Abstract
The heterogeneous morphologic features of canine plasmacytomas (PCTs) can make their differentiation from other round cell tumors challenging. Immunohistochemistry (IHC) for lambda (λ) and kappa (к) immunoglobulin (Ig) light chains is often equivocal because of high background staining. The chromogenic in situ hybridization (CISH) technique for light chains has shown higher sensitivity compared to IHC in human plasma cell tumors. Therefore, we aimed to validate automated CISH for light chains in canine tissues and to evaluate its diagnostic potential in canine PCTs, in conjunction with routinely used IHC markers. CISH for light chains demonstrated a clear signal in plasma cell populations of canine control tissues (lymph nodes, lymphoplasmacytic inflammation) showing a polyclonal pattern with a prevalence of λ-producing cells. CISH detected monotypic light chain expression in 33 of 53 (62%) PCTs, 31 expressing λ and 2 expressing к. CISH was more sensitive than IHC for λ light chain (58% vs. 47%, respectively) and more easily interpretable given the absence of confounding background staining. The absence of CISH staining for both λ and к in a considerable subset of tumors may be the result of lower light chain production by neoplastic cells. Multiple myeloma oncogene 1 (MUM1) was expressed by all but 2 PCTs (96%), which showed λ expression by CISH and IHC. The identification of poorly differentiated canine PCTs requires the assessment of a panel of IHC markers, with the potential support of CISH for Ig light chains.
Introduction
Plasma cell tumors are frequently diagnosed as extramedullary masses (plasmacytomas, PCTs) in dogs, involving primarily the skin and the oral mucosa. 24 Canine PCT morphology varies from well differentiated, with neoplastic cells resembling typical mature plasma cells, to neoplasms with blastic appearance. 36 For this reason, distinguishing poorly differentiated PCTs from other round cell tumors may represent a diagnostic challenge. 30
Immunohistochemistry (IHC) for multiple myeloma oncogene 1 (MUM1) is widely used to confirm the diagnosis of PCTs in dogs.24,30 MUM1 is a transcription factor involved in regulation of cell growth, especially B-cell differentiation. 13 Conventionally believed to be highly specific for canine PCTs, MUM1 expression has been demonstrated in cutaneous histiocytomas and in 21% of B-cell lymphomas.24,30 Therefore, MUM1 should be used in conjunction with histiocytic (e.g., ionized calcium-binding adaptor molecule 1, Iba1) and lymphoid markers to distinguish some undifferentiated round cell tumors.28,30 Canine PCTs are negative for the T-cell marker CD3, and variably positive for B-cell markers including CD79a (56–82%) and CD20 (19%).24,28 The common leukocyte antigen CD45RA is frequently expressed (94%), although the positivity varies among tumors and in different tumor areas. 28
IHC is frequently used for identification of lambda (λ) and kappa (к) immunoglobulin (Ig) light chains in PCTs.8,23 The vast majority of canine PCTs (97–100%) express the λ light chain, reflecting the normal ratio of Ig light chains produced by canine plasma cells.1,8,23 IHC to detect Ig light chains, although sensitive, may have low specificity for several reasons, including nonspecific adsorption of Ig onto the cell surfaces, internalization of Ig by phagocytes, and binding of Ig to different cell types via Fc receptors. 28 Moreover, the interpretation of results is often confounded by background staining, given the presence of Ig or light chains in the interstitium and because of nonspecific binding to proteins such as collagen. 38 PCR analysis of antigen receptor gene rearrangement (PARR) has been proposed as an adjunct tool to detect clonality in PCTs.27,32,33 A PARR based on Ig к locus demonstrated higher sensitivity compared to the Ig heavy chain locus, although limited literature is available about its specificity.32,33
In human pathology, IHC for light chains is now commonly replaced or associated with RNA in situ hybridization (ISH), which has been proven more sensitive to multiple myeloma, the most common plasma cell neoplasm in humans, and non-Hodgkin B-cell lymphomas.4,7,34 To our knowledge, no studies investigating the applicability of ISH for Ig light chains in canine PCTs have been reported. Therefore, we investigated the chromogenic in situ hybridization (CISH) method for λ and к light chains in canine tissues and evaluated its diagnostic potential in canine PCTs, comparing CISH results with routine IHC marker expression.
