within the afferent lymphatics and the subcapsular region.
Immunohistochemical staining was performed using alpha-fetoprotein (AFP), a placental alkaline phosphatase (PLAP), b cytokeratin, c carcinoembryonic antigen (CEA), d Wilms tumor 1 protein (WT1), e and vimentin. f Negative controls were obtained by omitting the primary antibodies. A positive reaction to AFP was observed in tumor cells within papillary, tubular, and cyst-like structures (Fig. 4). Positive reactions to PLAP appeared on the tumor cell surface and cytoplasm of approximately 20% of tumor cells, and these cells formed small foci (Fig. 5). Focal cytoplasmic staining for CEA was identified in the tubular neoplastic component, and there was diffuse intense staining of the cytokeratin. The tumor cells did not react to WT1 or vimentin.
For electron microscopy, the paraffin-embedded tumor tissues were deparaffinized in xylene, post-fixed in osmium tetroxide, embedded in epoxy resin, and stained with lead citrate and uranyl acetate. Ultrastructurally, the tumor cells had large nuclei with deep indentations, extensive rough endoplasmic reticulum, and small numbers of mitochondria. Occasionally, microvillus-like structures projected into the luminal spaces, and tight junctional complexes with desmosomes were present between the tumor cells (Fig. 6).
For lectin histochemistry, the following 8 types of biotinylated lectins g were used: concanavalin A (ConA), wheat germ agglutinin (WGA), peanut agglutinin (PNA), Ulex europaeus agglutinin I (UEA-1), Dolichos biflorus agglutinin (DBA), soybean agglutinin (SBA), Griffonia simplicifolia I (GS-1), and Bauhinia purpurea (BPA). Bovine abomasum was used as the positive control. Negative controls were obtained by omitting the biotinylated lectins. Results from the lectin histochemical analyses are shown in Table 1. Positive reactions to ConA appeared in the cytoplasm of the tumor cells. Positive reactions to WGA, PNA, DBA, SBA, GS-1, and BPA were weak in the cytoplasm but strong on the tumor cell surface. The tumor cells did not react to UAE-1.
Comparison of lectin histochemical findings.*
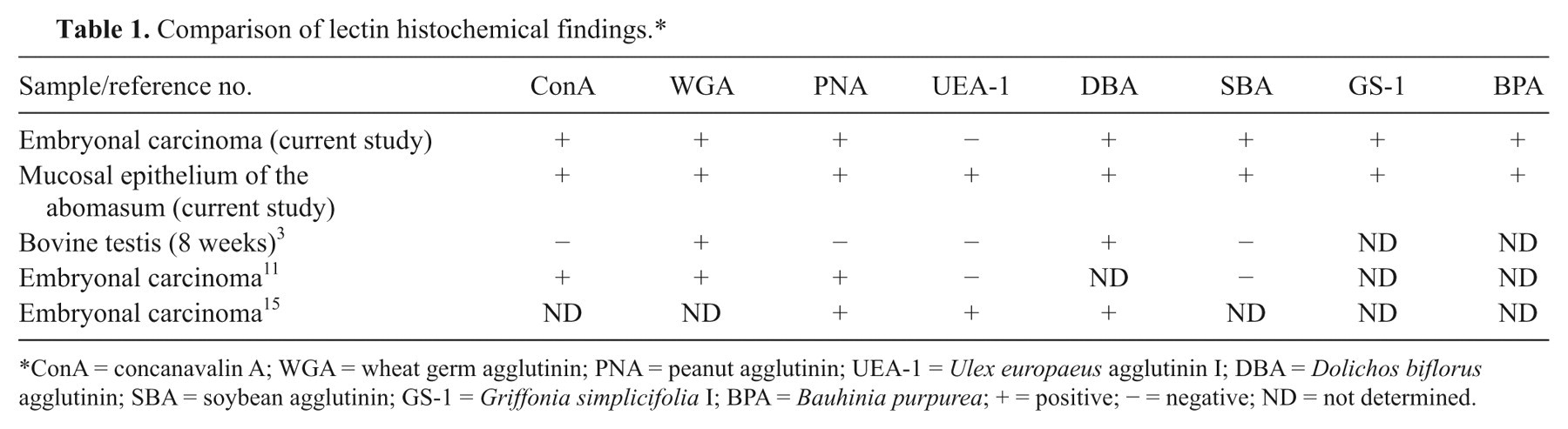
ConA = concanavalin A; WGA = wheat germ agglutinin; PNA = peanut agglutinin; UEA-1 = Ulex europaeus agglutinin I; DBA = Dolichos biflorus agglutinin; SBA = soybean agglutinin; GS-1 = Griffonia simplicifolia I; BPA = Bauhinia purpurea; + = positive; − = negative; ND = not determined.
