Abstract
A diagnosis of light-chain multiple myeloma was made in an 11-year-old male American Shorthair cat. The cat showed atypical plasma cell infiltration in the bone marrow, biclonal gammopathy caused by polymerization of myeloma protein (M-protein), and Bence-Jones proteinuria. The M-protein in the serum of the cat was analyzed by using 12% sodium dodeyl sulfate (SDS) polyacrylamide gel electrophoresis with Coomassie brilliant blue staining. An intense band with a size of 27 kDa, the size of the immunoglobulin light chain, was clearly observed, whereas the band corresponding to the immunoglobulin heavy chain (59 kDa) was undetectable. The 27-kDa band was confirmed to be an immunoglobulin light chain by Western blotting by using antibodies for feline immunoglobulin. These data suggested that the neoplastic plasma cells produce light chain only, leading to the diagnosis of light-chain multiple myeloma in the cat.
Keywords
Multiple myeloma is a malignant plasma-cell dyscrasia characterized by bone-marrow plasmacytosis. Malignant plasma cells produce an abnormal monoclonal immunoglobulin; it is the most common cause of monoclonal gammopathies in humans and other animals, including dogs and cats. 4,9,12 The monoclonal immunoglobulin produced may be an entire immunoglobulin of any class, a heavy chain only, or a light chain only (light-chain multiple myeloma, also called Bence-Jones myeloma). Multiple myeloma is an uncommon neoplasm of cats and accounts for less than 1 % of feline hematopoietic neoplasms. 3,7 Limited information is thus available regarding the production of immunoglobulin in feline malignant plasma cells, and myeloma producing light chain solely has not been previously reported. In this study, light-chain multiple myeloma was diagnosed in the case of a cat.
An 11-year-old male American Shorthair cat was admitted to the Veterinary Medical Teaching Hospital, Nippon Veterinary and Life Science University, with anorexia, fever, and a convulsion observed the day before admission. In clinical examination, the cat was overweight (5.9 kg) and had a fever (40.0°C). On the day of presentation, complete blood cell counts (CBC), serum biochemistry, and urinalysis were performed. The CBC was normal, except for nonregenerative anemia (hematocrit, 23%; reference range, 30%–45%) and lymphopenia (835/μl; reference range, 1,500–7,000/μl), suggesting chronic disease and stress. In serum biochemistry, marked hyperproteinemia (12.9 g/dl; reference range, 5.4–7.8 g/dl) caused by hyperglobulinemia (9.5 g/dl; reference range, 2.6–5.1 g/dl) was detected. The albumin globulin (A G) ratio was 0.36 (reference range, 0.45–1.19). These data suggested that the cat had monoclonal or polyclonal gammopathy. To distinguish these gammopathies, serum analysis was performed by using cellulose acetate electrophoresis. Although, unexpectedly, a sharp biclonal peak was identified in the y region (Fig. 1A), the presence of biclonal gammopathy was considered to be responsible for the hyperglobulinemia. In the serum biochemistry panel of the liver, the values of alanine aminotransferase (111 U/L; reference range, 6–83 U/L), aspartate aminotransferase (785 U/L; reference range, 26–43 U/L), and alkaline phosphatase (186 U/L; reference range, 2593 U/L) were mildly to markedly increased. The serum biochemistry panel of the kidneys was normal. Serum tests for feline immunodeficiency virus and feline infectious peritonitis virus antibodies and for feline leukemia virus antigen were negative. The urinalysis of a voided urine sample showed abnormal findings, including 3+ proteinuria and a specific gravity of 1.040, considering that glomerulopathy could be present.