Materials and methods
Sample collection and histopathology
We retrieved 53 formalin-fixed, paraffin-embedded (FFPE) samples from canine round cell tumors with a diagnosis of cutaneous, mucocutaneous, or oral PCT from the archives of the Istituto Zooprofilattico Sperimentale delle Venezie (Padua, Italy). Samples had been collected surgically and submitted to the histopathology laboratory between 2015 and 2019. Three pathologists (G. Foiani, C. Zanardello, and A. Carminato) reexamined the selected samples to confirm the diagnosis. The 53 PCTs were collected from 52 dogs, including 48 cutaneous and 5 oral tumors; 35 dogs were females and 17 were males (female-to-male ratio 2:1). The dogs included 17 of mixed breeds, 3 Labrador Retrievers, 3 Boxers, 3 English Setters, 3 West Highland White Terriers, 2 Beagles, 2 Dachshunds, 2 Epagneul Bretons, 2 Yorkshire Terriers, 1 Border Collie, 1 Cavalier King Charles Spaniel, 1 Cocker Spaniel, 1 Dobermann, 1 German Shepherd, 1 Golden Retriever, 1 Great Dane, 1 Jack Russel Terrier, 1 Belgian Tervuren, 1 Pit Bull Mastiff, 1 Poodle, 1 Segugio Italiano, 1 Shar Pei, 1 Shih-Tzu, and 1 Weimaraner. The median age at the time of diagnosis was 10 y (range: 5–17 y). Eighteen of 48 cutaneous PCTs were located on the head, more commonly in the ear (n = 7), lip (n = 4), and chin (n = 3), 18 on the extremities, 10 of which were on the foot, 9 on the trunk, and 3 on the perineum. Of the 5 oral tumors, 3 were located on the gingiva and 2 on the tongue. Two oral PCTs occurred in one dog in different gingival areas, 1 y apart (6- to 7-y-old).
Selected PCTs were histologically classified as cleaved, asynchronous, polymorphous-blastic, mature, or hyaline types. 23 Additional histologic findings such as the presence of amyloid and of intravascular neoplastic cells were also recorded. Nine canine diffuse large B-cell lymphomas (DLBCLs; 8 nodal and 1 intestinal) and 6 non-lymphoid cutaneous tumors (3 melanomas and 3 histiocytic sarcomas) were included in our study, to evaluate light chain CISH performance in non-plasmacytoid tumors.
CISH
CISH was performed in automation (BenchMark ULTRA system; Roche, Ventana Medical Systems). A series of canine control tissues, normally containing plasma cells, was used to develop the CISH protocol: 4 lymph nodes (2 normal and 2 reactive), 2 samples of intestine with lymphoplasmacytic enteritis, and 1 sample of gingiva with lymphoplasmacytic gingivitis. Four-μm thick FFPE tissue sections were mounted onto adhesive glass slides (TOMO; Matsunami). After deparaffinization in aqueous-based detergent solution (EZ Prep; Ventana), target site unmasking was performed by incubating the slides in ULTRA cell conditioning solution 2 (Ventana) for 16 min at 86°C, followed by ISH protease 3 (Ventana) treatment, at 36°C for 8 min. Sections were then incubated with INFORM lambda and kappa probes (Ventana), which are ready-to-use cocktails of fluorescein-labeled oligonucleotide probes, designed for detection of messenger RNA (mRNA) of human λ and к light chains. Denaturation was performed at 82°C for 8 min, followed by hybridization at 46°C for 1 h. After washing 3 times with buffer SSC (Ventana) for 8 min, the hybridization reaction was detected (ISH iVIEW blue detection kit; Ventana). Finally, sections were counterstained with red counterstain II (Ventana), at 36°C for 4 min. Positive signals were detected as blue cytoplasmic deposits. When tumor samples were subjected to CISH, sections of normal and/or reactive lymph nodes were included in each run.
λ and к light chains gene sequence alignment
The sequences of the human light chain CISH probes utilized were not disclosed for patent reasons. Therefore, sequences encoding the constant regions of λ and к light chains of canine and human Igs were retrieved from the Ensembl v.99 online database (https://www.ensembl.org; Table 1). 10 Sequence alignment for both Ig λ and к light chain constant regions was performed comparing 3 different canine sequences with the human counterpart; in addition to a general sequence, sequences from 2 different breeds were also used, to predict possible breed-related variations. To establish the overall and specific gene sequence identity of canine and human light chain constant regions, sequences were aligned using BLAST (https://blast.ncbi.nlm.nih.gov) and BLAST/BLAT (http://www.ensembl.org/). For the Ig λ light chain, the full sequence of chromosome 26 (~ 9,529 nucleotides [nt]) was used as the alignment region for dogs; a specific segment of 1,662 nt encoding for the constant region of λ light chain (IGLC2) was used for humans (located on chromosome 22). For the Ig к light chain, the entire available sequence of canine chromosome 17 (4,197 nt) was aligned over a shorter (1,723 nt) segment of human chromosome 38, encoding for the constant region of к light chain (IGKC).