Embryonal carcinoma is a tumor of primitive appearance with anaplastic epithelial cells that resemble those of early embryonic development arranged in solid, glandular, papillary, and tubular patterns. 10,19 In order to diagnose embryonal carcinoma in domestic animals, positive immunohistochemical staining for AFP is reported to be useful. 10 Alpha-fetoprotein is a glycoprotein that is normally synthesized by the fetal liver and yolk sac, 1 and is also expressed in some cases of malignant neoplasms such as hepatocellular carcinoma, embryonal carcinoma, yolk sac tumor, 18 and AFP-producing gastric carcinoma. 8 In addition, anti-human AFP has been shown to cross-react with many species including cattle, cat, dog, pig, sheep, rat, and monkey. 1,5
Some studies have shown PLAP to be a useful marker for the diagnosis of embryonal carcinoma in human beings, reporting PLAP positivity in approximately 90% of human embryonal carcinoma cases. 16,19 Thus, PLAP may also be a useful marker for the diagnosis of embryonal carcinoma in domestic animals.
Tumors in calves, like those in children, are relatively uncommon; some cases of mesothelioma and malignant lymphoma have been reported. 13,14 In addition, testicular tumors reported in calves have included an interstitial cell tumor in a cryptorchid, 9 a Sertoli cell tumor, 17 and a yolk sack carcinoma. 5 In the current case, differential diagnoses of embryonal carcinoma, yolk sac tumor, mesothelioma, and AFP-producing gastric carcinoma were considered. A diagnosis of yolk sac tumor is justifiable only when one of the many distinctive patterns of yolk sac tumor is identified. 17 These features were lacking in the current case. The fact that tumor cells in the current study stained negative for polysaccharide, vimentin, and WT1, and positive for AFP and PLAP excluded mesothelioma. Result from polysaccharide stain and immunohistochemical analyses are shown in Table 2. The basic histological pattern in the present case was similar to that of embryonal carcinoma and AFP-producing gastric carcinoma, and positive immunohistochemical staining of AFP was a characteristic finding of both tumors. 8,19,20 In human beings, immunostaining for PLAP is usually positive in embryonal carcinoma 16,19 and positive in about half of AFP-producing gastric carcinoma. 6 Carcinoembryonic antigen is usually positive in AFP-producing gastric carcinoma 8 and negative in embryonal carcinoma, 19 though a case of CEA-positive embryonal carcinoma was reported. 2 In the present case, macroscopically visible masses were not present at the lumen of the abomasum, and mucus production was not observed with polysaccharide stains or by electron microscopy. These findings are dissimilar to the distinctive characteristics of AFP-producing gastric carcinoma. 6 Based on the macroscopic, histopathologic, and ultrastructural appearance and on the positive immunohistochemical staining for AFP and PLAP, the present tumor was diagnosed as embryonal carcinoma in the abdominal cavity. The testes were considered to be the origin of the tumor in the present case because the testes were not found at necropsy.
Comparison of staining patterns of various neoplasms.*

AFP= alpha-fetoprotein; PLAP = placental alkaline phosphatase; WT1 = Wilms tumor 1 protein; CEA = carcinoembryonic antigen; + = positive; − = negative; ND = not determined.
Furthermore, lectin histochemistry of embryonal carcinoma has not been examined in domestic animals, leaving the utility of this method for diagnosis of embryonal carcinoma uncertain. In the current report, results were recorded and compared with the results of human embryonal carcinoma 11,15 and with 8-week-old bovine testis. 3 Results from the present case were similar to human embryonal carcinoma except for SBA. In human beings, ConA is a helpful marker for diagnosing embryonal carcinoma and yolk sac tumors, whereby it is positive for embryonal carcinoma and negative for yolk sac tumors. The findings in the current case suggest that ConA may be a useful marker for diagnosing embryonal carcinoma in cattle. More studies are necessary to evaluate the utility of lectin histochemistry on embryonal carcinoma. To the authors’ knowledge, this is the first description of embryonal carcinoma in cattle. For the diagnosis of germ cell tumors in calves, embryonal carcinoma should be included in a differential diagnosis.
Footnotes
a.
Nichirei, Tokyo, Japan.
b.
Zymed Laboratories, San Francisco, CA.
c.
AE1/3; Dako Denmark A/S, Glostrup, Denmark.
d.
All5; Dako Denmark A/S, Glostrup, Denmark
e.
6F-H2; Dako Denmark A/S, Glostrup, Denmark.
f.
V9; Dako Denmark A/S, Glostrup, Denmark.
g.
EY Laboratories, San Mateo, CA.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