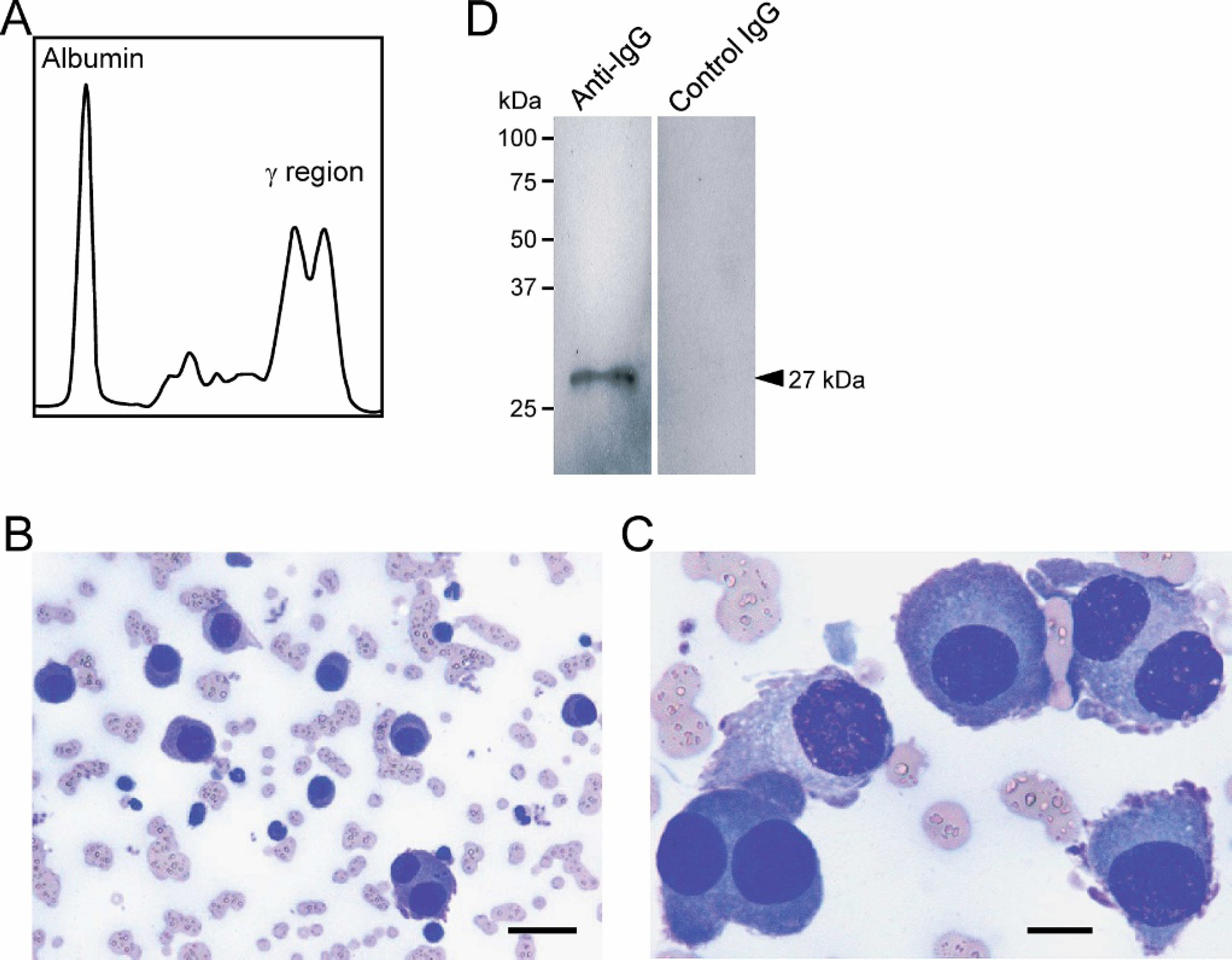
Diagnostic findings of multiple myeloma in the cat.
A bone marrow aspiration biopsy was then performed on the day of presentation because of a suspicion that the cat had multiple myeloma, because of an abnormality in the serum biochemistry, including hyperproteinemia, hyperglobulinemia, decreased A/G ratio, and a clonal peak pattern of serum electrophoresis. In the light microscopic findings of a bone marrow smear stained with Wright-Giemsa, marked plasmacytosis (>50% of bone marrow cells), with anisocytosis of the plasma cells was observed in focal regions of a slide (Fig. 1B), and other regions had moderately increased numbers of the plasma cells (approximately 20% of bone marrow cells). These plasma cells had “flame cell” morphology, characterized by peripheral pink-stained cytoplasmic processes, and displayed atypical morphology, including anisokaryosis, variable nucleus: cytoplasm (N:C) ratios, and several binucleated cells (Fig. 1C). A urine sample was thus analyzed for the detection of Bence-Jones protein with Western blotting. Urine from the cat was diluted with phosphate-buffered saline (PBS) to the protein concentration of 150 μg/ml and boiled for 5 min in 1 × SDS polyacrylamide gel electrophoresis (SDS-PAGE) sample buffer. An aliquot was applied to 12% SDS-PAGE (300 ng protein/lane), transferred onto a polyvinylidene fluoride membrane, a and blotted with fluorescein isothiocyanate (FITC)-labeled sheep anti-feline immunoglobulin (Ig) G antibody, b which recognizes both heavy and light chains of feline IgG, or FITC-labeled sheep IgG (control). The fluorescent signal of the immunoreactive proteins was visualized by using the VersaDoc 5000 imaging system. c As shown in Fig. 1D, an immunoreactive single band was clearly observed at the size of 27 kDa, which is the corresponding size of the immunoglobulin light chain, by an anti-feline IgG antibody, whereas, no band was observed with the control IgG, which indicated that the urine Bence-Jones protein was positive in the cat. No obvious multiple punctate lytic lesion was identified on survey skeletal radiographs. The cat's condition was then diagnosed as multiple myeloma based on the identification of clonal gammopathy, atypical plasma-cell infiltration in the bone marrow, and Bence-Jones proteinuria.