Accession identifiers of sequences used for alignment, grouped by species and immunoglobulin type. Genetic information was retrieved from the Ensembl v.99 database (https://www.ensembl.org).

IGKC = immunoglobulin kappa light chain constant region; IGLC = immunoglobulin lambda light chain constant region.
IHC
Automated IHC was performed (BenchMark ULTRA system; Ventana) using primary antibodies anti-λ light chain (CONFIRM anti-lambda rabbit polyclonal primary antibody; Ventana), MUM1, CD3 (Dako), CD20 (Thermo Scientific), CD45RA (Dr. Peter Moore, UC-Davis School of Veterinary Medicine), and Iba1 (FUJIFILM, Wako Pure Chemical; Table 2). Briefly, 3-μm thick FFPE tissue sections were mounted on adhesive glass slides, deparaffinized in aqueous-based detergent solution (EZ Prep; Ventana), and subjected to heat-induced antigen retrieval. After detection, sections were counterstained with Mayer hematoxylin, dehydrated, and mounted (Eukitt; Kaltek). Positive controls from canine tissues (Table 2) and tumor sections with omission of the primary antibodies were included in each run.
Panel of antibodies applied to canine plasmacytomas.
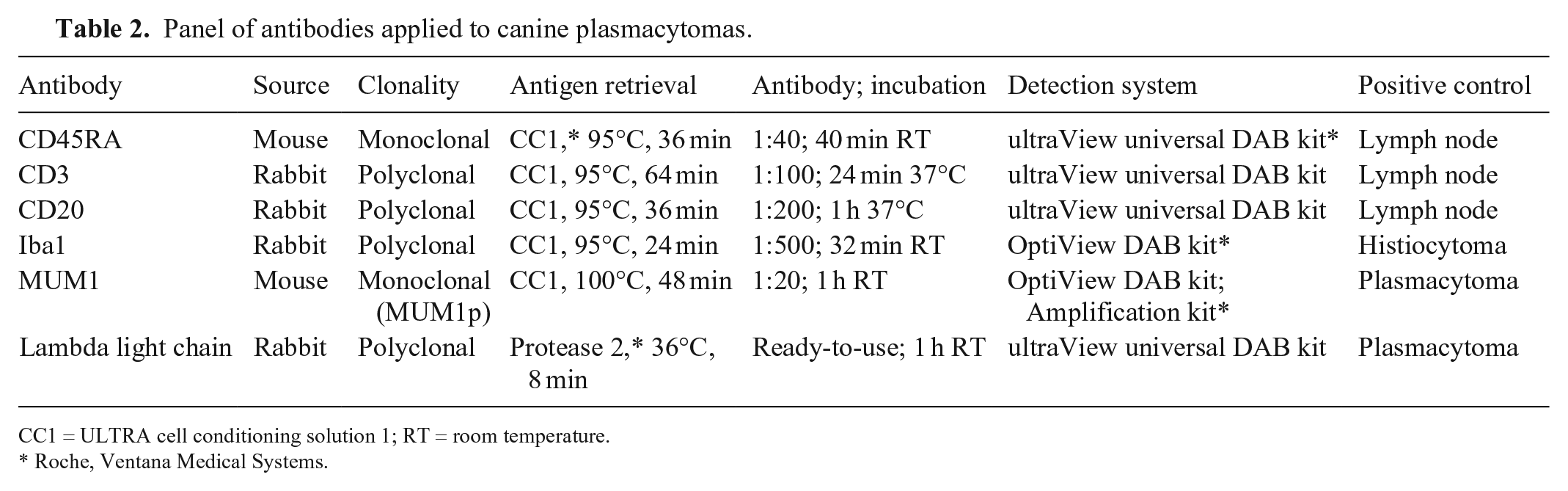
CC1 = ULTRA cell conditioning solution 1; RT = room temperature.
Roche, Ventana Medical Systems.
Results
Histopathology
PCTs were histologically classified as cleaved (18 of 53, 34%), asynchronous (18 of 53, 34%), polymorphous-blastic (10 of 53, 19%), mature (6 of 53, 11%), or hyaline (1 of 53, 2%) types. 23 In 5 of 18 cleaved PCTs, neoplastic cells had features of transition to the hyaline type (large quantity of lightly eosinophilic cytoplasm and eccentrically located crescent-shaped nuclei). 8 Six of 53 (11%) tumors had abundant amyloid deposition, and intravascular neoplastic cells were detected in 3 of 53 (6%) tumors.
CISH
In all canine control tissues used for protocol development, a polyclonal plasma cell population was detected, with a predominance of λ light chain expression. No reaction was observed in non-lymphoid cells. For both light chains, CISH positivity was identified as an intense blue chromogen deposit occupying all of the cytoplasm of plasma cells, and sometimes obscuring the nucleus. Background staining was low or absent. In lymph nodes, positive plasma cells expressed λ and к in a ratio of ~ 9:1 and were mainly observed in the medullary cords (Figs. 1, 2). A few scattered positive cells were also detected in the capsule, in the interfollicular space, and within germinal centers (Figs. 3, 4). In samples affected by lymphoplasmacytic enteritis and gingivitis, a polyclonal inflammatory population expressing both light chains was observed.
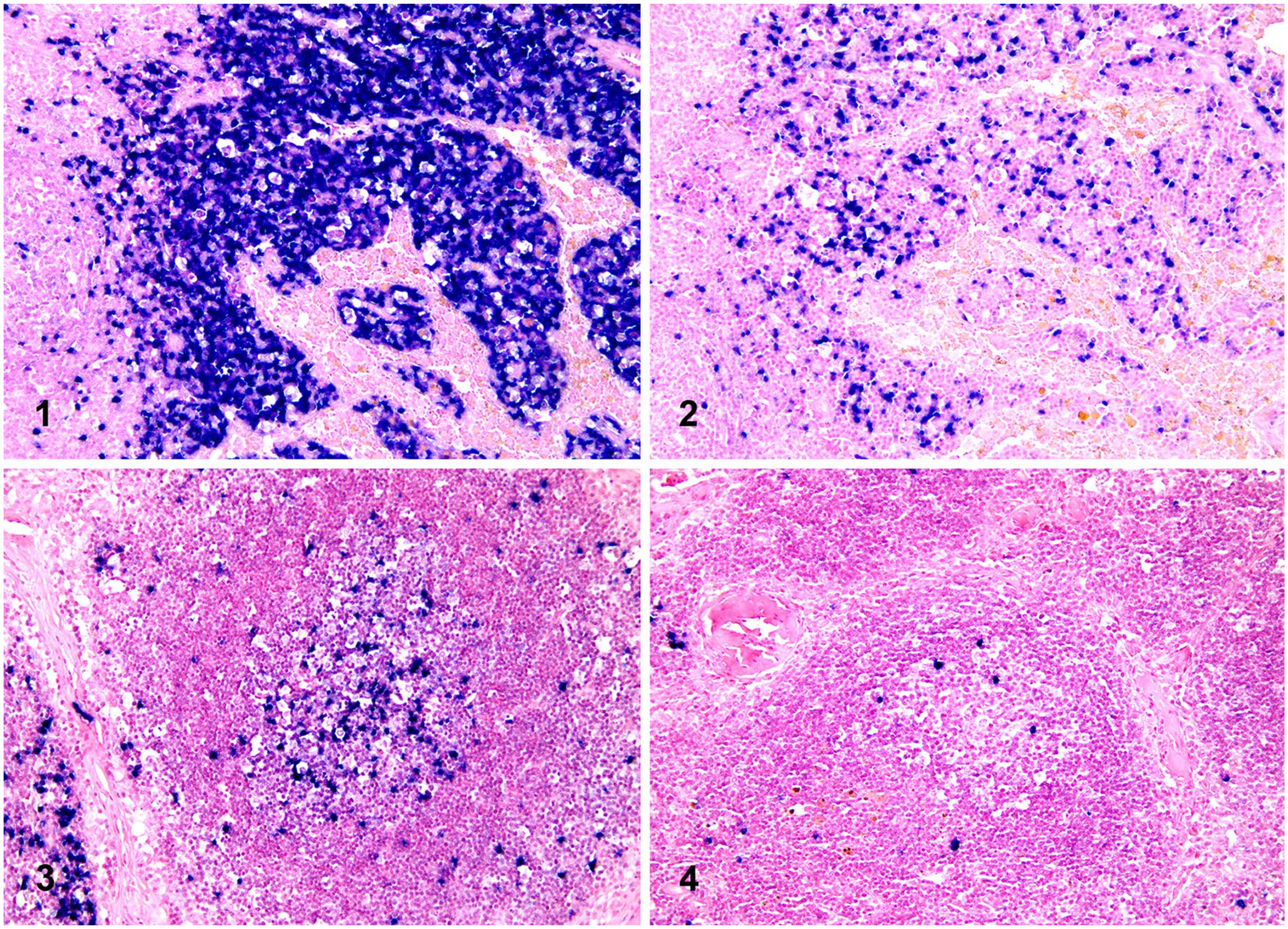
Chromogenic in situ hybridization (CISH) for lambda (λ) and kappa (κ) light chains in canine lymph node sections (ISH iVIEW blue detection kit, red counterstain II; Ventana).