The peak in the y region on electrophoresis analysis in multiple myeloma is typically single; however, some cases of multiple myeloma have been reported to have a biclonal peak in humans, as well as in cats. 1,5,11,15 Biclonal gammopathy is likely to occur by the polymerization of immunoglobulin, 1 a neoplastic transformation of a cell clone undergoing immunoglobulin class switching 8 or independent transforming events yielding proliferation of unrelated plasma cell clones. 16 To examine the possibility that the biclonal gammopathy is caused by the polymerization of produced immunoglobulin, serum of the case was analyzed by agarose gel electrophoresis under the non-reducing condition and the mildly reducing condition that dissociates polymerized immunoglobulin. Sera were diluted with PBS or PBS that contained 2-mercaptoethanol (2ME), a reducing agent, to a final concentration of 0.1 M. After sera were incubated at 37°C for 2 hr, 5-μl aliquots were electrophoresed on a 1 % agarose gel, followed by staining with Coomassie brilliant blue (CBB) and densitometric tracing. A sharp biclonal peak was identified in the γ region (right of the pattern; - electrode side) under the non-reducing condition (Fig. 2; upper panel), whereas, one of the more cathode side peaks was integrated into a single peak at the more anode side peak (the side of albumin; + electrode side) under the reducing condition (Fig. 2; lower panel). Although some part of the second peak seems to be remaining as a shoulder peak in the reducing condition, it could reflect some incomplete dissociation of the multimers in the mildly reducing condition. Therefore, the biclonal peak found in this case was considered to represent the polymerization of immunoglobulin rather than the class switching of immunoglobulin or the biclonal nature of plasma-cell clones.
The myeloma protein (M-protein) was then analyzed by higher resolution electrophoresis. Sera from the patient and a normal cat were examined by 12% SDS-PAGE (300 ng protein/lane) followed by CBB staining (Fig. 3A). In contrast to normal cat serum, an intense band of 27 kDa, the size of the immunoglobulin light chain, was clearly observed and the band corresponding to the immunoglobulin heavy chain (59 kDa) was undetectable in the serum of the affected cat. These findings suggested that the malignant plasma cells produced the light chain but not the heavy chain of immunoglobulin. To confirm the 27-kDa paraprotein as an immunoglobulin light chain, Western blotting by using an antibody for feline IgG (the same antibody used for the detection of urine Bence-Jones protein) was performed after separation of serum proteins with 12% SDS-PAGE (300 ng protein/lane) (Fig. 3B; left panel). In the affected cat, an immunoreactive band of 27 kDa was clearly detected that was far more intense than that in normal serum, indicating that the paraprotein in the case was a light chain of immunoglobulin. Consistent with the results of protein electrophoresis shown in Fig. 3A, no detectable band of 59 kDa was observed in the affected cat serum by the same Western blotting (Fig. 3B; left panel), as well as that using feline-specific mouse monoclonal antibody for immunoglobulin heavy chain d (Fig. 3B; right panel). On the basis of these data, a diagnosis of light-chain multiple myeloma was made in the cat.