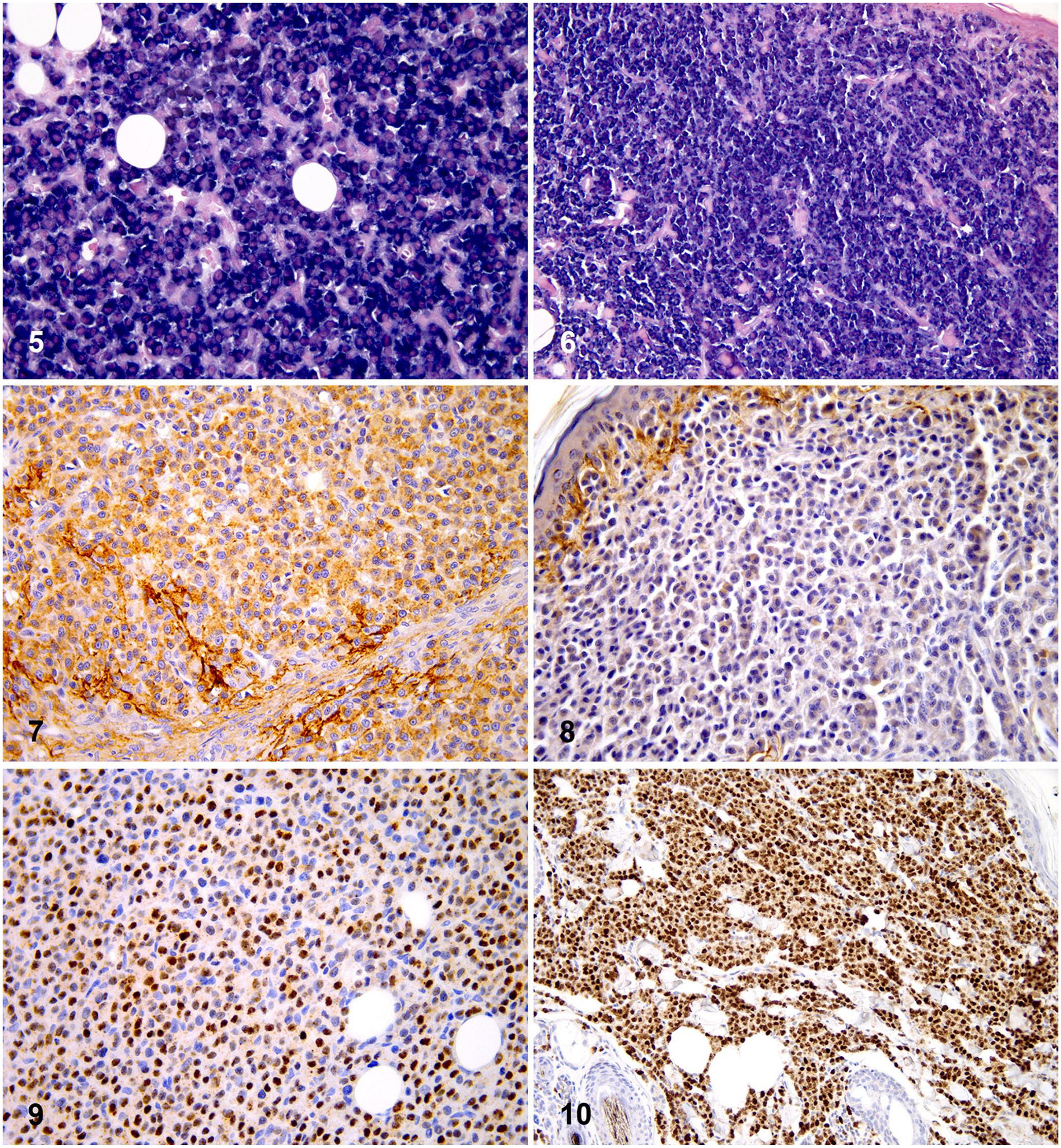
Thirty-three of 53 (62%) PCTs had monotypic light chain production demonstrable by CISH, 31 of 53 (58%) expressing λ (Fig. 5) and 2 of 53 (4%) expressing к light chains (Fig. 6). Tumor cells were diffusely and uniformly stained, except for 3 of 31 λ-positive cases in which staining was multifocal. In 20 of 53 (38%) PCTs, a clear CISH signal was not detectable in tumor cells. Most cases, including CISH-negative PCTs, had a polyclonal population of plasma cells at the periphery of lesions, serving as positive internal controls.
Comparison of chromogenic in situ hybridization (CISH) and immunohistochemical (IHC) findings in 2 canine cutaneous plasmacytomas (PCTs).
In DLBCLs, light chain expression was demonstrated by CISH in 2 of 9 (22%) cases, 1 of which (nodal) expressed λ and the other (intestinal) the к chain; λ and к expression was multifocal and less intense than control or PCT cases. In the remaining 8 DLBCLs, rare λ- and к-positive scattered cells were observed. All of 6 non-lymphoid tumors (3 melanomas and 3 histiocytic sarcomas) were negative for both λ and к chains.
λ and к light chains gene sequence alignment
We found that alignment of the full canine chromosome 26 sequence with the human IGLC2 sequence returned an overall identity of 58%. However, we detected 3 large segments of nucleotides returning a higher identity. The 3 λ light chain sequences with higher identity of 476 and 498 nt had the following identities: 86.5% in region 2 of chromosome 26, 86.2% in region 7 of chromosome 26, and 86.2% in region 6 of chromosome 26. Additionally, we detected shorter sequences of 11–28 nt with high identity within the aligned sequences. Specifically, continuous nt ranging from 238 to 266 (CACAGCAGCTTCAGCTGCCTGGTCACGCA) returned an identity of 89.7% (6-nt difference from the human sequence).
The overall alignment between canine chromosome 17 and the human IGKC sequence elicited an identity of 71.9–72.7%. The continuous area of the canine к light chain sequence with the highest identity of 96.2% was sited between nucleotides 1–26 (TGCCTCTGTTGTGTGCTTGCTGAATA) with a gap of 2 nt compared to the human counterpart.
IHC
Twenty-five of 53 (47%) PCTs had λ light chain cytoplasmic expression by IHC (Fig. 7), which was multifocally distributed in 9 cases. Nineteen (36%) tumors were negative, and 9 (17%) tumors were characterized by small peripheral areas of positivity that were considered as non-informative for diagnostic purposes (Fig. 8). In general, moderate-to-high levels of background staining were observed.
Fifty-one (96%) PCTs had unequivocal nuclear positivity for MUM1 (Figs. 9, 10). In addition to the strong nuclear staining, weak-to-moderate granular cytoplasmic positivity was frequently present. In the remaining 2 of 53 (4%) tumors, MUM1 positivity was weak and focally distributed in a small number (< 10%) of neoplastic cells, thus these samples were considered negative.
Of note, 6 of 53 (11%) PCTs with negative or non-informative IHC for λ light chains, and both cases with negative MUM1 IHC, had clear CISH positivity for λ light chains. All of 20 cases negative for both light chains by CISH were also negative (14 cases) or non-informative (6 cases) for λ light chains by IHC.
Overall, 20 (38%) cases demonstrated mainly membranous CD20 positivity of neoplastic cells, which was multifocal in 17 cases. Moreover, in most CD20-negative tumors, scattered positive cells were observed, often with large or multiple nuclei. Infiltration of CD3- or CD20-positive small mature lymphocytes was frequent, dispersed throughout lesions or clustered peripherally and around vessels. CD45RA expression was observed in 42 of 53 (79%) cases, with a multifocal pattern in 28 of them. All tumors were negative for Iba1 and CD3.
All 9 DLBCLs were CD45RA and CD20 positive. MUM1 was multifocally expressed in 2 of 9 (22%) DLBCLs, with both nuclear and cytoplasmic staining patterns. The 2 cases were the same ones that had CISH positivity for one light chain. In the other 7 DLBCLs, rare MUM1-positive cells were scattered throughout the neoplasms. The 3 melanomas and 3 histiocytic sarcomas were negative for all of the IHC markers tested, except for strong and diffuse membranous and cytoplasmic positivity for Iba1 in the histiocytic sarcomas.