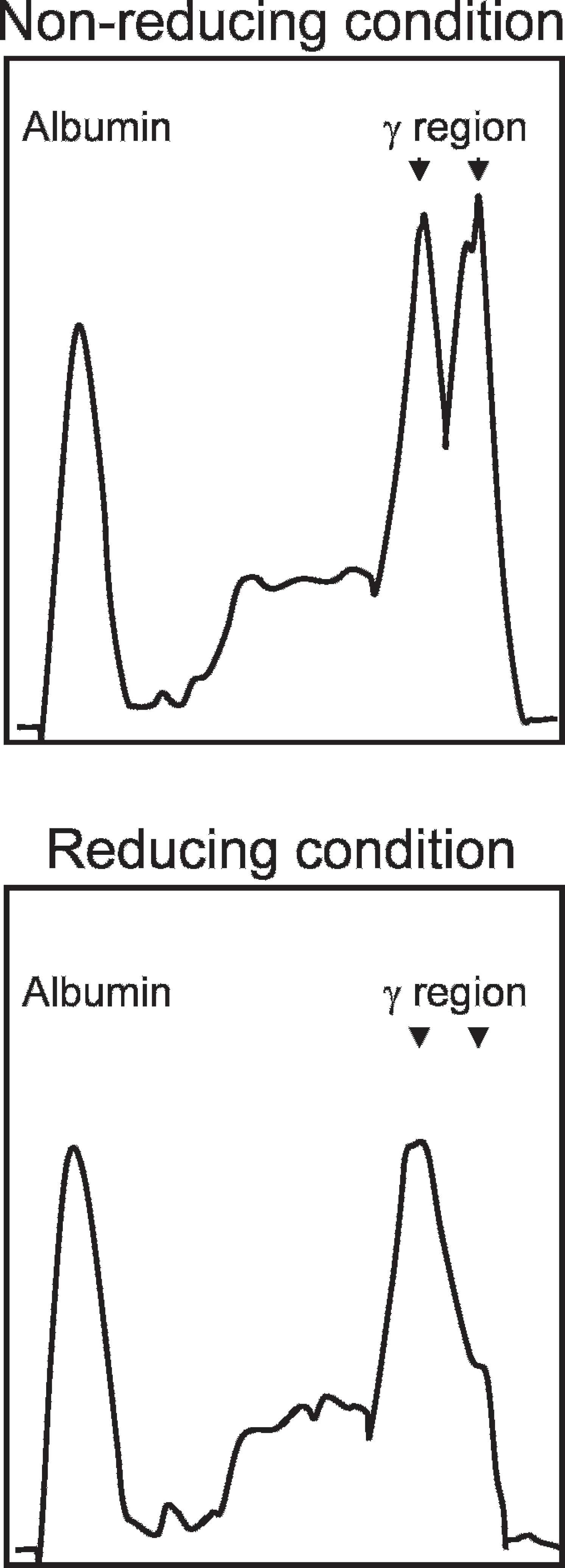
Serum agarose gel electrophoresis under non-reducing and mildly reducing conditions. A sharp biclonal peak (indicated by arrowheads) was identified in the y region under the nonreducing condition
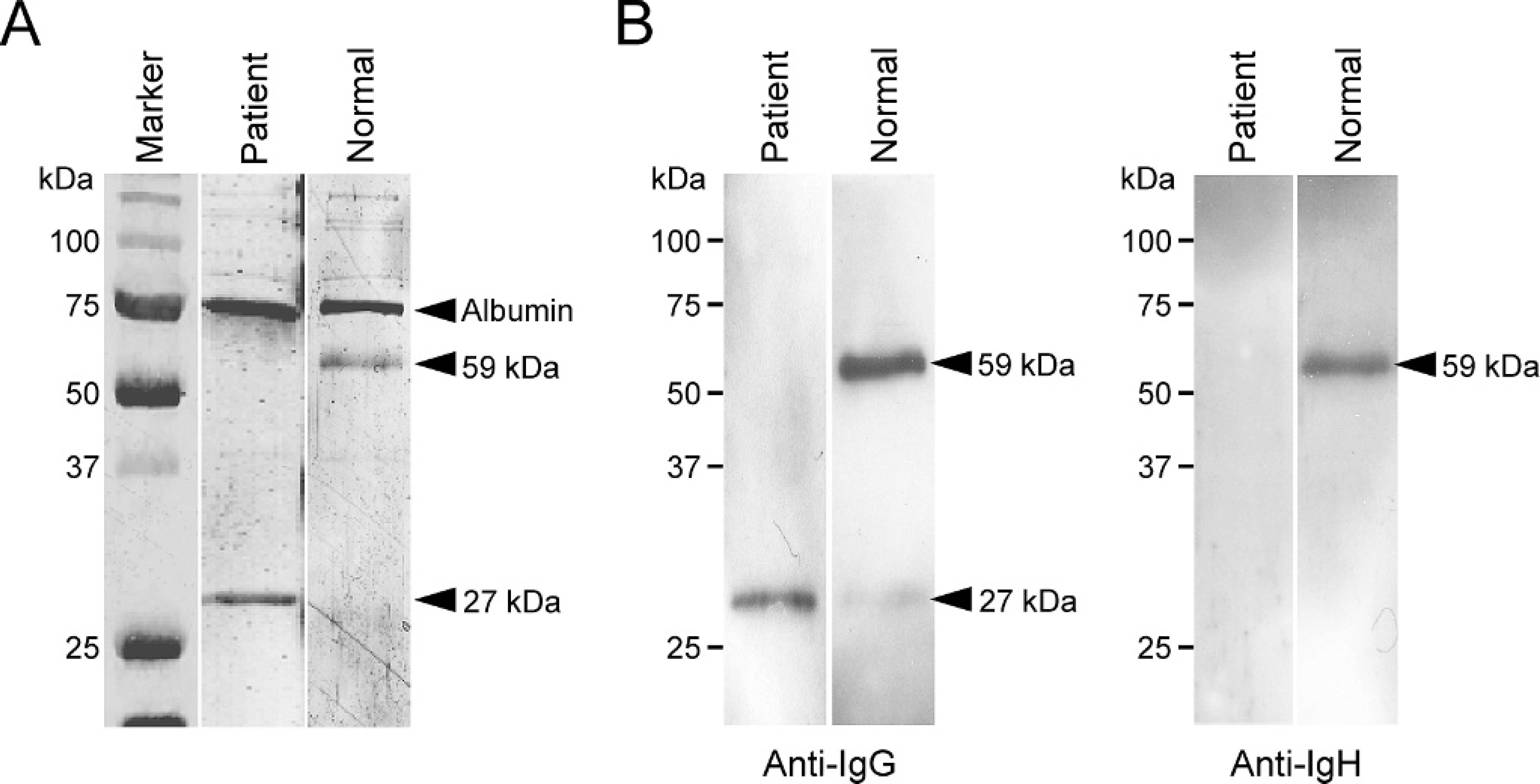
Detection of light-chain gammopathy.
The cat underwent treatment with prednisolone, but no other chemotherapy, because of deterioration of the systemic condition. The cat died 2 days after the first presentation, despite supportive therapy.
Proliferation of malignant plasma cells in bone marrow might interfere with the normal production of erythrocytes, resulting in nonregenerative anemia. Elevated liver enzymes may reflect metastasis or ischemia from altered blood viscosity because of M-protein, and intense proteinuria with an increased specific gravity may indicate the presence of glomerulopathy because of light chains nephrotoxicity. These pathophysiologic abnormalities could contribute to the deterioration of systemic condition in the cat.
The most common M-proteins in multiple myeloma are IgG and IgA in humans, as well as in dogs, 4,10,13 and other classes are rare. Reports of multiple myeloma that define the class of M-protein in cats have been infrequent; there are reports of IgG, IgA, and IgM, 1,6,14,17 but no previous report of light-chain multiple myeloma was found. In cases of human light-chain multiple myeloma, the detection of serum monoclonal gammopathy was relatively rare (found in 18% of patients) in spite of Bence-Jones proteinuria because of the efficient renal excretion of free light chain. 10 A case of light-chain myeloma without serum monoclonal gammopathy has been also reported in a dog. 2 In the present case, the prevention of light-chain excretion by polymerization of the chain may be associated with the existence of gammopathy in serum.
acknowledgements. This research was supported partially by a Grant-in-Aid for Scientific Research (18580323) and “Academic Frontier” Project for Private Universities: matching fund subsidy (2005-2009) from Ministry of Education, Culture, Sports, Science and Technology of Japan (MEXT).
Footnotes
a.
Millipore, Bedford, MA
b.
American Qualex, San Clemente, CA
c.
Bio-Rad, Richmond, CA
d.
Serotec, Oxford, UK