Discussion
In normal and hyperplastic canine lymph nodes, we obtained a strong and clear CISH signal in the cytoplasm of λ- and к-producing plasma cells, with absent or low background staining that did not affect the interpretation of results. Plasma cell localization resembled the pattern of distribution reported for human tissues. 37 In addition, a subset of germinal center cells compatible with centrocytes and centroblasts were also stained, demonstrating that CISH can detect light chain expression in non-plasmacytoid B cells, as reported for humans. 21 In samples of lymphoplasmacytic inflammation, we observed a polyclonal pattern of concurrent λ and к light chain expression. In all analyzed samples, most plasma cells expressed λ chains, reflecting the normal ratio of Ig light chain production in canine plasma cells, which is ~ 9 λ to 1 к. 1
The INFORM lambda and kappa probes utilized in our work came from ready-to-use cocktails of fluorescein-labeled oligonucleotide probes, designed for detecting mRNA of human λ and к light chains. Unfortunately, information about the probe sequences was protected by patent and could not be retrieved. However, alignment of the canine and human DNA sequences for the constant regions of λ and к light chains returned identities of several areas that were > 80%, which is regarded as the threshold for cross-reactivity of markers raised against human molecules with the corresponding mammalian species of interest. 16 Others 19 also have found that Ig λ light chains have a higher degree of homology with the corresponding human sequences, especially near the midpoint of the human locus. In the lack of specific information regarding CISH probe targets, к light chain canine sequences are difficult to compare with the human counterpart, given that their organization is complex and bears several areas with inverted loci, pseudogenes, and block duplications. 19
Likely, the commercial probes that we used contain oligonucleotides identifying highly preserved regions of light chains. This hypothesis was supported by the expression patterns observed in normal canine tissues that paralleled those reported for human tissues and were indicative of the specificity of the CISH reaction in dogs. This hypothesis is further substantiated by the observation that conserved sequences of the cytoplasmic domains of other molecules with relevant immune functions (such as CD3, CD79a, and CD20) are characterized by high homology among mammalian species, and that human antibodies raised against these molecules have demonstrated effective cross-reactivity in several species, including dogs. 11 Thus, it is not surprising that similar conditions exist for the constant regions of Ig light chain sequences.
In the canine PCTs that we studied, CISH detected monotypic light chain expression in 62% of the selected cases, providing additional evidence of their monoclonal origin. In agreement with human studies, 4 CISH was more sensitive than IHC in detecting λ light chain expression (58% vs. 47% of cases, respectively). The absence of confounding background staining, together with higher signal intensity, represented a crucial advantage of the CISH over IHC for detecting light chains.4,34 However, we detected a lower overall percentage of light chain–positive PCTs than expected, either by CISH (58% λ- and 4% к-positive) or IHC (47% λ-positive). All of 20 CISH-negative cases were correspondingly negative or non-informative by IHC for λ chain. In the 2 largest studies on canine PCTs,8,23 IHC positivity for λ or к light chain was considered necessary for a definitive diagnosis, reporting slightly different ratios between the λ- and к-stained tumors (64 of 64, 100% λ-positive; 114 of 117, 97% λ-positive; 3 of 117, 4% к-positive cases). Other authors reported small percentages of non-stained tumors diagnosed as PCTs by morphology: 1 of 26 (4%) cases, which was also negative for Ig heavy chains IgG, IgM, and IgA (among 22 of 26, 85% λ-positive; 3 of 26, 12% к-positive cases) and 2 of 29 (7%) cases (among 20 of 29, 69% λ-positive; 7 of 29, 24% к-positive cases).6,17 Our study contains technical differences in the IHC method from previous work on canine tissues, which may have contributed to these conflicting results. Specifically, we carried out our IHC procedure for light chains using an automatic stainer, which should improve staining quality. 5 The ready-to-use antibody solution that we used (CONFIRM anti-lambda rabbit polyclonal primary antibody) has not been reported previously, to our knowledge. However, the close correspondence between CISH and IHC results suggests that negative cases are probably not related to the method. We could speculate that preanalytical processing (such as low or excessive fixation) may have altered protein and mRNA integrity. 9 Nevertheless, the presence of intensely stained normal plasma cells in most cases served as positive internal controls, for both CISH and IHC. Despite our negative results, the plasma cell origin of these tumors was strongly supported by cell morphology, MUM1 positivity, and Iba1 negativity. Therefore, the possibility that these tumors produce little-to-no amount of light chains is a likely hypothesis that needs to be further investigated. In humans, oligosecretory and non-secretory multiple myelomas have been described,14,20,22,29,31,35 and there are rare reports of cutaneous PCTs negative for both light chains. 3
CISH detected light chain expression in 2 of 9 (22%) DLBCLs, with a multifocal and rather less intense staining than PCTs, which may be interpreted as indicative of the relatively low levels of light chain mRNA expression in lymphoma cells compared with neoplastic plasma cells. 4 The small percentage of positives obtained in our study is not surprising given that CISH for light chains also demonstrated a lower sensitivity for lymphomas compared to plasma cell tumors in humans, and CISH sensitivity is related to the B-cell lineage and maturation stage.4,25
In our study, a high percentage (96%) of PCTs were MUM1 positive, in agreement with previous reports (93.5–100%).24,30 Given this high sensitivity, IHC for MUM1 is routinely used to confirm the plasma cell origin in canine tumors, often replacing the IHC for light chains.12,24 However, the specificity of MUM1 for PCTs has recently been questioned by a study that demonstrated consistent expression of MUM1 in a series of 20 canine histiocytomas. 30 The product of the MUM1/IRF4 gene is a member of the interferon regulatory factor family of transcription factors, involved in B-cell differentiation and T-cell activation. 13 In addition to plasma cells, MUM1 is also expressed in subsets of germinal center B cells, activated T cells, macrophages, and dendritic cells.13,18 In our study, MUM1 expression was also detected in 2 of 9 (22%) DLBCLs, confirming previous results obtained in DLCBLs, other B-cell lymphomas (follicular and with plasmablastic differentiation), and anaplastic lymphomas. 24 Noteworthy, both MUM1-positive DLBCLs had concurrent light chain expression, as detected by CISH. These findings were suggestive of possible plasmablastic differentiation that was further supported by the morphology of large cells with a cytoplasmic hof. Therefore, according to our findings and to previous reports, 30 MUM1 expression alone is not sufficient for the confirmation of plasma cell origin and needs to be interpreted with caution and with the support of cell morphology and of additional diagnostic procedures including other markers and/or CISH. In the 2 PCTs that were negative for MUM1 (4%), a plasma cell origin was recognized by CISH and IHC expression for λ light chain. Therefore, these methods could be helpful in cases with negative or non-informative MUM1 staining.
The expression of CD20 and CD45RA was observed in 38% and 79% of PCTs, respectively, and staining pattern was characterized by heterogeneous distribution in the neoplastic population. In previous studies, the percentage of positive PCTs was lower (19%) for CD20 and higher (94%) for CD45RA.24,28 These discrepancies could be the result of a different sensitivity of the methods or to case selection. In humans, despite conflicting results of early studies, the pan–B-cell marker CD20 is expressed in a subset (13–22%) of multiple myelomas, and therapies with anti-CD20 monoclonal antibody have been attempted.15,26
On the basis of the histologic classification, 23 the most frequent histologic types of PCTs were cleaved and asynchronous (both with a frequency of 34%), in agreement with previous studies. 24 Most tumors had a mixture of histologic features, which made the definition of a specific type difficult in some cases. A second classification system for canine PCTs, including the monomorphous-blastic type, has been proposed by other authors. 8 Both systems do not seem to correlate with biologic behavior, in contrast to the original classification system of human multiple myeloma from which they derive.2,23 Considering the lack of prognostic significance, together with the difficulty in applying these classification systems, we believe that the definition of a more useful and simplified morphologic classification for PTCs should be developed. 8
Amyloid extracellular deposition was present in 11% of cases in our series, similar to previous studies (8.4–12.5%).8,24 Intravascular neoplastic cells were detected in a lower percentage of cases (6%) in comparison with results of another study (16%). 12 Neither of these 2 histologic features has been demonstrated to be correlated with prognosis.8,12
Based on our data, IHC for MUM1 continues to be the most sensitive marker for canine PCT diagnosis. However, given a lower specificity than previously believed, MUM1 testing should be supported by other markers. The diagnostic algorithm of poorly differentiated canine PCTs should include a panel of different IHC markers and, ideally, the support of CISH for Ig light chains.
Footnotes
Acknowledgements
We thank the technical staff at the Laboratory of Histopathology of the Istituto Zooprofilattico Sperimentale delle Venezie of Padua, Italy, for their assistance with this study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
